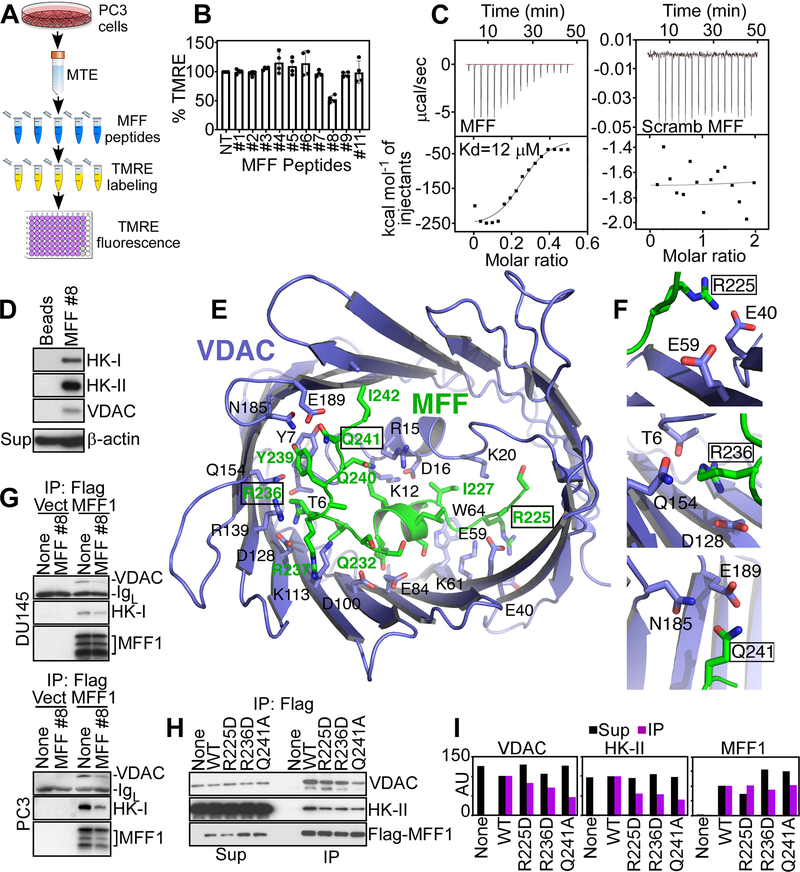
Figure 2.
Peptidyl mimicry of MFF-VDAC1 complex. A, Schematic diagram of high-throughput screening of MFF-derived peptides for inhibition of mitochondrial inner membrane potential. MTE, mitochondrial extracts. B, The conditions are as in (A) and MFF-derived peptides (20 μM for 40 min) were screened for modulation of mitochondrial membrane potential by TMRE staining. Mean±SD (n=2). C, Isothermal titration calorimetry binding data of WT (MFF, left) or scrambled (Scramb, right) MFF peptide to human recombinant VDAC1. Representative experiment (n=4). D, PC3 cell extracts were fractionated on beads-coupled MFF peptide #8 by affinity chromatography and bound proteins were identified by Western blotting. Sup, supernatant. E, Structural model of VDAC1-MFF peptide #8 complex generated with the CABS-Dock server. The MFF peptide (green stick) binds the interior cavity of VDAC1 (blue carton/stick) in an extensive network of interactions blocking access to solutes. F, Predicted contact sites of MFF residues R225, R236 or Q241 with VDAC1. G, DU145 or PC3 cells transfected with vector (Vect) or Flag-MFF1 cDNA were immunoprecipitated (IP) with an antibody to Flag, treated with MFF peptide #8 and analyzed by Western blotting. IgL, Ig light chain. H and I, PC3 cells transfected with WT Flag-MFF or Flag-MFF R225D, R236D or Q241A mutant cDNA were immunoprecipitated (IP) with an antibody to Flag and immune complexes were analyzed by Western blotting (H) and quantified by densitometry (I). AU, arbitrary units.