Fig. 8. Immunogenicity profiles of thin film vaccine validate use in oral immunization strategies.
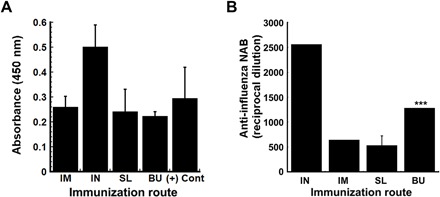
Individual samples of heat-inactivated serum collected 28 days after immunization from BALB/c mice were evaluated for influenza-specific IgG by ELISA. The average optical density read from samples obtained from each treatment group are presented to serve as a measure of relative antibody concentration. Each assay was validated by readings obtained from a sample collected from mice immunized with the same strain of influenza with an established anti-influenza antibody titer of 1:40 (+ control, Emory). Readings obtained from samples collected from mice given saline (negative controls) were subtracted from all absorbances (A). Neutralization capacity of antibodies was assessed by serial dilution of heat-inactivated serum with a fixed amount of H1N1 influenza virus before infection of MDCK (Madin-Darby canine kidney) cells. The reciprocal dilution plotted for each treatment group reflects the dilution at which the ability of the virus to infect target cells was reduced by 50% (B). In each panel, results are expressed as average values ± SEM and are representative of groups containing four mice per immunization route. ***P < 0.001, two-tailed Student’s t test. IM, intramuscular; IN, intranasal; SL, sublingual; BU, buccal.
