Abstract
Vanadium is a transition metal that, being ubiquitously distributed in soil, crude oil, water and air, also found roles in biological systems and is an essential element in most living beings. There are also several groups of organisms which accumulate vanadium, employing it in their biological processes. Vanadium being a biological relevant element, it is not surprising that many vanadium based therapeutic drugs have been proposed for the treatment of several types of diseases. Namely, vanadium compounds, in particular organic derivatives, have been proposed for the treatment of diabetes, of cancer and of diseases caused by parasites. In this work we review the medicinal applications proposed for vanadium compounds with particular emphasis on the more recent publications. In cells, partly due to the similarity of vanadate and phosphate, vanadium compounds activate numerous signaling pathways and transcription factors; this by itself potentiates application of vanadium-based therapeutics. Nevertheless, this non-specific bio-activity may also introduce several deleterious side effects as in addition, due to Fenton's type reactions or of the reaction with atmospheric O2, VCs may also generate reactive oxygen species, thereby introducing oxidative stress with consequences presently not well evaluated, particularly for long-term administration of vanadium to humans. Notwithstanding, the potential of vanadium compounds to treat type 2 diabetes is still an open question and therapies using vanadium compounds for e.g. antitumor and anti-parasitic related diseases remain promising.
Keywords: Vanadium, Biological properties, Cancer therapy, Insulin-enhancing agents, Antiparasitic activity
1. Introduction
Progress in the chemistry of vanadium, namely in the search of its therapeutic applications has been exponential during the last 10–15 years, and several reviews have been published, a few of them during the last five years [1], [2], [3], [4], [5], [6], [7], [8], [9], [10], [11], [12], [13], as well as a few others in distinct areas, not so well covered in the more recent reviews [14], [15], [16], [17], [18], [19], [20]. In this work we will make a review on medical applications of vanadium complexes, not trying to systematically cover all publications, but to make a more comprehensive text. The recent book edited by Michibata [20] includes chapters of ‘Vanadium Effects on Bone Metabolism’, ‘Vanadium in Cancer Prevention’, Cardiovascular Protection with Vanadium Compounds’ and ‘Inhalation Toxicity of Vanadium’, thus these topics will only be shortly addressed.
1.1. Distribution, essentiality and potential toxicity of vanadium
Considering relative elemental abundance in our planet's crust, vanadium (0.019%) is the 18th element, thus not far from that of zinc (0.008%) [6]. It is ubiquitously distributed in soil, fossils (particularly in crude oil), water, air and living organisms. In sea water vanadium exists mainly in the form of H2VO4 − and is the 2nd most abundant transition element, being about two orders of magnitude more abundant than iron [6]. The usual V concentrations in drinking water are in the 10 nM range, but groundwater from volcanic areas may contain much higher amounts, up to ca. 2.5 μM [6], and contamination is frequently observed in rivers, lakes and seas ([11] and references therein).
Many metal ions have a general tendency to interact with biomolecules, therefore, it is not surprising that natural evolution has incorporated many of them into performing a wide variety of tasks and playing crucial roles in living beings. Since nature has made such extensive use of metal ions, the questions: “Can metal ions be incorporated into drugs? And can coordination chemistry be used with advantage for medicinal purposes?” naturally arise and are the subject of Medicinal Inorganic Chemistry [7], [21]. Vanadium, being ubiquitously present in our planet's crust also found roles in biological systems, and this work focuses on the potential uses of vanadium-based therapeutic compounds in Medicine.
Several groups of organisms accumulate vanadium and/or employ vanadium in life processes. On the basis of its high amount found in the earth’ crust, namely in pre-biotic conditions and when life first appeared, and of its known use in biochemistry, vanadium is likely to have an essential role in most if not all living beings, namely in humans [2], [6]. The average vanadium concentration in the human body is approximately 0.3 μM and with equilibrium between the amount excreted from the body and the constant supply of some vanadium via food and drinking water may easily be maintained [22].
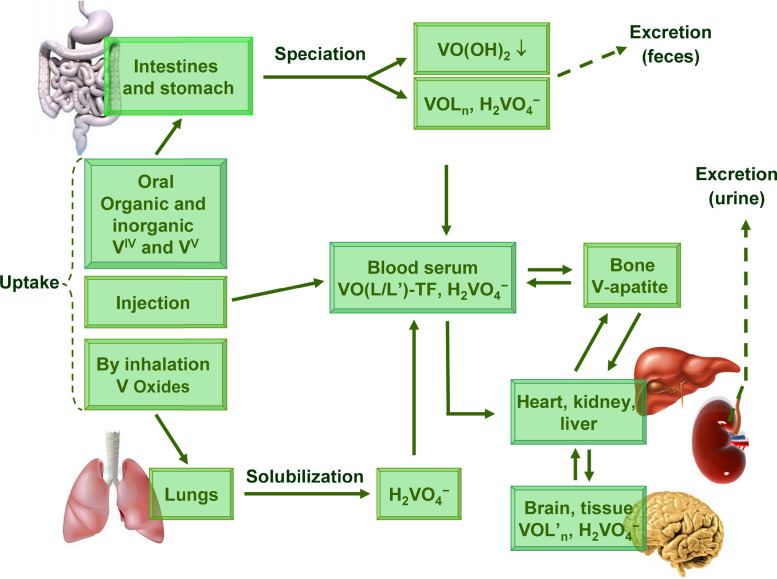
The main routes of vanadium uptake and distribution in the human body are sketched in Fig. 1 (adapted from [6]). Non-occupational vanadium exposure is predominantly from the food supply and typical daily doses consumed by humans have been estimated at 0.01–0.03 mg V/day [5]. Dietary uptake is a quite ineffective process because most of the dietary vanadium is usually excreted with the feces (see below).
Fig. 1.
Uptake and distribution of vanadium compounds in the body.
Source: Adapted from Ref. [6].
The main natural sources for vanadium loads in the atmosphere are continental dust evolving from weathering of soils, marine aerosols and volcanic emissions [11], [23], [24]. Other major sources for aerial contamination are from mining, industrial enterprises and burning of crude oil. In non-polluted areas, the concentration of vanadium oxides (VOx) is in the range of 50 ng m−3, but pollution, where combustion of petroleum and oil are the main contributors to aerial VOx, can raise the V levels up to >103 ng m−3 in urban and industrial areas in particular [25], [26].
Worker exposure to vanadium has largely resulted from inhalation of vanadium pentoxide in workplace dust [27]. Respiratory effects are the most common problems thereof, but many organs can suffer adverse effects [28]. Vanadium oxides are readily absorbed in the lungs and after solubilization in the form of vanadate, H2VO4 −, enter the blood stream. The maximum allowable concentration in breathing air amounts to 0.05 mg m−3. In case of one-time exposure, the limit values for immediate danger to health for an average human are 7 mg of vanadium (intravenous application), and 35 mg of V m−3 in breathing air (inhalation) [5], [6].
Normally, food does not constitute a potential hazard. The average contents of V in food are ca. 30 μg kg−1; the no-effect level for oral intake is 10 mg kg−1 per day [6]. For relevant references see Costa Pessoa and Tomaz [7], the two volumes of ‘Vanadium in the Environment’ [29], the chapter of ‘Inhalation Toxicity of Vanadium’ by Assem and Levy [30], the review of Olopade and Connor [13] and all references provided therein.
2. Biological actions
2.1. Enzyme inhibition
Many studies developed in vitro or in experimental animal models have revealed that vanadium compounds display insulin mimetic properties such as mitogenic effects, stimulatory and inhibitory action on cell differentiation and numerous metabolic effects.
In vitro studies in cell-free systems have shown the effects of vanadium derivatives on the activity of many enzymes, especially those related to phosphate reactions.
Vanadium inhibits several ATPases with different potency [31]. Moreover, vanadium compounds also inhibit different phosphatases such as alkaline phosphatase, acid phosphatase and tyrosine–protein phosphatases [32]. Other enzymes inhibited by vanadium are ribonucleases, phosphodiesterases, phosphoglucomutase and glucose-6-phosphatase [33].
The PTPases activate or inhibit intracellular signaling pathways triggering different biological effects in a cascade way. In biological systems the level of phosphorilated proteins is a balance between the activity of kinases and phosphatases, showing that both types of enzymes have an important role in the regulation of cellular processes. The phosphatases that hydrolyze phosphoproteins are classified in two groups: with affinity for serine–threonine phosphate proteins (PS/TPases) and those with affinity to tyrosine-phosphate proteins (PTPases). All of them catalyze the hydrolysis of phosphate esters. The PS/TPases are metalloenzymes that produce the direct attack of a water molecule to the P atom of the substrate. On the contrary, the PTPases are not metalloenzymes and their active site has a cysteine residue and in their catalytic mechanism a fosfocysteine is formed as an intermediate. Moreover, these enzymes are structural and mechanistically different from the acid and alkaline phosphatases as well as those that hydrolize phosphate-esters of small molecules such as glucose-6-phosphate [34]. Vanadium compounds can oxidize this cysteine residue by the formation of free radicals regulating in that way many biological events [35], [36], [37].
The alkaline phosphatases are bound membrane glycoproteins codified by different genes and classified as specific tissue phosphatases and non-specific tissue phosphatases. These enzymes hydrolyze phosphate monoesters from small molecules and proteins and catalyze the transference of phosphate groups to hydroxyl groups of organic molecules. The mechanism of action occurs through the phosphorylation of a serine residue of the active site, followed by the transference of the phosphate group to a water molecule or to an organic acceptor [38].
The ability of vanadium to inhibit these enzymes is closely related to the physicochemical properties of vanadate and phosphate (see Section 3.2). Structurally, vanadate is similar to phosphate but it is important to emphasize that vanadate shows a greater flexibility in its coordination geometry. Both anions participate in similar reactions. The generation of a stable transition state between the vanadate and the enzyme has been considered the reason for its inhibitory effect to the enzyme activity. Studies by X-ray diffraction demonstrated that vanadate binds in the active site of the enzyme with trigonal bipyramid geometry. Assays with vanadium compounds of different coordination geometry suggested that the complexes with five coordination were more potent inhibitors than those with coordination numbers six or seven [39]. Nevertheless, there are other factors that can affect the enzymatic activity [39], [40].
The VIVO2+ cation also inhibits different enzymes and in some cases with a greater potency than vanadate. One proposed mechanism for this inhibition is based on the possibility of VIVO to adopt a trigonal bipyramid, bind in the active site of the enzymes and thus cause inhibition of the activity [33], [41].
2.2. Enzyme activation
In most cases, enzyme activation by vanadium follows an indirect mechanism. Vanadium can stimulate the enzyme activity through the formation of complexes with ligands that resemble the structure of physiological substrates. For instance, the glucose-6-phosphate dehydrogenase is activated by vanadate. Another mechanism to regulate enzymatic activity is related to the enzymes activated or inhibited through the phosphorylation of tyrosine residues. Vanadate also forms esters with Tyr residues mimicking their phosphorylation process with a great impact in several biological events. The activation by vanadate of some cytosolic kinases and the insulin receptor (IR) has impact on the balance between phosphorylation–dephosphorylation processes. In vitro experiments with cells in culture show that VCs may cause or enhance insulin mimetic actions such as stimulation of protein phosphorylation in Tyr residues increasing the level of phosphoinositides, stimulate cell proliferation in different cell lines, promote the uptake of small molecules and ions, etc. On the contrary, vanadium compounds also can exhibit antitumor effects as well as toxic and transforming actions in different cell lines. Moreover, it is worthy to know that globally the underlying mechanisms of action of vanadium compounds are still poorly understood [14], [42], [43], [44], [45], [46].
Vanadium compounds are well-known for their antidiabetic effects both on glucose and lipid metabolism, but the mechanisms are still not completely understood. It has been previously reported that VO(acac)2, VO(maltolato)2 and NaVO3, attenuated basal lipolysis in 3T3L1 adipocytes assessed by glycerol release as a marker of lipolysis [47]. Though both Akt and ERK pathways are activated by the VCs, but only Akt activation by these three vanadium compounds plays a role in their antilipolytic effect. On the other hand, VO(acac)2 can block cell cycle progression at the G1/S phase via a highly activated ERK signal pathway in human hepatoma HepG2 cells. The results indicated that similar activated pathways may lead to differential biological consequences for cancer cells and adipocytes, indicating that VCs may be used in the prevention and treatment of both diabetes and cancer. The action of insulin depends on its binding to the hormone receptor and its activation. Then the transduction of the signal involves a series of protein kinases such as MAPK and S6 kinases, as well as lipid kinases such as PI3-K. The insulin mimetic effects of vanadium may be caused by the stimulation of the autophosphorylation of the insulin receptor but in different manner than the hormone [48], [49], [50], [51], [52], [53]. These effects depend on the tissue and the animal species. For instance, in rat lung cells and rabbit and mouse diaphragm cells vanadium did not cause autophosphorylation of insulin receptor [54], [55]. On the other hand, the inhibition of tyrosine kinase activity of insulin receptor by vanadate did not suppress the effects of vanadate on glucose metabolism, suggesting a post-receptor action [52]. VCs that show insulin-like effects, stimulate kinases of the signal transduction pathways used by insulin beyond IR and IRS-1such as MAPKs and PI3K.
In CHO cells, which do not express insulin receptors [56], vanadate and oxidovanadium(IV) stimulate the phosphorylation of proteins of the insulin pathway such as ERKs, p70s6k and p90rsk, which in turn phosphorylate and regulate the action of transcription factors, many of them related with cell proliferation. PI3-K is other enzyme that regulates metabolic and mitogenic effects of insulin. Vanadium promotes the activity of this enzyme [57]. Moreover, the activation of the pathway ras-MAPK by VOSO4 seems to depend on the activity of PI3-K [58], [59]. On the other hand, in Swiss 3T3 mouse fibroblasts, vanadate stimulates S6 ribosomal kinase (p90S6K or p90rsk) [60].
Most of the results support the hypothesis that PTPases inhibition may be the main mechanism by which vanadium compounds exert insulin mimetic or insulin enhancing effects [40], [61], [62], [63].
It was also suggested that vanadate may cause some of the insulin effects through the activation of a cytosolic kinase (CytPTK) [63]. This can be considered an alternative via to promote insulin like effects.
Another interesting field is the effects of vanadium on cell proliferation and differentiation. Growth factors stimulate cell proliferation though the activation of different receptors that in turn trigger intracellular signaling pathways which promote the action of different transcription factors. Insulin stimulates Ras-MAPK pathway and as a sequence cascade events, insulin exerts its mitogenic actions. In the same way, different agents able to activate the signal transduction pathways of insulin can cause cellular proliferation. Different in vitro studies of cells in culture have demonstrated that VCs display a biphasic behavior toward the mitogenesis: concentration between (1) 2.5 and 25 μM usually enhanced cell proliferation while (2) concentrations over 50 μM inhibited this event [64], [65].
Vanadate stimulates DNA synthesis in fibroblasts, behaves as a growth factor mimetic agent promoting the transition of the cells from G0 to G1 phase in the cell cycle [66], [67]. Moreover, vanadate potentiates the proliferative effect of IGF-I in osteoblasts in culture [68]. At the mitogenic doses, vanadate also showed a dose-dependent increase in alkaline phosphatase (a marker of mature osteoblasts) in cultured calvarial cells and stimulated bone collagen synthesis.
Different VIVO-complexes with organic ligands regulate osteoblast differentiation, displaying a stimulatory action on collagen type I synthesis [69].
The mitogenic actions of vanadium compounds may be mediated by the MAK pathway. In fact, as it has been previously mentioned, V indirectly activates some kinases such as ERKs and PI3-K. Sequentially, the activated ERKs promote the action of some transcription factors and convey the cells to enter into the cell cycle [58], [59]. PI3K activation is also related to the mitogenic actions of vanadium because there is a crossing between the two pathways. PI3K activates PKB and in this way it can support for the mitogenic actions of VCs [70].
Other pathways activated by vanadium that may be related to its proliferative effect are those of phosphoinositides and heterotrimeric protein G. At 100 μM, V increases the levels of inositol mono-, di- and tri-phosphate through mechanisms dependent or non-dependent of tyrosine kinases [71]. On the other hand there are cases where vanadate stimulates the proteins G pathway but at very high concentrations (3–10 mM). These concentrations are very high comparing with those that cause proliferative effects in cell cultures [72].
2.3. Physiological roles of vanadium
The report of Cantley et al. [73] that endogenous vanadate inhibits Na+ and K+ ATPase in vitro was made in 1977 and since then many other physiological roles have been ascribed to vanadium, such as mitogenic actions, stimulation of bone cell proliferation, bone collagen synthesis and noradrenaline release from pulmonary artery, inhibition of oxidative drug demethylation, stimulation of oncogen expression and histamine release from mast cells, as well as toxic and cytostatic actions, antidiabetic actions and neoplastic transformations. The possibility that vanadium compounds (VCs) may regulate osteogenesis and possibly be able of counteracting the bone damaging actions of glucocorticoids has also been suggested [74], [75], [76].
As mentioned, one relevant prospective role of VCs is in diabetes mellitus (DM) treatments. An important feature of vanadium's insulin mimetic (IM) action is the multiplicity of its effects and these were reviewed in e.g. [12], [17], [18], [63], [74], [75], [76], [77], [78], [79]. In both spontaneously and chemically induced diabetic animals, vanadium alleviate not only the primary symptoms of diabetes, high blood glucose and triglyceride and cholesterol levels (although tissue vanadium levels normally do not correlate with the glucose-lowering effect), but also prevents or reverses several of the secondary complications: sorbitol accumulation [80], [81], cataract development [82], [83], impaired thyroid hormone [84], [85], alterations in kidney morphology [86], [87] and adrenal hypertrophy [87]. In partially pancreatectomized rats [88] vanadium administration reversed insulin resistance by restoration of muscle glycogen synthesis; in STZ-diabetic rats it stimulated basal hexose uptake in muscle and liver [89] and even in non-diabetic rat muscle vanadium therapy increased sensitivity to insulin stimulation of glycolysis and glycogen synthesis [90], impaired antioxidant status and excessive food and fluid intake [91], [92].
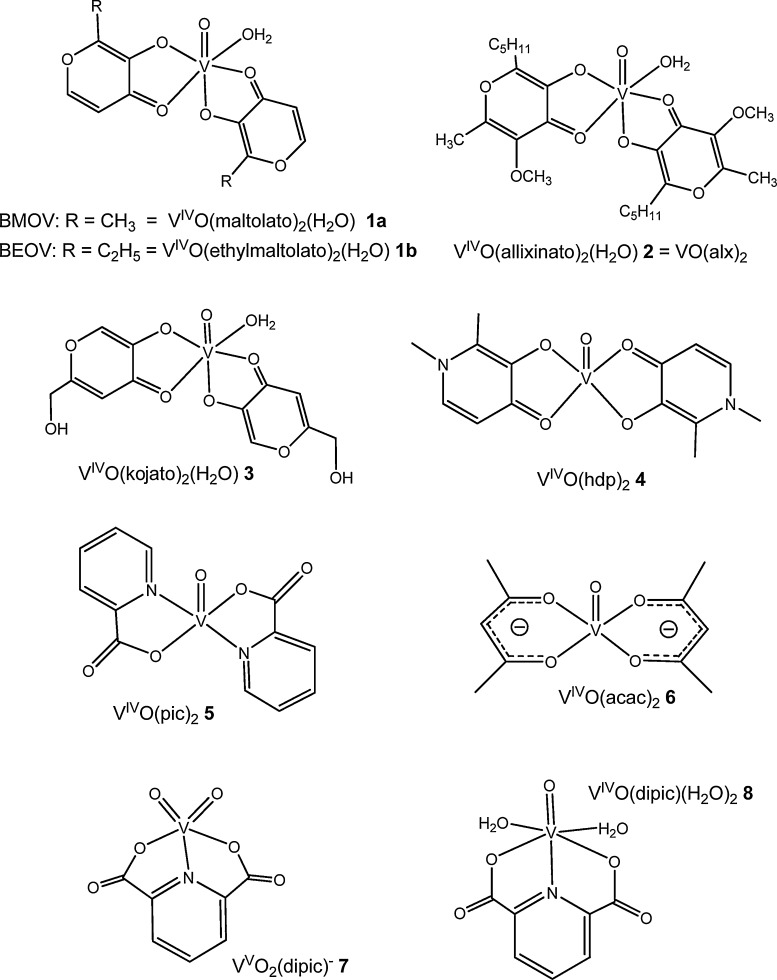
It was also reported that vanadate prevented the decline in cardiac performance due to diabetes [84] and cardiomyopathy [84]. With streptozotocin-induced (STZ) diabetic rats, isolated working heart parameters of left ventricular developed pressure and rate of pressure development also indicated that BMOV (1a, Fig. 2 ) administration resulted in significant attenuation of this heart dysfunction [93].
Fig. 2.
Some vanadium compounds that have been reported to exhibit insulin-like effects [7].
Since secondary complications of diabetes are known to have increased oxidative stress as etiologic components [90], the possibility that vanadium's insulin mimesis is partially due to changes in pro-oxidant/oxidant balance was also considered [94], [95]. An imbalance of production of ROS over functional antioxidant defenses has been associated with several pathological conditions [96], e.g. cardiovascular disease [97], rheumatoid and osteoarthritis [96], diabetes mellitus [98], tumor angiogenesis and proliferation [99], cataract formation [100], and inflammation [101].
Curcumin, a compound known for its neuroprotective action [102], has demonstrated efficacy in all of these pathological conditions [103], while VO(cur)2 was more effective as an anti-cancer agent, compared to uncomplexed curcumin or VOSO4 alone, it was more than twice as effective as curcumin alone as an anti-arthritic agent, and was more than four times as effective as curcumin alone in inhibiting smooth muscle cell proliferation [104]. Probably a relevant aspect deserving care is that neither curcumin nor VO(cur)2 were effective in reducing plasma glucose levels in STZ-diabetic rats. In contrast, there are reports on the capacity of curcumin to facilitate lowering of plasma glucose or to decrease insulin resistance. The difference might be that these experiments by Thompson et al. [104] were obtained upon acute oral administration of curcumin and VO(cur)2 [104], while the data by other researchers are from oral gavage on a daily basis for 6–7 weeks of curcumin to STZ rats [105] or db/db diabetic mice [106].
One interesting compound is VO(alx)2 (compound 2, Fig. 2). The group of Sakurai prepared several VCs and evaluated their biological functions in vitro and in vivo [17], [107], demonstrating that whether administration was by acute oral gavage or by acute i.p. injection, VO(alx)2 could be one of the most promising complexes for the treatment of diabetes [108]. The pharmacological functions of VO(alx)2 include hypoglycemic effects and the improvement of hyperinsulinemia, hypercholesterolemia and hypertension [108], [109]. VO(alx)2 significantly induced V uptake in the insulin-responsive tissues such as the adipose, liver and muscle tissues [110]. VO(alx)2 also affects the tyrosine phosphorylation of IRβ and IRS, leading to the activation of PI3K–Akt signaling and the translocation of GLUT4 to the plasma membrane. Moreover, it was also demonstrated that VO(alx)2 regulates the DNA binding activity of the FoxO1 transcription factor, which in turn controls the expression of gluconeogenesis genes in response to Akt activation [111]. Overall, VO(alx)2 improves diabetes, obesity and hypertension by enhancing insulin sensitivity and leptin resistance in obesity-linked type 2 diabetic KKAy mice and was proposed as a promising VC to treat both type 1 and obesity-linked type 2 diabetes [109].
As a final comment the reader should also consider the interesting review by Aureliano and Crans [112], who summarized proteins that interact with vanadates, giving more focus to decavanadates, and showing that vanadate biological effects are not only due to monomeric vanadate. Namely, decavanadate interacts with the polyphosphate, nucleotide and inositol 3-phosphate binding sites of enzymes.
3. Therapeutic applications of vanadium compounds (VCs)
Concerning applications of VCs as therapeutic agents, treatment of diabetes has been one of the main focuses. In fact, vanadium compounds have been reported to exhibit insulin-like effects and among the VCs tested as small molecule insulin-mimetics, or insulin-enhancers, VIVO(maltolato)2 (1a, BMOV, Fig. 2), and VIVO(Etmaltolato)2 (1b, BEOV, Fig. 2), have been extensively studied. BMOV and BEOV may be taken orally and both lower plasma glucose levels in streptozotocin-induced (STZ) diabetic rats [76], BEOV having completed Phase I and IIa clinical trials. Although essentially successful, the clinical trials have provisionally been abandoned [113].
Examples of VCs that have been reported to have potential as antitumor agents are the inorganic complex Metvan [VIVO(SO4)(4,7-Mephen)2], vanadocene dichloride [(η5-Cp)2VIVCl2] [14], [114], a V-cysteine complex [115] and a semicarbazone derivative [116] and VO(acac)2 (see Section 4). Several VCs have also been reported to have prospective usefulness as anti-parasitic [117], [118], [119], [120], [121], [122], spermicidal [123], [124], anti-viral [125], anti-HIV [126], [127], [128], [129], [130], and anti-tuberculosis [131], [132], [133] agents (see below).
3.1. Uptake, speciation and biodistribution of vanadium compounds
Fig. 1 summarizes some of the main aspects associated with uptake and distribution of VCs in the human body. Dietary forms of vanadium are either vanadate, H2VO4 −, present mainly in drinking water, and VIVO-compounds {VIVOL}. Free VIVO2+ ( (VIVO(OH)5]2+) is not much relevant, since most of it would precipitate as VIVO(OH)2, only allowing for very low (nanomolar) concentration of ‘free’ VIVO2+ {actually [VIVO(OH)3]− and [(VIVO)2(OH)5 −]n} at the best (see below). Vanadate(V) is partially reduced in the stomach, and in the slightly alkaline medium of the intestines precipitated in the form of VIVO(OH)2. Thus, most of the vanadium supplied by nutritional sources is secreted with the feces in the form of sparingly soluble vanadyl hydroxide VIVO(OH)2 [6].
Vanadium can also enter the blood stream by injection or infusion, either intentionally when injected intravenously or intraperitoneally, or accidentally when present as a ‘contaminant’ in infusion solutions [134]. Besides speciation, vanadium compounds ending up in the blood stream either after resorption in the gastrointestinal tract, or via the lungs, or by infusion/injection, are subjected to redox interconversion between VV and VIV, depending on the oxygen tension and the presence of redox-active agents. The main transporter in blood for both anionic vanadate(V), cationic VIVO2+, and neutral or charged VIVOL species is transferring [135].
Values in the range 0.2–15 nM were determined [136] for the vanadium concentration in human blood plasma. Upon oral treatment of human patients with VCs, the concentration of V in blood serum depends on the regime and amount of compound given, and much higher values have been reported, ca. 1–10 μM [76], [113], [137]. In rats treated with BMOV [138], [139] or BEOV [140] (1a or 1b in Fig. 2) maximum values of ca. 40–60 μM were found in their blood plasma.
Plasma vanadium contents in rats may be considered to decline in three phases. The 1st phase is a rapid decline with a half-life t 1/2 of 1 h, followed by a second intermediate decline (t 1/2 ≈ 26 h) and a third slow one with t 1/2 ≈ 10 days. Vanadium contents in blood are thus reduced to about 30% within the first 24 h [5], [134]. Clearing occurs directly via urinary excretion, as well as after distribution over tissues, and about 50% of the vanadium is found in urine after ca. 12 days. The residence time of vanadium in bones, where it may replace phosphorus in hydroxyapatite, is ca. 1 month [113], corresponding to a half-life of 4–5 days.
Vanadium in biological systems may be present in the oxidation states +III (VIII), +IV (VIV) or +V (VV) [141]. For mammals VIV and VV are normally considered more relevant as VIII is very susceptible to oxidation; however, VIII-hTF species (hTF = human serum transferrin) have been reported to be quite air-stable [142], [143]. At the very low concentrations such as those present in blood plasma VV exists mainly as H2VO4 − and HVO4 2−, often referred as VO3 −, or as monovanadate; at pH 7.4 and low concentrations of VV in blood plasma, and several potential ligands being present, it is not expected that divanadates (V2) or tetravanadates (c-V4) might form. In cells, usual physiological vanadate concentrations are also too low to allow for the formation of oligovanadates. Locally, however, concentration enhancement or template-directed nucleation [144] may take place, and the oligovanadate(s) then formed can interact with proteins and DNA. Cellular targets and processes probably affected by decavadates are: (1) membrane depolarization, (2) pumps, channels, receptors in cellular membrane and in endoplasmic reticulum membrane, (3) contractile system, (4) cytoskeleton structure and function, (5) mitochondria bioenergetics, (6) ROSs species changes and antioxidants responses, and (7) probably also nucleous [112].
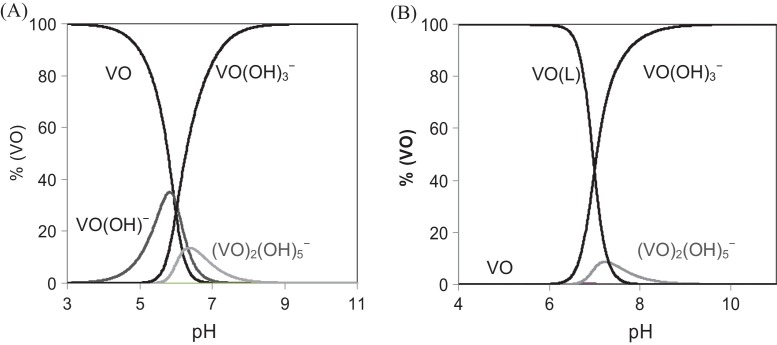
VIV normally exists as VIVO-species. VIVO2+ is quite susceptible to oxidation for pH > 3 and forms an insoluble hydroxide for pH > 4 (K sp ≈ 7.4 × 10−23) [145], [146]. At pH > 5 oligomeric vanadium species may be relevant, even at concentrations as low as 10 nM, see Fig. 3 . In the pH range 5–8 and at concentrations below 10 μM, VIVO2+ ( VIVO(H2O)5 2+) does not exist as such in this pH range, but VIV is soluble and is mainly in the form of VIVO(OH)3 − and [(VIVO)2(OH)5]n − [147] (Fig. 3). In blood serum probably VIVO2+ is totally bound to high molecular mass and low molecular mass bio-ligands also present; inside cells many bio-ligands are present that form complexes with VIVO2+ with binding constants probably higher than 109, thus most of the VIVO2+ should also be bound. However, the presence of a very small amount of VIVO(OH)3 − (free or bound?) cannot be ruled out and its relevance in physiological media should be taken into account.
Fig. 3.
(A) Distribution diagram of VIVO hydrolysis at 10 nM concentration. (B) The same but including the possibility of forming a VIVO(L) complex (concentration of L = 100 nM) with a conditional stability constant of 109; β values of the hydrolytic VIV-species were obtained from Ref. [147].
3.2. Vanadate/phosphate analogy
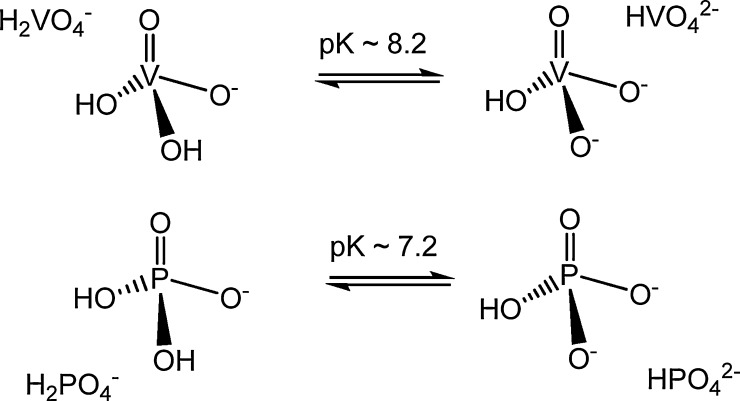
Monovanadate and phosphate are structural analog, monovanadate being only slightly larger than phosphate (see Fig. 4 ) [24].
Fig. 4.
Analogy between phosphate and vanadate. Besides monovanadate being structurally similar to phosphate, the acid-base equilibria operating and other types of reactions (e.g. V and/or P ‘ester’ formation) are similar. However, there are also some structural and pKa differences (see text) [6], [148].
From a geometrical point of view the two anions are not much different, vanadate(V) thus being a competitor of phosphate in sites commonly occupied by phosphate [23]. However, there are also significant differences:
-
-
due to the different pK a, at physiological pH and ionic strengths, vanadate is mostly present in the form of H2VO4 − {depending on pH the amount of HVO4 2− is ca. 1/6 (at pH ∼ 7.4) to 1/25 (at pH ∼ 6.8) of the amount of H2VO4 −}, while HPO4 2− and H2PO4 − exist in approximately equal amounts.
-
-
phosphorus can only attain the coordination number 5 in transitional states, while vanadium is much more versatile in this respect, easily forming stable complexes with coordination numbers 5 or 6. Therefore, once incorporated into the active site of a phosphate-dependent enzyme, taking the place of phosphate, the activity of this enzyme is inhibited [23].
Presently there is no solid basis for categorizing VCs, such as those depicted in Fig. 2, as harmful when administered in reasonable amounts. Instead, as mentioned above, vanadium in the form of H2VO4 −, is probably involved in the regulation of phosphate-dependent processes, such as metabolic processes involving phosphatases and kinases and the phosphate metabolism in general [32]. Thus, probably vanadium is an essential element. This plausible regulatory role of vanadium is certainly related to the structural and chemical similarity between phosphate and vanadate (see Fig. 4). Additionally, the participation of VIV and VV in leveling reactive oxygen species (ROS) suggests that vanadium can be beneficial in the treatment of several diseases and malfunctions related to ROS imbalances [23].
The competitive behavior of vanadate with respect to phosphate is likely the clue for the insulin-mimetic/insulin-enhancing effect of VCs. As mentioned, for pH > 6 at low concentrations, free VIVO2+ exists significantly as VIVO(OH)3 −, being structurally similar to VVO(OH)3 ( H3VO4). Inter-conversion between VIV and VV is thus fast and easy in physiological conditions, and it has been suggested that, besides monovanadate(V), VIVO(OH)3 − may also mimic phosphate [6].
3.3. Transport of vanadium in blood
Once in the blood stream, blood serum constituents will probably dominate vanadium speciation, and consequently the final cellular uptake by tissue cells and the targeting pathways for vanadium.
Speciation can encompass ligand exchange and/or redox-interconversion between VV and VIV (and, potentially, VIII) [143], [149]. For ligand exchange, the low molecular mass constituents lactate and citrate, and the high molecular mass constituents, transferrin (hTF), albumin and immunoglobulin G, are the more important ones, with hTF having a predominant role. Reducing agents such as ascorbate, glutathione and NADH can induce reduction of VV to VIV, and oxidants such as NAD+, O2, O2 2− and O2 − can convert VIV to VV.
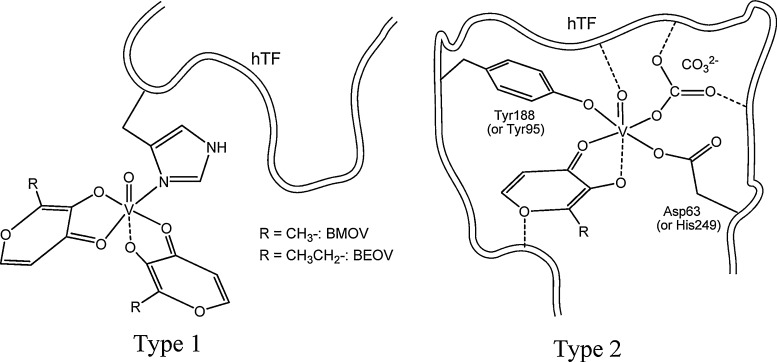
If vanadium is introduced in blood in the form of a prospective drug of composition VIVOLn (examples are some of the complexes of Fig. 2), part of the vanadium may cross the membrane of the erythrocyte cells [150], VV is reduced to VIV by cellular glutathione [151], [152], [153], [154] and binds to hemoglobin [153]. The vanadium compounds remaining in the blood plasma may bind to hTF forming either type 1 or type 2 species (see Fig. 5 ) or forming (VIVO)2hTF.
Fig. 5.
Two distinct proposed types of binding for VIVO(carrier)2 complexes to hTF. The possibility of the Type 1 binding to a protein was recently confirmed by the x-ray diffraction characterization of a lysozyme-VIVO(pic)2 adduct [155].
Source: Adapted from Ref. [143].
In holo-hTF, (FeIII)2hTF, the protein is in its ‘close conformation’, thus it is recognized by the transferrin receptors of the cell membranes, and FeIII is up-taken by cells as (FeIII)2hTF by a receptor-mediated endocytosis process. Regarding the cellular uptake of vanadium, present in a prospective drug of composition V IV OL n, several scenarios can apply for the up-take process:
-
-
The complex is stable enough to remain intact, and sufficiently lipophilic to slowly cross the cellular membrane via diffusion. Pyridinone complexes such as 4 in Fig. 2 enter erythrocytes by diffusion [150].
-
-
The ligands are detached, oxidation occurs, thus forming H2VO4 −, and vanadium may enter the cells as monovanadate via anion channels.
-
-
The ligand system is maintained, or is replaced in part or completely by transferrin to form VIVO(L)(hTF) (see right side of Fig. 5) and (VIVO)2hTF [7], [155], [156]. Namely (VIVO)2hTF is probably recognized by the cellular hTF receptors and internalized via endocytosis [149]; there is no information available regarding VIVO(L)(hTF), but in this case probably hTF does not form the ‘closed conformation’ required for adequate binding to the cell receptors.
-
-
Either VIVO2+, or vanadates, or VIVO(L)n species, bind holo-hTF, at available donor atoms of the protein; these species are probably recognized by the cellular hTF receptors and internalized via endocytosis [143], [157].
-
-
VIII species may form, e.g. (VIII)2hTF and or (VIII)(FeIII)hTF, which forms the ‘closed conformation’ and probably is recognized by the cellular hTF receptors and also internalized via receptor-mediated endocytosis [143].
-
-
The complex is stable enough to remain intact and contains one or more functions in the outer coordination sphere that are recognized by cell receptors; receptor-mediated uptake is then again a potential transport route.
-
-
Albumin receptors on the cell (e.g. adipocites) surface would also be likely vehicles if a VC binds to HSA [158].
It was also reported that human serum albumin (HSA) enhanced the insulin-like activity of a few VCs more that transferrin [159]. The VCs increased tyrosine phosphorilation in response to insulin, including IRβ and IRS1 [160]. This may be due to enhanced uptake by cells, but the subject deserves further studies.
3.4. Biodistribution in the body
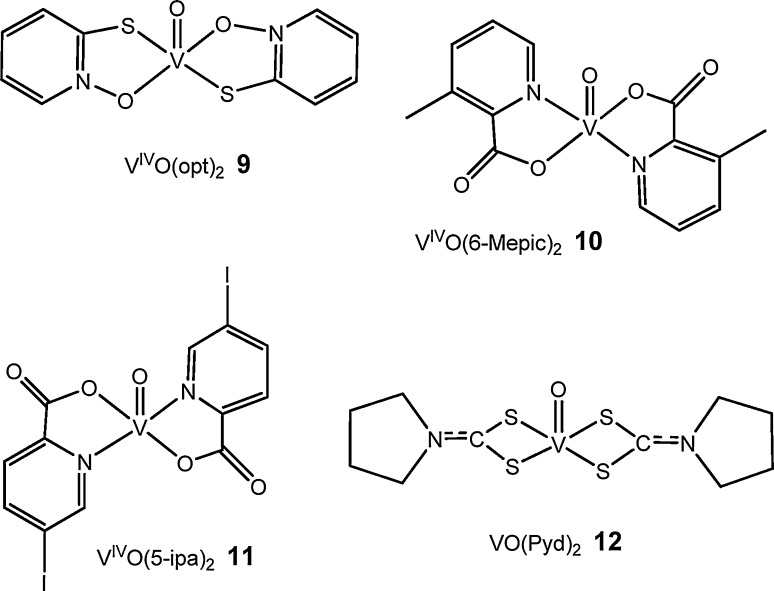
If we aim for the clinical use of VCs in the future, the organ distribution and metallokinetic features of vanadium, as well as of the corresponding complex forms, have to be well understood [16]. For these purposes, neutron activation analysis (NAA) has been one of the important techniques used to determine the total vanadium levels in organs of rats given VOSO4 or VCs with different coordination modes. In rats treated with VOSO4 vanadium was found in kidney > liver > bone > pancreas [161], [162], [163], while in rats treated with VCs such as VO(5-ipa)2 (Fig. 6 ) it was detected in bone > kidney > spleen > liver > pancreas [164]. Such differences between organ distribution of VOSO4 and VCs might suggest long-acting character of the complexes. Vanadium levels after 25 weeks of BMOV supplementation were comparable with those obtained after 10 weeks of VOSO4 feeding, but daily BMOV dose taken was about half [93].
Fig. 6.
Several additional insulin-enhancing vanadium compounds [164], [165], [166], [171].
BMOV treatment of diabetic rats at a concentration of 0.75 mg/mL in drinking water resulted in accumulation of V in bone, kidney, liver, muscle, and fat, without any mortality over a 25-week period, with a decrease of plasma glucose, %GHb, triglycerides and cholesterol. BMOV therapy effectively prevented the development of the myocardial dysfunction associated with STZ-induced diabetes in rats, even in those rats whose blood glucose levels remained above normal [93].
Total vanadium distribution in STZ-rats treated with VO(6-Mepic)2 was also examined by an NAA method [165], [166]. V accumulated in almost all tissues, particularly in bone and kidney, in the mitochondrial fraction of the liver and in the supernatant of the kidney of STZ-rats receiving VO(6-Mepic)2 by i.p., injections. Similar tendencies were observed in vanadium distribution in STZ-rats given the same VC by oral administration, except for an accumulation in the mitochondrial fraction of the liver. V accumulation in the bone of rats receiving i.p. injections of VO(6-Mepic)2 was remarkable: ≈3.3 times that in the kidney and ≈11 times that in the liver. These results were somewhat similar to the distribution of vanadium in rats treated with i.p. injections of VO(pic)2, where accumulation in bone was approximately 4.7 times that in the kidney and 35 times that in the liver. In rats treated with complexes such as VO(5-ipa)2, by NAA [74], [162], [164], [165] or radioisotope [167] determinations, V was found in kidney > liver > bone > pancreas.
In STZ rats treated with VO(5-ipa)2 by daily i.p. injections for 14 days, and then without administration of the VC for additional 14 days, the organ distribution of total V was: bone (0.32 μmol/g of wet tissue) > > kidney (0.08) > spleen (0.04) > adipose (0.02) ≈ liver ≈ pancreas > lung ≈ heart > blood cell ≈ serum > brain [16].
The long-term action of VO(6-Mepic)2 and of other VCs by oral administration is probably related to the accumulation of V mainly in the bone. Vanadium accumulated in the bone may be released gradually to other organs via the blood-stream after stopping the administration of the VC, and is able to normalize the blood glucose levels for a long period.
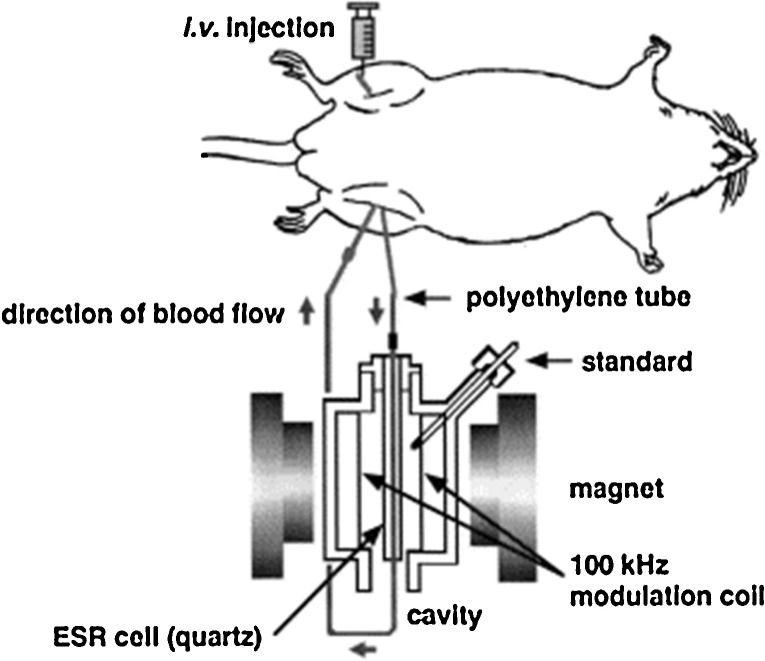
To access the metallokinetic features of VO complexes the group of Sakurai applied a BCM-EPR method, which allows the tracing of paramagnetic species in the blood (Fig. 7 ) [16], [168]. For example, VCs were given by a single i.v. injection to rats at 37 °C under anesthesia with pentobarbital, and the VIV-EPR spectra were measured at room temperature every 30 s. It was concluded that a major factor for the disappearance of EPR signals in the circulating blood due to VCs was not the participation of redox processes of VO species in the circulating blood, but their distribution to the tissues and elimination from the body [164]. Thus the VIVO species taken up into the blood are distributed to the short- and long-stay tissues, and then accumulated there or re-distributed in the bone, liver and kidney.
Fig. 7.
Scheme depicting how in vivo blood circulation monitoring-electron paramagnetic resonance (BCM-EPR) studies on rats are carried out [168]. The method was used to measure the real-time disposition of spin probes in the circulating blood of rats.
Source: Reproduced with permission from Ref. [168].
The real-time EPR analysis of VIVO species revealed that in terms of half-life (t 1/2), their clearance rate from the blood of rats given VOSO4 was higher than those given VCs, being 5 min in VOSO4-treated rats and 7–30 min in VC-treated rats [164]. The slow clearance rate of the VCs suggests that a higher distribution of vanadium in rat organs is obtained, which in turn indicates long-term acting normoglycemic effects upon stopping V administration.
The slow elimination of V from the circulating blood of rats indicated a different association of the VC with the blood components such as serum proteins or erythrocytes between rats given VO(5-ipa)2 and those given other VCs. By metallokinetic analysis it was concluded that VIVO species tended to be distributed in the peripheral tissues and gradually eliminated from the circulating blood. VCs with electron-withdrawing and donating groups remained significantly at higher levels and longer, due to their slower clearance rates from blood, suggesting that the high exposure and longer residence of the VC is associated with a higher normoglycemic effect in diabetic animals. In vitro IM activity, metallokinetic character and in vivo antidiabetic action of VO–picolinate complexes thus appear related to their chemical structures [169], [170].
Vanadium also accumulated in almost all tissues, especially in bone > kidney > liver > pancreas in rats given VO(pyd)2 12. In sub-cellular distributions of vanadium in rats treated with VO(pyd)2 or with VOSO4, vanadium was found most abundantly in the supernatant fraction of the kidney, while in the other three fractions no significant difference in vanadium distribution was found [171]. The hepatic distribution of vanadium in rats treated with VOSO4 differed from that in rats treated with VO(pyd)2: while the highest vanadium concentration was determined in the mitochondrial fraction of the liver in rats treated with VOSO4, the distribution of vanadium showed no significant difference among the four fractions in rats treated with VO(pyd)2 [171].
For BMOV and VOSO4 [167] disappearance of vanadium was fastest from blood, and slowest from bone, followed by kidney, then liver. The relatively slow clearance of V from plasma may be attributable to vanadium recirculation [140], as also seen with a much smaller dose of V by oral gavage [172], [173]; disappearance of vanadium was fastest from soft tissues (11–132 h versus 15–73 h) and slowest from bone (t 1/2 = 24 days for NH4VO3 versus t 1/2 = 30 days for BEOV) [140].
Chronic administration of a VO-compound has lead to bone tissue accumulating the highest amounts of vanadium. Suggestions have been made that V is probably in the form of vanadate(V) substituting phosphate in the mineralized hydroxyapatite structure of bone [174]. However, the measured bone tissue paramagnetism indicated that it also contains VIVO-species [175].
VIVO2+ may substitute Ca2+ and Mg2+ in bone tissue [176] and diabetic individuals have a higher incidence of bone fractures than non-diabetic ones. The effect of VO(acac)2 on bone structure, after chronic daily treatment of alloxan-induced diabetic rats for 35 consecutive days, and several parameters related to the bone characteristics were measured [177]. The authors reported that the plasma level of osteocalcin, a protein that may be used as a marker of osteoblast activity and is secreted by osteoblasts was decreased in diabetic rats (7.5 ± 1.9 ng/mL), but was partially restored upon VO(acac)2 treatment (ca. 12.6 ± 1.1 ng/mL), though still lower than the baseline level in non-diabetic rats (ca. 15.4 ± 4.4 ng/mL) and in non-diabetic rats treated with vanadium (ca. 15.1 ± 1.8 ng/mL). Additionally, while the treatment of diabetic rats with VO(acac)2 did not change the bone mineral density, overall it improved or normalized their strength, trabecular thickness and the mineral apposition rate [79], [177].
3.5. Vanadium in the treatment of diabetes
Background: Diabetes mellitus (diabetes), is associated with an impaired glucose and fatty acid metabolism, induced by a non-existent or insufficient insulin supply, or inadequate response to insulin. Insulin is produced by the β-cells of the Langerhans’ islets in the pancreas. Receptors on the β-cells are activated by glucose (and, synergistically, fructose), stimulating the secretion of insulin [178] that then targets cellular trans-membrane insulin receptors (IRs) to down-regulate glucose levels.
Type 1 and type 2 are the main manifestations of diabetes. According to estimates [179], up to 10% of the world population is suffering from diabetes. Type 1 (or juvenile) diabetes, corresponds to approximately 10% of diabetes cases, is the result of greatly reduced or even lacking production of insulin, commonly as a result of an autoimmune reaction which destroys the β-cells, or a damage of the pancreas by an accident. Type 2 diabetes normally develops in elderly (commonly > 60) individuals; insulin is still produced, but the insulin receptors (IRs) of the tissue cells no longer respond properly to insulin.
As mentioned in Section 2.2, the main potential of vanadium for the treatment of diabetes probably comes from the similarity between vanadate and phosphate highlighted above. However, unlike phosphate and phosphate esters, vanadate is not easily released once bound at the enzyme's active site. Additionally, in contrast to phosphorus, for which the 5-coordinate state, achieved on binding covalently to the active center, is just a transitional situation, vanadium forms quite stable 5- or 6-coordination compounds; thus, once bound at the active site, vanadates are not easily released and hence block off this site for phosphate, thus resulting in an inhibition of the respective enzyme [6]. Interestingly, the known examples of protein structures with vanadium bound in the active sites globally show that both the square pyramidal and the trigonal bipyramidal geometries can support the transfer of the phosphoryl group in phosphatases and other phosphorylases [180]. However, it is clear that there are much more reported examples of trigonal bipyramidal complexes in the active site than square pyramidal complexes. Considering that in small molecules the square pyramidal geometry is more common than the trigonal bipyramidal one, this observation supports the expectation that the trigonal bipyramidal geometry of VV is supported by the proteins with preference over the square pyramidal geometry. This is an interesting result, but probably is not very surprising as most of these phosphatases are build-up to support a 5-coordinate phosphate transition state.
3.5.1. Insulin-mimetic (-enhancing) vanadium compounds
Vanadium compounds that show insulin-like effects are frequently designated by insulin mimetics (IM); however, since in most situations there is some residual insulin production, the designation insulin-enhancing compounds are more adequate. For many years, in the treatment of diabetic animals and, sporadically, human individuals, VV and VIV compounds have been employed. VIVO2+ is quite susceptible for oxidation for pH > 3 and forms insoluble hydroxide for pH > 4. At pH > 5 oligomeric vanadium species may be relevant, even at concentrations as low as 10 nM (see Fig. 3).
Mainly due to the very low oral bioavailability of inorganic salts (ca. 1–2%), VIV complexes with organic ligands have been extensively explored. Organic ligands allow for a fine-tuning of the vanadium compound with respect to stability, rate of absorbance from the GI tract (when applied orally), targeting of and internalization by the tissue cells, and toxicity. A key compound in this regard has been BMOV [181], [182], 1a in Fig. 2, and derivatives thereof, such as the VIV-complexes formed with ethylmaltol (BEOV), 1b [113] and allixin (2) [108], [183]. The related complex with kojato (3) and the pyridinone complex 4 have also been prospective therapeutics. Several other complexes have been studied, such as VO(acac)2 6, and several picolinato and dipicolinato derivatives (Fig. 6).
Maltol is a naturally occurring compound and an approved food additive in many countries. BMOV and BEOV have so far been the only VCs to be subjected to clinical tests, essentially with encouraging response [113]. Several other VCs have been tested with cell cultures (in vitro) and with diabetic animals (in vivo), such as e.g. STZ rats and Zucker rats as models for diabetes 1 and 2, respectively.
Fig. 1 provides an overview of the uptake and distribution of vanadium compounds in the human body. When internalized orally, VCs are subjected to rather strong acidic conditions (pH ≈ 2) in the stomach; the potentially ‘destructive’ effects of such acidity can be circumvented by drug encapsulation. The saliva and the small intestine are slightly alkaline. Therefore, in addition to the expected speciation of the originally applied pro-drug, while crossing these organs with quite distinct pH conditions, the possible loss of ligand(s), oxidation (mainly in the oral cavity) and reduction (mainly in the GI tract) must be considered as unavoidable situations. Additionally, the original VC may have changes upon interaction with low and high molecular mass constituents, which may behave as ligands. These are abundantly present in the nutritional components, as well as in body fluids and secretions of body fluids stimulated by the intake of food. In fact, without participation of ligands, VCs would be converted into simple compounds: (i) VIVO2+ salts, probably then forming insoluble VIVO(OH)2 (for 4 < pH < 7.5), which will not be absorbed and is expelled with the feces, and (ii) VV-compounds, H2VVO4 − (in the pH range 7 ± 0.7) and VVO2 + (pH 2–3). The anion H2VVO4 − is expected to be absorbed in the small intestine and thus can be distributed throughout the body.
In addition to the oral uptake of inorganic vanadium compounds, inhalation and thus pulmonary ingestion of vanadium oxides, present in dust particles, is another possible route [184]. In the lungs, a substantial part of the VOx may be solubilized being converted to vanadates and thus also introduced into the blood stream.
Vanadium coordination compounds VO(carrier)n, with organic carrier ligands, such as those indicated in Fig. 2, Fig. 6, will also be involved in speciation when entering the body. At low and high pH the loss of one or all carrier ligands may take place, the compounds being converted to other distinct complexes or to inorganic vanadium. Alternatively, or additionally, the body's own ligands L′ can induce re-organization in the coordination sphere, forming VOL′n, or mixed (carrier)/L′ species, thus changing absorption properties.
The carrier ligand in VO(carrier)n compounds largely influences the efficacy of a vanadium compound by determining resorption, transport, and stability of the complex, and thus the bioavailability of the true antidiabetic species, possibly vanadate. Underlining the advantageous bioavailability and therapeutic efficacy of VO(carrier)n compounds [113], these have indeed been more effective than inorganic VCs.
The group of Sakurai tested several new drug delivery systems trying direct intestinal administration (e.g. into the ileum), involving enteric-coated capsulation (ECC), gelatine capsules and VO-biopolymer (poly-γ-glutamic acid, γ-pga) complexes, to overcome several of the gastro-intestinal absorption and/or irritation problems with vanadium inorganic salts [14], [17], [185], [186]. Such administration forms indeed improved absorption, ECC and γ-pga delivery systems giving better results than gelatine capsules. However, to our knowledge these promising results with diabetic mice did not lead to any clinical tests with humans.
Transdermal delivery has also been tried in diabetic rats both passively and by iontophoresis, using peroxovanadium, with and without complexation by 1,10-phenanthroline [76], [187], [188]. Increased blood levels of vanadium were achieved, but blood glucose reduction was reported to be modest.
The VO(carrier)n compounds are inherently susceptible to hydrolysis and administration of these VCs is likely to result in loss of ligand, and extensive work has been carried out aimed at determining the actual active species [1], [107], [111], [189], [190], [191], [192], [193], [194], [195], [196]. Studies using BMOV and BEOV demonstrated that upon administration of the complex, the maltol ligand separates from the metal ion, and transport proteins such as transferrin are likely to play key roles in the distribution of vanadium intracellularly [1], [7], [113], [194], [197], [198], [199].
Due to transmetalation processes and cellular compartmentation it is also likely that other ligands in addition to transferrin are involved in the biological effects of V compounds. Thus, most probably the VC taken will partially or completely hydrolyze in the GI and/or in blood serum, and vanadium will bind to bio-ligands, namely to hTF. Either by endocytosis, by diffusion, or by the anion channels (vanadate), vanadium may be uptaken by cells and then be again subjected to speciation and redox leveling. Its availability for subsequent therapeutic or toxicity effects will depend on several factors, namely the amount uptaken, the type of tissue and if the carrier ligand is still present or not, allowing leveling of effects and/or targeting to the sites of therapeutic action. In any case the final intracellular break down of the complex most probably occurs to allow for the display of vanadium's physiological effects.
3.5.2. The mode of action of vanadate in glucose homeostasis
Most cells contain insulin receptors incorporated in their membranes. The IR is a tetrameric trans-membrane protein, being tyrosine kinases, with two α subunits exposed to the outside and the two β subunits (IRβ) exposed to the cytosol. When insulin docks to the α subunits, the tyrosine residues of the intracellular β subunit IRβ become phosphorylated, this corresponding to the activation of IRβ. The cytosolic insulin-signaling cascade is initiated in this way, and involves several post-receptor events, ending up with the activation of the glucose transporter (GLUT4). Thus, the phosphorilation of IRβ triggers the phosphorylation of IR substrates, IRS, intracellular proteins containing tyrosine residues. The activation of the IRS in turn initiates a signaling cascade, in the course of which kinases such as phosphatidylinositol 3-kinase (PI3K), protein kinase B (PKB/Akt), and glycogen synthase kinase-3β (GSK3β) are activated [3], [183]. PKBs, which bind phosphate through Tyr or Ser residues, and then target GLUT4, the glucose transporter [3], [6], which is translocated to the cell surface and intakes glucose. Once in the cytosol, glucose is either broken down to CO2 and H2O, or triggered by activated GSK3β, polymerized to glycogen.
There is increasing consensus that the vanadate-phosphate analogy is relevant for the insulin enhancing action and mode of operation of vanadium compounds, and the most widely accepted mode of action for V compounds thus far is attributed to the inhibition of protein tyrosine phosphatases.
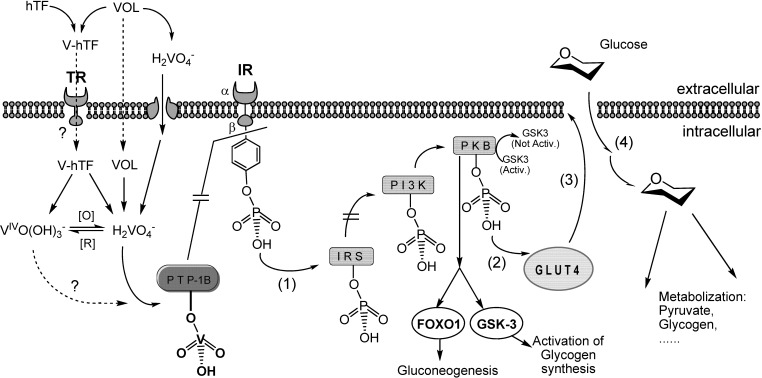
In the absence of insulin or in the case of inadequate insulin response of the IR, phosphorylation of the tyrosine residues in the IRβ subunits of the IRs is counteracted by a protein tyrosine phosphatase (PTP-1B). As a result the signaling breaks down, and so does the cellular uptake of glucose. If vanadate is present inside the cell, as the result of uptake of a vanadium compound (see Fig. 8 ), then VVO2(OH)2 − H2VO4 − (and/or VIVO(OH)3 −) will be available inside the cells. Due to its similarity to phosphate, vanadate binds to the active site of PTP-1B and consequently deactivates (inhibits) it, thus maintaining this enzyme in the phosphorylated state [200], thereby restoring/keeping active the signal transduction paths for glucose uptake [201] (Fig. 8). This possibility was confirmed for e.g. BMOV [202] 1a and for VO(dhp)2 4 [203].
Fig. 8.
Simplified sketch of the possible mechanism of action of VCs. The internalization of glucose by the glucose transporter GLUT4, is triggered by the phosphorylated insulin receptor (IR). In the absence of insulin or insufficient insulin response, protein tyrosine phosphatase 1B (PTP-1B) dephosphorylates the IR, and the glucose intake is stopped. By binding to PTP-1B vanadate may block PTP-1B, this restoring the signaling path. (1) Phosphate remaining bound to IRβ, the insulin receptor substrate (IRS), the phosphatidylinositol 3-kinase (PI3K, which activates protein kinase B, PKB, also known as Akt) remain phosphorilated, thus the signaling path is kept active and (2) activation of the glucose transporter remains in operation, as well as (3) translocation of GLUT4, and (4) cellular uptake of glucose by GLUT4.
VO complexes inhibit PTP-1B and activate phosphatidylinositol 3-kinase → Akt signaling through the enhancement of tyrosine phosphorylation of IRβ and IRS [3]. The particular case of BMOV action was reviewed by Srivastava and coworkers [204] who showed that, besides being better than inorganic V-compounds in inducing phosphorylation of PKB, GSK-3 and FOXO1, a key upstream transducer of BMOV is probably the transactivation of IGF-1R. In this way and through the activation of the PI3K pathway, the phosphorylation of PKB is mediated as well as its downstream targets which regulate glucose transport, glycogen synthesis and gluconeogenesis. The group of Crans [205] also suggested that activation of IR signaling by both insulin and BMOV administration involves increased association of IR with specialized nanoscale membrane micro-domains. The observed insulin-like activity of BMOV (or of its decomposition products) would be due to changes in cell-surface membrane lipid order rather than due to direct interaction with the IR.
In the case of VO(acac)2 it is not certain whether it directly stimulates the IR tyrosine kinase activity, or if its acts indirectly through activation of other tyrosine kinases or inhibition of tyrosine phosphatases [160]. In fact, VO(acac)2 was reported to exhibit synergism with insulin, to regulate the Tyr phosphorylation levels of the IR and of IRS-1 [160], [206] and in some conditions to act as an uncompetitive inhibitor of PTP-1B [206]. It was also reported that VO(acac)2 in the presence of HSA also activates, and much more than VOSO4, the phosphorylation of IRβ [207].
Vanadate may bind to several other protein tyrosine phosphatases inside the cell, inhibiting their action, or to protein kinases, stimulating their action. However, kinase activation does not appear to be involved with the pharmacological effects of vanadium, at least for autophosphorylation of the insulin receptor, other phosphotyrosine phosphatases, or PI3K [207], [208], [209], [210], [211], [212]. Whether VIVO(OH)3 − (≈H3VIVO4 −) may indeed have a role similar to that of VV vanadates (HnVVO4 (3−n)−) is not known. If both can bind to the O-atom of the Tyr side groups, then redox reactions involving the two forms may also be operating (the consequences of these processes cannot be easily anticipated).
The mode of action for V compounds being associated to the inhibition of protein tyrosine phosphatases, it is also relevant to understand that some VCs are reversible inhibitors, whereas others are irreversible by modifying the protein through redox processes [213]. The recent review of Crans et al. [180] of coordination environment of vanadate bound to protein tyrosine phosphatases concluding that actual differences between the coordination environments are very small and presumably less critical than generally anticipated, is an interesting observation whose consequences on the subject under discussion cannot easily be anticipated.
The group of Crans et al. has also proposed that interactions of VCs with cellular redox processes are important in the anti-diabetic effects of VCs [190], VCs cause increases in ROS and RNS (reactive nitrogen species) via multiple mechanisms [214], [215]. Thus, alternatively, or additionally, ROS produced by vanadates (VIVO(OH)3 − or H2VO4 −) may be responsible for e.g. the inhibition of PTP-1B by oxidatively targeting the Cys residue present in it. Cys has to be present in its reduced form for full activity of PTP-1B [6]. The group of Crans also proposed that the compound stability and the ability to interact with cellular redox reactions are key aspects for the insulin-enhancing activity exerted by VCs [5]. Moreover, the possibility that membrane interactions are influenced by the ligand was also suggested, and that such membrane effects may affect uptake and action of the VCs [207], [216].
3.5.3. Experiments with humans
Both inorganic and organic forms of vanadium have been tested in human subjects [217], [218], [219], [220], [221]. Doses used are normally comparatively lower than those in experimental animals, and thus only modest improvements in insulin and glucose metabolism were seen within a few weeks upon the start of the trials. The major drawback reported was gastrointestinal (GI) distress. Since only a small amount of vanadium is absorbed, generally <2% of an oral dose [222], a major goal of research has been the development of appropriate ligands both to improve absorption and thus decrease the dose required, and to attenuate the gastric irritation.
BEOV completed Phase I and then advanced to Phase IIa clinical trials. In the Phase I trial, the safety and tolerability of orally administered increasing single doses between 10 mg and 90 mg of BEOV were tested with 40 non-diabetic volunteers. No adverse effects were found throughout the trial period, which included a comparison study between BEOV and VOSO4 upon comparable levels of vanadium intake, and demonstrated increased uptake and longer residence time for the complex [76], [117].
The results reported for the nonlinear pharmacokinetics of BMOV after oral intake, in which the maximum plasma concentration of vanadium (C max) and the area under the curve (AUC), increased in a non-proportional way with the five oral doses (10–90 mg), and the apparent oral clearance (dose/AUC) decreased significantly as the dose was increased, suggesting that both the oral absorption and first-pass elimination of BMOV are capacity-limited processes through the gastrointestine and liver. Feeding had quite negative effect on availability of BEOV, suggesting ligand substitution by food components [76].
Other major observations in the Phase I study were the safety and tolerability of pharmacologically relevant doses of BEOV: there were no adverse health effects in any of the volunteers; gastrointestinal, liver and kidney function, blood parameters such as hemoglobin and bilirubin levels, all remained within their normal limits throughout the study. Overall bioavailability of vanadium from BEOV was three times that of VOSO4 [223], also consistent with previous data obtained with animals [224]. Noteworthy, vanadium absorption after administration of 75 mg BEOV in the fasted state was approximately 13 times higher than from administration of the same dose in the fed state. Tolerability was comparable in both fed and fasted states, with no clinically significant adverse events or changes in the safety parameters assessed.
The objectives of the Phase IIa trial were to assess the safety and efficacy of a 20 mg (equivalent, on a molar basis, to ∼12.5 mg of hydrated VOSO4, or to ∼3 mg as V), daily dose of orally administered BEOV over a 28-day treatment period in type 2 diabetic individuals, with a 14-day non-treatment follow-up. Glycosylated hemoglobin change, FPG, response to oral glucose tolerance and glycosylated hemoglobin (%HbA1c, percent hemoglobin A1c) were some of the controlled outcome parameters included. Seven subjects were treated with BEOV in the fasted state and two were given a placebo control [76].
Globally a positive treatment effect was observed in most of the treated patients, such that reductions in fasting blood glucose were observed when compared to the two placebo subjects. Response to an oral glucose load generally improved in the treated diabetic subjects compared to controls. Glycosylated hemoglobin increased as a percentage of total hemoglobin in the placebo controls. In the treated subjects the change in %HbAIc during treatment was variable, but two of the patients showed consistent decrease. According to the several measures done, overall, the dose of BEOV, given daily to type 2 diabetic subjects for about one month, clearly affected diabetic symptomatology.
3.5.4. Comments on IM therapeutic vanadium compounds
In terms of pharmacological effects, as emphasized above, one very relevant action of vanadium, as monovanadate, is the inhibition of the active sites of phosphatases and related enzymes involved in the hydrolysis of phosphate esters. Vanadium indeed appears to be a particularly effective inhibitor of the phosphatase that deactivates IR's kinase active site; moreover, it inhibits a variety of phosphatases which control numerous cellular functions. Consequently, it is possible that vanadium may have deleterious side effects at doses used in pharmacological studies [208].
Additionally, partly due to the ability to generate ROS, and also because of the mixture of inhibiting and enhancing effects in several biologically relevant processes, which exert nonspecific effects on different cell structures, VCs have many routes of action, sometimes diametrically opposite. Namely they may have both antitumor and carcinogenic properties. Additionally, not much is known regarding the effect of VCs on the immune system and inflammatory reactions [11].
Given the potential toxicity of vanadium, the biotransformations and biodistribution of vanadium-containing drugs are of outmost importance. A VC that would not readily hydrolyze in the gastrointestinal tract when given orally and could be directed to specific tissues might presumably overcome the mentioned deleterious side effects.
Thus, the potential of VCs to treat diabetes is still an open field. Ideally to avoid or at least minimize adverse side effects, VCs that may be directed to insulin-sensitive tissues (adipocytes, hepatocytes, and skeletal muscle) need to be developed. Partial or complete hydrolysis of the chelate ligands in these tissues could then release the bioactive form locally.
Whether the tight connection between adverse effects and beneficial effects can be solved is not yet established, but if the toxic side effects of vanadium could be avoided it would have high potential pharmacological interest for the treatment of diabetes.
3.6. Vanadium in the treatment of cancer
In recent years the anticancer properties of VCs have been noticed, but the underlying mechanisms are also not well understood.
The main targets for the antitumor effects of vanadium are the disruption of cellular metabolism through the generation of ROS, the alterations of cellular organelles such as lysosomes, mitochondria, the spindle proteins such as actin and tubulin, some signal transduction pathways, cyclins and caspases which in turn play a role in cell cycle arrest and apoptosis. Moreover, cell proliferation can also be disturbed by genotoxic effects of vanadium exerted at the nuclei of the cells and on DNA damage.
In different cancer cell lines, some VCs act inhibiting cell proliferation in the whole range of tested concentrations. These VCs were then evaluated as potentially antitumor agents [14], [44] Identification of appropriate models for in vivo and in vitro preclinical testing of inhibitors of tumor angiogenesis and progression is vital to the successful development of anticancer therapeutics. Although the focus is on human molecular targets, most preclinical in vivo efficacy testing has been done with mice.
In an interesting study [225], hepatic pre-neoplasia was induced in male Sprague-Dawley rats, and the levels of modified DNA bases 8-hydroxy-2′-deoxyguanosine (8-OHdG), a potential marker involved in the initiation of carcinogenesis, were measured upon supplementation of NH4VO3 in drinking water, at a dose of 0.5 ppm during 4 consecutive weeks. The formation of 8-OHdG decreased in the pre-neoplastic rat liver. Moreover, in a long-term DEN plus PB regimen, vanadium limits in situ MT expression with a concomitant decrease in MT immunoreactivity. Treatment by NH4VO3 restored hepatic levels of essential trace elements and decreased nodular incidence and nodule multiplicity in the rats treated with DEN plus PB. Globally, the study provided evidence supporting the chemopreventive potential of vanadium in limiting neoplastic transformation during the preneoplastic stages of hepatocarcinogenesis in rats [225].
Tumor cells derived from human placenta HTB-14, the murine tumor cell line MDAY-D2 and endothelium murine cells EDMA were inhibited by 5–50 μM sodium vanadate; the cells in active proliferation were more sensible to the inhibitory action of the VV-derivatives [226].
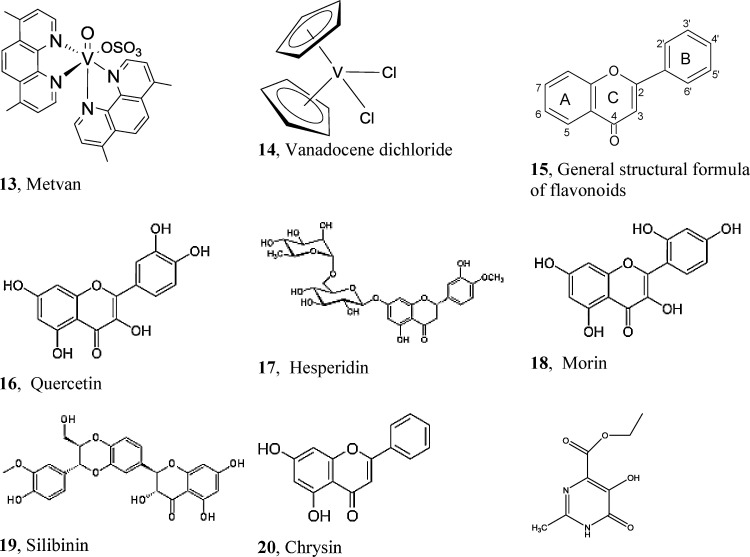
Several VCs with potential application in cancer treatment have been synthesized. Among VIV-derivatives, Metvan (Fig. 9 ) was identified as one of the most promising multitargeted anticancer VC with apoptosis-inducing activity. At nanomolar and low micromolar concentrations, Metvan induces apoptosis in different tumoral cell lines of human origin such as leukemia cells, multiple myeloma cells and solid tumor cells derived from glioblastoma, breast cancer, ovarian, prostate and testicular cancer patients [14], [114], [227], [228]. Another advantage of Metvan is that it is highly effective against ovarian cancer and testicular cancer cell lines resistant to cisplatin. Its action is probably associated with the generation of ROS and depletion of glutathione together with a loss of mitochondrial transmembrane potential. The metastatic properties of some tumor cells such as human malignant glioblastoma and breast cancer were also highly inhibited or impaired by Metvan. Moreover, Metvan showed advantages of significant antitumor activity, delayed tumor progression and increased survival time in models of severe combined immunodeficient mouse xenograft of human malignant glioblastoma and breast cancer.
Fig. 9.
Several anti-tumor vanadium complexes or compounds which form anti-tumor vanadium complexes [14], [114], [143], [227], [228], [230], [236], [237], [239], [240], [241], [242], [243], [244], [245].
The broad spectrum anticancer activity of Metvan together with favorable pharmacodynamic features and lack of toxicity emphasizes that this VIV-compound has potential to be the first vanadium complex as an alternative to platinum-based chemotherapy [114].
On the other hand, Vanada- and niobatricarbadecaboranyl monohalide complexes proved to be potent cytotoxic agents against murine and human leukemia and lymphoma growth as well as HeLa suspended uterine carcinoma [229]. The vanadium complex decreased the proliferation of several tumor cell lines: KB nasopharynx, Hepe liver, HCT-8 ileum and 1-A9 ovary solid carcinomas. In human HL-60 promyelocytic leukemia cells the compound reduced the growth of KB nasopharynx, Hepe liver, HCT-8 ileum and 1-A9 ovary solid carcinomas. In these cells DNA and purine de novo syntheses were significantly inhibited or suppressed activities of DNA polymerase alpha and PRPP-amido transferase, but the activity of topoisomerase I and II was not inhibited. The complex activated caspases 3, 6 and 9 which conveyed the cells to apoptosis.
Another interesting group of compounds with antitumoral properties are three vanadocene derivatives (see e.g. 14 in Fig. 9) [230]. These induce apoptosis in human cancer cells by an as yet unknown mechanism. In human HepG2 cells, a several vanadocene complexes triggered activation of the c-fos promoter without affecting the minimum promoter containing p53 response elements or the GADD45 promoter. These results indicated that the apoptotic signal of vanadocenes is not triggered by primary DNA damage and it does not require p53 induction, thereby disproving the hypothesis that it mechanistically resembles the cytotoxic action of cisplatin [231]. On the other hand, vanadocenes are efficient agents against human testicular cell lines [232].
In a systematic study of several metallocenes containing vanadium (vanadocenes as well as of several other metals) were tested on the human testicular cancer cell lines Tera-2 and Ntera-2. Interestingly, only the VIV-containing metallocenes exhibited significant cytotoxicity leading the tumoral cells to apoptosis within 24 h [232].
To protect biomolecules against the attack of free radicals and/or to suppress the resultant injure, there are numerous natural free radical scavengers and antioxidants. Among antiradicals, flavonoids are a group of natural occurring polyphenols predominantly synthesized by higher plants [233]. Chemically, flavonoids have the general structure corresponding to 15 in Fig. 9, with a 15-carbon skeleton, consisting of two phenyl rings (A and B) and one heterocyclic ring (C). This carbon structure is frequently abbreviated as C6-C3-C6.
Metal chelation may have important roles in the improvement of both the antioxidant properties and the antitumoral behavior of flavonoids [234] and this binding influences the antioxidant and antitumoral properties of flavonoids.
The phenolic —OH substituents and the electronic resonance between A- and B rings of flavonoids are the basis for their antioxidant and biological activities. Besides, the —OH groups located on B ring are the most significant factors that influence their scavenging properties toward the reactive oxygen species (ROS) [234], [235].
The antioxidant and antitumoral properties of a series of flavonoids (quercetin, hesperidin, morin, silibinin, chrysin) and their complexes with oxidovanadium(IV) was investigated. In a model of two murine osteoblastic cell lines in culture, quercetin (16), displayed antitumor properties since it caused an inhibition of cell proliferation in the tumoral cell line UMR195 while it did not show any deleterious action in the non-transformed osteoblasts MC3T3-E1. Nevertheless, the VIV-complex was more deleterious in the tumoral osteoblasts [236].
The VIV-complex with hesperidin (17), when tested on the osteosarcoma cell line UMR106 as well as on the Caco-2 human colon adenocarcinoma, showed promissory anticarcinogenic properties [237].
Morin (18), is also a natural compound with attracting healthy properties. Epidemiological studies already have indicated that adequate intakes of flavonoid-rich foods may reduce the risk of coronary heart disease and certain cancers [238]. The antioxidant and anticancer activity of morin and the improvement of these capacities were investigated by chelation to oxidovanadium(IV), and the antiproliferative effects, morphological changes and ROS generation in normal (mouse) MC3T3-E1 and tumoral (rat) UMR106 bone cell lines in culture were studied. Morin and its VIV-complex behaved as good antioxidant agents for some of the radicals and that the complexation improved this behavior. A considerable variation in sensitivity was observed in the breast cancer cells but non-specificity was found for the treatment of osteosarcoma. Moreover, the compounds did not affect the normal proliferation of the breast epithelial mammal cells. The mechanistic studies demonstrated that the complex did not generate ROS and did not produce any damage of DNA. The plasmatic membrane was observed to be damaged only in the SKBR3 cell line. In contrast, the perturbation of the mitochondrial membrane potential and the activation of caspase 3/7 for the breast tumor cells revealed an apoptotic cell death process.
Moreover, two new complexes of flavonoids with oxidovanadium(IV) have thoroughly been investigated in the MG-63 cell line, a very good model for osteosarcoma, the most common form of bone cancer and to study the effects of potential anticancer drugs. Silibinin or silybin (19) is one of the popular dietary supplements that has been extensively studied for its antioxidant, hepatoprotective and anti-cancer properties. Silibinin has also demonstrated potent antiproliferative effects against various malignant cell lines [239], [240], [241], [242].
It was reported that in UMR106 rat osteosarcoma cell line silibinin caused a decrease of cell proliferation ca. 40% at 100 μM, while its effect on the non-transformed osteoblasts MC3T3-E1 was less than 10% at the same concentration [243]. In MG-63 human osteosarcoma cell line, its VIV-complex, Na2[VIVO(silibinin)2]·6H2O, VOsil, inhibited the viability of the osteosarcoma cells in a dose dependent manner with a greater potency than silibinin or the inorganic VIV. The VOsil also displayed a concentration effect both in cyto- and genotoxicity processes. Distribution of the redox status of the cells determined through the increase in ROS level and the decrease in GSH/GSSG ratio were the main events involved in the deleterious effects of the complex on tumoral osteosarcoma cells. Moreover, the complex caused cell cycle arrest and the activation of caspase 3 that triggered apoptosis [244]. Comparing the antitumoral effectiveness of VOsil, with those of the reference metallo-drug (cisplatin) in MG-63 cells: (a) at the lower tested concentrations (2.5 and 10 μM), cisplatin and VOsil did not have any effect on cell viability; (b) at 25 μM, cisplatin and VOsil were similar as inhibitory agents of cell proliferation, since both of them produced ca. 15% decrease in cell viability; (c) at the middle tested concentration (50 μM), VOsil was less deleterious than cisplatin (60% versus 23% survival), whereas at 100 μM the differences were 33% versus 21% of surviving cells, respectively.
Another interesting flavonoid complex with potential antitumor properties is with chrysin (20): [VIVO(chrysin)2EtOH]2 (VOchrys). Oxidovanadium(IV), chrysin and VOchrys caused a concentration-dependent inhibition of cell viability in MG-63 osteosarcoma cells, VOchrys being the strongest antiproliferative agent in this cell line [245] having a lower IC50 value (16 μM) than cisplatin (43 μM) in this cell line (Etcheverry, unpublished results).
From the comparison of the antitumoral actions of VOchrys, VOsil and cisplatin in the human osteosarcoma cell line MG-63 it could be concluded that VOchrys was the strongest antiproliferative agent, ROS and the alterations in the GSH/GSSG ratio being relevant for the main mechanism of action of VOchrys. When the oxidative stress was diminished by additions of ROS scavengers (vitamin C plus vitamin E) or GSH, the viability experiments demonstrated beneficial effects. Besides, VOchrys caused disruption of the mitochondria membrane potential (MMP), increased the level of caspase 3 and produced DNA fragmentation, as measured by the sub-G1 peak in cell cycle arrest experiments. All these events finally convey the human osteosarcomal cells to apoptosis.
Several other organically chelated vanadium compounds potentiate pharmacological actions over those of simple vanadium salts [246], [247]. For example, vanadium complexes with quinoline and pyridinone ligands were synthesized and tested for the antiproliferative activity toward U937 cells. The antiproliferative activity of quinoline complexes toward U937 cells showed little dependence on the length and shape of the alkyl chain. In contrast, a good correlation was found between the IC50 values and partition coefficients (log P) values of pyridinone complexes. One member of the latter group showed an IC50 value smaller than cisplatin. Besides, this complex triggered apoptosis but through a pathway independent of caspase activation since inhibitors of caspase-3, -8, and -9 did not affect the antiproliferative activity [248].
The complex VO(acac)2 was reported to block cell cycle progression permanently at G1 phase in a dose- and time-dependent manner in HepG2 cells. This was further evidenced by the growth regulatory signals during the G1 stage. Neither O2 • − nor the H2O2 levels were increased, but perhaps not surprisingly the levels of phosphorylated extracellular signal-regulated protein kinase (ERK) and that of Akt were highly activated. Globally, the authors suggested that VO(acac)2-induced proliferation inhibition is caused by G1/S cell cycle arrest, which resulted from the decreased level of phosphorylated pRb in its active hypophosphorylated form, via a highly activated ERK signal in HepG2 cells. The results presented here provided new insight into the development of vanadium compounds as potential anticancer agents [249].
A vanadium(III)-l-cysteine compound showed significant anti-tumor effects on 3,4-benzopyrone induced soft tumors [250], [251]. This VIII-Cys compound, or its oxidation products, at a concentration of 10−3 M, showed inhibition of NEP activity as potent as thiorphan at 10−3 M, while VIVOSO4 in the same concentration exhibited less than 50% inhibitory activity than that of thiorphan at 10−3 M. Additionally, the antimetastatic effects of the VIII-Cys compound, Cys and VIVOSO4 were examined on Wistar rats treated with 3,4-benzopyrene. The results revealed that 1 prevents significantly lung metastases (only 9.5% of animals treated with the VIII-Cys compound showed metastases), whereas 47–52% of the rats of the control group and those treated with Cys and VIVOSO4 exhibited metastases [115].
Several VV-complexes were screened for their potential cytotoxic activity in two different cell lines [252]. The toxic effects were incubation time and concentration dependent and specific for each compound and type of cells. The HeLa tumor cells were more sensitive to drug effects than the 3T3-L1 fibroblasts. According to the IC50 values and the results on reversibility to drug effects, the VV-MHCPE system (MHCPE, 20 in Fig. 9) show higher toxicity in the tumor cells than in non-tumor cells, this indicating potential antitumor activity.
Another interesting group of vanadium compounds are complexes of oxidovanadium(IV) with ligands that hold multiple donor atoms able to coordinate with metal centers. Strong chelating ligands are very important in living systems since they can facilitate the uptake and transport of metals inside the cells. The role of low-molecular-mass compounds involving multidentate oxygen donors is of great value. Oxodiacetate (oda), O(CH2COO−)2, is a very versatile ligand since the O donor atoms can be placed on different orientations. Oda holds an OOO donor group and can coordinate metal ions by forming chelate rings [253]. Vanadium compounds once absorbed in mammals are stored mainly in bones with a half-life of ca. 5 days [254].
These results enhanced the scientific interest in the biological effects of vanadium in hard tissues. VIVO-complexes of oda, VIVO(oda) 22, as well as VIVO(oda)(bipy) 23 and VIVO(oda)(phen) 24 (Fig. 10 ), displayed important effects in bone related cells in culture. All these compounds were tested on two osteoblast-like cell lines in culture (MC3T3E1 derived from mouse calvaria and UMR106 derived from a rat osteosarcoma cells). VIVO(oda) caused inhibition of cellular proliferation in both cell lines, but the cytotoxicity was stronger in the normal (MC3T3E1) than in the tumoral (UMR106) osteoblasts. The compound also produced some effects on cell differentiation tested through the specific activity of alkaline phosphatase of the UMR106 cells because they expressed a high activity of this enzyme. As with other vanadium compounds, VIVO(oda) behaved as an inhibitory agent of osteoblast differentiation [255]. Besides the dose-dependent decrease in cell proliferation, VIVO(oda) caused morphological and actin alterations.
Fig. 10.
Structural formulae of VIVO-oda compounds [259].
VIVO(oda) increased the level of ROS which correlated with a decreased in GSH/GSSG ratio. Besides, VIVO(oda) induced a dissipation of the mitochondria membrane potential (MMP) and promoted an increase in ERK cascade phosphorylation, which is involved in the regulation of cellular death and survival. All the effects were more pronounced in MC3T3-E1 than in UMR106 cells. Using specific inhibitors of different intracellular signaling pathways, it could be demonstrated that VIVO(oda) stimulated ERKs phosphorilation by induction of upstream kinases of ERK pathway by free radicals. Clearly, the results suggested that the apoptotic actions of this complex were mediated by the involvement of oxidative stress, MMP alterations and ERK pathway [256].
Moreover, the related VO(oda)(phen) 24 in the same osteoblastic model caused inhibition of cellular proliferation in both cell lines (MC3T3E1and UMR106), but the cytotoxicity was stronger in the normal than in the tumoral osteoblasts [257]. On the contrary, VO(oda)(bipy) 23 was statistically stronger in the tumoral cells [258]. More recently the effects of VO(oda) (22), VO(oda)(bipy) (23) and VO(oda)(phen) (24) (Fig. 10) on MG-63 human osteosarcoma cell line were reported [259].
All three complexes depicted in Fig. 10 caused a concentration dependent inhibition of cell viability. The antiproliferative action of 24 could be observed in the whole range of concentrations (from 2.5 to 100 μM), while that for 22 and 23 showed a decrease of cell viability only at higher concentrations. VO(oda)(phen) caused strong cytotoxicity affecting lysosomes and mitochondria metabolism from the lowest tested dose (2.5 μM), while VO(oda) and VO(oda)(bipy) produced these effects at higher concentrations. No DNA damage could be detected by the comet assay with VO(oda) at 2.5–10 μM, while VO(oda)(phen) and VO(oda)(bipy) induced DNA damage at 2.5 and 10 μM, respectively (p < 0.01). ROS levels increased at 10 μM of VO(oda)(phen) and only at 100 μM of VO(oda) or VO(oda)(bipy). Moreover, VO(oda)(phen) and VO(oda)(bipy) triggered apoptosis as a relevant mechanism of cell death.
Nuclease activity of the three compounds 22–24 revealed that DNA cleavage caused by VO(oda)(bipy) and VO(oda) was similar, while VO(oda)(phen) showed a stronger effect. Within this series of compounds a good relationship between the bioactivity of the complexes and their structures is noticed. VO(oda)(phen) presented the most potent antitumor action in human osteosarcoma cells followed by VO(oda)bipy and then by VO(oda) according to the number of intercalating heterocyclic moieties [260].
Several VV-compounds were reported to exhibit antitumoral properties in various experimental systems [11], [14], [44], [201], [261] and Korbecki et al. [11] critically reviewed some of these cases.
One interesting report used in vitro and in vivo models to characterize the anti-tumor properties of VV-complexes with hydroxilamido and amino acids (Fig. 11 ) [262].
Fig. 11.
Structure of VV-hydroxylamido-amino acid complexes [262].
The complexes VO(NH2O)2(Val) and VO(NH2O)2(Met) inhibited the proliferation of two osteoblast cell lines in culture, MC3T3-E1 and UMR106 with different potency. Besides, the complexes were deleterious on the viability of an in vivo model of zebrafish eggs. A good correlation between the in vitro and in vivo experiments was supported by the IC50 values for both complexes, considering the results in the osteoblast studies and the IC50 values obtained with the in vivo model of toxicity (FET test). Moreover, the cytotoxicity in cell cultures showed a decrease in mitochondrial enzymatic activities and DNA damage could also be observed by comet formation in MC3T3-E1 cells while this effect could not be observed in the osteosarcoma cells. Additionally, VO(NH2O)2(Val) greatly enhanced ROS levels in MC3T3-E1 and UMR106 cells, suggesting an important role for oxidative stress in the toxicity mechanism of this compound. Since VO(NH2O)2(Val) was more stable as well as less cytotoxic, both in cell cultures and in the in vivo model than VO(NH2O)2(Met), it is considered a promissory compound to be further investigated as a potential antitumoral agent. Moreover, these results emphasize the potential of zebrafish as an attractive complementary platform for drug development.
Globally the results concerning the anti-tumor properties of VCs are promising with respect to their possible introduction into the therapy due to their low IC50 (several μM, depending on cell line and VC), antiproliferative and proapoptotic effects. There are many possible targets of VCs. The text above, the review of Korbecki et al. [11] and references therein summarize most of the relevant data.
On the other hand, VCs may also have carcinogenic properties and stimulate tumor development, as well as anti-apoptotic properties on cancer cells, thus several mechanisms also existing for VCs presenting carcinogenic action.
The effects of vanadium compounds depend on many factors, namely the pro-apoptotic or anti-apoptotic effect of VCs depend largely on the cell type, but also on the type of VC and its dose. The key protein, defects of which may drastically change the effects of VCs, is p53 (a large number of tumor cell types have defects in the gene encoding this protein) [11]. In p53-defective cells (tumor cells or non-tumor p53-knock out cells), VCs inhibit the cell cycle and thus induce apoptosis [263]. Activation of NF-κB by ROS generated by VCs enhances the apoptotic effect [264]. In contrast, in p53-functional cells, disturbed phosphorylation of p53 leads to inhibition of apoptosis [265]. In addition, VCs stimulate the cell cycle, thus inhibiting apoptosis, as both processes are mutually connected [263]. Moreover, NF-κB activation inhibits apoptosis of tumor cells. One relevant issue is that frequently VCs cause much more DNA damage in tumor cells compared to non-tumor cells when present at the same levels. Extensive DNA damage leads to apoptosis of tumor cells while a less intensive damage evoked by vanadium compounds in non-tumor cells may stimulate synthesis and activation of repair enzymes, thus protecting those cells from apoptosis [11]. The above processes promote tumor cell growth at early stages of the disease and have an antitumor effect in the advanced stages of cancer. In fact, there are studies in animals treated with carcinogens that suggest that VCs used at low levels have selective effects on the tumor cells [266], [267].
3.7. Other medicinal uses
3.7.1. Antiparasitic vanadium compounds
In 2011 a comprehensive review on the current status of the potentiality of vanadium compounds in the treatment of neglected tropical diseases and its background was published [9]. This work reviewed the research carried out till end of 2010 on VCs as prospective agents against Trypanosoma cruzi, Leishmania spp. and Entamoeba histolytica, the protozoan parasites that are the causative agents of American trypanosomiasis (Chagas disease), leishmaniasis and amoebiasis, respectively; a brief description of the diseases was included, thus it is the recommended reference for this purpose. This review was the kickoff for further related work and was followed by the inclusion of the topic in recent reviews and thematic chapters [6], [10], [268].
The present review includes further work carried out on the topic. Although research efforts on the medicinal chemistry of vanadium have mainly focused whether on improving biodistribution and tolerability of the vanadium insulin-enhancing core or on developing potential anti-tumor compounds, in recent years some research groups dedicated efforts to the development of prospective agents toward the potential treatment of parasitic diseases produced by trypanosomatid and E. histolityca parasites.
Vanadium compounds evaluated in vitro on a related protozoan parasite, Trypanosoma brucei, will be also addressed in the current review. Human African trypanosomiasis (sleeping sickness) is caused by parasites of the T. brucei complex (e.g. T. brucei gambiense and T. brucei rhodesiense). It is transmitted by the bite of the tsetse fly. It represents a major disease burden in sub-Saharan regions of Africa. It is also considered by the World Health Organization as one of the seventeen neglected tropical diseases together with Chagas disease and leishmaniases. The disease affects mostly poor populations living in remote rural areas of Africa. Untreated, it is usually fatal. Although controlled in the 1960s, the disease re-emerged due to lack in sustained surveillance in the 1980s. More recently, upon increased control the number of new reported cases has been diminishing slowly.
Only four drugs are registered for the treatment of human African trypanosomiasis: pentamidine, suramin, melarsoprol and eflornithine. They give rise to undesirable collateral toxic effects, show limited and variable efficacy depending on the type or stage of the disease and suffer from parasite's development of resistance [269], [270], [271], [272], [273].
In respect to anti-T. cruzi agents a typical medicinal chemistry research was carried out to rationally design prospective antitrypanosomal compounds that could be hits or lead compounds for further drug development. This research involved the design of two families of VIVO-compounds including polypyridyl ligands (NN) bearing DNA intercalating capacity (Fig. 12 ). This strategy pointed out at DNA as a potential parasite target. It was inspired in the fact that compounds able to generate nucleic acids modifications have received high attention in anti-tumor drug design and in the reported metabolic similarities between tumor cells and highly proliferative trypanosomatid parasites. For instance, some compounds that efficiently interact with DNA in an intercalative mode exert anti-trypanosomal activity [274], [275], [276].
Fig. 12.
Polypyridyl compounds selected as bidentate potentially intercalating ligands: dipyrido[3,2-a: 2′,3′-c]phenazine (dppz), 2,2′-bipyridine (bipy), 1,10-phenanthroline (phen), 5-amine-1,10-phenanthroline (aminophen), 5,6-epoxy-5,6-dihydro-1,10-phenanthroline (epoxyphen), 1,10-phenanthroline-5,6-dione (phendione), [1,2,5]thiadiazolo[3,4-f][1,10]phenanthroline (tdzp), included in complexes with T. cruzi activity.
Through this strategy two families of structurally related compounds have been synthesized, fully characterized and evaluated on the parasite: [VIVO(SO4)(H2O)2(NN)] and [VIVO(L-2H)(NN)] (Fig. 13 ).
Fig. 13.
Schematic general structure of the two newly synthesized VIVO families of compounds [117], [118], [277], [278], [279], [280].
The activity on T. cruzi of the [VIVO(SO4)(H2O)2(NN)] series is shown in Table 1 . The NN ligands showed activity on the parasite [117], [277]. Their coordination to the VIVO core led to activities in the same range than the reference tripanocidal drug Nifurtimox (IC50 6.0 μM) and to slight changes in activity for the dppz, phendione and epoxyphen complexes; noteworthy, the tdzp complex showed a significantly improved antitrypanosomal activity in respect to free tdzp. Although the complexes are structurally analogous, the nature of the NN ligand included in the coordination sphere was determinant for the antitrypanosomal activity.
Table 1.
In vitro activity on T. cruzi (Dm28c strain or Tulahuen 2 epimastigotes) of the [VIVO(SO4)(H2O)2(NN)] and [VIVO(L-2H)(NN)] complexes [117], [118], [277], [278], [279], [280].
| Compound | IC50 ± SD (μM) | Compound | IC50 ± SD (μM) |
|---|---|---|---|
| [VIVO(SO4)(H2O)2(dppz)] | ca. 3 | [VIVO(L1-2H)(phen)] | 2.0 |
| [VIVO(SO4)(H2O)2(tdzp)] | 15.6 ± 4.88 | [VIVO(L2-2H)(phen)] | 3.1 |
| [VIVO(SO4)(H2O)2(phendione)] | 7.24 ± 5.27 | [VIVO(L3-2H)(phen)] | 2.3 |
| [VIVO(SO4)(H2O)2(epoxyphen)] | 21.91 ± 9.94 | [VIVO(L4-2H)(phen)] | 1.6 |
| [VIVO(L5-2H)(phen)] | 3.8 | ||
| [VIVO(L1-2H)(bipy)] | 73 | [VIVO(L1-2H)(aminophen)] | 0.50 ± 0.02 |
| [VIVO(L2-2H)(bipy)] | 84 | [VIVO(L2-2H)(aminophen)] | 0.27 ± 0.09 |
| [VIVO(L3-2H)(bipy)] | 76.2 ± 9.7 | [VIVO(L3-2H)(aminophen)] | 0.52 ± 0.02 |
| [VIVO(L4-2H)(bipy)] | 161 ± 57 | [VIVO(L4-2H)(aminophen)] | 0.51 ± 0.02 |
| [VIVO(L5-2H)(bipy)] | 58.0 ± 14.8 | [VIVO(L5-2H)(aminophen)] | 0.51 ± 0.02 |
| [VIVO(L1-2H)(dppz)] | 13 | [VIVO(L1-2H)(epoxyphen)] | 1.0 ± 0.1 |
| [VIVO(L2-2H)(dppz)] | 19 | [VIVO(L2-2H)(epoxyphen)] | 0.9 ± 0.2 |
| [VIVO(L3-2H)(dppz)] | 4.70 ± 3.35 | [VIVO(L3-2H)(epoxyphen)] | 1.2 ± 0.3 |
| [VIVO(L4-2H)(dppz)] | 12.7 ± 8.27 | [VIVO(L4-2H)(epoxyphen)] | 3.9 ± 0.6 |
| [VIVO(L5-2H)(dppz)] | 5.05 ± 1.60 | [VIVO(L5-2H)(epoxyphen)] | 1.1 ± 0.3 |
IC50, concentration required for inhibition of 50% of culture growth.
Atomic force microscopy (AFM) studies using pBR322 plasmid as model molecule confirmed that the complexes modified the tertiary structure of the plasmid which indicates that they interact with DNA (Fig. 14 ). Although only a reduced series of structurally related compounds was compared and AFM studies alone are not enough to judge the ability of the compounds to affect DNA, a correlation between the anti-T. cruzi activity and the intensity of the effect observed on the plasmid appeared to exist [277].
Fig. 14.
AFM images showing the modifications suffered by pBR322 plasmid DNA (a) due to the interaction with (b) [VIVO(SO4)(H2O)2(tdzp)] [277].
Additional parasite targets could be affected by the complexes due to the presence of the VIVO core. Although not demonstrated it was hypothesized that coordination to vanadium may in some way improve bioavailability by acting as a carrier system, improving cell uptake due to changes in solubility and/or lipophilicity of the phenanthroline derivatives upon coordination.
A larger series of heteroleptic bioactive [VIVO(L-2H)(NN)] compounds was obtained where L are tridentate salicylaldehyde semicarbazone derivatives depicted in Fig. 15 , where NN is either bipy, dppz, phen, epoxyphen or aminophen [278], [279], [280].
Fig. 15.
Tridentate salicylaldehyde semicarbazone derivatives selected as coligands in [VIVO(L-2H)(NN)] compounds [278], [279], [280].
This series includes several compounds with very interesting biological properties. The in vitro anti-T. cruzi activity resulted mainly dependent on the nature of the NN ligand and almost independent of the chemical nature of the substituents R1 and R2 in the salicylaldehyde moiety (Table 1 and Fig. 16 ). The approximate order of activity was [VIVO(L-2H)(bipy)] < [VIVO(L-2H)(dppz)] < [VIVO(L-2H)(phen)] < [VIVO(L-2H)(epoxyphen)] < [VIVO(L-2H)(aminophen)]. The semicarbazone ligands showed IC50 values higher than 100 μM and the VIVO-compounds were in general much more active than the polypyridyl compounds included in their coordination spheres. Seventeen of the twenty-five complexes were more active than the Nifurtimox (IC50 (μM): 6.0 (Dm28c strain), 7.7 (Tulahuen 2 strain)). The compounds including aminophen were the most active ones showing IC50 values in the submicromolar range and being 9.6- to 18.5-fold more active than aminophen itself, as measured by their IC50 values in μM units. The selectivity to the parasite in respect to mammalian cells was also studied for the most relevant compounds. The phen compounds showed selectivity indexes SI (IC50 mammalian cell model/IC50 T. cruzi) ranging between 38 and 596 using THP-1 human acute monocytic leukemia (ATCC) cells as mammalian cell models [278]. The epoxyphen and aminophen compounds showed SI values (IC50 J774 murine macrophages/IC50 T. cruzi in the range 10–41 and 96–185, respectively. Furthermore, none of the aminophen complexes displayed relevant toxicity against J-774 macrophages at doses down to 50 μM, depicting in all cases much higher SI values than the corresponding value for aminophen itself. According to accepted guidelines for the development of anti-T. cruzi drugs these compounds deserve further studies to establish their therapeutic potentiality [281].
Fig. 16.
Graphical comparison of the IC50 values on T. cruzi of the five families of [VIVO(L-2H)(NN)] compounds: (a) NN = bipy, dppz and phen, IC50 Nfx (Nifurtimox) (Dm28c strain) 6.0 μM; (b) NN = epoxyphen and aminophen, IC50 Nfx (Tulahuen 2 strain) 7.7 μM.
Source: Values taken from Ref. [281].
Having twenty-five structurally related compounds, a QSAR (quantitative structure activity relationship study) study was carried out to get a design guide for further antitrypanosomal drug development. This study is particularly relevant since QSAR studies are still not very common in inorganic medicinal chemistry research due to the difficulty to get a high number of structurally related compounds [280]. Lipophilic effects are among the factors controlling transport to and interaction with biological receptors. Lipophilicity was experimentally determined using reversed-phase TLC experiments where the stationary phase, precoated TLC-C18, may be considered to simulate lipids of biological membranes or receptors, and the mobile phase, MeOH:DMF:Tris–HCl buffer pH 7.4 (85:5:10, v/v/v), resembles the aqueous biological milieu. The R f values in this system were converted to R M via the relationship: R M = log10[(1/R f) − 1] and reported. The complexes resulted more lipophilic than the NN ligands.
A statistically satisfactory correlation between IC50 on T. cruzi and the parameter R M and the indicator variable I col, a structural descriptor of the NN co-ligands, was obtained [280]:
The indicator variable I col takes into account the potentially different biological behavior of the selected NN scaffolds. The highly electrophilic epoxyphen may react with biological nucleophiles from proteins and DNA through the epoxide-moiety, while the nucleophile aminophen may react with biological electrophiles through the amino-moiety. Moreover, the π-expanded systems dppz, phen and phen derivatives may also intercalate to DNA.
In summary, the equation highlights the relevance of the lipophilic parameter on the desired bioactivity. Additionally it also reflects the significance of the structure of the co-ligand on the anti-T. cruzi effect. Accordingly complexes with DNA-modifying co-ligands with the higher I col values, i.e. aminophen and epoxyphen, were the most cytotoxic for the parasite.
A parabolic relationship between biological response and lipophilicity was obtained and, from this correlation an optimum R M value was determined which may be a design guide for future development of new compounds bearing a better biological profile.
When trying to access the mechanism of action of the compounds, DNA interaction of the complexes was assessed through different techniques: agarose gel electrophoresis, viscosity measurement of DNA solutions and AFM [280].
In addition, a metabolomic study was performed by incubating the parasite with the two most active VIVO-compounds [VIVO(L2-2H)(epoxyphen)] and [VIVO(L2-2H)(aminophen)] in order to study the changes in relevant biochemical pathways promoted by them. In particular, the consumption of glucose by trypanosomatid protozoa is characterized by the excretion of reduced products such as succinate, pyruvate or lactate not only in anerobiosis, but also under aerobic conditions. A compartmentation of the glycolytic enzymes, most of which are placed in an organelle called the glycosome, and a relative inefficacy of the respiratory chain has been described. Previous research on T. cruzi epimastigotes metabolism using [l-13C] glucose as substrate described the detection by 13C NMR of the carboxylates succinate, pyruvate, acetate and lactate and the amino acids Ala and Gly as main catabolites of glucose [282]. Therefore, the modifications in the excreted metabolites produced by the incubation of the parasite with the selected VIVO-compounds were followed by 1H NMR spectroscopy. An increment of excreted Gly, acetate and Ala was observed. Acetate and Gly are catabolites produced in the parasite single mitochondrion. Alanine is produced in the cytosol of the parasite cell from pyruvate that is originated in a specific parasite organelle called glycosome, via phosphoenolpyruvate. This pyruvate is also used, into the mitochondrion, in the like-Krebs cycle (Fig. 17 ) [283]. Consequently, an increment in the amount of Ala could be indicative of an increment of cytosolic pyruvate due to a deficient mitochondrion uptake emerging of deleterious effects on mitochondrion integrity. Consequently, the mitochondrion could be pointed out as a potential target for these complexes [280].
Fig. 17.
Schematic representation of carbon source metabolism in trypanosomes.
The presence of the VIVO core together with the semicarbazone and NN co-ligands seemed to be determinant of the activity. The [VVO2(L-H)] complexes, where L are the salicylaldehyde semicarbazone derivatives L1–L5 showed no antitrypanosomal activity [279].
Concerning the question if the vanadium center is needed in order to have bioactive compounds, the previously obtained compounds with aromatic amine N-oxide derivatives showed a significantly higher activity for the VIVO-compounds that for analogous CuII and PdII compounds [284]. These results are in agreement with the previously described observations which suggested that the complexes probably not only interact with DNA but also affect relevant metabolic pathways in the parasite.
Recently, the genomes of the parasites T. cruzi, T. brucei and Leishmania major have been decoded and showed a high degree of similarity [285]. Thus, clinically useful drugs affecting common enzymatic targets or metabolic pathways in these trypanosomatid parasites are being sought [286]. The emerging broad-spectrum chemical scaffolds could offer an innovative and less expensive approach for anti-parasitic drug discovery [287]. Therefore, some of the VIVO-compounds described above were also tested either on T. brucei or on different Leishmania species in order to recognize metal-based moieties that could inhibit T. cruzi or T. brucei or Leishmania spp. growth by acting on common targets.
The series of five [VO(L-2H)(phen)] compounds were evaluated on L. panamensis and L. chagasi promastigotes and intracellular amastigotes. Only [VO(L1-2H)(phen)] and [VO(L3-2H)(phen)] showed promising antileishmanial activities. They were more active against the promastigote form than on the intracellular amastigote form of the parasites. In particular, they showed low IC50 values on promastigotes of L. panamensis (IC50 2.74 and 2.75 μM, respectively), on intracellular amastigotes of L. panamensis (IC50 19.52 and 20.75 μM, respectively) and showed low toxicity on THP-1 mammalian cells (IC50 188.55 and 88.13 μM, respectively) [278].
The [VIVO(L-2H)(dppz)] compounds with L = L3–L5 (Fig. 15) showed dose dependent growth inhibitory activity toward the infective form of T. brucei brucei strain 427, cell line 449. They show IC50 values in the low micromolar range (2.8 ± 0.1, 3.0 ± 0.2 and 1.5 ± 0.1, respectively). On the other hand, the bipy analogous complexes showed no significant activity against the same parasite. Both series showed a similar behavior against T. cruzi and T. brucei. These results are encouraging in respect to the proposed strategy for the development of compounds that could show activity toward both genetically related parasites [279].
Using a similar strategy other mixed-ligand bioactive VIVO-compounds were developed including phen as co-ligand. For this purpose the semicarbazone ONO co-ligand was changed by other ONO or NNO tridentate compounds.
In the case of the complexes depicted in Fig. 18 the mixed-ligand VIVO complex 26 showed a more than 10-fold improvement of IC50 against T. cruzi (Dm28c) with respect to the ligand HL6 (IC50 120.8 μM) and an IC50 value (10.7 μM) in the same order of that of Nifurtimox (IC50 6.0 μM). The complex is almost ten times more active than [VVO(L6)] 25. To explain the high inhibitory activity against the parasite of 26 as compared to 25, the interaction of this compound with DNA due to the presence of the phen ligand was postulated [288] (Fig. 19 ).
Fig. 18.
VIVO- and VVO2-complexes of the bioactive tridentate ligand 2-(benzothiazol-2-yl-hydrazonomethyl) phenol (L6) [288].
Fig. 19.
Oxidovanadium(IV) N-acylhydrazone complexes where L7 = LASSBio1064 = (E)-N′-(2-hydroxybenzylidene-4-chlorobenzohydrazide [289].
Two related VIVO N-acylhydrazone complexes [VIVO(L7-2H)(phen)]·(H2O) 27 and [VIVO(L7-2H)(H2O)] 28, where L7 = LASSBio1064 = (E)-N′-(2-hydroxybenzylidene-4-chlorobenzohydrazide were synthesized and evaluated on T. cruzi and L. major [289]. In this case, being L7 active on T. cruzi, both VIVO-complexes were also active in vitro against the epimastigote form of T. cruzi (Tulahuen 2 strain), showing IC50 values of the same order or significantly lower than that of Nifurtimox (IC50: L7 2.1; 27 6.0; 28 1.7 μM). On the other hand, L7 was inactive on L. major promastigotes and therefore again only the mixed-ligand VIVO-complex 27, which includes phen in its coordination sphere, showed activity on L. major promastigotes (IC50 value 22.1 μM). In addition, the ligand and both metal compounds showed low toxicity on peritoneal macrophage cells used as mammalian cell models (IC50 > 100 μM). It was hypothesized that the higher activity of these VCs may be related to either an easier uptake and/or to their higher efficacy toward their targets. On the other hand, inside cells the vanadium speciation may yield species containing or not the original ligands. The phen moiety may indeed be relevant in this respect since it improves the biological activity of the metal-based systems probably by acting as DNA intercalator. Indeed, DNA interaction ability of the phen complex was demonstrated by AFM, fluorescence spectroscopy and viscosity measurements.
There are two additional recent reports of VCs bearing inhibitory effects on Leishmania spp. growth. One of them established the growth inhibition of L. tarentolae promastigotes in vitro due to decavanadate (NH4)6V10O28·6H2O. The authors suggested the inhibition of phosphatases as a probable mechanism of action [290].
The other report involves a completely different strategy [291]. In the mammalian host, the Leishmania parasites live inside macrophages. The resistance to Leishmania or the healing ability of leishmaniasis is related to Th1 response by macrophages. Thus, compounds that activate macrophages for the Th1 pathway could show interesting leishmanicidal activity. In this context, native or chemically modified polysaccharides have been studied to potentiate the immune response. A previous research by the same authors showed that VIVO complexation of galactomannan (GMPOLY), isolated from the lichen Ramalina celastri, exacerbated the cytotoxicity to intracellular Leishmania [9]. The in vitro effects of galactomannan (GALMAN-A) isolated from seeds of Mimosa scabrella and its oxidovanadium(IV/V) complex on macrophage activation and leishmanicidal activity on Leishmania (L.) amazonensis intramacrophages amastigotes were confirmed. Both are potent activators of macrophages favoring the upregulation of Th1 response in macrophages, responsible of leishmanicidal effects.
In respect to VCs as prospective agents for the treatment of amoebiasis, Maurya et al. developed a large series of vanadium compounds with activity against E. histolytica that was discussed in the previous review [9]. Expanding this work a new series of VIVO- and VV-complexes of binucleating bis(dibasic tridentate ONS donor) ligand systems showing antiamoebic activity was reported (Fig. 20 ) [292].
Fig. 20.
VVO2-complexes of binucleating bis(dibasic ONS donor ligands) showing activity in vitro on E. histolytica[292].
The synthesized VVO2-complexes along with the ligands were screened for antiamoebic activity in vitro against the most common HM1:IMSS a highly virulent strain of E. histolytica. The complexes and the ligands showed IC50 values in the micromolar range. In particular, compounds 29, 30 and 31 (Fig. 20) are more active than the reference antiamoebic drug Metronizadol (IC50: 29 0.353; 30 0.092; 31 0.85; Metronidazol 1.68 μM). The reason why the VV-complexes are more active than the ligands is not clear. Authors hypothesized about the possibility that complexation might favor permeation of the drugs through the lipid layer of the cell membrane. VIVO-complexes with the same ligands resulted poorly active.
3.7.2. Antiviral vanadium compounds
Only scarce work has also been reported on vanadium compounds showing activity on certain viruses, bacteria and fungi.
3.7.2.1. Anti-HIV vanadium complexes
The effect of VCs on human immune deficiency virus (HIV) was reviewed by Sun et al. [293]. Studies showed that vanadium–thiourea complexes and vanadium substituted polyoxotungstates exhibited potent anti-HIV properties toward infected immortalized T-cells [127], [294].
In particular, the vanadium-substituted polyoxidotungstate [(VIVO)2(VVO)(SbW9O33)2]11− exhibited antiviral activity not only against HIV-1 in vitro but also against the viruses causing influenza, Dengue fever and SARS (severe acute respiratory syndrome). At physiological conditions, this ‘Keggin sandwich’ decompose into vanadate(V) and tungstate(VI), which were suggested to be the real active species [294], [295].
As it is known that VIV-complexes are not much stable under physiological conditions, porphyrinato ligands were used to stabilize the VIVO-unit and to get water-soluble oxidovanadium(IV)-porphyrins [128]. Porphyrins have a rigid square planar scaffold which could prohibit the demetalation reaction. The VIV-porphyrins prepared are stable in GSH containing solutions, and their inhibitory effects on HIV-1(BaL) replication in Hut/CCR5 cells were evaluated. All of these complexes showed anti-viral activities compared to the vehicle control, but one of them, water soluble and containing aminosulfonyl functional groups (32 in Fig. 21 ), exhibited the highest potency at the 5 μM level with over 97% inhibition.
Fig. 21.
VIVO-porphyrin complex with anti-HIV properties [128].
HIV-1 reverse transcriptase (RT) is one of the major targets for anti-HIV drugs, and binding of VCs to RT has been reported [296]. The inhibitory power of 32 toward HIV-1RT was measured using an ELISA method, and significant RT inhibition was observed after 30 min incubation, as compared with the drug-free control. Computational stimulation of the interaction(s) between the VIV–porphyrin complexes and different molecular targets of HIV-1, revealed that 32 shows preferential binding to CD4 protein, with a binding energy suggesting that 32 may exhibit the anti-HIV-1 activity by blocking the entrance of the virus to its targeted host cells (e.g. Hut/CCR5) [297].
Oxidovanadium(IV) cyclam complexes were also developed as potential anti-HIV agents. The xylylbicyclam complexes 33 and 34 (Fig. 22 ) were highly active against HIV-1 (IIIB) and HIV-2 (ROD) strains (IC50 values in the range 1–5 μM for 33 and 0.1–0.3 μM for 34). However, the oligomeric VIVO–cyclam complexes showed to be inactive [129].
Fig. 22.
VIVO-xylylbicyclam anti-HIV compounds [129].
The authors proposed the binding of the complexes to a receptor protein that HIV can use to infect T lymphocytes, called chemokine receptor type 4 (CXCR-4), as the mechanism of antiviral action. Docking studies of the most active compound 33 showed that binding of it to a model of the human CXCR-4 receptor might occur via coordination of vanadium to the carboxylate of Asp171 of the protein, accompanied by a weak attractive interaction of the oxidovanadium moiety to Trp 195 and by hydrophobic interaction between tryptophan residues and the bicyclam rings. The EPR data in aqueous solutions revealed two different environments for the VIV center in 33 and 34 and some exchange of axial sulfato and chloride ligands with solvent. The different nature of the VIV species present is probably the reason for the distinct biological activity of the chlorido and sulfato complexes.
3.7.2.2. Vanadium compounds active on other viruses
Vanadium compounds showing activity in vitro on other viruses responsible of viral infections such as influenza, Dengue fever and SARS have been previously reviewed [201]. For instance, antiviral activity was observed for vanadate dependent haloperoxidase, isolated from the alga Corallina inaequalis, and its mutant Pro395Asp/Leu241Val/Thr343Ala. High activity was reported for both enveloped (Herpes simplex) and non-enveloped viruses (Coxsackievirus B4). In addition, activity against Gram-positive (Staphylococcus aureus) and Gram-negative bacteria (Pseudomonas aeruginosa) was observed [298].
3.7.3. Antibacterial vanadium compounds
3.7.3.1. Activity of vanadium compounds against Mycobacterium tuberculosis
Only a few reports on anti-tuberculosis activity of VCs can be found in the literature. A very slight potentiation of the activity on M. tuberculosis H 37 R v of the bioactive 2-aminobenzoylhydrazide by generation of VIVO-complexes was reported several years ago [299].
More recently, VIVO- and VV-complexes of three thiosemicarbazone derivatives (Htsc), [VO(acac)(tsc)] and [VO2(tsc)], were evaluated on the pan-susceptible M. tuberculosis (MTB) H37Rv ATCC 27294 strain. Only [VO(acac)(L8-H)], where L8 = 2-acetylpyridine-N(4)-phenylthiosemicarbazone and acac = acetylacetonate, showed an interesting minimum inhibitory concentration (MIC) result (MIC 90% 3.6 μM) (Fig. 23 ). The activity of L8 (MIC 90% 57.7 μM) was enhanced by binding to the VIVO-core. The results of unspecific cytotoxicity on mammalian cells models were not reported [300].
Fig. 23.
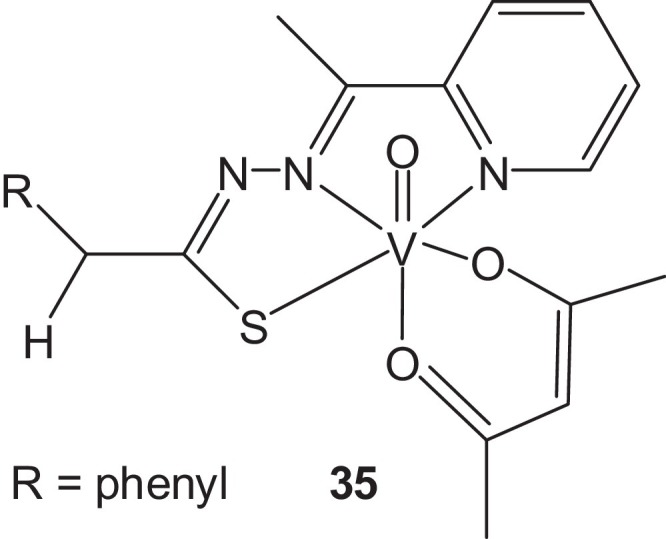
Structure of 35 [VO(acac)(L8-H)], where L8 = 2-acetylpyridine-N(4)-phenylthiosemicarbazone and acac = acetylacetonate [300].
Good biological results have been recently obtained with VIVO and VV homoleptic complexes of 8-hydroxyquinoline (8HQ) and heteroleptic ones including picolinato or Schiff bases as co-ligands [133]. 8HQ is a privileged structure, which is present in many bioactive natural products, and has been used as pharmacophore in the design of bioactive compounds against different diseases. As shown in this work it has also anti-MTB activity. When tested on the pan-susceptible M. tuberculosis H37Rv ATCC 27294 strain the complexes 36, 37, 38 and 39 (Fig. 24 ) showed low MIC values (90% inhibition) but similar to that of 8-hydroxyquinoline (MIC: 36 2.9; 37 4.1; 38 3.4; 39 3.7; 8HQ 2.5 μM). They qualify as potential anti-MTB agents, having lower MICs than some drugs commonly used to treat tuberculosis. Being 8HQ active on MTB, the authors suggested a non-specific mechanism of action. The inclusion of 8HQ as ligand in VCs might improve its bioavailability enhancing the ability to penetrate the lipid-rich hydrophobic cell wall of the mycobacteria.
Fig. 24.
8-Hydroquinoline anti-TB bioactive vanadium compounds [133].
3.7.3.2. Activity of vanadium compounds against other bacteria and fungi
The activity of VCs against other bacteria and fungi has been also reported. Only some representative examples are included below.
The antibacterial activity of different polyoxometalates (PMs) against Helicobacter pylori, the Gram-negative bacterium which is a principal gastric pathogen of humans, was investigated [301]. The compounds were evaluated on drugs-susceptible and resistant H. pylori strains. Among the studied compounds the polyoxometalate Keggin-structural K5[SiVVW11O40] showed bacteriostatic activity.
Ciprofloxacin is a quinolone type antibiotic that targets the DNA topoisomerase (gyrase) of bacterial type II. Heteroleptic VIVO-complexes with this antibiotic as ligand and various amino acids (dl-Ala, l-Tyr, l-Trp, l-Glu and l-Leu) were evaluated against Gram-positive (S. aureus, Baccilus subtilis, Serratia marcescens), and Gram-negative (P. aeruginosa, Escherichia coli) bacteria. In most of the cases a significant increase of the antibacterial activity of ciprofloxacin due to complexation was observed [302].
Metal complexes of the Schiff base derived from condensation of 1H-indole-3-carboxaldehyde and 1-phenyl-2,3-dimethyl-4-amino-3-pyrazolin-5-one (L9) were tested for their in vitro antibacterial activity against Gram-positive bacteria (S. aureus, K. pneumoniae and L. monocytogenes), Gram-negative bacteria (E. coli, P. aeruginosa and Salmonella typhimurium) and anti-fungal activity (Candida albicans and Aspergillus flavus) strains. The coordination to metal ions and, in particular to VIVO-center in [VO(L9)2(H2O)]SO4, led to an increase in antibacterial and antifungal activity [303].
VIVO-complexes with Schiff bases derived from condensation of 5-(phenyl/substituted phenyl)-2-hydrazino-1,3,4-thiadiazoles and indoline-2,3-dione showed higher in vitro antifungal (Aspergillus niger, Colletotrichum falcatum, Colletotrichum pallescence) and antibacterial against Gram-negative (Escherichia coli, Salmonella typhi) and Gram-positive (S. aureus, Bacillus subtilis) activities than free ligands [304].
4. Conclusions
Vanadium being relevant in several biological processes, many of its complexes with organic derivatives have been proposed for the treatment of several types of diseases, although no compound is presently used in the clinic. Namely, as reviewed here and reported in many previous publications, vanadium compounds have been proposed for the treatment of diabetes, cancer, diseases caused by parasites, for anti-viral and anti-bacteria use and for as diverse types of therapeutic uses as cardio-protection and to improve cardiac performance.
There has been substantial progress with respect to the speciation of VCs in blood serum, but a lot remains to be done concerning in situ real-time investigation on the fate of a VC. In fact, besides questions on their speciation in the gastrointestinal tract, even small VCs cannot be expected to simply diffuse through membranes to get to the target tissues. They must undergo translocation from the bloodstream through the endothelium of capillaries into the interstitial fluid [79]. Binding to transferrin and/or albumin might be relevant in this respect, but further research must address the mechanisms by which these processes might occur. Thus, a deeper knowledge of the in vivo speciation of VCs should help to better understand the mechanisms of transport, targeting and the mode of action, and give support to the design of efficient vanadium-based drugs.
Despite the many advances, the not yet well-established mode of action of VCs remains a challenge. Many of the reported therapeutic effects are related to the similarity of vanadate and phosphate, thus to the inhibition of the active sites of phosphatases and related enzymes resulting thereof; kinase activation may also be relevant, as well as the ability to generate ROS.
As discussed in many publications, vanadium compounds activate numerous signaling pathways and transcription factors, this simultaneously potentiating application of vanadium compounds in therapeutics, but also exerting nonspecific diametrically opposite effects on different cell structures. Additionally, still not much is known regarding global toxicity issues and effects of VCs on the immune system and inflammatory reactions, particularly if long-term administration is required. Potential carcinogenicity and correlation between the use of VCs and effects on the free-radical cell background are also issues to address.
Globally vanadium compounds have potential pharmacological interest for the treatment of diabetes, assuming that its toxic side effects can be overcome. Drugs that when given orally do not readily hydrolyze in the gastrointestinal tract and could be directed to specific tissues might possibly overcome certain deleterious side effects. Thus, the use of VCs to treat type 2 diabetes is still an open question.
Given the importance of cancer and parasitic-related diseases such as Amoebiasis or Chagas’, the non-adequacy or increasing resistance of pathogens against the presently used therapies, and the potentially lower costs of vanadium-based drugs, the prospective use of VCs in these areas appears promising. The combination of VCs with other drugs for the enhancement of the therapeutic effects is also an area deserving much more attention.
The ability to overcome the balance existing between the therapeutic action and adverse effects exerted by VCs, by shifting this balance toward the beneficial side, is the crucial issue for the future use of vanadium compounds in Medicine.
Acknowledgements
The authors thank Fundação para a Ciência e Tecnologia (FCT) and PEst-OE/QUI/UI0100/2013 (Portugal) UNLP (11X690), CONICET (PIP1125) and ANPCyT (PICT 2008-2218) (Argentina) and PEDECIBA Química and ANII (Uruguay). SBE is member of the Carrera del Investigador, CONICET, Argentina.
Contributor Information
Joao Costa Pessoa, Email: joao.pessoa@ist.utl.pt.
Susana Etcheverry, Email: etcheverry@biol.unlp.edu.ar.
Dinorah Gambino, Email: dgambino@fq.edu.uy.
References
- 1.Kiss T., Jakusch T., Hollender D., Dornyei A., Enyedy E.A., Costa Pessoa J., Sakurai H., Sanz-Medel A. Coord. Chem. Rev. 2008;252:1153. [Google Scholar]
- 2.Rehder D. John Wiley & Sons; New York: 2008. Bioinorganic Vanadium Chemistry. [Google Scholar]
- 3.Sakurai H., Yoshikawa Y., Yasui H. Chem. Soc. Rev. 2008;37:2383. doi: 10.1039/b710347f. [DOI] [PubMed] [Google Scholar]
- 4.Rehder D. Dalton Trans. 2013;42:11749. doi: 10.1039/c3dt50457c. [DOI] [PubMed] [Google Scholar]
- 5.Willsky G.R., Chi L.H., Godzala M., Kostyniak P.J., Smee J.J., Trujillo A.M., Alfano J.A., Ding W.J., Hu Z.H., Crans D.C. Coord. Chem. Rev. 2011;255:2258. doi: 10.1016/j.ccr.2011.06.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rehder D. Future Med. Chem. 2012;4:1823. doi: 10.4155/fmc.12.103. [DOI] [PubMed] [Google Scholar]
- 7.Costa Pessoa J., Tomaz I. Curr. Med. Chem. 2010;17:3701. doi: 10.2174/092986710793213742. [DOI] [PubMed] [Google Scholar]
- 8.Jakusch T., Costa Pessoa J., Kiss T. Coord. Chem. Rev. 2011;255:2218. [Google Scholar]
- 9.Gambino D. Coord. Chem. Rev. 2011;255:2193. [Google Scholar]
- 10.Rehder D. In: Sigel A., et al., editors. Vol. 13. Springer Science + Business Media; Dordrecht: 2014. (Interrelations Between Essential Metal Ions and Human Diseases, Metal Ions in Life Sciences). [DOI] [Google Scholar]
- 11.Korbecki J., Baranowska-Bosiacka I., Gutowska I., Chlubek D. Acta Biochim. Polon. 2012;59:195. [PubMed] [Google Scholar]
- 12.Smith D.M., Pickering R.M., Lewith G.T. Q. Int. J. Med. 2008;101:351. doi: 10.1093/qjmed/hcn003. [DOI] [PubMed] [Google Scholar]
- 13.Olopade J.O., Connor J.R. Curr. Top. Toxicol. 2011;7:34. [Google Scholar]
- 14.Evangelou A.M. Crit. Rev. Oncol./Hematol. 2002;42:249. doi: 10.1016/s1040-8428(01)00221-9. [DOI] [PubMed] [Google Scholar]
- 15.Shechter Y., Goldwaser I., Mironchik M., Fridkin M., Gefel D. Coord. Chem. Rev. 2003;237:3. [Google Scholar]
- 16.Sakurai H., Kojima Y., Yoshikawa Y., Kawabe K., Yasui H. Coord. Chem. Rev. 2002;226:187. [Google Scholar]
- 17.Sakurai H., Katoh A., Yoshikawa Y. Bull. Chem. Soc. Jpn. 2006;79:1645. [Google Scholar]
- 18.(a) Hulley P., Davison A. J. Trace Elem. Exp. Med. 2003;16:281. [Google Scholar]; (b) Dabrowiak J.C. Wiley & Sons; Chichester, UK: 2009. Metals in Medicine. [Google Scholar]
- 19.Tracey A.S., Willsky G.R., Takeuchi E.S. CRC Press; Boca Raton: 2007. Vanadium: Chemistry, Biochemistry, Pharmacology and Practical Applications. [Google Scholar]
- 20.Michibata H., editor. Vanadium: Biochemical and Molecular Biological Approaches. Springer; Dordrecht: 2012. [Google Scholar]
- 21.Orvig C., Abrams M.J. Chem. Rev. 1999;99:2201. doi: 10.1021/cr980419w. [DOI] [PubMed] [Google Scholar]
- 22.Kordowiak A.M., Holko P. Post. Biol. Kom. 2009;36:361. [Google Scholar]
- 23.Barcelaux D.G. J. Toxicol. Clinic. Toxicol. 1999;37:265. doi: 10.1081/clt-100102425. [DOI] [PubMed] [Google Scholar]
- 24.Rehder D. Sigel A., Sigel H., Sigel R.K., editors. Met. Ions Life Sci. 2013;13:139–159. doi: 10.1007/978-94-007-7500-8_5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cohen M.D. ACS Symp. Ser. 974. 2007. p. 217. [Google Scholar]
- 26.Scior T., Guevara-García A., Bernard P., Do Q.-T., Domeyer D., Laufer S. Mini Rev. Med. Chem. 2005;5:995. doi: 10.2174/138955705774575264. [DOI] [PubMed] [Google Scholar]
- 27.Irsigler G.B., Visser P.J., Spangenberg P.A. Am. J. Ind. Med. 1999;35:366. doi: 10.1002/(sici)1097-0274(199904)35:4<366::aid-ajim7>3.0.co;2-n. [DOI] [PubMed] [Google Scholar]
- 28.Rom W.N., editor. Environmental and Occupational Medicine Little. Brown and Co.; Boston, MA: 1992. [Google Scholar]
- 29.Nriagu J.O., editor. vol. 30. John Wiley & Sons; New York: 1998. (Wiley Series in Advances in Environmental Science and Technology). [Google Scholar]
- 30.Assem F.L., Levy L.S. In: Vanadium: Biochemical and Molecular Biological Approaches. H Michibata H., editor. Springer; Dordrecht: 2012. pp. 209–224. [Google Scholar]
- 31.Nechay B.R., Saunders J.P. J. Environ. Pathol. Toxicol. 1978;2:247. [PubMed] [Google Scholar]
- 32.McLauchlan C.C., Hooker J.D., Jones M.A., Dymon Z., Backhus E.A., Greiner B.A., Dorner N.A., Youkhana M.A., Manus L.M. J. Inorg. Biochem. 2010;104:274. doi: 10.1016/j.jinorgbio.2009.12.001. [DOI] [PubMed] [Google Scholar]
- 33.Stankiewics P.J., Tracey A. In: Sigel H., Sigel A., editors. vol. 31. Marcel Dekker, Inc.; New York: 1995. pp. 249–286. (Metal Ions in Biological Systems. Vanadium and Its Role in Life). [Google Scholar]
- 34.Zhang Z.Y. Crit. Rev. Biochem. Mol. Biol. 1998;33:1. doi: 10.1080/10409239891204161. [DOI] [PubMed] [Google Scholar]
- 35.Crans D.C. In: Vanadium Compounds. Chemistry, Biochemistry, and Therapeutic Applications. Tracey A., Crans D.C., editors. American Chemical Society; Washington, DC: 1998. pp. 82–103. [Google Scholar]
- 36.Posner B.I., Yang C.R., Shaver A. In: Vanadium Compounds. Chemistry, Biochemistry, and Therapeutic Applications. Tracey A., Crans D.C., editors. American Chemical Society; Washington, DC: 1998. pp. 316–328. [Google Scholar]
- 37.Chang L., Karin M. Nature. 2001;410:37. doi: 10.1038/35065000. [DOI] [PubMed] [Google Scholar]
- 38.Van Hoof V.O., De Broe M.E. Crit. Rev. Clin. Lab. Sci. 1994;31:197. doi: 10.3109/10408369409084677. [DOI] [PubMed] [Google Scholar]
- 39.Crans D.C., Tracey A. In: Vanadium Compounds. Chemistry, Biochemistry, and Therapeutic Applications. Tracey A., Crans D.C., editors. American Chemical Society; Washington, DC: 1998. pp. 2–29. [Google Scholar]
- 40.Kustin K. In: Vanadium Compounds. Chemistry, Biochemistry, and Therapeutic Applications. Tracey A., Crans D.C., editors. American Chemical Society; Washington, DC: 1998. pp. 170–185. [Google Scholar]
- 41.Peters K.G., Davis M.G., Howard B.W., Pokross M., Rastogi V., Diven C., Greis K.D., Eby-Wilkens E., Maier M., Evdokimov A., Soper S., Genbauffe F. J. Inorg. Biochem. 2003;96:321. doi: 10.1016/s0162-0134(03)00236-8. [DOI] [PubMed] [Google Scholar]
- 42.Shechter Y., Karlish S.J. Nature. 1980;284:556. doi: 10.1038/284556a0. [DOI] [PubMed] [Google Scholar]
- 43.Stern A., Yin X., Tsang S.S., Davison A., Moon J. Biochem. Cell Biol. 1993;71:103. doi: 10.1139/o93-018. [DOI] [PubMed] [Google Scholar]
- 44.Djordjevic C. In: Metal Ions in Biological Systems. Vanadium and Its Role in Life. Sigel H., Sigel A., editors. Marcel Dekker, Inc.; New York: 1995. pp. 595–615. [Google Scholar]
- 45.Domingo J.L. Mol. Cell. Biochem. 2000;203:185. doi: 10.1023/a:1007067011338. [DOI] [PubMed] [Google Scholar]
- 46.Srivastava A.K. Mol. Cell. Biochem. 2000;206:177. doi: 10.1023/a:1007075204494. [DOI] [PubMed] [Google Scholar]
- 47.Liu J.C., Yu Y., Wang G., Wang K., Yang X.G. Metallomics. 2013;5:813. doi: 10.1039/c3mt00001j. [DOI] [PubMed] [Google Scholar]
- 48.Fantus I.G., Kadota S., Deragon G., Foster B., Posner B.I. Biochemistry. 1989;28:8864. doi: 10.1021/bi00448a027. [DOI] [PubMed] [Google Scholar]
- 49.Tamura S., Brown T.A., Dubler R.E., Larner J. Biochem. Biophys. Res. Commun. 1983;113:80. doi: 10.1016/0006-291x(83)90434-5. [DOI] [PubMed] [Google Scholar]
- 50.Tamura S., Brown T.A., Whipple J.H., Fujita-Yamaguchi Y., Dubler R.E., Cheng K., Larner J. J. Biol. Chem. 1984;259:6650. [PubMed] [Google Scholar]
- 51.Bernier M., Laird D.M., Lane M.D. J. Biol. Chem. 1988;263:13626. [PubMed] [Google Scholar]
- 52.Shisheva A., Shechter Y. Biochemistry. 1992;31:8059. doi: 10.1021/bi00149a041. [DOI] [PubMed] [Google Scholar]
- 53.Kadota S., Fantus I.G., Deragon G., Guyda H.J., Hersh B., Posner B.I. Biochem. Biophys. Res. Commun. 1987;147:259. doi: 10.1016/s0006-291x(87)80115-8. [DOI] [PubMed] [Google Scholar]
- 54.Srivastava A.K., Chiasson J.L. Biochim. Biophys. Acta. 1989;996:13. doi: 10.1016/0167-4838(89)90087-3. [DOI] [PubMed] [Google Scholar]
- 55.Strout H.V., Vicario P.P., Saperstein R., Slater E.E. Endocrinology. 1989;124:1918. doi: 10.1210/endo-124-4-1918. [DOI] [PubMed] [Google Scholar]
- 56.Pandey S.K., Anand-Srivastava M.B., Srivastava A.K. Biochemistry. 1998;37:7006. doi: 10.1021/bi9726786. [DOI] [PubMed] [Google Scholar]
- 57.Molero J.C., Martinez C., Andres A., Satrustegui J., Carrascosa J.M. FEBS Lett. 1998;425:298. doi: 10.1016/s0014-5793(98)00258-0. [DOI] [PubMed] [Google Scholar]
- 58.Pandey S.K., Chiasson J.L., Srivastava A.K. Mol. Cell. Biochem. 1995;153:69. doi: 10.1007/BF01075920. [DOI] [PubMed] [Google Scholar]
- 59.Pandey S.K., Theberge J.F., Bernier M., Srivastava A.K. Biochemistry. 1999;38:14667. doi: 10.1021/bi9911886. [DOI] [PubMed] [Google Scholar]
- 60.Novak-Hofer I., Thomas G. J. Biol. Chem. 1985;260:10314. [PubMed] [Google Scholar]
- 61.Fantus I.G., Deragon G., Lai R., Tang S. Mol. Cell. Biochem. 1995;153:103. doi: 10.1007/BF01075924. [DOI] [PubMed] [Google Scholar]
- 62.Nantel A., Mohammad-Ali K., Sherk J., Posner B.I., Thomas D.Y. J. Biol. Chem. 1998;273:10475. doi: 10.1074/jbc.273.17.10475. [DOI] [PubMed] [Google Scholar]
- 63.Shechter Y. Diabetes. 1990;39:1. doi: 10.2337/diacare.39.1.1. [DOI] [PubMed] [Google Scholar]
- 64.Cortizo A.M., Etcheverry S.B. Mol. Cell. Biochem. 1995;145:97. doi: 10.1007/BF00935481. [DOI] [PubMed] [Google Scholar]
- 65.Etcheverry S.B., Cortizo A.M. In: Vanadium Compounds. Chemistry, Biochemistry, and Therapeutic Applications. Tracey A., Crans D.C., editors. American Chemical Society; Washington, DC: 1998. pp. 270–276. [Google Scholar]
- 66.Smith J.B. Proc. Natl. Acad. Sci. U.S.A. 1995;80:6162. [Google Scholar]
- 67.Wice B., Milbrandt J., Glaser L. J. Biol. Chem. 1987;262:1810. [PubMed] [Google Scholar]
- 68.Lau K.H., Tanimoto H., Baylink D.J. Endocrinology. 1988;123:2858. doi: 10.1210/endo-123-6-2858. [DOI] [PubMed] [Google Scholar]
- 69.Etcheverry S.B., Barrio D.A. In: Vanadium: The Versatile Metal. American Chemical Society Series 974, vol. 15. Kustin K., Costa Pesoa J., Crans D.C., editors. 2007. p. 204. [Google Scholar]
- 70.Wilkinson M.G., Millar J.B. FASEB J. 2000;14:2147. doi: 10.1096/fj.00-0102rev. [DOI] [PubMed] [Google Scholar]
- 71.Randazzo P.A., Olshan J.S., Bijivi A.A., Jarett L. Arch. Biochem. Biophys. 1992;292:258. doi: 10.1016/0003-9861(92)90077-a. [DOI] [PubMed] [Google Scholar]
- 72.Paris S., Pouyssegur J. J. Biol. Chem. 1987;262:1970. [PubMed] [Google Scholar]
- 73.Cantley L.C., Josephson L., Warner R., Yanagisawa M., Lechene C., Guidotti G. J. Biol. Chem. 1977;252:7421. [PubMed] [Google Scholar]
- 74.Sakurai H., Yasui H., Adachi Y. Expert Opin. Invest. Drug. 2003;12:1189. doi: 10.1517/13543784.12.7.1189. [DOI] [PubMed] [Google Scholar]
- 75.Thompson K.H., Orvig C. J. Chem. Soc., Dalton Trans. 2000:2885. [Google Scholar]
- 76.Thompson K.H., Orvig C. J. Inorg. Biochem. 2006;100:1925. doi: 10.1016/j.jinorgbio.2006.08.016. [DOI] [PubMed] [Google Scholar]
- 77.Orvig C., Thompson K.H., Battell M., McNeill J.H. Met. Ions Biol. Syst. 1995;31:575. [PubMed] [Google Scholar]
- 78.Nechay B.R. Annu. Rev. Pharmacol. Toxicol. 1984;24:501. doi: 10.1146/annurev.pa.24.040184.002441. [DOI] [PubMed] [Google Scholar]
- 79.Makinen M.W., Salehitazangi M. Coord. Chem. Rev. 2014;279:1. doi: 10.1016/j.ccr.2014.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lohr J.W., Bennett M.I., Pochal M.A., McReynolds J., Acara M., Willsky G.R. Res. Commun. Chem. Pathol. Pharmacol. 1991;72:191. [PubMed] [Google Scholar]
- 81.Saxena A.K., Srivastava P., Kale R.K., Baquer N.Z. Biochem. Int. 1992;26:59. [PubMed] [Google Scholar]
- 82.Thompson K.H., McNeill J.H. Res. Commun. Chem. Pathol. Pharmacol. 1993;80:187. [PubMed] [Google Scholar]
- 83.Ugazio G., Bosia S., Burdino E., Grignolo F. Res. Commun. Mol. Pathol. Pharmacol. 1994;85:313. [PubMed] [Google Scholar]
- 84.Heyliger C.E., Tahiliani A.G., McNeill J.H. Science. 1985;227:1474. doi: 10.1126/science.3156405. [DOI] [PubMed] [Google Scholar]
- 85.Ramanadham S., Mongold J.J., Brownsey R.W., Cros G.H., McNeill J.H. Am. J. Physiol. 1989;257:H904. doi: 10.1152/ajpheart.1989.257.3.H904. [DOI] [PubMed] [Google Scholar]
- 86.Mongold J.J., Cros G.H., Vian L., Tep A., Ramanadham S., Siou G., Diaz J., McNeill J.H., Serrano J.J. Pharmacol. Toxicol. 1990;67:192. doi: 10.1111/j.1600-0773.1990.tb00812.x. [DOI] [PubMed] [Google Scholar]
- 87.Dai S., Thompson K.H., Vera E., McNeill J.H. Pharmacol. Toxicol. 1994;75:265. doi: 10.1111/j.1600-0773.1994.tb00359.x. [DOI] [PubMed] [Google Scholar]
- 88.Rossetti L., Laughlin M.R. J. Clin. Invest. 1989;84:892. doi: 10.1172/JCI114250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Meyerovitch J., Farfel Z., Sack J., Shechter Y. J. Biol. Chem. 1987;262:6658. [PubMed] [Google Scholar]
- 90.Challiss R.A.J., Leighton B., Lozeman F.J., Budohoski L., Newsholme E.A. Biochem. Pharmacol. 1987;36:357. doi: 10.1016/0006-2952(87)90294-2. [DOI] [PubMed] [Google Scholar]
- 91.Nelson W.O., Karpishin T.B., Rettig S.J., Orvig C. Can. J. Chem. 1988;66:123. [Google Scholar]
- 92.Clevette D.J., Orvig C. Polyhedron. 1990;9:151. [Google Scholar]
- 93.Yuen V.G., Orvig C., Thompson K.H., McNeill J.H. Can. J. Phys. Pharmacol. 1993;71:270. doi: 10.1139/y93-042. [DOI] [PubMed] [Google Scholar]
- 94.Srivastava P., Saxena A.K., Kale R.K., Baquer N.Z. Res. Commun. Chem. Pathol. Pharmacol. 1993;80:283. [PubMed] [Google Scholar]
- 95.Thompson K.H., McNeill J.H. In: Trace Elements in Man and Animals 9: Proc. 9th Int. Symp. Trace Elem. Man Animals. Fischer P.W.F., L’Abbe M.R., Cockell K.A., Gibson R.S., editors. NRC Research Press; Ottawa: 1997. pp. 349–350. [Google Scholar]
- 96.Halliwell B., Gutteridge J.M.C. Methods Enzymol. 1990;186:1. doi: 10.1016/0076-6879(90)86093-b. [DOI] [PubMed] [Google Scholar]
- 97.Lusis A.J. Nature. 2000;407:233. doi: 10.1038/35025203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Brownlee M. Nature. 2001;414:813. doi: 10.1038/414813a. [DOI] [PubMed] [Google Scholar]
- 99.Han D.C., Lee M.-Y., Shin K.D., Jeon S.B., Kim J.M., Son K.-H., Kim H.-C., Kim H.-M., Kwon B.-M. J. Biol. Chem. 2004;279:6911. doi: 10.1074/jbc.M309708200. [DOI] [PubMed] [Google Scholar]
- 100.Rice-Evans C.A., Diplock A.T. Free Radic. Biol. Med. 1993;15:77. doi: 10.1016/0891-5849(93)90127-g. [DOI] [PubMed] [Google Scholar]
- 101.Blake D.R., Allen R., Lunec J. Br. Med. J. 1987;43:371. doi: 10.1093/oxfordjournals.bmb.a072188. [DOI] [PubMed] [Google Scholar]
- 102.LoPachin R.M., Gavin T., Geohagen B.C., Zhang L.H., Casper D., Lekhraj R., Barber D.S. J. Neurochem. 2011;116:132. doi: 10.1111/j.1471-4159.2010.07091.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.NCI DCPC chemoprevention branch and agent development committee. J. Cell. Biochem. 1996;26S:72. [Google Scholar]
- 104.Thompson K.H., Bohmerle K., Polishcuk E., Martins C., Toleikis P., Tse J., Yuen V., McNeil J.H., Orvig C. J. Inorg. Biochem. 2004;98:2063. doi: 10.1016/j.jinorgbio.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 105.Na L.X., Zhang Y.L., Li Y., Liu L.Y., Li R., Kong T., Sun C.H. Nutr. Metab. Cardiovasc. Dis. 2011;21:526. doi: 10.1016/j.numecd.2009.11.009. [DOI] [PubMed] [Google Scholar]
- 106.Seo K.I., Choi M.S., Jung U.J., Kim H.J., Yeo J., Jeon S.M., Lee M.K. Mol. Nutr. Food Res. 2008;52:995. doi: 10.1002/mnfr.200700184. [DOI] [PubMed] [Google Scholar]
- 107.Kawabe K., Yoshikawa Y., Adachi Y., Sakurai H. Life Sci. 2006;78:2860. doi: 10.1016/j.lfs.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 108.Adachi Y., Yoshida J., Kodera Y., Katoh A., Takada J., Sakurai H. J. Med. Chem. 2006;49:3251. doi: 10.1021/jm060229a. [DOI] [PubMed] [Google Scholar]
- 109.Adachi Y., Yoshikawa Y., Yoshida J., Kodera Y., Katoh A., Takada J., Sakurai H. Biochem. Biophys. Res. Commun. 2006;345:945. doi: 10.1016/j.bbrc.2006.05.003. [DOI] [PubMed] [Google Scholar]
- 110.Sakurai H., Katoh A., Kiss T., Jakusch T., Hattori M. Metallomics. 2010;2:670–682. doi: 10.1039/c0mt00025f. [DOI] [PubMed] [Google Scholar]
- 111.Hiromura M., Nakayama A., Adachi Y., Doi M., Sakurai H. J. Biol. Inorg. Chem. 2007;12:1275. doi: 10.1007/s00775-007-0295-x. [DOI] [PubMed] [Google Scholar]
- 112.Aureliano M., Crans D.C. J. Inorg. Biochem. 2009;103:536. doi: 10.1016/j.jinorgbio.2008.11.010. [DOI] [PubMed] [Google Scholar]
- 113.Thompson K.H., Lichter J., LeBel C., Scaife M.C., McNeil J.H., Orvig C. J. Inorg. Biochem. 2009;103:554. doi: 10.1016/j.jinorgbio.2008.12.003. [DOI] [PubMed] [Google Scholar]
- 114.D’Cruz O.J., Uckun F.M. Expert Opin. Invest. Drug. 2002;11:1829. doi: 10.1517/13543784.11.12.1829. [DOI] [PubMed] [Google Scholar]
- 115.Papaioannou A., Manos M., Karkabounas S., Liasko R., Evangelou A.M., Correia I., Kalfakakou V., Costa Pessoa J., Kabanos T.A. J. Inorg. Biochem. 2004;98:959. doi: 10.1016/j.jinorgbio.2004.02.011. [DOI] [PubMed] [Google Scholar]
- 116.Noblía P., Vieites M., Parajón-Costa B.S., Baran E.J., Cerecetto H., Draper P., Gonzalez M., Piro O.E., Castellano E.E., Azqueta A., Ceráin A.L., Monge-Vega A., Gambino D. J. Inorg. Biochem. 2005;99:443. doi: 10.1016/j.jinorgbio.2004.10.019. [DOI] [PubMed] [Google Scholar]
- 117.Benítez J., Guggeri L., Tomaz I., Costa Pessoa J., Moreno V., Lorenzo J., Aviles F.X., Garat B., Gambino D. J. Inorg. Biochem. 2009;103:1386. doi: 10.1016/j.jinorgbio.2009.07.013. [DOI] [PubMed] [Google Scholar]
- 118.Benítez J., Guggeri L., Tomaz I., Arrambide G., Navarro M., Costa Pessoa J., Garat B., Gambino D. J. Inorg. Biochem. 2009;103:609. doi: 10.1016/j.jinorgbio.2008.10.018. [DOI] [PubMed] [Google Scholar]
- 119.Ananth K.M., Kanthimathi M., Nair B.U. Trans. Met. Chem. 2001;26:333. [Google Scholar]
- 120.Maurya M.R., Khan A.A., Irfan I., Azam A., Kumar A., Costa Pessoa J. Eur. J. Inorg. Chem. 2009:5377. [Google Scholar]
- 121.Maurya M.R., Khan A.A., Azam A., Ranjan S., Mondal N., Kumar A., Avecilla F., Costa Pessoa J. Dalton Trans. 2010;39:1345. doi: 10.1039/b915752b. [DOI] [PubMed] [Google Scholar]
- 122.Maurya M.R., Kumar A., Bhat A.R., Azam A., Bader C., Rehder D. Inorg. Chem. 2006;45:1260. doi: 10.1021/ic050811+. [DOI] [PubMed] [Google Scholar]
- 123.Ghosh P., D’Cruz O.J., DuMez D.D., Peitersen J., Uckun F.M. J. Inorg. Biochem. 1999;75:135. doi: 10.1016/s0162-0134(99)00046-x. [DOI] [PubMed] [Google Scholar]
- 124.D’Cruz O.J., Waurzyniak B., Uckun F.M. Toxicology. 2002;170:31. doi: 10.1016/s0300-483x(01)00508-x. [DOI] [PubMed] [Google Scholar]
- 125.Shigeta S., Mori S., Yamase T., Yamamoto N., Yamamoto N. Biomed. Pharmacother. 2006;60:211. doi: 10.1016/j.biopha.2006.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Yasui H., Adachi Y., Katoh A., Sakurai H. J. Biol. Inorg. Chem. 2007;12:843. doi: 10.1007/s00775-007-0239-5. [DOI] [PubMed] [Google Scholar]
- 127.D’Cruz O.J., Dong Y.H., Uckun F.M. Biochem. Biophys. Res. Commun. 2003;302:253. doi: 10.1016/s0006-291x(03)00161-x. [DOI] [PubMed] [Google Scholar]
- 128.Wong S.Y., Sun R.W.Y., Chung N.P.Y., Lin C.L., Che C.M. Chem. Commun. 2005:3544. doi: 10.1039/b503535j. [DOI] [PubMed] [Google Scholar]
- 129.Ross A., Soares D.C., Covelli D., Pannecouque C., Budd L., Collins A., Robertson N., Parsons S., De Clercq E., Kennepohl P., Sadler P.J. Inorg. Chem. 2010;49:1122. doi: 10.1021/ic9020614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Timerbaev A.R., Hartinger C.G., Keppler B.K. Trends Anal. Chem. 2006;25:868. [Google Scholar]
- 131.David S., Barros V., Cruz C., Delgado R. FEMS Microbiol. Lett. 2005;251:119. doi: 10.1016/j.femsle.2005.07.044. [DOI] [PubMed] [Google Scholar]
- 132.Maiti A., Ghosh S. J. Inorg. Biochem. 1989;36:131. doi: 10.1016/0162-0134(89)80020-0. [DOI] [PubMed] [Google Scholar]
- 133.Correia I., Adão P., Roy S., Wahba M., Matos C., Maurya M.R., Marques F., Pavan F.R., Leite C.Q.F., Avecilla F., Costa Pessoa J. J. Inorg. Biochem. 2014;141:83. doi: 10.1016/j.jinorgbio.2014.07.019. [DOI] [PubMed] [Google Scholar]
- 134.Heinemann G., Fichti B., Vogt W. Clin. Pharmacol. 2003;55:241. doi: 10.1046/j.1365-2125.2003.01711.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Gorzsás A., Andersson I., Pettersson L. Eur. J. Inorg. Chem. 2006:3559. [Google Scholar]
- 136.Heinemann G., Vogt W. Clin. Chem. 1996;42:1275. [PubMed] [Google Scholar]
- 137.Willsky G.R., Goldfine A.B., Kostyniak P.J., McNeill J.H., Yang L.Q., Khan H.R., Crans D.C. J. Inorg. Biochem. 2001;85:33. doi: 10.1016/s0162-0134(00)00226-9. [DOI] [PubMed] [Google Scholar]
- 138.Zhang S.Q., Zhong X.Y., Chen G.H., Lu W.L., Zhang Q. J. Pharm. Pharmacol. 2008;60:99. doi: 10.1211/jpp.60.1.0013. [DOI] [PubMed] [Google Scholar]
- 139.Kiss T., Jakusch T., Bouhsina S., Sakurai H., Enyedy E. Eur. J. Inorg. Chem. 2006:3607. [Google Scholar]
- 140.Thompson K.H., Liboiron B.D., Sun Y., Bellman K.D.D., Setyawati I.A., Patrick B.O., Karunaratne V., Rawji G., Wheeler J., Sutton K., Bhanot S., Cassidy C., McNeil J.H., Yuen V.G., Orvig C. J. Biol. Inorg. Chem. 2003;8:66. doi: 10.1007/s00775-002-0388-5. [DOI] [PubMed] [Google Scholar]
- 141.Rehder D. Angew. Chem. Int. Ed. 1991;30:148. [Google Scholar]
- 142.Bertini I., Canti G., Luchinat C. Inorg. Chim. Acta: Bioinorg. Chem. 1982;67:L21–L23. [Google Scholar]
- 143.Costa Pessoa J., Gonçalves G., Roy S., Correia I., Mehtab S., Santos M.F.A., Santos-Silva T. Inorg. Chim. Acta. 2014;420:60. [Google Scholar]
- 144.Schemberg J., Schneider K., Demmer U., Warkentin E., Müller A., Ermler U. Angew. Chem. Int. Ed. 2007;46:2409. doi: 10.1002/anie.200604858. [DOI] [PubMed] [Google Scholar]
- 145.Ducret L. Bull. Soc. Chim. Fr. 1948:658. [Google Scholar]
- 146.Ducret L. Ann. Chim. S12. 1951;V6:723. [Google Scholar]
- 147.Vilas Boas L.F., Costa Pessoa J. In: Wilkinson G., Gillard R.D., McCleverty J.A., editors. vol. 3. Pergamon Press; Oxford: 1987. pp. 453–583. (Comprehensive Coordination Chemistry). [Google Scholar]
- 148.Schmidt H., Andersson I., Rehder D., Pettersson L. Chem. Eur. J. 2001;7:251. doi: 10.1002/1521-3765(20010105)7:1<251::aid-chem251>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 149.Mehtab S., Gonçalves G., Roy S., Tomaz A.I., Santos-Silva T., Santos M.F.A., Romão M.J., Jakusch T., Kiss T., Costa Pessoa J. J. Inorg. Biochem. 2013;121:187. doi: 10.1016/j.jinorgbio.2012.12.020. [DOI] [PubMed] [Google Scholar]
- 150.Delgado T.C., Tomaz A.I., Correia I., Costa Pessoa J., Jones J.G., Geraldes C.F., Castro M.M. J. Inorg. Biochem. 2005;99:2328. doi: 10.1016/j.jinorgbio.2005.08.014. [DOI] [PubMed] [Google Scholar]
- 151.Costa Pessoa J., Tomaz I., Kiss T., Buglyó P. J. Inorg. Biochem. 2001;84:259. doi: 10.1016/s0162-0134(00)00233-6. [DOI] [PubMed] [Google Scholar]
- 152.Costa Pessoa J., Tomaz I., Kiss T., Kiss E., Buglyó P. J. Biol. Inorg. Chem. 2002;7:225. doi: 10.1007/s007750100289. [DOI] [PubMed] [Google Scholar]
- 153.Sanna D., Serra M., Micera G., Garribba E. Inorg. Chem. 2014;53:1449. doi: 10.1021/ic402366x. [DOI] [PubMed] [Google Scholar]
- 154.Garner M., Reglinski J., Smith W.E., McMurray J., Abdullah I., Wilson R. J. Biol. Inorg. Chem. 1997;2:235. [Google Scholar]
- 155.Jakusch T., Hollender D., Enyedy É.A., González C.S., Montes-Bayón M., Sanz-Medel A., Costa Pessoa J., Tomaz I., Kiss T. Dalton Trans. 2009:2428. doi: 10.1039/b817748a. [DOI] [PubMed] [Google Scholar]
- 156.Gonçalves G., Castro M.M.C.A., Avecilla F., Kiss T., Jakusch T., Tomaz I., Correia I., Veiros L.F., Palacio L., Maestro M., Garcia M.H.V., Costa Pessoa J. Dalton Trans. 2013;42:11841. doi: 10.1039/c3dt50553g. [DOI] [PubMed] [Google Scholar]
- 157.Sanna D., Micera G., Garribba E. Inorg. Chem. 2013;52:11975. doi: 10.1021/ic401716x. [DOI] [PubMed] [Google Scholar]
- 158.Gouranton E., El Yazidi C., Cardinault N., Amiot M.J., Borel P., Landrier J.F. Food Chem. Toxicol. 2008;46:3832. doi: 10.1016/j.fct.2008.10.006. [DOI] [PubMed] [Google Scholar]
- 159.Santos M.F.A., Correia I., Oliveira A.R., Garribba E., Costa Pessoa J., Santos-Silva T. Eur. J. Inorg. Chem. 2014:3293. [Google Scholar]
- 160.Ou H., Yan L., Mustafi D., Makinen M.W., Brady M.J. J. Biol. Inorg. Chem. 2005;10:874. doi: 10.1007/s00775-005-0037-x. [DOI] [PubMed] [Google Scholar]
- 161.Nakai M., Watanabe H., Fujiwara C., Kakegawa H., Satoh T., Takada J., Matsushita R., Sakurai H. Biol. Pharm. Bull. 1995;18:719. doi: 10.1248/bpb.18.719. [DOI] [PubMed] [Google Scholar]
- 162.Sakurai H., Tsuchiya K., Nukatsuka M., Sofue M., Kawada J. J. Endocrinol. 1990;126:451. doi: 10.1677/joe.0.1260451. [DOI] [PubMed] [Google Scholar]
- 163.Sakurai H., Tsuchiya K., Nukatsuka M., Kawada J., Ishikawa S., Yoshida H., Komatsu M. J. Clin. Biochem. Nutr. 1990;8:193. [Google Scholar]
- 164.Takino T., Yasui H., Yoshitake A., Hamajima Y., Matsushita R., Takada J., Sakurai H. J. Biol. Inorg. Chem. 2001;6:133. doi: 10.1007/s007750000182. [DOI] [PubMed] [Google Scholar]
- 165.Fujimoto S., Fujii K., Yasui H., Matsushita R., Takada J., Sakurai H. J. Clin. Biochem. Nutr. 1997;23:113. [Google Scholar]
- 166.Tracey A.S., Gresser M.J. Proc. Natl. Acad. Sci. U.S.A. 1986;83:609. doi: 10.1073/pnas.83.3.609. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Setyawati I.A., Thompson K.H., Yuen V.G., Sun Y., Battell M., Lyster D.M., Vo C., Ruth T.J., Zeisler S., McNeill J.H., Orvig C. J. Appl. Physiol. 1998;84:569. doi: 10.1152/jappl.1998.84.2.569. [DOI] [PubMed] [Google Scholar]
- 168.Yasui H., Takechi K., Sakurai H. J. Inorg. Biochem. 2000;78:185. doi: 10.1016/s0162-0134(00)00002-7. [DOI] [PubMed] [Google Scholar]
- 169.Nakano H., Nishioka M., Sangen O., Yamamoto Y. J. Polym. Sci., Polym. Chem. Ed. 1981;19:2919. [Google Scholar]
- 170.Yasui H., Tamura A., Takino T., Sakurai H. J. Inorg. Biochem. 2002;91:327. doi: 10.1016/s0162-0134(02)00443-9. [DOI] [PubMed] [Google Scholar]
- 171.Sakurai H., Watanabe H., Tamura H., Yasui H., Matsushita R., Takada J. Inorg. Chim. Acta. 1998;283:175. [Google Scholar]
- 172.DiStefano J.J., III, Laidlaw E.M. Am. J. Physiol. (Reg. Int. Comp. Physiol. 15) 1984;246:R651. [Google Scholar]
- 173.Al-Bayati M.A., Raabe O.G., Giri S.B., Knaak J.B. J. Am. Coll. Toxicol. 1991;10:233. [Google Scholar]
- 174.Etcheverry S.B., Apella M.C., Baran E.J. J. Inorg. Biochem. 1984;20:269. doi: 10.1016/0162-0134(84)85025-4. [DOI] [PubMed] [Google Scholar]
- 175.Dikanov S.A., Liboiron B.D., Thompson K.H., Vera E., Yuen V.G., McNeill J.H., Orvig C. J. Am. Chem. Soc. 1999;121:11004. [Google Scholar]
- 176.Williams R.J.P. Eur. J. Biochem. 1985;150:231. doi: 10.1111/j.1432-1033.1985.tb09013.x. [DOI] [PubMed] [Google Scholar]
- 177.Zhang S.Q., Chen G.H., Lu W.L., Zhang Q. J. Bone Miner. Metab. 2007;25:293. doi: 10.1007/s00774-007-0759-7. [DOI] [PubMed] [Google Scholar]
- 178.Kyriazis G.A., Sounddarapandian M.M., Tyrberg B. Proc. Natl. Acad. Sci. U.S.A. 2012;109:2713. doi: 10.1073/pnas.1115183109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Scully T. Nature. 2012;458:S2. doi: 10.1038/485s2a. [DOI] [PubMed] [Google Scholar]
- 180.Crans D.C., Tarlton M.L., McLauchlan C.C. Eur. J. Inorg. Chem. 2014;2014:4450–4468. [Google Scholar]
- 181.McNeill J.H., Yuen V.G., Hoveyda H.R., Orvig C. J. Med. Chem. 1992;35:1489. doi: 10.1021/jm00086a020. [DOI] [PubMed] [Google Scholar]
- 182.Levina A., Lay P.A. Dalton Trans. 2011;40:11675. doi: 10.1039/c1dt10380f. [DOI] [PubMed] [Google Scholar]
- 183.Hiromura M., Adachi Y., Machida M., Hattori M., Sakurai H. Metallomics. 2009;1:92. [Google Scholar]
- 184.Rondini E.A., Walters D.M., Bauer A.K. Part. Fibre Toxicol. 2010;7:1. doi: 10.1186/1743-8977-7-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Sakurai H. Chem. Rec. 2002;2:237. doi: 10.1002/tcr.10029. [DOI] [PubMed] [Google Scholar]
- 186.Fugono J., Yasui H., Sakurai H. J. Pharm. Pharmacol. 2001;53:1247. doi: 10.1211/0022357011776531. [DOI] [PubMed] [Google Scholar]
- 187.Brand R.M., Hamel F.G. Int. J. Pharm. 1999;183:117. doi: 10.1016/s0378-5173(99)00071-x. [DOI] [PubMed] [Google Scholar]
- 188.Brand R.M., Duensing G., Hamel F.G. Int. J. Pharm. 1997;146:115. [Google Scholar]
- 189.Crans D.C., Mahroof-Tahir M., Johnson M.D., Wilkins P.C., Yang L.Q., Robbins K., Johnson A., Alfano J.A., Godzala M.E., Austin L.T., Willsky G.R. Inorg. Chim. Acta. 2003;356:365. [Google Scholar]
- 190.Buglyo P., Crans D.C., Nagy E.M., Lindo R.L., Yang L.Q., Smee J.J., Jin W.Z., Chi L.H., Godzala M.E., Willsky G.R. Inorg. Chem. 2005;44:5416. doi: 10.1021/ic048331q. [DOI] [PubMed] [Google Scholar]
- 191.Hanson G.R., Sun Y., Orvig C. Inorg. Chem. 1996;35:6507. doi: 10.1021/ic960490p. [DOI] [PubMed] [Google Scholar]
- 192.Fukui K., Fujisawa Y., Ohya-Nishiguchi H., Kamada H., Sakurai H. J. Inorg. Biochem. 1999;77:215. doi: 10.1016/s0162-0134(99)00204-4. [DOI] [PubMed] [Google Scholar]
- 193.Dikanov S.A., Liboiron B.D., Orvig C. J. Am. Chem. Soc. 2002;124:2969. doi: 10.1021/ja011104s. [DOI] [PubMed] [Google Scholar]
- 194.Liboiron B.D., Thompson K.H., Hanson G.R., Lam E., Aebischer N., Orvig C. J. Am. Chem. Soc. 2005;127:5104. doi: 10.1021/ja043944n. [DOI] [PubMed] [Google Scholar]
- 195.Willsky G.R., Godzalla M.E., Kostyniak P.J., Chi L.H., Gupta R., Yuen V.G., McNeill J.H., Mahroof-Tahir M., Smee J.J., Yang L.Q., Lobernick A., Watson S., Crans D.C. In: Kustin K., Pessoa J.C., Crans D.C., editors. ACS Symp. Ser. 974; Washington, DC; 2007. p. 93. [Google Scholar]
- 196.Sakurai H. In: Kustin K., Pessoa J.C., Crans D.C., editors. Vanadium: The Versatile Metal. ACS Symp. Ser. 974; Washington, DC; 2007. p. 110. [Google Scholar]
- 197.Harris W.R. Biochemistry. 1985;24:7412. doi: 10.1021/bi00346a057. [DOI] [PubMed] [Google Scholar]
- 198.Bordbar A.-K., Creagh A.L., Mohammadi F., Haynes C.A., Orvig C. J. Inorg. Biochem. 2009;103:643. doi: 10.1016/j.jinorgbio.2008.10.009. [DOI] [PubMed] [Google Scholar]
- 199.Sanna D., Micera G., Garribba E. Inorg. Chem. 2009;48:5747. doi: 10.1021/ic802287s. [DOI] [PubMed] [Google Scholar]
- 200.Wei Y., Zhang C., Zhao P., Yang X., Wang K. J. Inorg. Biochem. 2011;105:1081. doi: 10.1016/j.jinorgbio.2011.05.008. [DOI] [PubMed] [Google Scholar]
- 201.Rehder D. John Wiley & Sons; Chichester, UK: 2008. Bioinorganic Vanadium Chemistry. [Google Scholar]
- 202.Li M., Ding W., Baruah B., Crans D.C., Wang R. J. Inorg. Biochem. 2008;102:1846. doi: 10.1016/j.jinorgbio.2008.06.007. [DOI] [PubMed] [Google Scholar]
- 203.Domingues N., Pelletier J., Ostenson C.G., Castro M.M.C.A. J. Inorg. Biochem. 2014;135:115. doi: 10.1016/j.jinorgbio.2013.11.005. [DOI] [PubMed] [Google Scholar]
- 204.Vardatsikos G., Mehdi M.Z., Srivastava A.K. Int. J. Mol. Med. 2009;24:303. doi: 10.3892/ijmm_00000233. [DOI] [PubMed] [Google Scholar]
- 205.Winter P.W., Al-Qatati A., Wolf-Ringwall A.L., Schoeberl S., Chatterjee P.B., Barisas B.G., Roess D.A., Crans D.C. Dalton Trans. 2012;41:6419. doi: 10.1039/c2dt30521f. [DOI] [PubMed] [Google Scholar]
- 206.Makinen M.W., Rivera S.E., Zhou K.I., Brady M.J. In: Vanadium: The Versatile Metal. ACS Symp. Ser. 947, Washington, DC. Kustin K., Crans D.C., Costa Pessoa J., editors. 2007. pp. 82–92. [Google Scholar]
- 207.Li J.P., Elberg G., Crans D.C., Shechter Y. Biochemistry. 1996;35:8314. doi: 10.1021/bi960209i. [DOI] [PubMed] [Google Scholar]
- 208.Vincent J.B., Love S. Biochim. Biophys. Acta. 2012;1820:362. doi: 10.1016/j.bbagen.2011.07.003. [DOI] [PubMed] [Google Scholar]
- 209.Chen J., Ostenson C.G. Biochem. Biophys. Res. Commun. 2004;325:555. doi: 10.1016/j.bbrc.2004.10.077. [DOI] [PubMed] [Google Scholar]
- 210.Drake P.G., Balbis A., Wu J., Bergeron J.J., Posner B.I. Am. J. Physiol. Endocrinol. Metab. 2000;279:E266. doi: 10.1152/ajpendo.2000.279.2.E266. [DOI] [PubMed] [Google Scholar]
- 211.Comi R.J., Grunberger G., Gorden P. J. Clin. Invest. 1987;79:453. doi: 10.1172/JCI112833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Hundberg A., Vaag A., Vinten J., Beck-Nielson H. Diabetologia. 1993;36:668. doi: 10.1007/BF00404079. [DOI] [PubMed] [Google Scholar]
- 213.Salmeen A., Andersen J.N., Myers M.P., Meng T.C., Hinks J.A., Tonks N.K., Barford D. Nature. 2003;423:769. doi: 10.1038/nature01680. [DOI] [PubMed] [Google Scholar]
- 214.Minasi L.A., Willsky G.R. J. Bacteriol. 1991;173:834. doi: 10.1128/jb.173.2.834-841.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Cortizo A.M., Caporossi M., Lettieri G., Etcheverry S.B. Eur. J. Pharmacol. 2000;400:279. doi: 10.1016/s0014-2999(00)00356-3. [DOI] [PubMed] [Google Scholar]
- 216.Crans D.C., Trujillo A., Pharazyn P., Cohen M. Coord. Chem. Rev. 2011;7:2178. [Google Scholar]
- 217.Boden G., Chen X., Ruiz J., van Rossum G.D.V., Turco S. Metabolism. 1996;45:1130. doi: 10.1016/s0026-0495(96)90013-x. [DOI] [PubMed] [Google Scholar]
- 218.Goldfine A.B., Simonson D.C., Folli F., Patti M.E., Kahn C.R. J. Clin. Endocrinol. Metab. 1995;80:3311. doi: 10.1210/jcem.80.11.7593444. [DOI] [PubMed] [Google Scholar]
- 219.Cohen N., Halberstam M., Shlimovich P., Chang C.J., Shamoon H., Rossetti L. J. Clin. Invest. 1995;95:2501. doi: 10.1172/JCI117951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Goldfine A.B., Willsky G.R., Kahn C.R. In: Vanadium Compounds: Chemistry, Biochemistry and Therapeutic Applications. Tracey A.S., Crans D.C., editors. Oxford University Press; New York: 1998. pp. 353–367. [Google Scholar]
- 221.Halberstam M., Cohen N., Shlimovich P., Chang C.J., Shamoon H., Rossetti L. J. Clin. Invest. 1996;45:659. doi: 10.1172/JCI117951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Nielsen F.H. In: Sigel H., Sigel A., editors. vol. 31. Marcel Dekker, Inc.; New York: 1995. pp. 543–574. (Metal Ions in Biological Systens). [Google Scholar]
- 223.James M.M., Charles S.C. J. Biol. Chem. 1982;257:4362. [Google Scholar]
- 224.Shisheva A., Gefel D., Shechter Y. Diabetes. 1992;41:982. doi: 10.2337/diab.41.8.982. [DOI] [PubMed] [Google Scholar]
- 225.Chakraborty T., Chatterjee A., Saralaya M.G., Chaterjee M. J. Biol. Inorg. Chem. 2006;11:855. doi: 10.1007/s00775-006-0128-3. [DOI] [PubMed] [Google Scholar]
- 226.Cruz T.F., Morgan A., Min W. Mol. Cell. Biochem. 1995;153:161. doi: 10.1007/BF01075933. [DOI] [PubMed] [Google Scholar]
- 227.Dong Y., Narla R.K., Sudbeck E., Uckun F.M. J. Inorg. Biochem. 2000;78:321. doi: 10.1016/s0162-0134(00)00060-x. [DOI] [PubMed] [Google Scholar]
- 228.Narla R.K., Chen C.L., Dong Y.H., Uckun F.M. Clin. Cancer Res. 2001;7:1094. [PubMed] [Google Scholar]
- 229.Hall I.H., Durham R.W., Tram M., Mueller S., Rachandran B.M., Sneddon L.G. J. Inorg. Biochem. 2003;93:125. doi: 10.1016/s0162-0134(02)00565-2. [DOI] [PubMed] [Google Scholar]
- 230.Vinklárek J., Pavlík I., Cernosek Z. Met. Based Drug. 1997;4:207. doi: 10.1155/MBD.1997.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Aubrecht J., Narla R.K., Ghosh P., Stanek J., Uckun F.M. Toxicol. Appl. Pharmacol. 1999;154:228. doi: 10.1006/taap.1998.8592. [DOI] [PubMed] [Google Scholar]
- 232.Ghosh P., D’Cruz O.J., Narla R.K., Uckun F.M. Clin. Cancer Res. 2000;6:1536. [PubMed] [Google Scholar]
- 233.Moridani M.Y., Pourahmad J., Bui H., Siraki A., O’Brien P.J. Free Rad. Biol. Med. 2003;34:243. doi: 10.1016/s0891-5849(02)01241-8. [DOI] [PubMed] [Google Scholar]
- 234.Ferrer E.G., Williams P.A.M. In: Handbook on Flavonoids: Dietary Sources, Properties and Health Benefits. Pandi-Perumal S.R., Daniel P., Cardinali, editors. Nova Science Publishers; New York: 2011. pp. 145–190. (Chapter 3) [Google Scholar]
- 235.Kim J.D., Liu L., Guo W., Meydani M. J. Nutr. Biochem. 2006;17:165. doi: 10.1016/j.jnutbio.2005.06.006. [DOI] [PubMed] [Google Scholar]
- 236.Ferrer E.G., Salinas M.V., Correa M.J., Naso L., Barrio D.A., Etcheverry S.B., Lezama L., Rojo T., Williams P.A.M. J. Biol. Inorg. Chem. 2006;11:791. doi: 10.1007/s00775-006-0122-9. [DOI] [PubMed] [Google Scholar]
- 237.Etcheverry S.B., Ferrer E.G., Naso L., Rivadeneira J., Salinas V., Williams P.A. J. Biol. Inorg. Chem. 2008;13:435. doi: 10.1007/s00775-007-0332-9. [DOI] [PubMed] [Google Scholar]
- 238.Yang C.S., Landau J.M., Huang M.T., Newmark H.L. Annu. Rev. Nutr. 2001;21:381. doi: 10.1146/annurev.nutr.21.1.381. [DOI] [PubMed] [Google Scholar]
- 239.Ge Y., Zhang Y., Chen Y., Li Q., Chen J., Dong Y., Shi W. Int. J. Mol. Sci. 2011;12:4861. doi: 10.3390/ijms12084861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Agarwal C., Wadhwa R., Deep G., Biedermann D., Gažák R., Křen V., Agarwal R. PLOS ONE. 2013;8:e60074. doi: 10.1371/journal.pone.0060074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Singh R.P., Agarwal R. Curr. Cancer Drug Targets. 2004;4:1. doi: 10.2174/1568009043481605. [DOI] [PubMed] [Google Scholar]
- 242.Mokhtari M.J., Motamed N., Shokrgozar M.A. Cell Biol. Int. 2008;32:888. doi: 10.1016/j.cellbi.2008.03.019. [DOI] [PubMed] [Google Scholar]
- 243.Naso L.G., Ferrer E.G., Butenko N., Cavaco I., Lezama L., Rojo T., Etcheverry S.B., Williams P.A.M. J. Biol. Inorg. Chem. 2011;16:653. doi: 10.1007/s00775-011-0769-8. [DOI] [PubMed] [Google Scholar]
- 244.Leon I.E., Porro V., Di Virgilio A.L., Naso L.G., Williams P.A., Bollati-Fogolín M., Etcheverry S.B. J. Biol. Inorg. Chem. 2014;19:59. doi: 10.1007/s00775-013-1061-x. [DOI] [PubMed] [Google Scholar]
- 245.Leon I.E., Di Virgilio A.L., Porro V., Muglia C.I., Naso L.G., Williams P.A., Bollati-Fogolin M., Etcheverry S.B. Dalton Trans. 2013;42:11868. doi: 10.1039/c3dt50524c. [DOI] [PubMed] [Google Scholar]
- 246.Goldwaser Y., Qian S., Gershonov E., Fridkin M., Shechter Y. Mol. Pharmacol. 2000;58:738. doi: 10.1124/mol.58.4.738. [DOI] [PubMed] [Google Scholar]
- 247.Islam M.N., Kumbhar A.A., Kumbhar A.S., Zeller M., Butcher R.J., Dusane M.B., Joshi B.N. Inorg. Chem. 2010;49:8237. doi: 10.1021/ic9025359. [DOI] [PubMed] [Google Scholar]
- 248.Yamaguchi T., Watanabe S., Matsumura Y., Tokuoka Y., Yokoyama A. Bioorg. Med. Chem. 2012;20:3058. doi: 10.1016/j.bmc.2012.02.063. [DOI] [PubMed] [Google Scholar]
- 249.Fu Y., Wang Q., Yang X.G., Yang X.D., Wang K. J. Biol. Inorg. Chem. 2008;13:1001. doi: 10.1007/s00775-008-0387-2. [DOI] [PubMed] [Google Scholar]
- 250.Liasko R., Kabanos T.A., Karkabounas S., Malamas M., Tasiopoulos A.T., Stefanou D., Collery P., Evangelou A. Anticancer Res. 1998;18:3609. [PubMed] [Google Scholar]
- 251.Evangelou A., Karkabounas S., Kalpouzos G., Malamas M., Liasko R., Stefanou D., Vlahos A.T., Kabanos T.A. Cancer Lett. 1997;119:221. doi: 10.1016/s0304-3835(97)00278-4. [DOI] [PubMed] [Google Scholar]
- 252.Faneca H., Figueiredo V.A., Tomaz A.I., Gonçalves G., Avecilla F., Pedroso de Lima M.C., C-Geraldes C.F.G., Costa Pessoa J., Castro M.M.C.A. J. Inorg. Biochem. 2009;103:601. doi: 10.1016/j.jinorgbio.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 253.Del Río D., Galindo A., Vicente R., Mealli C., Ienco A., Masi D. Dalton Trans. 2003;9:1813. [Google Scholar]
- 254.Thompson K.H., Tsukada Y., Xu Z., Battell M., McNeill J.H., Orvig C. Biol. Trace Elem. Res. 2002;86:31. doi: 10.1385/BTER:86:1:31. [DOI] [PubMed] [Google Scholar]
- 255.Rivadeneira J., Barrio D.A., Etcheverry S.B., Baran E.J. Biol. Trace Elem. Res. 2007;118:159. doi: 10.1007/s12011-007-0025-9. [DOI] [PubMed] [Google Scholar]
- 256.Rivadeneira J., Di Virgilio A.L., Barrio D.A., Muglia C.I., Bruzzone L., Etcheverry S.B. Med. Chem. 2010;6:9. doi: 10.2174/157340610791208754. [DOI] [PubMed] [Google Scholar]
- 257.León I.E., Etcheverry S.B., Parajón-Costa B.S., Baran E.J. Biol. Trace Elem. Res. 2012;147:403. doi: 10.1007/s12011-012-9322-z. [DOI] [PubMed] [Google Scholar]
- 258.León I.E., Etcheverry S.B., Parajón-Costa B.S., Baran E.J. J. Mex. Chem. Soc. 2013;57:175. [Google Scholar]
- 259.León I.E., Butenko N., Di Virgilio A.L., Muglia C.I., Baran E.J., Cavaco I., Etcheverry S.B. J. Inorg. Biochem. 2014;134:106. doi: 10.1016/j.jinorgbio.2013.10.009. [DOI] [PubMed] [Google Scholar]
- 260.Yodoshi M., Odoko M., Okabe N. Chem. Pharm. Bull. 2007;55:853. doi: 10.1248/cpb.55.853. [DOI] [PubMed] [Google Scholar]
- 261.Djordjevic C., Wampler G.L. J. Inorg. Biochem. 1985;25:51. doi: 10.1016/0162-0134(85)83007-5. [DOI] [PubMed] [Google Scholar]
- 262.León I.E., Di Virgilio A.L., Barrio D.A., Arrambide G., Gambino D., Etcheverry S.B. Metallomics. 2012;4:1287. doi: 10.1039/c2mt20091k. [DOI] [PubMed] [Google Scholar]
- 263.Zhang Z., Chen F., Huang C., Shi X. J. Environ. Pathol. Toxicol. Oncol. 2002;21:223. [PubMed] [Google Scholar]
- 264.Parrondo R., de las Pozas A., Reiner T., Rai P., Perez-Stable C. Mol. Cancer. 2010;9:182. doi: 10.1186/1476-4598-9-182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Morita A., Yamamoto S., Wang B., Tanaka K., Suzuki N., Aoki S., Ito A., Nanao T., Ohya S., Yoshino M., Zhu J., Enomoto A., Matsumoto Y., Funatsu O., Hosoi Y., Ikekita M. Cancer Res. 2010;70:257. doi: 10.1158/0008-5472.CAN-08-3771. [DOI] [PubMed] [Google Scholar]
- 266.Ray R.S., Ghosh B., Rana A., Chatterjee M. Int. J. Cancer. 2007;120:13. doi: 10.1002/ijc.22277. [DOI] [PubMed] [Google Scholar]
- 267.Chakraborty T., Swamy A.H., Chatterjee A., Rana B., Shyamsundar A., Chatterjee M. J. Cell. Biochem. 2007;101:244. doi: 10.1002/jcb.21169. [DOI] [PubMed] [Google Scholar]
- 268.Rehder D. In: Reedijk J., Poeppelmeier K., editors. vol. 3. Elsevier; Oxford: 2013. pp. 819–834. (Comprehensive Inorganic Chemistry II). [Google Scholar]
- 269.http://www.who.int/trypanosomiasis_african/en/ (accessed 19.09.14).
- 270.Simarro P.P., Diarra A., Ruiz Postigo J.A., Franco J.R., Jannin J.G. PLoS Negl. Trop. Dis. 2011;5:e1007. doi: 10.1371/journal.pntd.0001007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Hotez P.J., Molyneux D.H., Fenwick A., Kumaresan J., Ehrlich Sachs S., Sachs J.D., Savioli L. N. Engl. J. Med. 2007;357:1018. doi: 10.1056/NEJMra064142. [DOI] [PubMed] [Google Scholar]
- 272.Delespaux V., de Koning H.P. Drug Resist. Updates. 2007;10:30. doi: 10.1016/j.drup.2007.02.004. [DOI] [PubMed] [Google Scholar]
- 273.Nussbaum K., Honek J., Cadmus C.M.C.v.C., Efferth T. Curr. Med. Chem. 2010;17:1594. doi: 10.2174/092986710790979953. [DOI] [PubMed] [Google Scholar]
- 274.Kinnamon K., Steck E.A., Rane E.S. Antimicrob. Agents Chemother. 1979;15:157. doi: 10.1128/aac.15.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Sánchez-Delgado R.A., Anzellotti A., Suárez L. In: Sigel H., Sigel A., editors. vol. 41. Marcel Dekker; New York: 2004. pp. 379–419. (Metal Ions in Biological Systems). [PubMed] [Google Scholar]
- 276.Navarro M., Gabbiani G., Messori L., Gambino D. Drug Discov. Today. 2010;15:1070. doi: 10.1016/j.drudis.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 277.Benítez J., Correia I., Becco L., Fernández M., Garat B., Gallardo H., Conte G., Kuznetsov M.L., Neves A., Moreno V., Costa Pessoa J., Gambino D. Z. Anorg. Allg. Chem. 2013;639:1417. [Google Scholar]
- 278.Benítez J., Becco L., Correia I., Milena Leal S., Guiset H., Costa Pessoa J., Lorenzo J., Tanco S., Escobar P., Moreno V., Garat B., Gambino D. J. Inorg. Biochem. 2011;105:303. doi: 10.1016/j.jinorgbio.2010.11.001. [DOI] [PubMed] [Google Scholar]
- 279.Fernández M., Becco L., Correia I., Benítez J., Piro O.E., Echeverria G.A., Medeiros A., Comini M., Lavaggi M.L., González M., Cerecetto H., Moreno V., Costa Pessoa J., Garat B., Gambino D. J. Inorg. Biochem. 2013;127:150. doi: 10.1016/j.jinorgbio.2013.02.010. [DOI] [PubMed] [Google Scholar]
- 280.Fernández M., Varela J., Correia I., Birriel E., Castiglioni J., Moreno V., Costa Pessoa J., Cerecetto H., González M., Gambino D. Dalton Trans. 2013;42:11900. doi: 10.1039/c3dt50512j. [DOI] [PubMed] [Google Scholar]
- 281.Romanha A.J., Lisboa de Castro S.de M., Correia Soeiro N., Lannes-Vieira J., Ribeiro I., Talvani A., Bourdin B., Blum B., Olivieri B., Zani C., Spadafora C., Chiari E., Chatelain E., Chaves G., Calzada J.E., Bustamante J.M., Freitas L.H., Jr., Romero L., Bahia M.T., Lotrowska M., Soares M., Gumes Andrade S., Armstrong T., Degrave W., de Araújo Andrade Z. Mem. Inst. Oswaldo Cruz. 2010;105:233. doi: 10.1590/s0074-02762010000200022. [DOI] [PubMed] [Google Scholar]
- 282.Cazzulo J.J. FASEB. 1992;6:3153. doi: 10.1096/fasebj.6.13.1397837. [DOI] [PubMed] [Google Scholar]
- 283.Bringaud F., Rivière L., Coustou V. Mol. Biochem. Parasitol. 2006;149:1. doi: 10.1016/j.molbiopara.2006.03.017. [DOI] [PubMed] [Google Scholar]
- 284.Benítez D., Lavaggi M.L., Gambino D., Torre M.H., Cerecetto H., González M. Med. Chem. Res. 2012;21:1439. [Google Scholar]
- 285.El-Sayed N.M., et al. Science. 2005;309:404. doi: 10.1126/science.1112181. [DOI] [PubMed] [Google Scholar]
- 286.Ribeiro I., Sevcsik A.M., Alves F., Diap G., Don R., Harhay M.O., Chang S., Pecoul B. PLoS Negl. Trop. Dis. 2009;3:e484. doi: 10.1371/journal.pntd.0000484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 287.Chibale K. ARKIVOC. 2002;IX:93. [Google Scholar]
- 288.Machado I., Fernández M., Becco L., Garat B., Brissos R.F., Zabarska N., Gamez P., Marques F., Correia I., Costa Pessoa J., Gambino D. Inorg. Chim. Acta. 2014;420:39. [Google Scholar]
- 289.Benítez J., Cavalcanti de Queiroz A., Correia I., Amaral Alves M., Alexandre-Moreira M.S., Barreiro E.J., Moreira Lima L., Varela J., González M., Cerecetto H., Moreno V., Costa Pessoa J., Gambino D. Eur. J. Med. Chem. 2013;62:20. doi: 10.1016/j.ejmech.2012.12.036. [DOI] [PubMed] [Google Scholar]
- 290.Turner T.L., Nguyen V.H., McLauchlan C.C., Dymon Z., Dorsey B.M., Hooker J.D., Jones M.A. J. Inorg. Biochem. 2012;108:96. doi: 10.1016/j.jinorgbio.2011.09.009. [DOI] [PubMed] [Google Scholar]
- 291.Adriazola I.O., doAmaral A.E., Amorim J.C., Lourenço Correia B., Oliveira Petkowicz C.L., Ramalho Mercê A.L., Rodrigues Noleto G. J. Inorg. Biochem. 2014;132:45. doi: 10.1016/j.jinorgbio.2013.09.017. [DOI] [PubMed] [Google Scholar]
- 292.Maurya M.R., Haldar C., Alam Khan A., Azam A., Salahuddin A., Kumar A., Costa Pessoa J. Eur. J. Inorg. Chem. 2012;15:2560. [Google Scholar]
- 293.Sun R.W.Y., Ma D.L., Wong E.L.M., Che C.M. Dalton Trans. 2007:1. doi: 10.1039/b705079h. [DOI] [PubMed] [Google Scholar]
- 294.Shigeta S., Mori S., Kodama E., Kodama J., Takahashi K., Yamase T. Antiviral Res. 2003;58:265. doi: 10.1016/s0166-3542(03)00009-3. [DOI] [PubMed] [Google Scholar]
- 295.Yamase T., Ishikawa E., Fukaya K., Nojiri H., Taniguchi T., Atake T. Inorg. Chem. 2004;43:8150. doi: 10.1021/ic049669n. [DOI] [PubMed] [Google Scholar]
- 296.Thompson K.H., Orvig C. Coord. Chem. Rev. 2001;219:1033. [Google Scholar]
- 297.Totrov M., Abagyan R. Proteins. 1997;(Suppl. 1):215. doi: 10.1002/(sici)1097-0134(1997)1+<215::aid-prot29>3.3.co;2-i. [DOI] [PubMed] [Google Scholar]
- 298.Renirie R., Dewilde A., Pierlot C., Wever R., Hober D., Aubry J.-M. J. Appl. Microbiol. 2008;105:264. doi: 10.1111/j.1365-2672.2008.03742.x. [DOI] [PubMed] [Google Scholar]
- 299.Mait A., Ghosh S. J. Inorg. Biochem. 1989;36:131. doi: 10.1016/0162-0134(89)80020-0. [DOI] [PubMed] [Google Scholar]
- 300.da P.I., Maia S., Pavan F.R., Leite C.Q., Lemos S.S., de Sousa G.F., Batista A.A., Nascimento O.R., Ellena J., Castellano E.E., Niquet E., Deflon V.M. Polyhedron. 2009;28:398. [Google Scholar]
- 301.Inoue M., Segawa K., Matsunaga S., Matsumoto N., Oda M., Yamase T. J. Inorg. Biochem. 2005;99:1023. doi: 10.1016/j.jinorgbio.2005.01.010. [DOI] [PubMed] [Google Scholar]
- 302.Patel M.N., Patel S.H., Chhasatia M.R., Parmar P.A. Bioorg. Med. Chem. Lett. 2008;18:6494. doi: 10.1016/j.bmcl.2008.10.068. [DOI] [PubMed] [Google Scholar]
- 303.Rosu T., Pahontu E., Ilies D.C., Georgescu R., Mocanu M., Leabu M., Shova S., Gulea A. Eur. J. Med. Chem. 2012;53:380. doi: 10.1016/j.ejmech.2012.03.046. [DOI] [PubMed] [Google Scholar]
- 304.Sahani M.K., Yadava U., Pandey O.P., Sengupta S.K. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2014;125:189. doi: 10.1016/j.saa.2014.01.041. [DOI] [PubMed] [Google Scholar]