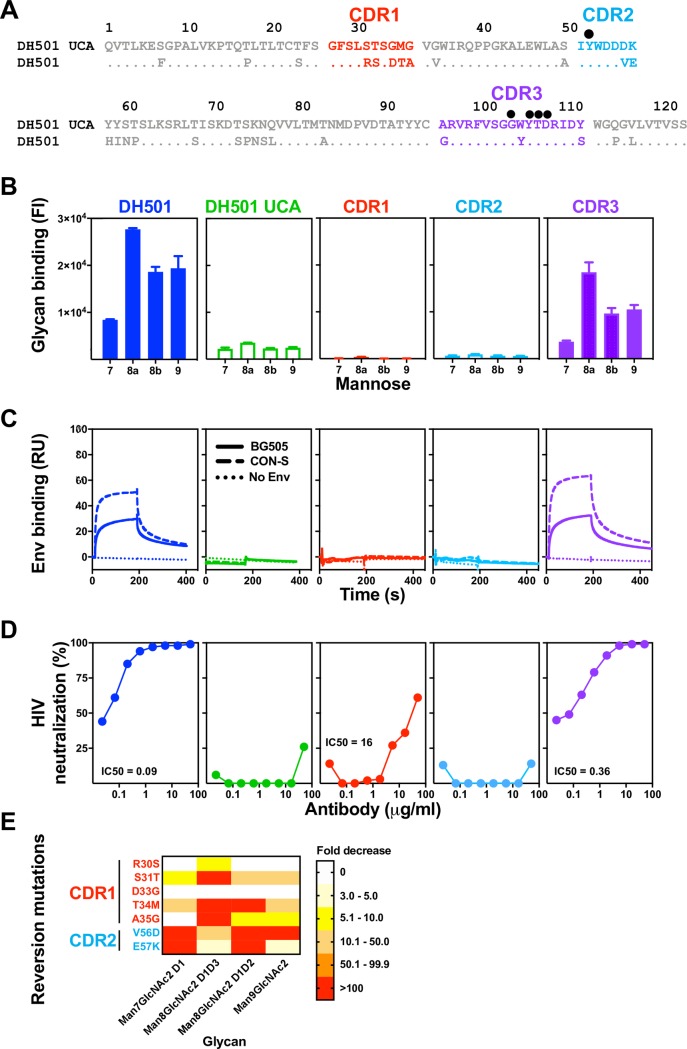
Fig 3. Somatic mutation of HCDR1 and HCDR2 is required for glycan reactivity and HIV-1 Env reactivity.
(A) ClustalW alignment of amino acids of DH501 and DH501 UCA VH. HCDRs are denoted based on the IMGT definition and color-coded. Identical residues are shown as dots. Amino acids resulting from somatic mutation are specified for DH501. Black circles above the sequence alignment denote amino acids that contact glycan in the crystal structure of DH501 in complex with Man9GlcNAc2. (B) Antibody binding to glycan was assessed for Man7GlcNAc2 D1 (7), Man8GlcNAc2 D1D3 (8a), Man8GlcNAc2 D1D2 (8b), Man9GlcNAc2 (9). Binding was assessed for DH501 with the HCDR1 (red), HCDR2 (light blue), or HCDR3 (purple) somatic mutations reverted to the UCA sequence. Column titles indicate the antibody analyzed in each functional assay. Antibodies are color-coded the same in B-D. The mean and standard error are shown for independent triplicate experiments. Positive binding based on negative control antibody binding is shown as a filled bar. Open bars indicate negative binding values. Positivity thresholds for 7, 8a, 8b, and 9 are 0.18x104, 0.32x104, 0.19x104, 0.18x104 respectively. (C) Trimeric HIV-1 envelope reactivity of DH501, DH501 UCA, and HCDR-reverted antibodies. IgG binding to CON-S SOSIP (dashed line) or BG505 6R.SOSIP.664 (solid line) was determined by SPR. IgG binding response measured with the buffer only control is represented as dotted lines. Representative data from two independent experiments are shown. (D) In vitro HIV-1 neutralization of kifunensine-treated, Man9GlcNAc2-enriched JR-FL in the TZM-bl assay. For antibodies with detectable neutralization titers, the IC50s in μg/mL are shown. (E) Single somatic mutations in the HCDR1 and 2 were reverted to the UCA sequence and binding to the free glycans as in B was assessed. The reversion mutations are color-coded based on their location shown in A. The heatmap shows the fold decrease in binding upon reversion of the single mutation. The ranges of fold decrease in glycan binding are color-coded as indicated in the legend.