Abstract
Background
Lymphatic filariasis (LF) is targeted for elimination by the year 2020. As of 2017, 67 of the 72 endemic countries have implemented annual Mass Drug Administration (MDA) for interrupting LF transmission. Transmission Assessment Survey (TAS) is the recommended protocol to evaluate the impact of MDA and to decide when to stop MDA in an Evaluation Unit (EU, population ≤2 million). As the human infection levels go down with repeated MDA rounds, it becomes a challenge to select the appropriate survey methods to assess transmission interruption. This study validates a standard protocol for molecular xenomonitoring of infection in vectors (MX) at an EU as a complementary tool for TAS to stop MDA and its utility for post-MDA or post-validation surveillance.
Methodology
The study was conducted in Cuddalore district, Tamil Nadu, India, which was found eligible for TAS after 15 annual rounds of MDA (4 with DEC alone and 11 with DEC plus albendazole). The district was divided into two EUs as per the TAS protocol and one EU was randomly selected for the study. A two-stage cluster design vector sampling, developed and validated at a sub-district level, was implemented in 30 randomly selected clusters in the EU. Female Culex quinquefasciatus were collected placing gravid traps overnight (1800–0600 hrs) inside the premises of systematically selected households. Pools of 20–25 blood-fed, semi-gravid and gravid Cx. quinquefasciatus were subjected to real-time quantitative PCR (polymerase chain reaction) assay for detecting Wuchereria bancrofti DNA. Pool infection rate (% of pools positive for W. bancrofti DNA), and the estimated prevalence of W. bancrofti DNA in mosquitoes and its 95% confidence interval were calculated. Additionally, in these 30 clusters, microfilaria (Mf) survey among individuals >5 years old was carried out. School-based TAS was conducted using Immunochromatographic Card Test (ICT) in the EU. Prepared itemized cost-menu for different cost components of MX survey and TAS were estimated and compared.
Results
MX survey showed that only 11 (3.1%) of the 358 pools (8850 Cx.quinquefasciatus females), collected from 30 clusters, were found positive for W. bancrofti DNA. The estimated vector infection rate was 0.13% (95% CI: 0.07–0.22%), below the provisional threshold (0.25%) for transmission interruption. Of 1578 children tested in the TAS, only four (0.25%) were positive for filarial antigenemia, and it is well below the critical cut-off (18 positives) for stopping MDA. Among 9804 persons tested in the 30 clusters, only four were found positive for Mf (0.04%; 95% CI: 0.01–0.1%). The Mf-prevalence was <1% threshold for transmission interruption in humans. The estimated costs for TAS and MX per EU were $14,104 USD and $14,259 USD respectively.
Conclusions
The result of MX protocol was in good agreement with that of TAS, providing evidence to recommend MX as a complementary tool to TAS to decide on stopping MDA. MX can also be a potential surveillance tool for post-MDA and post-validation phases as it could detect sites with residual infection and risk of resurgence of transmission. MX is economically feasible as its cost is slightly higher than that of TAS.
Author summary
Lymphatic filariasis (LF), commonly known as “elephantiasis” is caused by filarial parasites and transmitted among humans by mosquitoes. This parasitic infection results in chronic diseases such as swelling of limbs and hydrocele. Global programme to eliminate lymphatic filariasis (GPELF), launched by the World Health Organization (WHO) in 2000 endorsed the mass treatment of all the people above 2 years of age in the endemic areas with a single dose of anti-filarial drugs administered annually for a minimum period of 5 years. WHO also recommended transmission assessment survey (TAS) protocol to assess the impact of mass treatment and to decide on stopping mass treatment. The protocol aims at screening young children who were born after the mass treatment for filarial infection. If the number of infected children is smaller than the pre-defined number, mass treatment can be stopped. The same protocol is followed for periodical assessment to verify whether there are any new infections. Alternatively, vector infection levels by molecular xenomonitoring (MX, detection of parasite DNA in the mosquitoes) can be used to verify whether there are any infected mosquitoes. This tool has been applied in many studies and there is a provisionally established mosquito infection threshold level (0.25%) below which transmission is interrupted. This can be an alternative tool for TAS. We validated this method at district level by collecting filariasis transmitting mosquitoes from 30 villages/wards and compared the results with those of TAS. There was good agreement between the decisions based on TAS and MX in our study. Though in the EU both vector and human infection levels were below their respective threshold levels, the mosquito infection in individual sites was above the threshold, indicating residual hotspots and risk of resurgence. In addition, we estimated the cost of conducting MX and TAS for their economic feasibility and found that the cost of MX is only marginally higher than that of school-based TAS. Thus, our study results provide recommendations to use MX as a tool complementary to TAS (i) for taking a decision on stopping MDA, (ii) for monitoring post-MDA and post-validation surveillance programme, and (iii) for remapping areas to initiate MDA.
Introduction
The global programme to eliminate lymphatic filariasis (GPELF) launched in 2000 with the goal to eliminate LF as a public health problem by 2020 [1, 2] has made significant progress since its inception. About 7.1 billion cumulative treatments have been given towards interrupting transmission of LF through mass drug administration (MDA) of albendazole in combination with diethylcarbamazine (DEC) or ivermectin (2-drug regimen). At least 550 million people no longer require treatment out of about 1.3 billion people at risk of infection in 2000. As of 2017, 54 of the 72 LF endemic countries have fully implemented MDA, 13 countries have started MDA but have not up-scaled to all endemic districts and in the remaining 5 countries MDA is yet to be started [3]. WHO has formally acknowledged the claim of successfully eliminating LF as a public health problem in 11 countries (Cambodia, Cook Islands, Maldives, Niue, Sri Lanka, Togo, Vanuatu, Thailand, Egypt, Tonga and Marshall Islands) and in another 10 countries MDA has been stopped and post-MDA surveillance is in progress [4].
Transmission assessment survey (TAS) is the WHO-recommended protocol [5] to decide when to stop MDA (TAS 1) and to determine whether levels of infection have been sustained below the target threshold [antigen (Ag) prevalence <2% in 6 to 7-years-old children] after stopping MDA (‘post-MDA surveillance’). TAS is done in an evaluation unit (EU, an MDA implementation unit with a population not exceeding 2 million) if it has completed at least five rounds of MDA with an effective coverage (> 65%) and has recorded a microfilaria (Mf)-prevalence of < 1% or an Ag-prevalence of <2% in all the sentinel and spot-check sites. The sentinel and spot-check sites (geographical areas, each with a population of at least 500 people) are selected to collect parasitological data to monitor the impact of the MDA programme. While the sentinel sites remain the same, different spot-check sites are chosen for each assessment over the course of the MDA programme [5].
In India, if the assessment in all the sentinel and spot check sites indicates that the Mf-prevalence is below 1%, the Mf-prevalence is determined in 10 additional randomly selected sites to decide on conducting TAS [6]. In all 10 sites, Mf-prevalence should be below 1% for the area to conduct a TAS. TAS involves screening 6–7 year-old children through school or community-based surveys for detecting filarial antigen, as the children should have been protected from LF infection if MDA succeeded in interrupting transmission. When the Ag-prevalence in children is less than 2%, the EU is considered to have passed TAS and MDA will be stopped. If TAS has failed, MDA will continue.
The current recommendation for post-MDA surveillance is repeating TAS twice at 2–3 years (TAS 2) and 4–6 years (TAS 3) after stopping MDA. Evaluation unit that has cleared all the three TAS will be covered under post-validation surveillance for at least 5 years [4]. Post-MDA surveillance and mapping areas suspected to be LF endemic are required to prepare dossier for validation of LF elimination. However, factors related to sensitivity and evaluation unit size for TAS have been reported to limit its usefulness for confidently demonstrating transmission interruption in post-MDA phase [7–9]. A few countries including Sri Lanka [8], Tanzania [10], American Samoa [11, 12] and Ghana [13] have reported evidence of transmission despite passing TAS (< 2% Ag-prevalence in 6–7 year-old children) suggesting that TAS may be inadequate for taking the decision to stop MDA, when the infection in humans is at a very low level. Therefore there is an urgent need for more sensitive high-throughput and cost-effective diagnostic tools for detecting low infection signals during post-MDA situations [14].
India, the largest LF endemic country in the world (accounts for one-third of the global LF-burden [15] with 630 million people at risk of infection [16]), is one of the few countries that implemented MDA first for LF elimination following WHO guidelines [16]. The National Vector Borne Diseases Control Programme (NVBDCP) has implemented MDA with DEC alone from 2004, and co-administered DEC with albendazole from 2008 in all the 256 endemic districts (implementation units, IU) spread over 16 states and 5 union territories. At least 5 annual rounds of MDA with DEC plus albendazole have been completed in all the IUs. MDA was stopped in 100 IUs after passing TAS 1 (IU with more than 2 million population split into 2 or more EUs) and these IUs are currently under post-MDA surveillance; 25 IUs are being subjected to TAS 1 and in the remaining 131 IUs MDA is continuing. Currently the programme faces challenges as these IUs either failed TAS 1 (Ag-prevalence above the critical threshold of 2% for stopping MDA) or are not eligible for TAS as the infection levels remain above the critical threshold. The national programme has proposed to implement accelerated LF elimination strategy with MDA of a 3-drug regimen (DEC, albdendazole, ivermectin) to give a new impetus to the ongoing activities and achieve the ambitious goal of LF elimination by 2020 [16]. Since the effect of 3-drug regimen on adult worms is not yet fully understood, the currently recommended TAS in children based on antigen detection for 2-drug regimen may not be appropriate for deciding to stop or continue MDA with 3-drug regimen. Therefore, it is necessary to identify new target populations (human / mosquito), infection indicators, sampling strategies, and/or thresholds to determine when it is safe to stop MDA with 3-drug regimen.
Molecular xenomonitoring (MX, monitoring the presence of parasite DNA in mosquitoes) of infection in vectors has been recognized as a tool complementary to TAS and is used for monitoring recrudescence of infection in post-MDA [17, 18] and validation phases [19–21] when the infection is at a level lower than that detectable by Ag or Mf-testing. Vector infection of <0.25% for Culex, <1% for Anopheles and <0.1% for Aedes are suggested as provisional threshold levels to decide on stopping MDA [22]. MX involves collection of a large number of vector mosquitoes using appropriate collection methods, sampling techniques to obtain representative sample and detecting parasite DNA in pools of mosquitoes using PCR. Various collection methods have been employed and tested for their efficiency to capture different vector species: gravid traps [8, 19, 23–27], CDC light traps and battery powered aspirators for Cx. quinquefasciatus [28], AGT, a variant of OviART & pyrethrum spray method for Anopheles gambiae [13, 29, 30], and BG sentinel traps for Aedes polynesiensis [12]. MX technique is shown to be more efficient and sensitive in detecting parasite DNA than Mf-testing in humans [26, 31] and assessing the residual or renewed foci of infection after several rounds of MDA [8, 12, 19, 21, 26, 31–35]. LF endemic countries confirmed or supported the findings of TAS with MX on the basis of the absence of transmission during post-MDA or validation phases [19–21, 25]. Despite a number of studies which compared the decisions of TAS with those of MX, application of MX in operational settings requires an assessment of cost in relation to TAS and feasibility in terms of availability of adequate laboratory facilities and specially trained personnel [26].
In an earlier study, we developed and validated (i) a two-stage cluster design-based sampling strategy for collecting Cx. quinquefasciatus through gravid traps [19] and (ii) a cost-effective quantitative PCR assay for detecting W. bancrofti DNA in Cx. quinquefasciatus for MX at a sub-district level [36]. The present study aims to demonstrate the usefulness of the MX protocol at an evaluation unit level by comparing the results obtained from data for MX with those of microfilaremia in humans collected from the same communities, and also with the results of school-based TAS to provide a comprehensive assessment of the interruption of transmission. The usefulness and feasibility of MX as a complementary tool to TAS and for post-validation surveillance and its scope as a complementary tool for remapping areas suspected to be LF endemic for inclusion in the LF elimination programmes are discussed.
Methodology
Study area
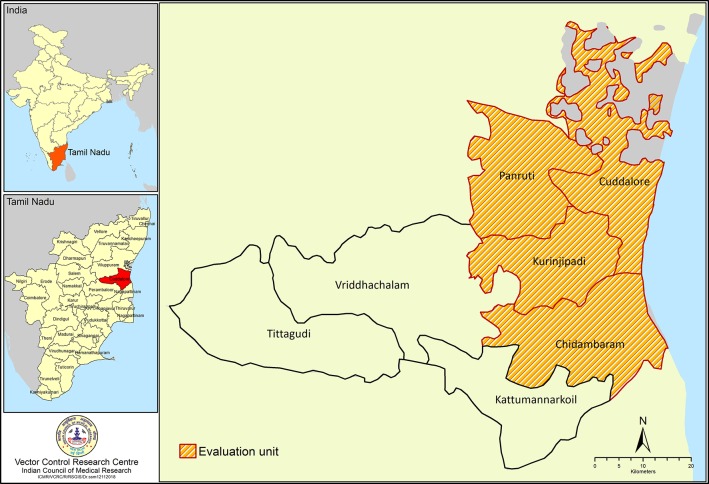
This study was conducted between 2015–2016 in an IU in Cuddalore district (administrative unit), in Tamil Nadu, India, which is endemic for Cx. quinquefasciatus transmitted Wuchereria bancrofti. This district has undergone 15 annual rounds (4 rounds of DEC alone and 11 rounds of DEC plus albendazole) of MDA during 1996–2014 with a break in 2005, 2006, 2008 and 2011. The programme reported MDA coverages (no. received the drug out of total population) were above 90% in all the rounds (S1 Table), whereas the coverage (no. received out of total no. of persons interviewed) by an independent assessment team in 2014 reported that it ranged from 70 to 83% (mean: 77%) with a consumption rate of 37–66% (mean: 54%, those swallowed the drug out of total no. of persons interviewed) in four randomly selected sites in the IU (S2 Table). As per national guidelines, pre-TAS assessment was done in two-stages. In stage 1, surveys were conducted in four sentinel and four spot-check sites. Mf prevalence was below 1% in all the 8 sites. In stage 2, Mf-survey was continued in 10 additional random sites (only in India). The Mf-prevalence was below 1% in each additional random site also, thereby the IU got qualified for TAS. The IU consists of seven sub-districts (Cuddalore, Chidambaram, Kurinchipadi, Panruti, Kattumannarkoil, Thittakudi, and Virudhachalam) having a total population of 2.6 million [37] with 1.7 million residing in rural areas (villages) and the rest in urban areas (wards). Since the total population is above 2 million, the IU was divided into two EUs as per the WHO protocol for conducting TAS [5]. Of the two EUs, one was selected randomly for the study.
The selected EU has a population of 1.6 million and 397,468 households [37] administered by four sub-districts: (i) Cuddalore (population: 0.42 million), (ii) Chidambaram (population: 0.46 million), (iii) Kurinchipadi (population: 0.33 million) and (iv) Panrutti (population: 0.41 million) (Fig 1). Of these 58% live in rural villages and the remaining in urban wards.
Fig 1. Map showing sub-districts of the studied evaluation unit (EU) in Cuddalore district, Tamil Nadu, India.
Vector sampling
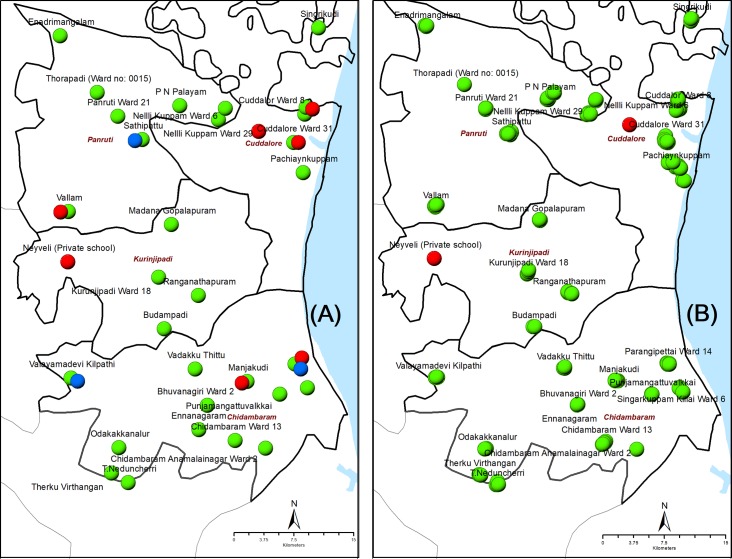
Vector surveys were carried out from January to August 2015 following a two-stage cluster sampling protocol, which was developed and validated at sub-district level for sampling Cx. quinquefasciatus mosquitoes and molecular xenomonitoring (MX) of filarial infection. A detailed description of the sampling methodology is reported elsewhere [19]. In stage 1, 30 clusters (village or ward) were selected randomly from 669 clusters in all sub-districts of the EU (Fig 2A). In stage 2 (household, HH), on an average 5 HHs were selected per cluster with probability proportional to size (HHs) of the selected cluster. In each selected HH, gravid traps (a VCRC modified version of the CDC gravid trap, Model 1712, John W. Hock Co., Gainesville, FL), with hay infusion that attracts Cx. quinquefasciatus [19], were placed outdoors within the household premises, at least 1 hour prior to sunset (1700 hrs) after obtaining oral consent from the residents. Geo-coordinates of all the mosquito collection points (HHs) were recorded using the Dell Axim X51 personal digital assistant (PDA) and mapped using ArcGIS (version 10.6, ESRI, Redlands, CA). The mosquito collection cages were removed from the traps the next morning (0630 hrs) and brought to the field laboratory. Mosquito collections in each selected HH were continued until a total of 50 Cx. quinquefasciatus female mosquitoes (except unfed) were caught or for a maximum of 3–4 nights.
Fig 2. Location of clusters (villages or wards) for mosquito collection and human blood survey in the evaluation unit.
The left panel shows the sites where mosquito and Mf-surveys were carried out (A) and the right-side panel shows the location of schools where the TAS was done (B). Clusters/schools that were negative for filarial infection in both humans and mosquitoes are shown in green, clusters with at least one mosquito pool positive for filarial infection or schools with at least one child positive for filarial antigen are shown in red and clusters with at least one Mf positive individual are shown in blue.
Collected mosquitoes were morphologically identified to species by experienced technical staff and female Cx. quinquefascatus further classified according to their abdominal appearance into gravid, semi-gravid, blood-fed and unfed. Unfed females were discarded; other ones were pooled by HH (25 or less mosquitoes/pool) in barcoded vials. Mosquito pools were dried at 95°C for a minimum of 15 minutes as described earlier [19] for later qPCR analysis. A maximum of 50 females (2 pools) were tested by HH. The target was to collect from all 30 clusters 7,500 females in 300 pools (30 clusters x 5 HHs per cluster x 25 mosquitoes per pool x 2 pools per HH = 7,500 mosquitoes).
Filarial DNA extraction from mosquito pools
Filarial parasite DNA was extracted from each mosquito pool following indigenously developed simple TE (Tris-EDTA) -based DNA extraction procedure using bead beating (BB) for grinding the mosquitoes [38]. The DNA samples thus obtained were coded and analyzed by real-time quantitative PCR as described elsewhere [19, 36]. Briefly, each real-time PCR reaction was performed with 12.5 μl of FastStart Essential DNA probes Master (Roche Diagnostics, Germany) along with 450 nmol/L of each primer: LDR1-5’ATTTTGATCATCTGGGAACGTTAATA-3’; LDR2-5’CGACTGTCTAATCCATTCAGAGTGA-3’ and 125 nmol/L probe (6 FAM-ATCTGCCCATAGAAATAACTACGGTGGATCTG-TAMRA) (IDT, USA) in a final volume of 25μl in 96-well MicroAmp optical plates (Roche Diagnostics, Germany). One microliter of the extracted DNA was used as template in each real-time PCR reaction as described earlier [39]. Each plate (Lightcycler 480 Multi- well 96) was run with forty samples in duplicate. Three concentrations (1 ng, 100 pg and 10 pg) of DNA each in duplicate were used as positive control. Three negative control (only water and no template DNA) in duplicate were run simultaneously.
Real-time quantitative PCR reactions were carried out to determine the cycle of quantification (Cq) values for each sample. Thermal cycling parameters used were 50°C for 2min, 95°C for 10 min followed by 40 cycles of 95°C for 15 sec and 60°C for 1min. Thermal cycling and data analysis were done with Light Cycler 96 (Roche, Germany) instrument using sequence detection system (SDS) software (Applied Biosystems). Cq values of samples ranging from 1.0–39.0 were considered positive, and samples that failed to reach the fluorescence threshold beyond 39 were considered indeterminate and repeated to confirm the negativity or positivity of those samples as described by Rao et al. [39].
Transmission Assessment Survey (TAS)
As required in the LF elimination programme, the TAS protocol described by WHO was used [5] to assess the impact for making decision on stopping MDA. List of schools with primary sections in the EU and the number of children enrolled in first and second grades in each school were obtained from the office of the district school education. Since the school enrolment rate in the EU was above 75%, school-based TAS was done considering the school (cluster) as a primary sampling unit, and children in grades I and II in the selected schools as secondary sampling unit. There were 887 primary schools with 36812 students in the first and second grades in this EU. The sample size and number of clusters for TAS was estimated using the survey sample builder (SSB) with an estimated non-response rate of 10%. As per SSB, a sample of 1556 children from 43 systematically selected clusters (Fig 2B) were chosen and targeted with a critical cut-off of 18 positives for deciding to stop MDA. All the selected children were tested for filarial antigenemia using the immonochromatographic card test (ICT) manufactured by BinaxNow (Scarborough, USA) [40] during the period from 26 March 2015 to 9 April 2015. From each child 100μl blood was collected and loaded directly on to the ICT cards. A team of 4 trained technicians conducted the survey and the results of the test were read at 10 minutes’ interval and positives were recorded.
Community surveys for microfilaraemia
Night blood survey was carried out from October 2016 to January 2017 to assess the prevalence of microfilaria (Mf) in each of the 30 sites where xenomonitoring was done (Fig 2A). All available and consenting persons, above 5 years old, in each systematically selected house were tested for Mf with 60μl thick blood smears prepared with blood collected by finger prick method between 1930 hrs and 2330 hrs, which was operationally feasible. Sample size (6085) was calculated for an expected Mf-prevalence of 1% (based on sentinel, spot-check and additional 10 random sites, S3 Table) with 0.25% precision and 95% CI. The calculated sample size after adjusting for a design effect of 1.5 was ~ 9200 persons. Considering a family size of four persons, 2300 houses were selected randomly with probability proportion to population size of the respective village. Blood slides were dehaemoglobinized with distilled water, dried, fixed in acidified methyl alcohol and stained with JSB-1 stain. Trained microscopists examined the blood slides and the number of Mf in each positive slide was recorded. All the positive slides and 5% of the negative slides selected randomly were cross-examined for quality assurance.
Data analysis
Mosquito ‘pool infection rate’ was calculated as the percentage of pools positive for W. bancrofti DNA relative to the number of pools screened by individual cluster, sub-district and for the EU as a whole. The prevalence of W. bancrofti DNA (maximum likelihood estimates with 95% confidence interval) was estimated with PoolScreen software 2.0.3 [41, 42]. Mf-prevalence in human was calculated as the ratio of number of blood slides positive for Mf to total number of slides examined. Kruskal-Wallis one-way analysis of variance was used to compare the difference in the density (number per trap-night) of Cx. quinquefasciatus (gravid, semi-gravid and freshy-fed) among sub-districts. Random effects logistic regression model was used to compare the differences across sub-districts in terms of (i) prevalence of Mf in human and (ii) percentage of Cx. quinquefasciatus: (a) captured by gravid traps out of all mosquito species, (b) with various abdominal status (blood-fed, semi-gravid, gravid, except unfed), and (c) pools positive for W. bancrofti DNA. Separate analysis was done for each of the above-mentioned parameters (response variable) with sub-districts and sites as fixed and random effects respectively. P values <0.05 were considered statistically significant.
Costing
Costs incurred in conducting MX and TAS were estimated per EU using itemized cost menu. For MX, the cost components include (i) personnel engaged for mosquito collection, preservation and processing, (ii) transport for the field surveys, (iii) field supplies (traps, vials, and barcodes) and (iv) lab processing of samples (reagents and primers).
The cost components for TAS include (i) personnel, (ii) transport to the field sites, and (iii) supplies (ICT, Binax, USA) and field supplies (lancets, cotton rolls, and surgical spirit). TAS was conducted jointly by the research team and the district health system. Therefore, the services of the health staff were included as ‘opportunity cost’ (the cost for diverted services by which the benefit of other services is lost) under ‘personnel’ head. In the present study, we have used the ICT kits from the national programme, donated by the WHO Regional Office in the country. Therefore, the cost of ICT was included as ‘opportunity cost’. Further, since the national programme currently uses filariasis test strip (FTS, Alere, Scarborough, ME) instead of ICT [40, 43], costing of TAS with FTS was also done for the purpose of comparison.
Ethics statement
The Institutional Human Ethics Committee of the ICMR-Vector Control Research Centre approved the study proposal. Survey teams explained the purpose of the study and study procedures to the selected household members. Written informed consent was obtained from all consenting adults and parents for children for collecting finger-prick blood to assess Mf and filarial Ag. Personal identity and results of tests were kept confidential. A list of persons detected positive for Mf or Ag was shared with the Deputy Director of Health Services of the District with a request to treat filarial infected persons as per the national programme guidelines of India. Since gravid traps for mosquito collection were placed outside the households and it did not interfere with any domestic activities within or around the households, the xenomonitoring part of the study does not involve any ethical issue, but oral consent was obtained from the head of the households.
Results
Sampling Cx. quinquefasciatus mosquitoes
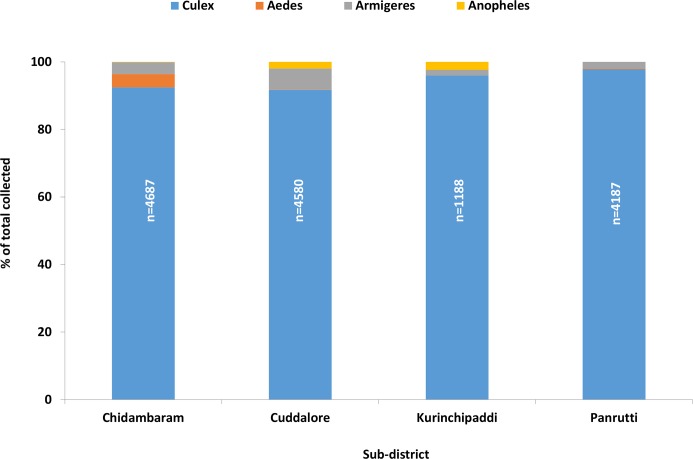
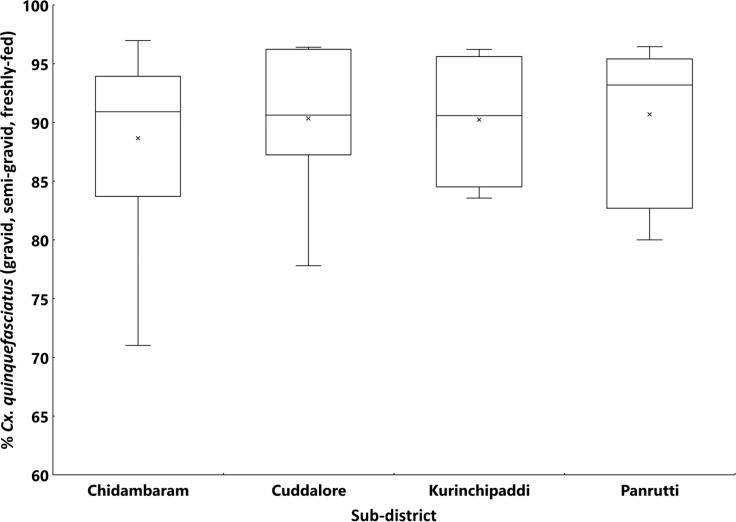
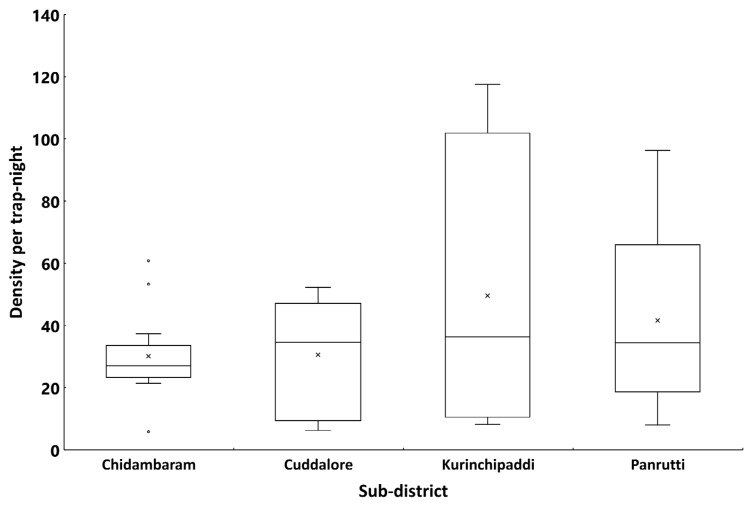
A total of 14,642 female mosquitoes comprising four species were collected spending 407 trap nights in 185 households spread over 30 clusters in the EU. Fig 3 presents the percentage composition of each species by sub-district. The filariasis vector, Cx quinquefasciatus, was the predominant species (94.1%), followed by Armigeres (3.8%), Aedes (1.4%) and Anopheles (0.8%). A similar pattern was observed in all the four sub-districts (>90% Cx. quinquefasciatus, Fig 3). Of the 13771 Cx. quinquefasciatus females collected in the EU, more than 90% were gravid, semi-gravid or freshly-fed in all the sub-districts (range: 71–97% in different clusters, Fig 4). The mean density of Cx. quinquefasciatus mosquitoes (gravid, semi-gravid and freshly-fed) in the EU was 30.8 per trap-night (range: 5.8–117.5 per trap-night in different clusters). The density averaged for the sub-districts ranged between 30.1 and 49.6 per trap-night (Fig 5). The sub-districts did not differ significantly in terms of percentage of Cx. quinquefasciatus among all species captured by gravid trap (P = 0.06), its percentage other than unfed (P = 0.32), and the density per trap-night (P = 0.95).
Fig 3. Mosquito species composition in gravid trap collection by sub-districts in the evaluation unit.
Fig 4. Box-plot showing the percentage Culex quinquefasciatus female mosquitoes (gravid and blood-fed, except unfed) among the total collected by gravid traps in different clusters by sub-district in the evaluation unit.
The horizontal line inside the box shows the median and the 'x' is the mean. The ends of the box are the 25th and 75th percentiles of the distribution of the percentage of mosquitoes. The whiskers extend to 1.5 times the height of the box (i.e. the interquartile range, IQR). If the data are distributed normally, approximately 95% of the data are expected to lie between the inner fences.
Fig 5. Density (Numbers of females/trap-night) of Culex quinquefasciatus female mosquitoes (gravid and blood-fed, except unfed) in different clusters by sub-districts in the evaluation unit.
The horizontal line inside the box shows the median and the 'x' is the mean. The ends of the box are the 25th and 75th percentiles of the distribution of the density. The whiskers extend to 1.5 times the height of the box (i.e. the interquartile range, IQR). If the data are distributed normally, approximately 95% of the data are expected to lie between the inner fences. Values above or below the upper or lower whiskers are labelled as outliers (o).
Vector infection rate
A total of 358 pools were formed out of 8850 gravid Cx. quinquefasciatus collected from 185 HHs (as against 150 HHs targeted) spread over 30 clusters in the EU. As many as 353 (98.6%) pools had a pool size of above 20 each as against the desired pool size of 25 [22]. Two mosquito pools were screened from each of 173 HHs (pools of 25 mosquitoes each were from 166 HHs, and of 5–25, median 24 females were from seven HHs). One mosquito pool (16–25, median 22, females) was screened from each of remaining 12 HHs. The qPCR (quantitative PCR) results by cluster and sub-district are summarized in Table 1. Of 358 pools tested, 11 (3.1%) pools from five clusters found positive for W. bancrofti DNA, representing three of the four sub-districts (range: 2.0 to 4.7%) in the EU. It is interesting to note that none of the 26 pools were screened positive in Kurinjipadi sub-district. Statistical analysis revealed that the pool infection rates did not differ significantly among sub-districts (P = 0.56).
Table 1. PoolScreen infection of Culex quinquefasciatus mosquitoes with Wuchereria bancrofti DNA in the evaluation unit during January-August 2015.
Clusters with one or more positive pools are shaded in orange.
| Cluster No. | Sub-district | Cluster | No. of houses sampled | No. of Cx. quinquefasciatus collected | No. of pools screened | No. of pools infected | Pool infection rate (%) | W. bancrofti DNA prevalence (95% CI) |
|---|---|---|---|---|---|---|---|---|
| 1 | Chidambaram | Bhuvanagiri Ward 2 | 3 | 150 | 6 | 0 | 0.00 | 0.0 (0.0–2.0) |
| 2 | Chidambaram Anamalainagar Ward 2 | 1 | 50 | 2 | 0 | 0.00 | 0.0 (0.0–4.2) | |
| 3 | Chidambaram Ward 13 | 5 | 217 | 9 | 0 | 0.00 | 0.0 (0.0–1.5) | |
| 4 | Ennanagram | 2 | 100 | 4 | 0 | 0.00 | 0.0 (0.0–2.7) | |
| 5 | Manjakudi | 12 | 541 | 22 | 1 | 4.55 | 0.18 (0.01–0.90) | |
| 6 | Odakakkanalur | 3 | 150 | 6 | 0 | 0.00 | 0.0 (0.0–2.0) | |
| 7 | Parangipettai Ward 14 | 5 | 250 | 10 | 4 | 40.00 | 1.9 (0.63–4.70) | |
| 8 | Punjamangattuvalkkai | 2 | 36 | 2 | 0 | 0.00 | 0.0 (0.0–5.8) | |
| 9 | Singarkuppam Killai Ward 6 | 4 | 200 | 8 | 0 | 0.00 | 0.0 (0.0–1.6) | |
| 10 | T.Neduncherri | 4 | 197 | 8 | 0 | 0.00 | 0.0 (0.0–1.6) | |
| 11 | Therku Virthangan | 3 | 150 | 6 | 0 | 0.00 | 0.0 (0.0–2.0) | |
| 12 | Vadakku Thittu | 4 | 200 | 8 | 0 | 0.00 | 0.0 (0.0–1.6) | |
| 13 | Velayamadevi Kilpathi | 8 | 390 | 16 | 0 | 0.00 | 0.0 (0.0–0.9) | |
| Chidambaram Sub-district | 56 | 2631 | 107 | 5 | 4.67 | 0.20 (0.07–0.45) | ||
| 14 | Cuddalore | Cuddalore Ward 31 | 9 | 450 | 18 | 3 | 16.67 | 0.71 (0.19–1.90) |
| 15 | Cuddalore Ward 8 | 13 | 623 | 26 | 0 | 0.00 | 0.0 (0.0–0.6) | |
| 16 | Nellikuppam Ward 29 | 5 | 250 | 10 | 0 | 0.00 | 0.0 (0.0–1.3) | |
| 17 | Nellikuppam Ward 6 | 5 | 250 | 10 | 0 | 0.00 | 0.0 (0.0–1.3) | |
| 18 | Pachia Kuppam | 21 | 1050 | 42 | 0 | 0.00 | 0.0 (0.0–0.3) | |
| 19 | Peria Kangankuppam | 5 | 250 | 10 | 1 | 10.00 | 0.40 (0.02–2.00) | |
| 20 | Singrikudi | 5 | 250 | 10 | 0 | 0.00 | 0.0 (0.0–1.3) | |
| Cuddalore Sub-district | 63 | 3123 | 126 | 4 | 3.17 | 0.13 (0.04–0.31) | ||
| 21 | Kurinjipadi | Buddampadi | 2 | 75 | 3 | 0 | 0.00 | 0.0 (0.0–3.2) |
| 22 | Kurunjipadi Ward 18 | 4 | 200 | 8 | 0 | 0.00 | 0.0 (0.0–1.6) | |
| 23 | Madana Gopalapuram | 4 | 200 | 8 | 0 | 0.00 | 0.0 (0.0–1.6) | |
| 24 | Ranganathapuram | 5 | 171 | 7 | 0 | 0.00 | 0.0 (0.0–1.8) | |
| Kurinjipadi Sub-district | 15 | 646 | 26 | 0 | 0.00 | 0.0 (0.0–0.6) | ||
| 25 | Panrutti | Enadrimangalam | 5 | 224 | 9 | 0 | 0.00 | 0.0 (0.0–1.4) |
| 26 | P N Palayam | 13 | 650 | 26 | 0 | 0.00 | 0.0 (0.0–0.5) | |
| 27 | Sathipattu | 13 | 650 | 26 | 0 | 0.00 | 0.0 (0.0–0.5) | |
| 28 | Thorapadi | 1 | 23 | 1 | 0 | 0.00 | 0.0 (0.0–6.6) | |
| 29 | Vallam | 13 | 650 | 26 | 2 | 7.69 | 0.31 (0.06–1.00) | |
| 30 | Panruti Ward 21 | 6 | 253 | 11 | 0 | 0.00 | 0.0 (0.0–1.3) | |
| Panrutti Sub-district | 51 | 2450 | 99 | 2 | 2.02 | 0.08 (0.01–0.27) | ||
| Overall for Evaluation Unit | 185 | 8850 | 358 | 11 | 3.07 | 0.13 (0.07–0.22) | ||
Pool Screen estimate revealed that the prevalence of W. bancrofti DNA for the EU (overall sub-districts) was 0.13% (95% CI: 0.07–0.22%) (Table 1). This rate is much lower than the suggested critical cut-off of 0.25% [22, 31, 44] for transmission to take place at sub-district and EU level. The parasite DNA rates as estimated by PoolScreen vary greatly among clusters (range: 0.0–1.9%). In four of the 30 clusters, the prevalence of filarial DNA in mosquitoes exceeded the provisional target of 0.25%. Moreover, surprisingly the estimated upper 95% CI for the prevalence of W. bancrofti DNA exceeded the provisional target in all the 30 clusters (Table 1).
Transmission assessment survey (TAS)
Table 2 shows the results of TAS by school and sub-district. A total of 1579 (including one invalid ICT test) children in grades I and II were tested from 43 schools selected for TAS. The actual number of children screened exceeded the target sample size of 1556. The excess sampling could be due to a higher non-response rate (10%) assumed in the calculation of sample size using SSB than the actual. Among the remaining 1578 children screened, only four (0.26%) were Ag positive by ICT and were located in two sub-districts. Three out of 34 children in one school and one out of 77 children in another school were Ag-positive. In one school the Ag-prevalence exceeded the critical level of 2%. Ag-prevalence ranged between 0.0 and 0.6% in the sub-districts. Since the number of Ag positive children is less than the critical cut-off (18), the EU passed TAS and qualified for stopping MDA. The village or ward in which schools selected for TAS were located were different from the clusters (villages or wards) selected for MX and Mf-surveys and hence there was no parallel data for comparison.
Table 2. Results of transmission assessment survey (TAS) in the selected schools in the evaluation unit conducted during April 2015.
Schools with one or more filarial antigen positive children are shaded in orange.
| School ID | Sub-district | Primary Health Centre | Health Sub-Centre | Location (government / Private School) | No. Blood Samples | ICT +ve | |||
|---|---|---|---|---|---|---|---|---|---|
| 1 | Chidambaram | Bhuvanagiri | Keelbhuvanagiri | Keezhbhuvanagiri (Govt.) | 12 | 0 | |||
| 2 | Kavarapattu | C.Kothangudi | C.Kothangudi (Govt.) | 22 | 0 | ||||
| 3 | Kavarapattu | C.Kothangudi | C.Kothangudi (Govt.) | 5 | 0 | ||||
| 4 | Kavarapattu | Kavarapattu | Kavarapattu (Govt.) | 35 | 0 | ||||
| 5 | Kavarapattu | Uthamasolamangalam | Sithalapadi (Govt.) | 8 | 0 | ||||
| 6 | Krishnapuram | Manjakollai | Manjakollai (Govt.) | 32 | 0 | ||||
| 7 | Orathur | Mathurandaganallur | Devangudi (Private) | 13 | 0 | ||||
| 8 | Orathur | Orathur | Paradhur Chavadi (Govt.) | 9 | 0 | ||||
| 9 | Orathur | Sathamangalam | Vadapakkam (Govt.) | 5 | 0 | ||||
| 10 | Orathur | Sathamangalam | Vaiyalur (Govt) | 9 | 0 | ||||
| 11 | Palayankottai | Nangudi | Vizhuperundhurai (Govt.) | 12 | 0 | ||||
| 12 | Palayankottai | Ramapuram | T. Viruthangam (Govt.) | 24 | 0 | ||||
| 13 | Palayankottai | Ramapuram | Therkupalayam (Govt.) | 31 | 0 | ||||
| 14 | Puduchattram | C.Pudupettai | Parangipettai (Private) | 101 | 0 | ||||
| 15 | Puduchattram | Periyapattu | Periyandikuzhi (Govt.) | 11 | 0 | ||||
| 16 | Sethiathoppu | Melvalayamadevi | Kathazhai (Govt.) | 24 | 0 | ||||
| 17 | Sethiathoppu | Nellikollai | Thurinjikollai (Private) | 14 | 0 | ||||
| 18 | Vallampadugai | Vallampadugai | Vallampadugai (Private) | 81 | 0 | ||||
| 19 | Vilagam | Thunisiramedu | Mugaiyur (Govt.) | 25 | 0 | ||||
| Chidambaram Sub-district | 473 | 0 | |||||||
| 20 | Cuddalore | Cuddalore Municipality | Pudhupalayam | Pudhupalayam (Private) | 63 | 0 | |||
| 21 | Cuddalore Municipality | Cuddalore OT | Cuddalore OT (Private) | 256 | 0 | ||||
| 22 | Cuddalore Municipality | Manjakuppam | Manjakuppam (Govt) | 13 | 0 | ||||
| 23 | Cuddalore Municipality | Thirupapuliyur | Thirupapadhiriyur (Govt.) | 33 | 0 | ||||
| 24 | Karaikadu | Karaikadu | Kannarapettai (Govt.) | 21 | 0 | ||||
| 25 | Karaikadu | Pachyankuppam | Vazhisodhipalayam (Govt.) | 17 | 0 | ||||
| 26 | Nellikuppam Municipality | Nellikuppam | Nellikuppam (Govt) | 12 | 0 | ||||
| 27 | Oraiyur | Aviyanur | Aviyanur (Govt.) | 23 | 0 | ||||
| 28 | Oraiyur | Chinnapettai | Chinnapettai (Govt.) | 26 | 0 | ||||
| 29 | Thiruvanthipuram | Thiruvanthipuram | Thiruvanthipuram (Govt.) | 34 | 3 | ||||
| 30 | Thukkanampakkam | Kalaiyur | Erandayiramvilagam (Govt.) | 15 | 0 | ||||
| Cuddalore Sub-district | 513 | 3 | |||||||
| 31 | Kurinjipadi | Neyveli Lignite Corporation | Neyveli Lignite Corporation | Neyveli (Private) | 77 | 1 | |||
| 32 | Puliyurkattusalai | Mathanagopalapuram | Peikanatham (Govt.) | 12 | 0 | ||||
| 33 | Puliyurkattusalai | Puliyurkattusalai | Puliyurkattsagai (Govt.) | 9 | 0 | ||||
| 34 | Thiruchopuram | Andarmullipallam | Andarmullipallam (Govt.) | 34 | 0 | ||||
| 35 | Thiruchopuram | Andarmullipallam | Thorapadi (Govt.) | 52 | 0 | ||||
| 36 | Thondamanatham | Sammattikuppam | Sammitikuppam (Govt.) | 12 | 0 | ||||
| 37 | Vadalur | Karunkuli | Kollakurdi(Govt) | 11 | 0 | ||||
| Kurinjipadi Sub-district | 207 | 1 | |||||||
| 38 | Panrutti | Marungur | Marungur | Kollukarankuttai Vallalar (Private) | 281 | 0 | |||
| 39 | Panruti Municipality | Panruti | Tharkavandimedu (Govt.) | 4 | 0 | ||||
| 40 | Perperiyankuppam | Keelkangeyankuppm | Athirikuppam (Govt.) | 13 | 0 | ||||
| 41 | Perperiyankuppam | PP Kuppam | Muthandikuppam (Govt.) | 16 | 0 | ||||
| 42 | VP Nallur | Thiruvamoor | Kolapakkam (Govt.) | 7 | 0 | ||||
| 43 | VP Nallur | Thiruvamoor | Thiruvamur (Govt.) | 65 | 0 | ||||
| Panrutti Sub-district | 386 | 0 | |||||||
| Overall for Evaluation Unit | 1579 | 4 | |||||||
Govt.—Government School
Private–Private School
Mf-prevalence
As many as 9804 persons (against 9200 targeted) from 2981 households (15.5% of 19179 HHs) located in the 30 randomly selected clusters were screened for Mf. The percentage of individuals sampled varied between 12 and 21% among clusters (Table 3). In the evaluation unit selected, only four individuals from three clusters of two sub-districts were screened positive for Mf (0.04%; 95% CI: 0.01–0.1%) with Mf counts ranging from 2 to 16 (mean: 8 per 60μl blood). Mf-prevalence in all the three clusters were less than 1%, indicating that the prevalence in the EU is below the critical threshold (<1%) for transmission to continue. Though the observed Mf-prevalence was below the critical threshold in all 30 clusters, their upper 95% confidence limits exceeded the critical level in 24 of these clusters. Mf-prevalence did not differ significantly (P = 0.81) between sub-districts (range: 0.0% to 0.07%).
Table 3. Microfiilaria (Mf) prevalence in clusters from which Culex quinquefasciatus mosquitoes were sampled during October 2016-January 2017.
Clusters with one or more positive individuals are shaded in orange.
| Cluster No. | Sub-district | Site Name | Population | No. of blood samples Screened | Fraction Sampled (%) | Mf-prevalence (95% CI) (Clopper-Pearson interval) |
|---|---|---|---|---|---|---|
| 1 | Chidambaram | Bhuvanagiri Ward 2 | 1152 | 142 | 12.3 | 0 (0.0–2.6) |
| 2 | Chidambaram Anamalai Nagar Ward 2 | 542 | 113 | 20.8 | 0 (0.0–3.2) | |
| 3 | Chidambaram Ward 13 | 2188 | 272 | 12.4 | 0 (0.0–1.3) | |
| 4 | Ennanagram | 774 | 120 | 15.5 | 0 (0.0–3.0) | |
| 5 | Manjakudi | 5949 | 745 | 12.5 | 0 (0.0–0.4) | |
| 6 | Odakakkanalur | 1092 | 145 | 13.3 | 0 (0.0–2.5) | |
| 7 | Parangipettai Ward 14 | 2271 | 275 | 12.1 | 0.4 (0.1–2.0) | |
| 8 | Punjamangattuvalkkai | 520 | 74 | 14.2 | 0.0 (0.0–4.8) | |
| 9 | Singarkuppam Killai Ward 6 | 1769 | 227 | 12.8 | 0.0 (0.0–1.6) | |
| 10 | T.Neduncherri | 1817 | 240 | 13.2 | 0.0 (0.0–1.5) | |
| 11 | Therku Virthangan | 1054 | 144 | 13.7 | 0.0 (0.0–2.5) | |
| 12 | Vadakku Thittu | 1738 | 220 | 12.7 | 0.0 (0.0–1.7) | |
| 13 | Velayamadevi Kilpathi | 3598 | 447 | 12.4 | 0.2 (0.01–1.2) | |
| Chidambaram Sub-district | 24464 | 3164 | 12.9 | 0.06 (0.01–0.2) | ||
| 14 | Cuddalore | Cuddalore Ward 31 | 3345 | 415 | 12.4 | 0.0 (0.0–0.9) |
| 15 | Cuddalore Ward 8 | 4543 | 547 | 12.0 | 0.0 (0.0–0.7) | |
| 16 | Nellikuppam Ward 29 | 1700 | 259 | 15.2 | 0.0 (0.0–1.4) | |
| 17 | Nellikuppam Ward 6 | 1722 | 220 | 12.8 | 0.0 (0.0–1.7) | |
| 18 | Pachia Kuppam | 7857 | 932 | 11.9 | 0.0 (0.0–0.4) | |
| 19 | Paria Kangankuppam | 1543 | 204 | 13.2 | 0.0 (0.0–1.8) | |
| 20 | Singrikudi | 1703 | 233 | 13.7 | 0.0 (0.0–1.6) | |
| Cuddalore Sub-district | 22413 | 2810 | 12.5 | 0.0 (0.0–0.13) | ||
| 21 | Kurinjipadi | Buddampadi | 973 | 127 | 13.1 | 0.0 (0.0–2.8) |
| 22 | Kurunjipadi Ward 18 | 1537 | 196 | 12.8 | 0.0 (0.0–1.9) | |
| 23 | Madana Gopalapuram | 2063 | 277 | 13.4 | 0.0 (0.0–1.3) | |
| 24 | Ranganathapuram | 2529 | 299 | 11.8 | 0.0 (0.0–1.2) | |
| Kurinjipadi Sub-district | 7102 | 899 | 12.7 | 0.0 (0.0–0.4) | ||
| 25 | Panrutti | Enadrimangalam | 2293 | 281 | 12.3 | 0.0 (0.0–1.3) |
| 26 | P N Palayam | 7317 | 875 | 12.0 | 0.0 (0.0–0.4) | |
| 27 | Sathipattu | 6018 | 714 | 11.9 | 0.3 (0.03–1.0) | |
| 28 | Thorapadi | 293 | 41 | 14.0 | 0.0 (0.0–8.6) | |
| 29 | Vallam | 6309 | 753 | 11.9 | 0.0 (0.0–0.5) | |
| 30 | Panruti Ward 21 | 2002 | 267 | 13.3 | 0.0 (0.0–1.4) | |
| Panrutti sub-district | 14622 | 2931 | 20.0 | 0.07 (0.01–0.2) | ||
| Overall for Evaluation Unit | 68601 | 9804 | 12.6 | 0.04 (0.01–0.10) | ||
A parallel comparison of the Mf-prevalence in these clusters with the prevalence of parasite DNA estimated by xenomonitoring showed that the prevalence estimates are comparable between the two survey methods (human vs mosquito) in one of the three clusters (95% CIs for Mf-prevalence in human and parasite DNA prevalence in vector overlap with each other).
Cost of surveys
Table 4 compares the costs of school-based TAS and MX survey for assessing LF transmission in the EU. The total cost for conducting MX at an EU level was estimated to be $14,259 USD (1USD = Rs.73.72) per EU. The major cost component was personnel, accounting for 64.6% of the total cost, followed by transportation (17.1%). The total cost for conducting TAS with ICT in the EU was estimated to be $14,103.8 USD and the cost per school was $328 USD, and cost per child was $9.06 USD. The major cost component for TAS was ‘supplies’, accounting for about 60.5% of the total cost, followed by ‘personnel’ (37.5%) and transportation (2%); ICT alone shares about 97.7% ($8443 USD) of the total cost of supplies (Table 4). The total cost of xenomonitoring is more than the school-based TAS using ICT by $155.2 USD.
Table 4. Costs (US $) of transmission assessment survey (TAS) and molecular xenomonitoring (MX) for assessing lymphatic filariasis transmission in the evaluation unit.
| Heads | Cost of school-based TAS with ICT (%) | Cost of MX (%) |
|---|---|---|
| Personnel | 5286.5 (37.5) | 9070.5 (63.6) |
| Transport | 287.7 (2.0) | 2442.9 (17.1) |
| Supplies | ||
| ICT | 8334.0 (59.1) | NA |
| Field supplies | 195.6 (1.4) | 1657.6 (11.6) |
| Sub-total (Supplies) | 8529.6 (60.5) | 1657.6 (11.6) |
| Lab processing | NA | 1088.0 (7.6) |
| Total cost | 14,103.8 | 14,259.0 |
NA—Not applicable
Discussion
This is the first MX evaluation study carried out at an EU level in India to monitor W. bancrofti infection in Cx. quinquefasciatus mosquitoes. The study compares the results of MX with those of human Mf-survey carried out via community-based sampling (not WHO protocol) of 30 clusters (villages or wards), and TAS via ICT and conducted through a school-based sampling of 43 schools in the EU. After 15 rounds of MDA, when mosquito infection levels are expected to be very low, gravid traps yielded enough mosquitoes for transmission assessment by MX. Further, MX implemented in 2015 revealed that the prevalence of W. bancrofti DNA in Cx. quinquefasciatus was below the 0.25% provisional critical threshold [22, 31, 44], suggesting interruption of transmission in the EU. The TAS in 2015 showed that the rate of antigen-positive children was below the 2% critical threshold, indicating as well interruption of transmission and that the EU is qualified for stopping MDA. Our data therefore validated MX as a complementary tool to stop MDA in the EU. One year later (2016), a blood survey for microfilaremia showed that “none of the clusters was above the 1% Mf-threshold”, thus confirming the results of MX and TAS.
MX and TAS for post-MDA surveillance and validation
Several countries with different epidemiological settings applied MX to generate evidence for absence of active transmission during post-MDA situation. Many of these studies reported that the prevalence of W. bancrofti DNA in An. gambiae [in Sierra Leone, Togo 20, 33, 45] or in Cx. quinquefasciatus [in Bangladesh and India, 19, 21, 46] are well below the provisional threshold for transmission interruption [1% and 0.25% for An. gambiae and Cx. quinquefasciatus respectively, 22]. The MX results in all the above studies corroborated with TAS findings (Ag-prevalence below the threshold of 2% among children) that led to stopping MDA in endemic districts after multiple rounds of MDA. However, a few studies in Sri Lanka [8, 26, 47] and in American Samoa [9, 12, 34] have shown that the prevalence of W. bancrofti DNA in Cx. quinquefasciatus (>0.25%) and Aedes polynesiensis (>0.1%) were above the provisional transmission thresholds for the respective vector-parasite combination, despite the Ag-prevalence by TAS in the EUs were below the critical threshold of 2%. In American Samoa, school-based third TAS and the community-based serological studies carried out 10 years after cessation of MDA confirmed recrudescence of LF transmission, as indicated by the MX before third TAS [9, 34].
Derua et al. [35] have reported that the prevalence of W. bancrofti DNA in An. gambiae complex and An. funestus group in Mafia islands, Tanzania were above the provisional thresholds (vector infection rate of 1.7%), where the antigen positivity (4%) among 6–9 year-old children was also above the threshold level, despite nine rounds of MDA.
Application of MX, prior to MDA in Conakry, Guinea, in West Africa revealed that the prevalence of W. bancrofti DNA by LAMP (loop-mediated isothermal amplification assay) in both An. gambiae and Cx. quinquefasciatus were above the provisional threshold for interruption of transmission, despite the absence of circulating filarial antigen in human population [32]. Based on the above findings, we conclude that MX can be used as a complement to TAS for stopping MDA, detecting residual foci of infection during post-MDA or post-validation phases and for mapping areas to initiate MDA.
MX for assessing residual hotspots
WHO, while recommending MX for post-MDA surveillance, suggested focussing on mosquito surveys on individual villages to provide an indication of the presence of residual infection (residual hotspots) [46] at village level [18]. Rao et al. [48], after analysing MX data by sites reported persistent infection in many sites in an EU in Sri Lanka that stopped MDA post-5 rounds after demonstrating transmission interruption through TAS. Our MX survey detected 5 clusters with residual infection compared to 3 clusters detected by community-based Mf-survey or 2 schools by Ag-survey through TAS, suggesting that MX was more sensitive than community-based Mf-survey or school-based Ag-survey by TAS for detecting residual infection in areas under post-MDA surveillance.
In all the 30 clusters, the upper 95% CI for W. bancrofti DNA prevalence exceeded the provisional vector infection threshold of 0.25% (Table 1). Considering only the cluster wise point estimates, the results of MX reveal that 13.3% (4/30 clusters; 95% CI: 3.8–30.7%) of the clusters in the EU are expected to exceed the suggested critical threshold of 0.25% infection in vector for transmission interruption. This means that on an average one would expect 89 of the 669 clusters in the EU to be under the risk of resurgence. Further, among the five clusters positive by MX, one cluster was also positive for Mf in humans. A post-hoc power analysis indicated that the sample size for this cluster is adequately powered (>90%) to reject the null hypothesis that the prevalence of infection in mosquitoes is <0.25%, the threshold for transmission interruption. This suggests that about 3.3% (1 out of 30 clusters sampled) of the 669 clusters (i.e. 20 clusters) in the EU are expected to have residual human Mf carriers and therefore are at risk of transmission. Our site wise analysis of the MX data suggests that MX could be a highly sensitive tool for detecting residual hotspots during post-MDA surveillance phase.
It is important that the residual hotspots in an EU are monitored and appropriate site-specific interventions, such as vector control or screening and treating individuals for filarial infection initiated to prevent resurgence of infection during post-MDA phase. Since MX will detect only those hotspots within the selected clusters, the presence of other such hotspots in the EU can be identified using the environmental risk factors as proxy indictors, as reported in a recent study in India [49].
However, it is important to note that the samples for MX, Mf and TAS are powered only to take a decision at an EU level and not at a cluster level. We have drawn inference about transmission or residual infection status at the cluster level (village, school) based on the estimated prevalence of infection (Mf and Ag) for individual clusters. Similar assessment has been made by others [8, 9, 26, 33, 48]. The individual cluster level estimate (for MX or TAS) could only give an indication, considering the small sample size for the individual clusters. Further studies with appropriate sample size for each cluster are warranted to provide quantitative assessment of transmission levels or residual infection. Such an estimate could be used as an additional criterion to supplement the TAS decision for stopping MDA in an EU, as had been recommended as ‘dual thresholds’ for stopping MDA in onchocerciasis control programmes in Africa [50].
Sampling: MX, Mf and TAS
Our data from both MX and Mf-surveys confirmed the presence of residual infection in a few of the communities in the EU, MX being more sensitive in detecting W. bancrofti persistence than the Mf-survey in humans (5 clusters positive by MX vs 3 clusters by Mf in human). MX detected filarial DNA in clusters where no infection was detected in humans by Mf-survey. Similar observation has also been reported in Sri Lanka [47]. This could be due to a higher efficiency of mosquitoes in picking up infection as reported earlier as a phenomenon of “limitation” [51–53]. However, MX did not show vector infection in two of the three clusters positive for Mf in human. The lack of concordance between MX and Mf-surveys at the cluster level could either be due to migration of individuals from endemic districts or the difference in the location of the HHs selected for the surveys. Lack of concordance has been reported elsewhere where both the surveys were carried out in the same HHs [31]. Such household level comparisons may not be realistic as the mosquitoes trapped are not necessarily from the same house where the traps are placed and the fact that MX sampling is not designed to take a decision at a household or cluster level.
The challenge with respect to MX is the availability of adequate manpower (technical expertise in mosquito identification and molecular assays) and laboratory facilities for processing the samples in the health system. With current promotion of global vector control response by the WHO, capacity building and strengthening of laboratories can be done, which will not only be useful for MX but also cover all vector borne diseases [54]. Community-based Mf-survey is operationally more challenging and time-consuming (invasive blood sampling, inconvenient survey time, mandatory written consent from volunteered individuals) than MX survey. MX is non-invasive and requires only verbal consent for placing gravid traps in the selected HHs.
In school-based TAS, the schools are selected systematically with probability proportional to the size of the target population (no. of children in I and II grades) in schools. In this selection procedure, though it provides equal probability of selecting schools, schools with large strengths share a major fraction of the sampled children. In our study, of the 43 schools selected for TAS, 35 (81.4%) are government schools and 8 (18.6%) are private schools. The eight private schools with a large strength shared 56.2% of the children (n = 1578) selected for TAS compared to 43.8% from 35 government schools. The above percentages represented the actual distribution of schools (80% government schools and 20% private schools) and children (46.9 vs 53.0%) in the EU. Children in the private institutions are mostly from middle class and above. Most of the government schools are spatially located in rural areas spread over the entire EU preferred by students from a low socio-economic background and at higher risk of infection (in terms of exposure to LF infection) [55–57]. Therefore, school-based TAS is likely to miss antigen positives from the small and highly focal residual clusters of transmission if the prevalence of infection is spatially variable within an EU. On the other hand, as for Mf-survey, a community-based TAS is practically more challenging (house-to-house visit, time of survey depending on the availability of residents, numbering and locating houses and defining area boundaries) and time-consuming than a school-based TAS, though it could minimize the risk of failing to detect the antigen positives among the children from the areas at high risk of transmission. Further, both Mf-survey and TAS are invasive and require written consent for drawing blood sample from each volunteered individual in a household.
Costs of MX and TAS
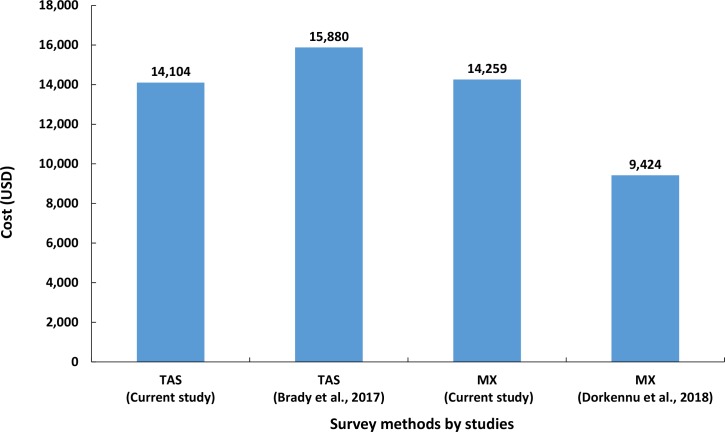
In the present study, the cost of MX was estimated to be $14259 USD per EU for 358 pools (Fig 6). As has been reported elsewhere [20, 58], a major part of the MX-cost incurred was towards allowances and wages for the personnel followed by transportation costs. The cost of MX in this study was higher than the estimate reported in Togo, African region ($11970.13 USD per EU for 210 pools) [20] which is inclusive of the cost of $2546.5 USD towards consultancy, training, ethics application, vehicle maintenance, data management, communication and shipment of samples to the central laboratory. The cost per EU after excluding all the above components is $9423.6 USD (Fig 6). Thus, the cost of per pool collected and processed was $44.9 USD in the African study against $40.8 USD in the current study. Despite the use of low-cost LAMP assay ($0.82 USD per test) [59], the cost per pool in the African study was marginally higher than that in the present study using TE based quantitative PCR assay ($3 USD per test). The higher cost could be due to the different methods (pyrethrum spray catches, human landing and exit trap collections) employed for collecting Anopheles mosquito vectors compared to gravid traps used for Culex mosquito in the present study.
Fig 6. Comparison of total cost per evaluation unit for transmission assessment survey (TAS) or molecular xenomonitoring (MX) incurred by studies carried out in different countries.
The cost of school-based TAS per EU ($14104 USD) in our study is lower than that derived ($16179 USD) for Asian countries (Fig 6) [58]. As ours is a research study, we did not include the cost of training the personnel. The cost in this study is less than the cost per EU for Asian region even after excluding the training component ($15880 USD). Alternatively, based on the findings of Brady et al. [58], had a community-based TAS been conducted in our study area, the estimated cost ($39971.0USD) would have been 2.83 times higher than that of the school-based TAS. If FTS were used instead of ICT, then the costs of school and community-based TAS per EU would be reduced by 43.6% and estimated to be $7941USD and $22,504USD, respectively. Though community-based TAS is more expensive than school-based TAS, it has been reported to be more sensitive in detecting residual hotspots [9]. However, the cost of community based TAS with FTS is 1.56 times higher than that of MX. Further, since MX is a non-invasive mosquito-based survey, and a more sensitive tool than school or community-based TAS in detecting residual hotspots in an EU, with a sample size powered enough to detect residual hotspots at a cluster-level, it could be a promising tool for remapping and monitoring transmission level during post MDA and validation phases. One of the limitations of the costing analysis is that this study was conducted in a research mode, which may not reflect the costing under programme, where there is scope for utilising the existing manpower after appropriate training, wherever available.
Conclusions
The results of this study demonstrating the validity of MX at an evaluation unit level provide evidence to recommend MX as a tool complementary to TAS for stopping MDA and to detect resurgence of infection during post-MDA surveillance or validation phases. WHO is reviewing available evidence from research studies and current country experiences to develop specific post-validation surveillance guidelines [4]. Epidemiological indicators such as antibody (Ab) in the younger age classes, antigen (Ag) among adults and infection in vectors are considered by WHO for post-validation surveillance. Mf-prevalence survey requires large samples and visiting individuals at night. On the other hand, MX is feasible and can assess not only the ongoing risk of transmission but also residual transmission hotspots and the risk of resurgence of infection. MX can also provide information on vector abundance to plan vector management, if required. Early detection of transmission risk using MX will be useful to initiate appropriate measures of vector control to prevent resurgence of infection during post-MDA and validation phases.
Supporting information
(DOCX)
(DOCX)
(DOCX)
Acknowledgments
We would like to acknowledge the field and laboratory team members, particularly Mr. V. Padmanaban, Technical Officer, Mr. R. Balasubramanian, Technical Officer and Mr. Y. Srinivas Murty, Technical Assistant for their technical assistance. We thank the Deputy Director of Health Services, Cuddalore, Directorate of Public Health and Preventive Medicine, Department of Health and Family Welfare, Tamil Nadu for the cooperation and support extended to the study. We are also grateful to all the school principals and teachers concerned for their assistance in conducting the transmission assessment survey and the parents who consented to their children’s participation in the study. We also acknowledge the residents of the villages in the evaluation unit of Cuddalore district, Tamil Nadu for their cooperation in placing the gravid traps on their household premises.
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
The author(s) received no specific funding for this work.
References
- 1.Ottesen EA. The global programme to eliminate lymphatic filariasis. Trop Med Int Health. 2000;5(9):591–4. 10.1046/j.1365-3156.2000.00620.x . [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. Progress report 2000–2009 and strategic plan 2010–2020 of the global programme to eliminate lymphatic filariasis: halfway towards eliminating lymphatic filariasis. Geneva: World Health Organization; 2010. [Google Scholar]
- 3.World Health Organization. Global programme to eliminate lymphatic filariasis: progress report, 2017. Wkly Epidemiol Rec. 2018;93(44):589–604. [Google Scholar]
- 4.World Health Organization. Validation of elimination of lymphatic filariasis as a public health problem. Geneva: World health organization; 2017. Available from: http://WHO/HTM/NTD/PCT/2017.01. [Google Scholar]
- 5.World Health Organization. Monitoring and epidemiological assessment of mass drug administration in the global programme to eliminate lymphatic fi lariasis: a manual for national elimination programmes. WHO/HTM/NTD/PCT/20114; 2011:1–79. [Google Scholar]
- 6.National Vector-Borne Disease Control Programme. Elimination of lymphatic filariasis, India: 2013–2014. National guidelines for transmission assessment survey (for district & state level health officials). 2014;http://nvbdcp.gov.in/Doc/TAS-National-Guidelines-2013-14.pdf. Accessed 27 Aug 2016.
- 7.Coulibaly Y, Coulibaly SY, Dolo H, Konate S, Diallo AA, Doumbia SS, et al. Dynamics of antigenemia and transmissionintensity ofWuchereria bancroftifollowingcessation of mass drug administration in aformerly highly endemic region of Mali. Parasites & Vectors. 2016;9(1):628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rao RU, Nagodavithana KC, Samarasekera SD, Wijegunawardana AD, Premakumara WD, Perera SN, et al. A comprehensive assessment of lymphatic filariasis inSri Lanka six years after cessation of mass drug administration. PLoS Negl Trop Dis. 2014;8(11):e3281 Epub 2014/11/14. 10.1371/journal.pntd.0003281 PNTD-D-14-00651 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sheel M, Sheridan S, Gass K, Won K, Fuimaono S, Kirk M, et al. Identifying residual transmission of lymphatic filariasis after mass drug administration: Comparing school-based versus community-based surveillance—American Samoa, 2016. PLoS Negl Trop Dis. 2018;12(7):e0006583 Epub 2018/07/17. 10.1371/journal.pntd.0006583 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rebollo MP, Mohammed KA, Thomas B, Ame S, Ali SM, Cano J, et al. Cessation of mass drug administration for lymphatic filariasis in Zanzibar in 2006: was transmission interrupted? PLoS Negl Trop Dis. 2015;9(3):e0003669 Epub 2015/03/31. 10.1371/journal.pntd.0003669 PNTD-D-14-01272 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lau CL, Won KY, Becker L, Soares Magalhaes RJ, Fuimaono S, Melrose W, et al. Seroprevalence and spatial epidemiology of Lymphatic Filariasis inAmerican Samoa after successful mass drug administration. PLoS Negl Trop Dis. 2014;8(11):e3297 Epub 2014/11/14. 10.1371/journal.pntd.0003297 PNTD-D-14-01061 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schmaedick MA, Koppel AL, Pilotte N, Torres M, Williams SA, Dobson SL, et al. Molecular xenomonitoring using mosquitoes to map lymphatic filariasis after mass drug administration in American Samoa. PLoS Negl Trop Dis. 2014;8(8):e3087 Epub 2014/08/15. 10.1371/journal.pntd.0003087 PNTD-D-14-00593 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Souza DK, Otchere J, Ahorlu CS, Adu-Amankwah S, Larbi IAD, E., McCarthy FA, et al. Low Microfilaremia Levels in Three Districts in Coastal Ghana with at Least 16 Years of Mass Drug Administration and Persistent Transmission of Lymphatic Filariasis. Tropical Medicine and Infectious Disease. 2018;3(4), 105 10.3390/tropicalmed3040105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Minetti C, LaCourse EJ, Reimer CB, Stothard JR. Focusing nucleic acid-based molecular diagnostics and xenomonitoring approaches for human helminthiases amenable to preventive chemotherapy. Parasitology. 2016. 10.1017/pao.2016.13. [DOI] [Google Scholar]
- 15.World Health Organization. Lymphatic filariasis infection and disease: Control strategies Report of a Consultative meeting held at the Universiti Sains Malaysia, Penang, Malaysia: 1994. [Google Scholar]
- 16.National Vector-Borne Disease Control Programme. Accelerated plan for elimination of lymphatic filariasis 2018: Directorate General of Health Services, Ministry of Health and Family Welfare, Govt. of India; [cited 2018]. Available from: nvbdcp.gov.in/WriteReadData/l892s/1031567531528881007.pdf.
- 17.World Health Organization. Defining the roles of vector control and xenomonitoring in the global programme to eliminate lymphatic filariasis Report of the Informal Consultation WHO/HQ, Geneva, 29−31 January 2002. Geneva: WHO/ Department of Communicable Disease Prevention CaE; 2002. [Google Scholar]
- 18.World Health Organization. Lymphatic Filariasis: A Handbook of Practical Entomology for National Lymphatic Filariasis Elimination Programmes. Geneva: World Health Organization,; 2013. Available from: http://apps.who.int/iris/bitstream/10665/87989/1/9789241505642_eng.pdf. [Google Scholar]
- 19.Subramanian S, Jambulingam P, Chu BK, Sadanandane C, Vasuki V, Srividya A, et al. Application of a household-based molecular xenomonitoring strategy to evaluate the lymphatic filariasis elimination program in Tamil Nadu, India. PLoS Negl Trop Dis. 2017;11(4):e0005519 Epub 2017/04/14. 10.1371/journal.pntd.0005519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dorkenoo MA, de Souza DK, Apetogbo Y, Oboussoumi K, Yehadji D, Tchalim M, et al. Molecular xenomonitoring for post-validation surveillance of lymphatic filariasis in Togo: no evidence for active transmission. Parasit Vectors. 2018;11(1):52 Epub 2018/01/25. 10.1186/s13071-017-2611-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Irish SR, Al-Amin HM, Paulin HN, Mahmood A, Khan RK, Muraduzzaman AKM, et al. Molecular xenomonitoring for Wuchereria bancrofti in Culex quinquefasciatus in two districts in Bangladesh supports transmission assessment survey findings. PLoS Negl Trop Dis. 2018;12(7):e0006574 Epub 2018/07/27. 10.1371/journal.pntd.0006574 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.World Health Organization. The role of polymerase chain reaction techniques for assessing lymphatic filariasis transmission. Geneva: World Health Organization,; 2009. Available from: http://apps.who.int/iris/bitstream/10665/87989/1/9789241505642_eng.pdf. [Google Scholar]
- 23.Irish SR, Moore SJ, Derua YA, Bruce J, Cameron MM. Evaluation of gravid traps for the collection of Culex quinquefasciatus, a vector of lymphatic filariasis in Tanzania. Trans R Soc Trop Med Hyg. 2013;107(1):15–22. Epub 2012/12/12. 10.1093/trstmh/trs001 . [DOI] [PubMed] [Google Scholar]
- 24.Irish SR, Stevens WM, Derua YA, Walker T, Cameron MM. Comparison of Methods for Xenomonitoring in Vectors of Lymphatic Filariasis in Northeastern Tanzania. Am J Trop Med Hyg. 2015;93(5):983–9. Epub 2015/09/10. 10.4269/ajtmh.15-0234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Moustafa MA, Salamah MMI, Thabet HS, Tawfik RA, Mehrez MM, Hamdy DM. Molecular xenomonitoring (MX) and transmission assessment survey (TAS) of lymphatic filariasis elimination in two villages, Menoufyia Governorate, Egypt. Eur J Clin Microbiol Infect Dis. 2017;36(7):1143–50. Epub 2017/02/06. 10.1007/s10096-017-2901-3 . [DOI] [PubMed] [Google Scholar]
- 26.Rao RU, Samarasekera SD, Nagodavithana KC, Punchihewa MW, Dassanayaka TD, P KDG, et al. Programmatic Use of Molecular Xenomonitoring at the Level of Evaluation Units to Assess Persistence of Lymphatic Filariasis in Sri Lanka. PLoS Negl Trop Dis. 2016;10(5):e0004722 Epub 2016/05/20. 10.1371/journal.pntd.0004722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thornton JH, Batengana BM, Eiras AE, Irish SR. Evaluation of collection methods for Culex quinquefasciatus, Aedes aegypti, and Aedes simpsoni in northeastern Tanzania. J Vector Ecol. 2016;41(2):265–70. Epub 2016/11/20. 10.1111/jvec.12221 . [DOI] [PubMed] [Google Scholar]
- 28.Ramesh A, Cameron M, Spence K, Hoek Spaans R, Melo-Santos MAV, Paiva MHS, et al. Development of an urban molecular xenomonitoring system for lymphatic filariasis in the Recife Metropolitan Region, Brazil. PLoS Negl Trop Dis. 2018;12(10):e0006816 Epub 2018/10/17. 10.1371/journal.pntd.0006816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Opoku M, Minetti C, Kartey-Attipoe WD, Otoo S, Otchere J, Gomes B, et al. An assessment of mosquito collection techniques for xenomonitoring of anopheline-transmitted Lymphatic Filariasis in Ghana. Parasitology. 2018;145(13):1783–91. Epub 2018/06/15. 10.1017/S0031182018000938 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dugassa S, Lindh JM, Lindsay SW, Fillinger U. Field evaluation of two novel sampling devices for collecting wild oviposition site seeking malaria vector mosquitoes: OviART gravid traps and squares of electrocuting nets. Parasit Vectors. 2016;9(1):272 Epub 2016/05/12. 10.1186/s13071-016-1557-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Farid HA, Morsy ZS, Helmy H, Ramzy RM, El Setouhy M, Weil GJ. A critical appraisal of molecular xenomonitoring as a tool for assessing progress toward elimination of Lymphatic Filariasis. Am J Trop Med Hyg. 2007;77(4):593–600. Epub 2007/11/06. [PMC free article] [PubMed] [Google Scholar]
- 32.Kouassi BL, de Souza DK, Goepogui A, Narh CA, King SA, Mamadou BS, et al. Assessing the presence of Wuchereria bancrofti in vector and human populations from urban communities in Conakry, Guinea. Parasit Vectors. 2015;8:492 Epub 2015/09/28. 10.1186/s13071-015-1077-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de Souza DK, Ansumana R, Sessay S, Conteh A, Koudou B, Rebollo MP, et al. The impact of residual infections on Anopheles-transmitted Wuchereria bancrofti after multiple rounds of mass drug administration. Parasit Vectors. 2015;8:488 Epub 2015/09/25. 10.1186/s13071-015-1091-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lau CL, Won KY, Lammie PJ, Graves PM. Lymphatic Filariasis Elimination in American Samoa: Evaluation of Molecular Xenomonitoring as a Surveillance Tool in the Endgame. PLoS Negl Trop Dis. 2016;10(11):e0005108 Epub 2016/11/02. 10.1371/journal.pntd.0005108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Derua YA, Rumisha SF, Batengana BM, Max DA, Stanley G, Kisinza WN, et al. Lymphatic filariasis transmission on Mafia Islands, Tanzania: Evidence from xenomonitoring in mosquito vectors. PLoS Negl Trop Dis. 2017;11(10):e0005938 Epub 2017/10/07. 10.1371/journal.pntd.0005938 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Vasuki V, Subramanian S, Sadanandane C, Jambulingam P, Khader MS. Molecular xenomonitoring of Wuchereria bancrofti in Culex quinquefasciatus from an endemic area: Comparison of two DNA extraction methods for realtime PCR assay. J Vector Borne Dis. 2016;53(1):77–80. Epub 2016/03/24. . [PubMed] [Google Scholar]
- 37.Office of the Registrar General & Census Commissioner I. Primary Census Abstract 2011. In: Ministry of Home Affairs GoI, editor. Delhi2011.
- 38.Vasuki V, Hoti SL, Sadanandane C, Jambulingam P. A simple and rapid DNA extraction method for the detection of Wuchereria bancrofti infection in the vector mosquito, Culex quinquefasciatus by Ssp I PCR assay. Acta Trop. 2003;86(1):109–14. Epub 2003/04/25. 10.1016/s0001-706x(02)00267-x . [DOI] [PubMed] [Google Scholar]
- 39.Rao RU, Atkinson LJ, Ramzy RM, Helmy H, Farid HA, Bockarie MJ, et al. A real-time PCR-based assay for detection of Wuchereria bancrofti DNA in blood and mosquitoes. Am J Trop Med Hyg. 2006;74(5):826–32. Epub 2006/05/12. [PMC free article] [PubMed] [Google Scholar]
- 40.Weil GJ, Lammie PJ, Weiss N. The ICT filariasis test: a rapid-format antigen test for diagnosis of bancroftian filariasis. Parasitology Today. 1997;13:401–4. 10.1016/s0169-4758(97)01130-7 [DOI] [PubMed] [Google Scholar]
- 41.Katholi CR, Toe L, Merriweather A, Unnasch TR. Determining the prevalence of Onchocerca volvulus infection in vector populations by polymerase chain reaction screening of pools of black flies. J Infect Dis. 1995;172:1414–7. 10.1093/infdis/172.5.1414 [DOI] [PubMed] [Google Scholar]
- 42.Katholi CR, Unnasch TR. Important experimental parameters for determining infection rates in arthropod vectors using pool screening approaches. Am J Trop Med Hyg. 2006;74(5):779–85. Epub 2006/05/12. . [PubMed] [Google Scholar]
- 43.Weil GJ, Curtis KC, Fakoli L, Fischer K, Gankpala L, Lammie PJ, et al. Laboratory and field evaluation of a new rapid test for detecting Wuchereria bancrofti antigen in human blood. Am J Trop Med Hyg. 2013;89(1):11–5. Epub 2013/05/22. 10.4269/ajtmh.13-0089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pedersen EM, Stolk WA, Laney SJ, Michael E. The role of monitoring mosquito infection in the Global Programme to Eliminate Lymphatic Filariasis. Trends Parasitol. 2009;25(7):319–27. Epub 2009/06/30. 10.1016/j.pt.2009.03.013 . [DOI] [PubMed] [Google Scholar]
- 45.Dorkenoo MA, Bronzan R, Yehadji D, Tchalim M, Yakpa K, Etassoli S, et al. Surveillance for lymphatic filariasis after stopping mass drug administration in endemic districts of Togo, 2010–2015. Parasit Vectors. 2018;11(1):244 Epub 2018/04/18. 10.1186/s13071-018-2843-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Swaminathan S, Perumal V, Adinarayanan S, Kaliannagounder K, Rengachari R, Purushothaman J. Epidemiological assessment of eight rounds of mass drug administration for lymphatic filariasis in India: implications for monitoring and evaluation. PLoS neglected tropical diseases. 2012;6(11):e1926 10.1371/journal.pntd.0001926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rao RU, Samarasekera SD, Nagodavithana KC, Dassanayaka TDM, Punchihewa MW, Ranasinghe USB, et al. Reassessment of areas with persistent Lymphatic Filariasis nine years after cessation of mass drug administration in Sri Lanka. PLoS Negl Trop Dis. 2017;11(10):e0006066 Epub 2017/10/31. 10.1371/journal.pntd.0006066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Rao RU, Samarasekera SD, Nagodavithana KC, Goss CW, Punchihewa MW, Dassanayaka TDM, et al. Comprehensive Assessment of a Hotspot with Persistent Bancroftian Filariasis in Coastal Sri Lanka. Am J Trop Med Hyg. 2018;99(3):735–42. Epub 2018/07/18. 10.4269/ajtmh.18-0169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Srividya A, Subramanian S, Sadanandane C, Vasuki V, Jambulingam P. Determinants of transmission hotspots and filarial infection in households after eight rounds of mass drug administration in India. Trop Med Int Health. 2018;23(11):1251–8. Epub 2018/08/29. 10.1111/tmi.13143 . [DOI] [PubMed] [Google Scholar]
- 50.WHO/APOC. Conceptual and operational framework of onchocerciasis elimination with ivermectin treatment. 2010.
- 51.Southgate BA. The significance of low density microfilaraemia in the transmission of lymphatic filarial parasites. J Trop Med Hyg. 1992;95:79–86. [PubMed] [Google Scholar]
- 52.Subramanian S, Krishnamoorthy K, Ramaiah KD, Habbema JD, Das PK, Plaisier AP. The relationship between microfilarial load in the human host and uptake and development of Wuchereria bancrofti microfilariae by Culex quinquefasciatus: a study under natural conditions. Parasitology. 1998;116 (Pt 3):243–55. 10.1017/s0031182097002254 . [DOI] [PubMed] [Google Scholar]
- 53.Stolk WA, Van Oortmarssen GJ, Subramanian S, Das PK, Borsboom GJ, Habbema JD, et al. Assessing density dependence in the transmission of lymphatic filariasis: uptake and development of Wuchereria bancrofti microfilariae in the vector mosquitoes. Med Vet Entomol. 2004;18(1):57–60. 10.1111/j.0269-283x.2004.0470.x . [DOI] [PubMed] [Google Scholar]
- 54.World Health Organization. Global vector control response 2017–2030. Geneva: World Health Organization; 2017. Available from: http://apps.who.int/iris/bitstream/10665/259205/1/9789241512978-eng.pdf?ua=1. [Google Scholar]
- 55.Houweling TA, Karim-Kos HE, Kulik MC, Stolk WA, Haagsma JA, Lenk EJ, et al. Socioeconomic Inequalities in Neglected Tropical Diseases: A Systematic Review. PLoS Negl Trop Dis. 2016;10(5):e0004546 Epub 2016/05/14. 10.1371/journal.pntd.0004546 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mutheneni SR, Upadhyayula SM, Kumaraswamy S, Kadiri MR, Nagalla B. Influence of socioeconomic aspects on lymphatic filariasis: A case-control study in Andhra Pradesh, India. J Vector Borne Dis. 2016;53(3):272–8. Epub 2016/09/30. . [PubMed] [Google Scholar]
- 57.Upadhyayula SM, Mutheneni SR, Kadiri MR, Kumaraswamy S, Nagalla B. A cohort study of lymphatic filariasis on socio economic conditions in Andhra Pradesh, India. PLoS One. 2012;7(3):e33779 Epub 2012/03/24. 10.1371/journal.pone.0033779 [pii]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Brady MA, Stelmach R, Davide-Smith M, Johnson J, Pou B, Koroma J, et al. Costs of Transmission Assessment Surveys to Provide Evidence for the Elimination of Lymphatic Filariasis. PLoS Negl Trop Dis. 2017;11(2):e0005097 Epub 2017/02/02. 10.1371/journal.pntd.0005097 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Takagi H, Itoh M, Kasai S, Yahathugoda TC, Weerasooriya MV, Kimura E. Development of loop-mediated isothermal amplification method for detecting Wuchereria bancrofti DNA in human blood and vector mosquitoes. Parasitol Int. 2011;60(4):493–7. Epub 2011/09/21. 10.1016/j.parint.2011.08.018 . [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)
(DOCX)
(DOCX)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.