Fig. 5. Adenovirus stability profiles of liquid film formulations during long-term storage.
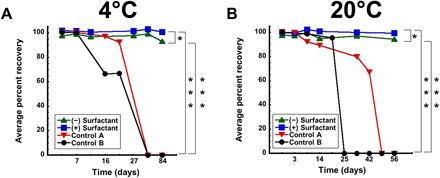
Virus (1.25 × 1012 vp) was placed in formulations 27 and 28 and stored in the liquid form in controlled environmental chambers held at 4°C (A) and 20°C (B) for 84 days. Replicate samples (n = 3) were collected at each time point, and live virus concentration was assessed by a standard infectious titer assay (19). Virus at the same concentration was also placed in two standard liquid formulations, and infectious titers under each storage condition were also assessed for comparison. Control A formulation consisted of 100 mM PBS (pH 7.4) and 10% glycerol. Control B formulation consisted of 100 mM PBS alone. In each panel, data represent the average ± SD. *P < 0.05, ***P < 0.001, two-tailed Student’s t test.
