Abstract
Newcastle disease (ND) is a contagious disease that affects most species of birds. Its causative pathogen, Newcastle disease virus (NDV), also exhibits considerable oncolytic activity against mammalian cancers. A better understanding of the pathogenesis of NDV will help us design efficient vaccines and novel anticancer strategies. GW3965, a widely used synthetic ligand of liver X receptor (LXR), induces the expression of LXRs and its downstream genes, including ATP-binding cassette transporter A1 (ABCA1). ABCA1 regulates cellular cholesterol homeostasis. Here, we found that GW3965 inhibited NDV infection in DF-1 cells. It also inhibited NF-κB activation and reduced the upregulation of proinflammatory cytokines induced by the infection. Further studies showed that GW3965 exerted its inhibitory effects on virus entry and replication. NDV infection increased the mRNA levels of several lipogenic genes but decreased the ABCA1 mRNA level. Overexpression of ABCA1 inhibited NDV infection and reduced the cholesterol content in DF-1 cells, but when the cholesterol was replenished, NDV infection was restored. GW3965 treatment prevented cholesterol accumulation in the perinuclear area of the infected cells. In summary, our studies suggest that GW3965 inhibits NDV infection, probably by affecting cholesterol homeostasis.
Keywords: Newcastle Disease Virus, Cholesterol Efflux, Severe Acute Respiratory Syndrome, Lipogenic Gene, Newcastle Disease Virus Infection
Introduction
Newcastle disease virus (NDV), the causative agent of Newcastle disease (ND), can infect at least 241 species of birds, and results in substantial economic loss to the poultry industry [27]. Newcastle disease virus, an enveloped, non-segmented, and negative-stranded RNA virus, belongs to the family of Paramyxoviridae. The genome of NDV encodes six structural proteins, including nucleoprotein (NP), phosphoprotein (P), large polymerase protein (L), matrix protein (M), hemagglutinin-neuraminidase (HN), and fusion (F) glycoprotein [19, 24, 39, 43]. Although commonly administrated with live NDV vaccines, Newcastle disease remains a severe burden for poultry production.
Viruses hijack the host machinery for their benefit; lipid metabolism is one of the features that the viruses exploit [37]. NDV infection disturbs the lipid metabolism in chickens [32]. Studies on various viruses demonstrate that the pharmacological agents targeting lipid metabolism can inhibit virus infection [30].
Liver X receptors (LXR-α and LXR-β) are DNA-binding transcription factors that belong to the nuclear receptor superfamily and act as sensors of lipid homeostasis [33]. LXR-α regulates cholesterol and lipid metabolism; its target gene ABCA1 promotes the efflux of cellular cholesterol to maintain sterol homeostasis [5, 38]. Cholesterol shapes the properties of lipid bilayers. It is the major component of lipid rafts, the platform for the assembly of various signaling molecule complexes [14, 35]. Lipid rafts are also required for the entry of some viruses, including severe acute respiratory syndrome (SARS) virus and Japanese encephalitis virus [21, 46].
In our studies, we showed that the LXR ligand GW3965 inhibited NDV infection. It inhibited NF-κB activation and decreased the upregulation of proinflammatory cytokines induced by the infection. GW3965 exerted its inhibitory effects on virus entry and replication. Meanwhile, NDV infection impaired ABCA1 gene expression, which could be restored by GW3965 treatment. ABCA1 overexpression inhibited NDV infection and reduced the cholesterol content in infected cells, whereas cholesterol replenishment could restore the infection. Filipin III staining showed that cholesterol was accumulated in the perinuclear area of the infected cells, whereas GW3965 treatment decreased the accumulation. Collectively, our studies suggested that GW3965 inhibited NDV infection, probably by affecting cholesterol homeostasis.
Materials and methods
Cell culture and virus
DF-1 cells were obtained from the American Type Culture Collection (ATCC, Manassas, VA, USA) and cultured (5 % CO2) in Dulbecco’s modified Eagle medium (DMEM, GIBCO) supplemented with 10 % fetal bovine serum (FBS, Invitrogen) at 37 °C. The NDV strain Herts/33 was obtained from China Institute of Veterinary Drug Control (Beijing, China). The virus was propagated in embryonated chicken eggs. The 50 % tissue culture infected dose (TCID50) was determined by the Reed-Muench method in DF-1 cells [31]. The cells were transfected with polyethylenimine (25 kDa; Sigma) according to the manufacturer’s instructions.
Reagents
GW3965 was purchased from Selleck (China). Cholesterol-water soluble was purchased from Sigma. Mouse monoclonal β-actin antibody (Sigma), goat anti-mouse IgG (Beyotime), and goat anti-rabbit IgG (Beyotime, China) were used in the studies. Filipin III was purchased from Cayman Chemical (U.S.A.). A mouse monoclonal antibody against NDV nucleocapsid protein (NP) was prepared in our laboratory.
Western blot
Cells were washed with PBS buffer, scraped and lysed with RIPA buffer (Tris-HCl 50 mM, pH 7.4, 1 mM EDTA, 150 mM NaCl, 1 % NP40, 5 mM NaF, 0.25 % sodium deoxycholate, 5 mM sodium orthovanadate, 0.4 % SDS, and 0.1 mM PMSF) containing protease inhibitor and phosphatase inhibitor (Thermo) for 2 h at 4 °C. The cell lysates were centrifuged at 14,000g for 15 min at 4 °C. The supernatant was collected and the protein concentration was determined by BCA protein assay kit (Thermo) according to the manufacturer’s instructions. Equal amount of protein samples were loaded and separated by SDS-PAGE. The proteins were then transferred to a nitrocellulose (NC) membrane (Bio-Rad). The membrane was blocked (5 % milk in PBS supplemented with 0.5 % Tween-20) at room temperature for 2 h followed by incubation with the primary antibody (1 h) and the secondary antibody (1 h) at room temperature, respectively. Immunoreactive bands were detected using an enhanced chemiluminescence system (Vazyme, China).
qRT-PCR
Total RNA was extracted using TRIzol (Invitrogen) according to the manufacturer’s instructions. One μg of total RNA was used to synthesize cDNA at 42 °C for 1 h with MMLV reverse transcriptase (Promega) using the random hexamer (18T) to transcribe host genes or specific primer to transcribe viral gRNA (genome RNA). Quantitative real-time PCR was carried out using AceQ qPCR SYBR Green Master Mix (Vazyme). The primer sequences used in the study are listed in Table 1. β-actin RNA was used as the internal control. The RNA copy numbers of proinflammatory cytokines and lipogenic genes were expressed as a ratio to β-actin RNA. The absolute copy number of viral gRNA was determined according to a previous study [29]. The qRT-PCR experiments were performed using the CFX96 Real-Time PCR system (Bio-Rad). Each assay was performed in triplicate.
Table 1.
Sequences of the primers used for qRT-PCR
| Name | Primer sequence (5’ to 3’) |
|---|---|
| IL-1β | Forward: GTTAATGATGAAGATGTTGATAGC |
| Reverse: GTTCCAGACACAGCAATC | |
| IL-6 | Forward: AGGACGAGATGTGCAAGAAG |
| Reverse: TGCTGTAGCACAGAGACTCG | |
| IL-8 | Forward: GCATTAGCACTCATTCTA |
| Reverse: CTCTCTTCCATCCTTTAG | |
| IL-10 | Forward: TGAACTTAACATCCAACTG |
| Reverse: ACCTCATCTGTGTAGAAG | |
| IFN-β | Forward: CTCGCAACCTTCACCTCAC |
| Reverse: CGCTGTAATCGTTGTCTTGGA | |
| ABCA1 | Forward: CTCATTACTGTGCTCATC |
| Reverse: CGTCTTCATCTTCATCATT | |
| LXR-α | Forward: CTCACTTCACAGAACTTG |
| Reverse: CTATGGTAGATGTCTTCAATA | |
| SREBP-1C | Forward: ACTTCATCAAGGCAGACT |
| Reverse: TGATGCTGGAGGTCTTAG | |
| PPAR-γ | Forward: AATGCCATCAGGTTTGGG |
| Reverse: GCGGGAAGGACTTTATGTATG | |
| FASN | Forward: AAGAGGAGAAGCAATATC |
| Reverse: CTGTGATGATGTAAGACT | |
| β-ACTIN | Forward: CTTCCAGCCATCTTTCTT |
| Reverse: ATATCCACATCACACTTCAT | |
| HMGCR | Forward: CTGGGTTTGGTTCTTGTTCA |
| Reverse: ATTCGGTCTCTGCTTGTTCA | |
| PLa-G (viral gRNA) | ACGATAAAAGGCGAAGGAGCA |
Cytokine ELISA
DF-1 cells were incubated with GW3965 (1 μM) during NDV infection. The supernatant was collected at 12 hpi. The concentrations of IL-1β, IL-6, IL-8, IFN-β, and TNF-α were determined using an enzyme-linked immunosorbent assay kit (Elabscience, China) according to the manufacturer’s instructions.
MTT assay
DF-1 cells cultured in a 96-well plate were treated with various concentrations of GW3965 (0, 0.1, 0.5, 1, 2, and 5 μM) for 24 h. The cells were washed. One hundred μl of DMEM containing 10 μl MTT (5 mg/ml) was added to each well. The plate was incubated for 4 h at 37 °C. The culture medium was removed, and 100 μl DMSO was added to each well. The plate was gently shaken for 10 min. The absorbance at 490 nm was determined by a spectrophotometer (Bio-Rad).
Cholesterol quantification
The cells were washed twice with PBS. The total cellular cholesterol was assayed using Tissue Total Cholesterol Assay Kit (Applygen, China) according to the manufacturer’s instructions.
Filipin III staining
DF-1 cells were grown on coverslips overnight and pretreated with GW3965 (1 μM) for 1 h. The cells were then infected with NDV (MOI = 1) in the presence of GW3965 (1 μM) for 12 h. The cells were fixed in 3 % paraformaldehyde and permeabilized with 0.5 % Triton X-100 for 10 min, followed by incubation in blocking buffer (5 % BSA in PBS buffer) for 30 min. The cells were then stained with filipin III (0.05 mg/mL) for 2 h at room temperature. The coverslips were mounted on slide glasses and observed under a fluorescence microscope (Nikon).
Statistical analysis
All data were determined in triplicate and are representative of at least two separate experiments. The results represent the means ± standard deviations of triplicate determination. The differences between means were considered significant at *p < 0.05 and very significant at **p < 0.01. All statistical analyses were performed by one-way ANOVA using the SPSS 16.0 software package.
Results
Synthetic LXR agonist GW3965 impedes NDV infection
Liver X receptors (LXR-α and LXR-β) are ligand-activated transcription factors that regulate host lipid metabolism and cholesterol homeostasis [15]. GW3965, a synthetic ligand, can effectively increase the expression of LXR-α and LXR-β [2, 41]. A study on HCV showed that GW3965 possesses an antiviral effect [44]. We therefore hypothesized that GW3965 might inhibit NDV infection. The cytotoxicity experiment showed that GW3965 did not cause significant cytotoxic effects in DF-1 cells at 0.1, 0.5, and 1 μM (Fig. 1A). We next explored the anti-NDV activity of GW3965 in DF-1 cells. The infected cells were incubated with different concentrations of GW3965 for 12h. The inhibitory effect of GW3965 was assessed by western blot, which demonstrated that GW3965 could significantly reduce the expression level of viral protein NP (Fig. 1B).
Fig. 1.
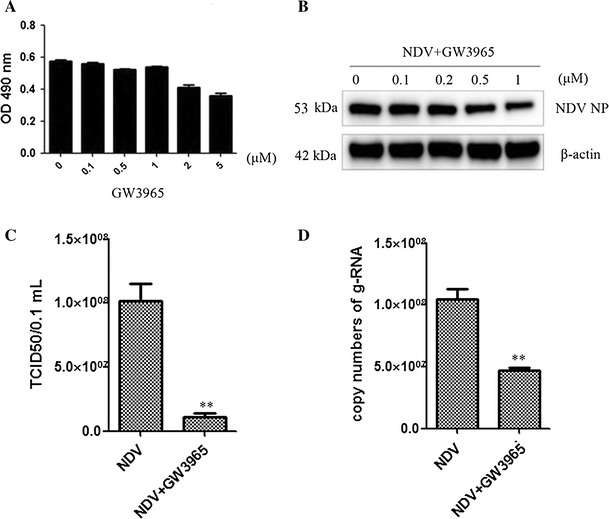
The effects of GW3965 on NDV infection. (A) The DF-1 cells were cultured with different concentrations of GW3965. Cell viability was determined by MTT assay at 24 h. (B) Synthetic LXR agonist GW3965 inhibited NDV infection in DF-1 cells. Cells were pretreated with GW3965 (1 μM) for 2 h, then infected with NDV (MOI = 1). GW3965 (1 μM) was continuously present during the infection. The expression level of the viral NP protein was determined by western blot. (C) The titers of the supernatant were determined by TCID50. (D) The viral gRNA in cells was detected by qRT-PCR assay
To confirm the inhibitory effect of GW3965 on NDV infection, we also analyzed the virus titer in the supernatant via TCID50 and viral genomic RNA (gRNA) in the cells via qRT-PCR. The virus titer was decreased in the GW3965-treated group compared to the control (Fig.1C). The viral gRNA copy number was also reduced significantly in proportion to the amount of GW3965 (Fig.1D). In summary, our results revealed that GW3965 had an inhibitory effect on NDV infection.
GW3965 inhibits NDV entry and replication
To explore whether GW3965 inhibited NDV attachment, we used qRT-PCR to detect the viral gRNA copy number. DF-1 cells were incubated with NDV (MOI = 1) at 4 °C for 1 h in the presence of GW3965 (1 μM). The cells were then collected and viral gRNA was prepared. The qRT-PCR assay showed that the viral gRNA copy number was not changed significantly, indicating that GW3965 treatment might not affect NDV attachment (Fig. 2A).
Fig. 2.
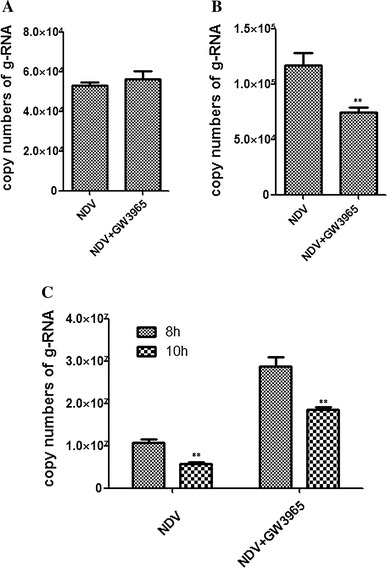
GW3965 affects NDV entry and replication. (A) GW3965 did not affect NDV attachment. DF-1 cells were incubated with NDV (MOI = 1) at 4 °C for 1 h in the presence of GW3965 (1 μM). The viral gRNA copy number was determined by qRT-PCR assay. (B) GW3965 inhibited NDV entry. DF-1 cells were first incubated with NDV (MOI = 1) at 4 °C for 1 h and were then cultured at 37 °C in the presence of GW3965 (1 μM) for 1 h. The cells were washed three times. The viral gRNA copy number was determined by qRT-PCR. (C) GW3965 inhibited NDV replication. After infected with NDV (MOI = 1), DF-1 cells were incubated with GW3965 (1 μM). Viral gRNA copy numbers were determined by qRT-PCR at 8 hpi and 10 hpi
We next determined whether GW3965 inhibited NDV entry. DF-1 cells were first incubated with NDV (MOI = 1) at 4 °C for 1 h and were then cultured at 37 °C in the presence of GW3965 (1 μM) for 1 h. The qRT-PCR result revealed that viral gRNA copy number was decreased significantly, indicating that GW3965 treatment reduced NDV entry (Fig. 2B).
To explore whether GW3965 inhibited NDV replication, the cells were treated with GW3965 after NDV infection and collected at 8 hpi and 10 hpi. The qRT-PCR result showed that viral gRNA copy numbers were decreased significantly at both time points, suggesting that GW3965 treatment also impaired the replication of NDV (Fig. 2C).
In summary, our results indicated that GW3965 affected NDV entry and replication, but not attachment. Due to technical limitations, we did not explore the effect of GW3965 on virus assembly and budding.
GW3965 inhibits NF-κB activation and decreases pro-inflammatory cytokine expression induced by infection
NF-κB, a multi-subunit nuclear transcription factor, regulates the transcription of various cytokines and host immune responses [7, 18, 26]. In quiescent cells, NF-κB is bound to its inhibitor IκB in the cytoplasm. Once stimulated, IκBα is phosphorylated and degraded. NF-κB is released and translocated into the nucleolus to regulate gene transcription. To explore the effect of GW3965 on the NF-κB signaling pathway in virus infection, the protein levels of IκBα, p65, phosphor-IκBα and phosphor-p65 were determined by Western blot. The results indicated that GW3965 suppressed the degradation of IκBα and the activation of NF-κB in a dose-dependent manner (Fig. 3).
Fig. 3.
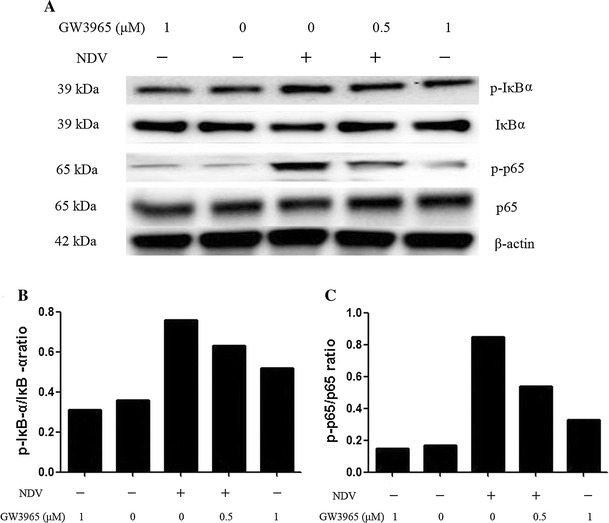
GW3965 inhibits NDV-induced NF-κB activation. (A) Cells were infected with NDV (MOI = 1) in the absence or presence of GW3965 (0.1, 0.5 and 1 μM) for 12 h. The protein levels of the NF-κB pathway were analyzed by Western blot with specific antibodies. β-actin was used as the loading control. (B and C) Quantifications of Western blots were performed with ImageJ software
NDV infection induces high-level expression of pro-inflammatory cytokines in many cells, which may contribute to inflammation responses and the pathogenesis of virus infection [8]. Many studies demonstrate that GW3965 exhibits a potent anti-inflammatory effect on the pathogenesis of various diseases by modulating the production of inflammatory cytokines [10, 45]. To explore whether GW3965 has an anti-inflammatory effect in DF-1 cells, we detected the expression of proinflammatory cytokines in infected cells. The qRT-PCR results showed that GW3965 significantly (p < 0.05) reduced the gene expression of IL-1β, IL-6, IL-8, TNF-α and IFN-β induced by NDV infection at 12 h compared to DMSO-treated cells (Fig. 4A-E). The concentrations of inflammatory cytokines in the supernatant were determined by ELISA. The result showed that GW3965 treatment also decreased the protein expression levels of inflammatory cytokines during NDV infection (Fig. 4F-J).
Fig. 4.
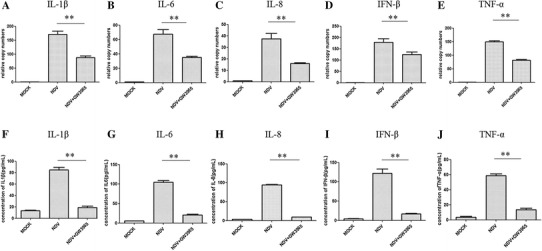
GW3965 reduces mRNA and protein expression levels of the proinflammatory cytokines in infected DF-1 cells. DF-1 cells were infected by NDV (MOI = 1) and incubated with GW3965 (1 μM). The cells were collected at 12 h post-treatment. The relative expression level of (A) IL-1β, (B) IL-6, (C) IL-8, (D) TNF-α and (E) IFN-β was determined by qRT-PCR. Values were normalized to internal control β-actin. The protein concentrations of (F) IL-1β, (G) IL-6, (H) IL-8, (I) IFN-β and (J) TNF-α in the supernatant were determined by ELISA assay
NDV infection increases LXR-α and its downstream lipogenic gene expression but decreases ABCA1 gene expression
To clarify whether NDV infection affected the expression of lipogenic genes, we examined the mRNA levels of these genes in DF-1 cells during the infection. We observed a significant induction of LXR-α gene expression in DF-1 cells compared with control cells. Significant upregulation of sterol regulatory element binding protein-1 (SREBP-1), peroxisome proliferator-activated receptor-γ1 (PPAR-γ1), and fatty acid synthase (FASN) genes were also observed (Fig. 5A) at different time points. Unexpectedly, the gene expression of ATP-binding cassette transporter A1 (ABCA1) was decreased during the infection (Fig. 5B). These results suggested that NDV infection disturbed host lipid metabolism. The upregulation of the lipogenic gene expression would provide the essential materials for virus propagation.
Fig. 5.
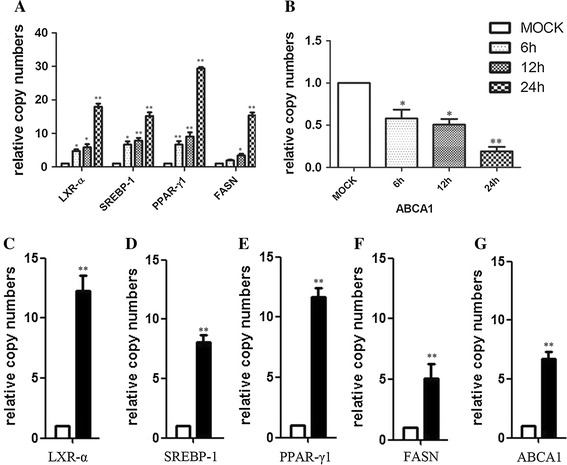
NDV infection disturbs LXR and its downstream lipogenic gene expression. (A-B) The effect of the NDV infection on the expression of lipogenic genes. DF-1 cells were infected with NDV (MOI = 1). Total RNA was extracted at 6, 12, and 24 hpi. The mRNA levels of LXR-α, SREBP-1, PPAR-γ1, FASN, and ABCA1 were determined. (C-G) The effect of GW3965 on the expression of lipogenic genes in infected DF-1 cells. DF-1 cells were infected with NDV (MOI = 1) in the presence or absence of GW3695 (1 μM) for 12 h. The relative mRNA levels of LXR-α, SREBP-1, PPAR-γ1, FASN, and ABCA1 were determined by qRT-PCR. The RNA copy number in NDV-infected cells was considered to be 1
GW3965 enhances LXR-α and its related lipogenic gene expression, including ABCA1
GW3965 treatment enhances the expression of LXRs and other lipogenic genes involved in lipid metabolic pathways. We next investigated the mRNA levels of lipogenic genes in NDV-infected cells that were treated with GW3965. The transcriptomic profile of treated cells revealed that GW3965 increased the mRNA levels of these genes in infected cells: LXRα (12.3-fold), SREBP-1 (8.1-fold), PPAR-γ1 (11.7-fold), FASN (6.3-fold), and ABCA1 (5.1-fold) (Fig. 5C-G).
ABCA1 is involved in the inhibition of NDV infection by regulating cholesterol
ABCA1 has been functionally implicated in cholesterol transport. ABCA1 is localized to the plasma membrane and intracellular compartments [17, 28]. As cholesterol is essential for virus entry, replication, and production, we hypothesized that ABCA1 might play an important role in the inhibition of NDV infection by regulating cholesterol efflux. To explore the function of ABCA1 in NDV infection, we overexpressed EGFP-ABCA1 (kindly provided by Prof. Pelkmans, University of Zurich) in DF-1 cells, followed by NDV infection. The western blot analysis showed that the NP protein expression level was significantly decreased in ABCA1-overexpressing cells (Fig. 6A, lane 2), whereas the virus infectivity could be restored when cholesterol was replenished (Fig. 6A, lane 3). The expression of EGFP and EGFP-ABCA1 were confirmed by western blot analysis (Fig. 6A). We also determined the cellular cholesterol content in the infected cells and in the infected cells with overexpressed ABCA1. The data demonstrated that the cholesterol content was not changed significantly in virus-infected cells, but overexpression of ABCA1 decreased the cholesterol content in both mock-infected and virus-infected cells (Fig. 6B). We found that cholesterol replenishment could attenuate the inhibitory effect of GW3965 on virus infectivity (Fig. 6C, lane 2 and 3). GW3965 also decreased the cholesterol content in both mock-infected and NDV-infected cells (Fig. 6D). The mRNA level of HMG-CoA reductase (HMGCR), a rate-limiting enzyme for cholesterol synthesis, was determined by qRT-PCR. The result illustrated that the gene expression of HMGCR was not changed significantly in infected cells (Fig. 6E).
Fig. 6.
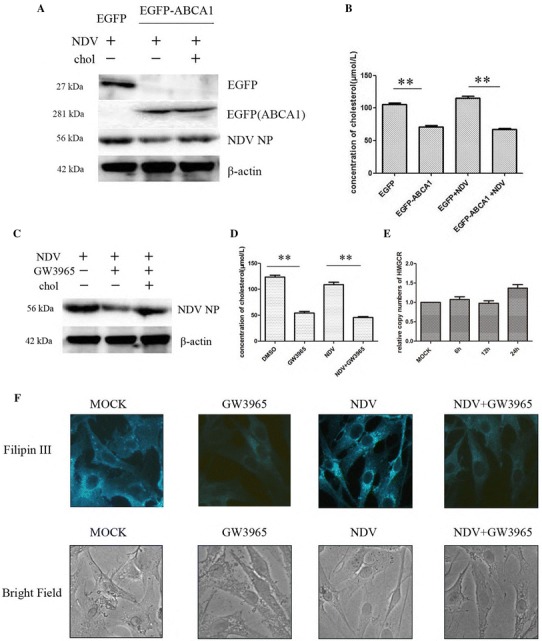
ABCA1 is involved in the inhibition of NDV infection. (A) Overexpression of ABCA1 inhibited NDV infection, whereas cholesterol replenishment restored NDV infectivity. DF-1 cells were transfected with EGFP (lane 1) or EGFP-ABCA1 (lane 2) for 24 h before the infection. The EGFP-ABCA1 over-expressing cells were incubated with 400 μg/ml cholesterol during the infection (lane 3). NDV-NP protein expression levels were determined at 12 hpi by Western blot. (B) Cholesterol contents were determined in mock-infected and NDV-infected cells that were previously transfected with EGFP or EGFP-ABCA1 at 12 hpi. (C) Cholesterol replenishment restored NDV infectivity in GW3965 (1 μM)-treated cells at 12 hpi. (D) Cholesterol contents were determined in mock-infected and NDV-infected cells (treated and non-treated by 1 μM GW3965) at 12 hpi. GW3965 treatment reduced the cholesterol content in DF-1 cells. (E) The cellular HMGCR mRNA level was determined by qRT-PCR in infected cells at different time points. (F) DF-1 cells were either mock-infected or infected with NDV (MOI = 1) in the presence or absence of GW3965. The cells were then fixed and stained with filipin III (0.05 mg/mL) at 12 hpi
We stained the mock-infected and virus-infected cells with filipin III to determine the distribution of cellular cholesterol. The results demonstrated that cholesterol accumulated in the perinuclear area of the infected cells compared to mock-infected cells (Fig. 6F), which was similar to the result in a previous study on West Nile virus [22]. GW3965 treatment decreased the cholesterol content in both mock-infected cells and NDV-infected cells.
Based on above results, we suspected that the anti-NDV effect of GW3965 might be associated with its ability to reduce the cholesterol content in DF-1 cells.
Discussion
NDV is a fatal pathogen for birds, causing high mortality in the poultry industry. Current control strategies against NDV are still inadequate. As obligate intracellular pathogens, viruses need to exploit cellular materials to facilitate the replication. Viruses can manipulate multiple metabolic pathways, including lipid metabolism [42]. Lipids are essential components in virus infection. During the infection, some viruses induce changes in cell membrane structures or utilize lipid synthetic enzymes to build a suitable microenvironment for infection at different infection stages [20].
Liver X receptors (LXRs) are nuclear receptors that are ubiquitously expressed in various tissues [1, 34, 36, 40]. LXRs play critical roles in regulating lipid metabolism. Several studies report that LXR is a promising target for antiviral drug development. GW3965 is a widely used ligand that specifically activates LXR-α and LXR-β in many cell lines. Our results showed that GW3965 inhibited NDV propagation in DF-1 cells. It decreased the viral protein expression level and viral gRNA level during the infection. Meanwhile, GW3965 treatment reduced the mRNA and protein expression levels of inflammatory cytokines (including IL-1β, IL-6, IL-8, IFN-β, and TNF-α) in infected DF-1 cells. GW3965 treatment also significantly induced LXR-α and its downstream lipogenic gene expression (SREBP-1, PPAR-γ1, ABCA1 and FASN) in infected DF-1 cells.
We found that NDV infection upregulated the expression of LXR-α and its downstream genes but impaired ABCA1 expression, which brought to our attention the function of ABCA1 in NDV infection. Cholesterol is an essential component of cellular membranes and is also vital for the integrity of various viruses, since the envelope membranes of these viruses are derived from the host membranes [12]. Generally, cells upregulate ABCA1 expression to promote cholesterol efflux and reduce cholesterol influx when the intracellular cholesterol concentration is increased [3]. To investigate the function of ABCA1 in NDV infection, we overexpressed ABCA1 in DF-1 cells. The expression level of NP protein was decreased significantly in ABCA1 overexpressing cells. Cellular cholesterol distribution was determined by filipin III staining, which showed that NDV infection caused the accumulation of cholesterol in the perinuclear area of the infected cells. Meanwhile, cholesterol content assay and qRT-PCR analysis on HMGCR mRNA showed that NDV infection did not increase cholesterol synthesis, implying that NDV might change cholesterol distribution to benefit the infection.
Our studies suggested that GW3965 inhibited NDV entry and replication. GW3965 treatment increases the expression of ABCA1. The content of cholesterol was decreased in GW3965-treated or ABCA1 over-expressing cells because of cholesterol efflux. Considering its regulation of cholesterol efflux, we suspected that ABCA1 might promote cholesterol efflux and affect cholesterol content in the cell membrane that was required for the optimal cell entry of NDV [23]. Several investigations have strongly suggested that lipids, including cholesterol and phospholipids, play critical roles in virus replication. The reason for the inhibitory effect of GW3965 on NDV replication might be multifaceted. One possible explanation is that cholesterol might be vital for producing an optimal lipid environment for virus replication, which could be disrupted by cholesterol depletion after ABCA1 was upregulated by GW3965 treatment. The accumulation of cholesterol in NDV-infected cells partially supported this possibility. In our studies, we did not determine the effect of GW3965 on virus assembly and budding due to the lack of a reliable method. It was shown that lipid rafts are important for ordered assembly and budding of infectious Newcastle disease virus particles [16]. Therefore, we suspected that GW3965 might affect NDV assembly and release, since cholesterol is an important component of lipid rafts.
HCV infection induces intracellular lipid accumulation and lipogenic gene expression, which is mediated by LXR. Viral NS5A protein, core protein, and viral replication increase LXR-α and its downstream lipogenic gene expression. Viral NS5A and core proteins do not interact with LXR-α, implying that they indirectly increase the transcriptional activity of LXR-α. Further study indicates that viral proteins and viral replication mediate LXR-α expression through the PI3K/AKT signaling pathway [9]. Nef, an accessory protein of HIV, inhibits the activity of ABCA1 in infected macrophages, which could suppress cholesterol efflux and result in the accumulation of intracellular cholesterol. Nef downregulates ABCA1 expression, which requires interaction between Nef and ABCA1 [25]. A new study revealed that Nef disrupts the interaction between the ER chaperone calnexin and ABCA1, which is essential for the functionality of ABCA1 [13]. Activation of Toll-like receptors 3 and 4 by E. coli and influenza virus infection blocks the induction of ABCA1 in culture macrophages and aortic tissue and therefore inhibits cholesterol efflux. Further experiments showed that the crosstalk between LXR and TLR signaling is mediated by IRF3, independent of the MyD-88-mediated pathway [4]. A new study demonstrated that the enhanced TLR4 signaling downregulates ABCA1 expression in cultured WT VSMCs, which is mediated by IRAK1 [11]. The previous study showed that the TLR3 signaling pathway can be induced by NDV infection [6]. We also observed the increased TLR4 gene expression during NDV infection (data not shown), implying the activation of the TLR4 signaling pathway. Further investigations are necessary to determine whether or how NDV proteins affect LXRs or ABCA1 gene expression. Furthermore, the signaling pathways that are involved in the induction of LXRs and downstream gene expression are worth further exploration.
In summary, our studies indicated that the LXR ligand GW3965 possesses anti-NDV activity. GW3965 treatment reduced NF-κB activation and decreased the upregulation of various proinflammatory cytokines to alleviate host immune response stresses. Treatment with GW3965 restored the expression of ABCA1, which could reduce the cellular cholesterol content. GW3965 regulated LXRs and expression of its downstream lipogenic genes. The mechanisms underlying the antiviral activity of GW3965 might be complex. Nevertheless, cholesterol efflux caused by GW3965 treatment might play an important role in inhibiting NDV infection.
Acknowledgments
This work was funded by the Agricultural Science and Technology Innovation Program (ASTIP) of Chinese Academy of Agricultural Sciences.
Contributor Information
Chan Ding, Email: shoveldeen@shvri.ac.cn.
Xiang Mao, Email: xmao@njau.edu.cn, Email: xmao@shvri.ac.cn.
References
- 1.Apfel R, Benbrook D, Lernhardt E, Ortiz MA, Salbert G, Pfahl M. A novel orphan receptor specific for a subset of thyroid hormone-responsive elements and its interaction with the retinoid/thyroid hormone receptor subfamily. Mol Cell Biol. 1994;14:7025–7035. doi: 10.1128/MCB.14.10.7025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bocchetta S, Maillard P, Yamamoto M, Gondeau C, Douam F, Lebreton S, Lagaye S, Pol S, Helle F, Plengpanich W, Guerin M, Bourgine M, Michel ML, Lavillette D, Roingeard P, le Goff W, Budkowska A. Up-regulation of the ATP-binding cassette transporter A1 inhibits hepatitis C virus infection. PLoS One. 2014;9:e92140. doi: 10.1371/journal.pone.0092140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bovenga F, Sabba C, Moschetta A. Uncoupling nuclear receptor LXR and cholesterol metabolism in cancer. Cell Metab. 2015;21:517–526. doi: 10.1016/j.cmet.2015.03.002. [DOI] [PubMed] [Google Scholar]
- 4.Castrillo A, Joseph SB, Vaidya SA, Haberland M, Fogelman AM, Cheng G, Tontonoz P. Crosstalk between LXR and toll-like receptor signaling mediates bacterial and viral antagonism of cholesterol metabolism. Mol Cell. 2003;12:805–816. doi: 10.1016/S1097-2765(03)00384-8. [DOI] [PubMed] [Google Scholar]
- 5.Chang YC, Lee TS, Chiang AN. Quercetin enhances ABCA1 expression and cholesterol efflux through a p38-dependent pathway in macrophages. J Lipid Res. 2012;53:1840–1850. doi: 10.1194/jlr.M024471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cheng J, Sun Y, Zhang X, Zhang F, Zhang S, Yu S, Qiu X, Tan L, Song C, Gao S, Wu Y, Ding C. Toll-like receptor 3 inhibits Newcastle disease virus replication through activation of pro-inflammatory cytokines and the type-1 interferon pathway. Arch Virol. 2014;159:2937–2948. doi: 10.1007/s00705-014-2148-6. [DOI] [PubMed] [Google Scholar]
- 7.Fitzpatrick SF, Tambuwala MM, Bruning U, Schaible B, Scholz CC, Byrne A, O’Connor A, Gallagher WM, Lenihan CR, Garvey JF, Howell K, Fallon PG, Cummins EP, Taylor CT. An intact canonical NF-kappaB pathway is required for inflammatory gene expression in response to hypoxia. J Immunol. 2011;186:1091–1096. doi: 10.4049/jimmunol.1002256. [DOI] [PubMed] [Google Scholar]
- 8.Fournier P, Arnold A, Wilden H, Schirrmacher V. Newcastle disease virus induces pro-inflammatory conditions and type I interferon for counter-acting Treg activity. Int J Oncol. 2012;40:840–850. doi: 10.3892/ijo.2011.1265. [DOI] [PubMed] [Google Scholar]
- 9.Garcia-Mediavilla MV, Pisonero-Vaquero S, Lima-Cabello E, Benedicto I, Majano PL, Jorquera F, Gonzalez-Gallego J, Sanchez-Campos S. Liver X receptor alpha-mediated regulation of lipogenesis by core and NS5A proteins contributes to HCV-induced liver steatosis and HCV replication. Lab Invest. 2012;92:1191–1202. doi: 10.1038/labinvest.2012.88. [DOI] [PubMed] [Google Scholar]
- 10.Guillem-Llobat P, Iniguez MA. Inhibition of lipopolysaccharide-induced gene expression by liver X receptor ligands in macrophages involves interference with early growth response factor 1. Prostaglandins Leukot Essent Fatty Acids. 2015;96:37–49. doi: 10.1016/j.plefa.2015.02.002. [DOI] [PubMed] [Google Scholar]
- 11.Guo L, Chen CH, Zhang LL, Cao XJ, Ma QL, Deng P, Zhu G, Gao CY, Li BH, Pi Y, Liu Y, Hu ZC, Zhang L, Yu ZP, Zhou Z, Li JC. IRAK1 mediates TLR4-induced ABCA1 downregulation and lipid accumulation in VSMCs. Cell Death Dis. 2015;6:e1949. doi: 10.1038/cddis.2015.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hotta K, Bazartseren B, Kaku Y, Noguchi A, Okutani A, Inoue S, Yamada A. Effect of cellular cholesterol depletion on rabies virus infection. Virus Res. 2009;139:85–90. doi: 10.1016/j.virusres.2008.10.009. [DOI] [PubMed] [Google Scholar]
- 13.Jennelle L, Hunegnaw R, Dubrovsky L, Pushkarsky T, Fitzgerald ML, Sviridov D, Popratiloff A, Brichacek B, Bukrinsky M. HIV-1 protein Nef inhibits activity of ATP-binding cassette transporter A1 by targeting endoplasmic reticulum chaperone calnexin. J Biol Chem. 2014;289:28870–28884. doi: 10.1074/jbc.M114.583591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Jiang H, Badralmaa Y, Yang J, Lempicki R, Hazen A, Natarajan V. Retinoic acid and liver X receptor agonist synergistically inhibit HIV infection in CD4+ T cells by up-regulating ABCA1-mediated cholesterol efflux. Lipids Health Dis. 2012;11:69. doi: 10.1186/1476-511X-11-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Karagianni P, Talianidis I. Transcription factor networks regulating hepatic fatty acid metabolism. Biochim Biophys Acta. 2015;1851:2–8. doi: 10.1016/j.bbalip.2014.05.001. [DOI] [PubMed] [Google Scholar]
- 16.Laliberte JP, McGinnes LW, Peeples ME, Morrison TG. Integrity of membrane lipid rafts is necessary for the ordered assembly and release of infectious Newcastle disease virus particles. J Virol. 2006;80:10652–10662. doi: 10.1128/JVI.01183-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lawn RM, Wade DP, Garvin MR, Wang X, Schwartz K, Porter JG, Seilhamer JJ, Vaughan AM, Oram JF. The Tangier disease gene product ABC1 controls the cellular apolipoprotein-mediated lipid removal pathway. J Clin Invest. 1999;104:R25–R31. doi: 10.1172/JCI8119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li W, Khor TO, Xu C, Shen G, Jeong WS, Yu S, Kong AN. Activation of Nrf2-antioxidant signaling attenuates NFkappaB-inflammatory response and elicits apoptosis. Biochem Pharmacol. 2008;76:1485–1489. doi: 10.1016/j.bcp.2008.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Linde AM, Munir M, Zohari S, Stahl K, Baule C, Renstrom L, Berg M. Complete genome characterisation of a Newcastle disease virus isolated during an outbreak in Sweden in 1997. Virus Genes. 2010;41:165–173. doi: 10.1007/s11262-010-0498-z. [DOI] [PubMed] [Google Scholar]
- 20.Lorizate M, Krausslich HG. Role of lipids in virus replication. Cold Spring Harbor Perspect Biol. 2011;3:a004820. doi: 10.1101/cshperspect.a004820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lu Y, Liu DX, Tam JP. Lipid rafts are involved in SARS-CoV entry into Vero E6 cells. Biochem Biophys Res Commun. 2008;369:344–349. doi: 10.1016/j.bbrc.2008.02.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mackenzie JM, Khromykh AA, Parton RG. Cholesterol manipulation by West Nile virus perturbs the cellular immune response. Cell Host Microbe. 2007;2:229–239. doi: 10.1016/j.chom.2007.09.003. [DOI] [PubMed] [Google Scholar]
- 23.Martin JJ, Holguera J, Sanchez-Felipe L, Villar E, Munoz-Barroso I. Cholesterol dependence of Newcastle Disease Virus entry. Biochim Biophys Acta. 2012;1818:753–761. doi: 10.1016/j.bbamem.2011.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Miller PJ, Kim LM, Ip HS, Afonso CL. Evolutionary dynamics of Newcastle disease virus. Virology. 2009;391:64–72. doi: 10.1016/j.virol.2009.05.033. [DOI] [PubMed] [Google Scholar]
- 25.Mujawar Z, Rose H, Morrow MP, Pushkarsky T, Dubrovsky L, Mukhamedova N, Fu Y, Dart A, Orenstein JM, Bobryshev YV, Bukrinsky M, Sviridov D. Human immunodeficiency virus impairs reverse cholesterol transport from macrophages. PLoS Biol. 2006;4:e365. doi: 10.1371/journal.pbio.0040365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Muzio M, Natoli G, Saccani S, Levrero M, Mantovani A. The human toll signaling pathway: divergence of nuclear factor kappaB and JNK/SAPK activation upstream of tumor necrosis factor receptor-associated factor 6 (TRAF6) J Exp Med. 1998;187:2097–2101. doi: 10.1084/jem.187.12.2097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nelson CB, Pomeroy BS, Schrall K, Park WE, Lindeman RJ. An outbreak of conjunctivitis due to Newcastle disease virus (NDV) occurring in poultry workers. Am J Public Health Nation’s Health. 1952;42:672–678. doi: 10.2105/AJPH.42.6.672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Neufeld EB, Remaley AT, Demosky SJ, Stonik JA, Cooney AM, Comly M, Dwyer NK, Zhang M, Blanchette-Mackie J, Santamarina-Fojo S, Brewer HB., Jr Cellular localization and trafficking of the human ABCA1 transporter. J Biol Chem. 2001;276:27584–27590. doi: 10.1074/jbc.M103264200. [DOI] [PubMed] [Google Scholar]
- 29.Qiu X, Yu Y, Yu S, Zhan Y, Wei N, Song C, Sun Y, Tan L, Ding C. Development of strand-specific real-time RT-PCR to distinguish viral RNAs during Newcastle disease virus infection. Sci World J. 2014;2014:934851. doi: 10.1155/2014/934851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ramezani A, Dubrovsky L, Pushkarsky T, Sviridov D, Karandish S, Raj DS, Fitzgerald ML, Bukrinsky M. Stimulation of liver X receptor has potent anti-HIV effects in a humanized mouse model of HIV infection. J Pharmacol Exp Ther. 2015;354:376–383. doi: 10.1124/jpet.115.224485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Reed LJ, Muench H. A simple method of estimating fifty percent endpoints. Am J Hyg. 1938;27:493–497. [Google Scholar]
- 32.Renli Q, Chao S, Jun Y, Chan S, Yunfei X. Changes in fat metabolism of black-bone chickens during early stages of infection with Newcastle disease virus. Animal. 2012;6:1246–1252. doi: 10.1017/S1751731112000365. [DOI] [PubMed] [Google Scholar]
- 33.Repa JJ, Liang G, Ou J, Bashmakov Y, Lobaccaro JM, Shimomura I, Shan B, Brown MS, Goldstein JL, Mangelsdorf DJ. Regulation of mouse sterol regulatory element-binding protein-1c gene (SREBP-1c) by oxysterol receptors, LXRalpha and LXRbeta. Genes Dev. 2000;14:2819–2830. doi: 10.1101/gad.844900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shinar DM, Endo N, Rutledge SJ, Vogel R, Rodan GA, Schmidt A. NER, a new member of the gene family encoding the human steroid hormone nuclear receptor. Gene. 1994;147:273–276. doi: 10.1016/0378-1119(94)90080-9. [DOI] [PubMed] [Google Scholar]
- 35.Simons K, Ikonen E. Functional rafts in cell membranes. Nature. 1997;387:569–572. doi: 10.1038/42408. [DOI] [PubMed] [Google Scholar]
- 36.Song C, Kokontis JM, Hiipakka RA, Liao S. Ubiquitous receptor: a receptor that modulates gene activation by retinoic acid and thyroid hormone receptors. Proc Natl Acad Sci USA. 1994;91:10809–10813. doi: 10.1073/pnas.91.23.10809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Syed GH, Amako Y, Siddiqui A. Hepatitis C virus hijacks host lipid metabolism. Trends Endocrinol Metab. 2010;21:33–40. doi: 10.1016/j.tem.2009.07.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Venkateswaran A, Laffitte BA, Joseph SB, Mak PA, Wilpitz DC, Edwards PA, Tontonoz P. Control of cellular cholesterol efflux by the nuclear oxysterol receptor LXR alpha. Proc Natl Acad Sci USA. 2000;97:12097–12102. doi: 10.1073/pnas.200367697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wilde A, McQuain C, Morrison T. Identification of the sequence content of four polycistronic transcripts synthesized in Newcastle disease virus infected cells. Virus Res. 1986;5:77–95. doi: 10.1016/0168-1702(86)90067-5. [DOI] [PubMed] [Google Scholar]
- 40.Willy PJ, Umesono K, Ong ES, Evans RM, Heyman RA, Mangelsdorf DJ. LXR, a nuclear receptor that defines a distinct retinoid response pathway. Genes Dev. 1995;9:1033–1045. doi: 10.1101/gad.9.9.1033. [DOI] [PubMed] [Google Scholar]
- 41.Wu Y, Yu DD, Hu Y, Cao HX, Yu SR, Liu SW, Feng JF. LXR ligands sensitize EGFR-TKI-resistant human lung cancer cells in vitro by inhibiting Akt activation. Biochem Biophys Res Commun. 2015;467:900–905. doi: 10.1016/j.bbrc.2015.10.047. [DOI] [PubMed] [Google Scholar]
- 42.Ye J. Reliance of host cholesterol metabolic pathways for the life cycle of hepatitis C virus. PLoS Pathog. 2007;3:e108. doi: 10.1371/journal.ppat.0030108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yusoff K, Tan WS. Newcastle disease virus: macromolecules and opportunities. Avian Pathol. 2001;30:439–455. doi: 10.1080/03079450120078626. [DOI] [PubMed] [Google Scholar]
- 44.Zeng J, Wu Y, Liao Q, Li L, Chen X, Chen X. Liver X receptors agonists impede hepatitis C virus infection in an Idol-dependent manner. Antiviral Res. 2012;95:245–256. doi: 10.1016/j.antiviral.2012.06.004. [DOI] [PubMed] [Google Scholar]
- 45.Zhang XQ, Even-Or O, Xu X, van Rosmalen M, Lim L, Gadde S, Farokhzad OC, Fisher EA. Nanoparticles containing a liver X receptor agonist inhibit inflammation and atherosclerosis. Adv Healthc Mater. 2015;4:228–236. doi: 10.1002/adhm.201400337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhu YZ, Cao MM, Wang WB, Wang W, Ren H, Zhao P, Qi ZT. Association of heat-shock protein 70 with lipid rafts is required for Japanese encephalitis virus infection in Huh7 cells. J Gen Virol. 2012;93:61–71. doi: 10.1099/vir.0.034637-0. [DOI] [PubMed] [Google Scholar]


