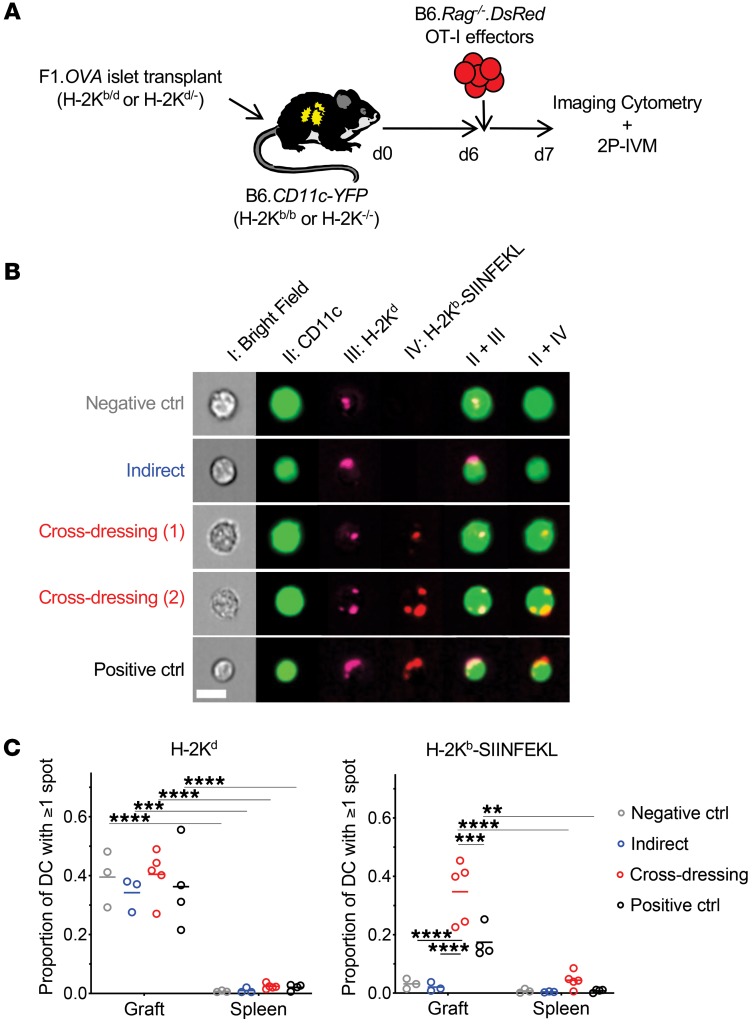
Figure 1. Host DCs are extensively cross-dressed with donor MHC class I–peptide complexes in islet allografts.
(A) F1.OVA H-2Kb–sufficient (H-2Kb/d) or F1.OVA H-2Kb-deficient (H-2Kd/–) islets were transplanted under the kidney capsule of B6.CD11c-YFP H-2Kb–sufficient (H-2Kb/b) or B6.CD11cYFP H-2Kb–deficient (H-2Kb–/–) recipients. 5 × 106 B6.Rag–/–.DsRed OT-I CD8+ effector T cells, which recognize the OVA peptide SIINFEKL bound to H-2Kb, were transferred i.v. 6 days later. One day after OT-I transfer, grafts were analyzed by imaging flow cytometry (B) and 2P-IVM (Figure 2). Control and experimental groups are shown in Table 1. (B) Leukocytes were isolated from transplanted allografts and analyzed by ImageStream. Intact H-2Kb–SIINFEKL complexes and donor H-2Kd molecules were identified as discreet spots on surface of host (CD11c-YFP) DCs. Representative images from each group are shown. (C) Proportion of host DCs positive for 1 or more spot of either H-2Kd or H-2Kb–SIINFEKL. In all groups, the majority of cells (~90%) carried only one spot, while the remainder had 2 to 5 spots (data not shown). The majority of DCs in the cross-dressing and control groups carried both MHC class I molecules. Each data point represents analysis of 1 transplanted animal. On average, 1071 (range = 100–3900) cells were analyzed per animal. **P < 0.01; ***P < 0.001; ****P < 0.0001. One-way ANOVA with Tukey’s multiple comparison test.