Abstract
Purpose
To determine if neurally adjusted ventilatory assist (NAVA) improves asynchrony, ventilatory drive, breath-to-breath variability and COMFORT score when compared to pressure support (PS).
Methods
This is a non-randomized short-term cross-over trial in which 12 pediatric patients with asynchrony (auto-triggering, double triggering or non-triggered breaths) were enrolled. Four sequential 10-min periods of data were recorded after 20 min of ventilatory stabilization (wash-out) at each of the following settings: baseline PS with the ventilator settings determined by the attending physician (1-PSb); PS after optimization (2-PSopt); NAVA level set so that maximum inspiratory pressure (P max) equaled P max in PS (3-NAVA); same settings as in 2-PSopt (4-PSopt).
Results
The median asynchrony index was significantly lower during NAVA (2.0 %) than during 2-PSopt (8.5 %, p = 0.017) and 4-PSopt (7.5 %, p = 0.008). In NAVA mode, the NAVA trigger accounted on average for 66 % of triggered breaths. The median trigger delay with respect to neural inspiratory time was significantly lower during NAVA (8.6 %) than during 2-PSopt (25.2 %, p = 0.003) and 4-PSopt (28.2 %, p = 0.0005). The median electrical activity of the diaphragm (EAdi) change during trigger delay normalized to maximum inspiratory EAdi difference was significantly lower during NAVA (5.3 %) than during 2-PSopt (21.7 %, p = 0.0005) and 4-PSopt (24.6 %, p = 0.001). The coefficient of variation of tidal volume was significantly higher during NAVA (44.2 %) than during 2-PSopt (19.8 %, p = 0.0002) and 4-PSopt (23.0 %, p = 0.0005). The median COMFORT score during NAVA (15.0) was lower than that during 2-PSopt (18.0, p = 0.0125) and 4-PSopt (17.5, p = 0.039). No significant changes for any variable were observed between 1-PSb and 2-PSopt.
Conclusions
Neurally adjusted ventilatory assist as compared to optimized PS results in improved synchrony, reduced ventilatory drive, increased breath-to-breath mechanical variability and improved patient comfort.
Electronic supplementary material
The online version of this article (doi:10.1007/s00134-012-2535-y) contains supplementary material, which is available to authorized users.
Keywords: Pediatrics, Neurally adjusted ventilatory assist, Pressure support, Mechanical ventilation, Patient–ventilator interaction, Comfort
Introduction
Dyspnea is one of the most frequent diagnosis for admission to the pediatric intensive care unit (PICU). It is associated with conditions in which the respiratory drive is increased or the respiratory system is subject to an increased mechanical load [1], a condition usually seen in mechanically ventilated patients. Asynchrony and decreased breath-to-breath variability have been associated with increased mechanical load and respiratory drive [2, 3]. Moreover, in children, asynchrony during assisted ventilation is common [4–6]. As a result, the approach to ventilatory support in children is usually pressure targeted ventilation, since it allows gas delivery to vary based on patient demand [4].
Neurally adjusted ventilatory assist (NAVA) is designed to improve synchrony [7, 8]. With NAVA, gas delivery is triggered, controlled and cycled by the diaphragmatic electromyogram signal (EAdi). The ventilator captures the change in the EAdi by a specially manufactured nasogastric tube equipped with ten electrodes. A number of studies in neonates and pediatric patients have demonstrated that patient ventilator synchrony is improved with the application of NAVA [4, 6], and studies in adults have shown that NAVA increases ventilatory variability compared to pressure support (PS) [9–11]. To maintain safety and patient comfort, large amounts of sedation are often necessary in ventilated PICU patients. To the best of our knowledge, however, there have been no studies directly examining the sedation needs of children being mechanically ventilated using NAVA compared to traditional ventilation modes.
We hypothesized that the use of NAVA would, compared to optimized PS, decrease asynchrony and ventilatory drive and increase breath-to-breath mechanical variability, leading to a decrease in dyspnea and therefore impacting the COMFORT score.
Patients and methods
This was a non-randomized short-term cross-over trial [12] conducted in the medical/surgical PICU. It was approved by the Ethics Committee of the Hospital Universitario Materno-Infantil La Paz (HULP-3132). Signed written informed consent was obtained from the parents of all patients.
Study protocol
A total of 12 patients endotracheally intubated and spontaneously triggering the mechanical ventilator were enrolled. There were no dropouts from the study. Patients ranged in age from newborns to 16 years. Each patient demonstrated major patient–ventilator asynchrony: ineffective triggering, auto-triggering and/or double triggering, which are described in detail in the Electronic Supplementary Material (ESM). All patients were ventilated using a Servo-i ventilator (Maquet Critical Care, Solna, Sweden) equipped with NAVA.
After informed consent was obtained, each patient was fitted with a specially manufactured nasogastric tube equipped with an array for sensing the electrical activity of the diaphragm (EAdi). There were no difficulties in positioning the NAVA-nasogastric tube. Four sequential 10-min periods of data were recorded after 20 min of ventilatory stabilization (wash-out) at each of the following settings: baseline PS with the ventilator settings determined by the attending physician (1-PSb); PS after optimization (2-PSopt); NAVA level set so that maximum inspiratory pressure (P max) equaled P max in PS (3-NAVA); same settings as in 2-PSopt (4-PSopt).
After recording 1-PSb, we optimized rise time, inspiratory termination criteria, pressure level, sensitivity and applied positive end-expiratory pressure (PEEP) according to the criteria described in the ESM.
Data collection
Pressure, flow, etCO2 (end-tidal carbon dioxide concentration in the expired air) and EAdi curves were recorded using the NAVA tracker v3.0 software (Maquet Critical Care AB, Solna, Sweden) for automated breath-by-breath analysis and direct visual evaluation of asynchronies. Major asynchronies were reported as the total number of asynchronous events (auto-triggered breaths, double triggering and non-triggered breaths) per minute and as an asynchrony index [AI = 100 × number of major asynchrony events/(ventilator respiratory rate + non-triggered breaths)] [13]. The COMFORT score [14] was evaluated at the end of each phase of the study. The sedation regime was that determined by the PICU attending physician in charge according to the PICU policies and guidelines (see ESM). Children had been on the same sedation and analgesia regime for at least 3 days before being admitted to the study. No changes in sedation or analgesia (Table 1) were made throughout the study. The following variables were used for data analysis: neurobreath (defined as a breath with a ΔEAdi >1.5 μV for more than 150 ms); a breath’s maximum EAdi (maxEAdi); a breath’s initial baseline inspiratory EAdi (minEAdi); a breath’s inspiratory pressure (P max). Neural inspiratory time (nTi) was defined as the time between minEAdi and maxEAdi, or between minEAdi and the end of the EAdi plateau; mechanical inspiratory time (mTi) was defined as the time with positive flow.
Table 1.
Basic clinical details on the patients
| ID | Age | PBW (kg) | Gender | Days on MV | Days in PICU | PaO2/FiO2 | Disease on admission | Reason for MV | Sedo-analgesic regime |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 4 months | 5.7 | Male | 71 | 98 | 422 | Congenital tracheal stenosis | ARF, respiratory ECMO |
DZP 0.15 mg/kg b.i.d. MTD 0.15 mg/kg b.i.d. |
| 2 | 4 months | 4.8 | Female | 198 | 198 | 137 | Acute liver failure. Neonatal hemochromatosis | MOF, liver transplantation. Pulmonary fibrosis |
MDZ 0.16 mg/kg/h FEN 1.6 μg/kg/h |
| 3 | 2 months | 4.6 | Male | 50 | 125 | 57 | Fallot tetralogy | Cardiogenic shock |
MDZ 0.15 mg/kg/h KET 0.075 mg/kg/h |
| 4 | 6 months | 5.3 | Male | 41 | 51 | No arterial access | AoCo, IVC, pulmonary artery banding | Aspiration pneumonia |
MDZ 1 mg/kg/h RMF 15 μg/kg/h |
| 5 | 3 months | 5 | Male | 8 | 15 | 176 | RSV bronchiolitis | ARDS |
MDZ 0.12 mg/kg/h KET 0.07 mg/kg/h |
| 6 | 6 months | 7.2 | Male | 21 | 32 | 158 | Influenza A (H1N1), pneumocistys carinii pneumonia, hyper IgM immunodeficiency | ARDS |
MDZ 0.25 mg/kg/h FEN 4 μg/kg/h |
| 7 | 7 weeks | 4.8 | Male | 9 | 18 | 178 | RSV bronchiolitis | ARDS |
MDZ 0.07 mg/kg/h FEN 0.7 μg/kg/h |
| 8 | 2.5 years | 13.9 | Male | 10 | 22 | 140 | Necrotizing pneumonia, empyema | Neumothorax, bronchopleural fistula |
DZP 0.25 mg/kg q.i.d MTD 0.33 mg/kg b.i.d. |
| 9 | 2 months | 5.3 | Male | 7 | 9 | 189 | RSV bronchiolitis | ARF |
DZP 0.15 mg/kg t.i.d. MOR 0.1 mg/kg t.i.d. |
| 10 | 9 days | 4.5 | Male | 6 | 13 | No arterial access | RSV bronchiolitis | ARF |
MDZ 0.25 mg/kg/h FEN 2.5 μg/kg/h |
| 11 | 7 months | 7.1 | Female | 76 | 92 | 258 | Acute liver failure | MOF, liver transplantation |
MDZ 0.5 mg/kg/h MTM 30 mg/kg q.i.d. |
| 12 | 7 years | 23 | Male | 1 | 21 | 220 | Dilated cardiomyopathy | Excort® Berlin heart left VAD implantation | MTM 30 mg/kg q.i.d. |
AoCo Aortic coartation, ARDS acute respiratory distress syndrome, ARF acute respiratory failure, b.i.d. two times a day, DZP diazepam, ECMO extracorporeal life support, FEN fentanyl, ID patient identification number, IVC interventricular communication, KET ketamine, MDZ midazolam, MOF multiorgan failure, MOR morphine, MTD methadone, MTM metamizol, MV mechanical ventilation, PaO 2/FiO 2 partial pressure oxygen in the blood/fraction of inspired oxygen at the beginning of the study, PBW predicted body weight, PICU pediatric intensive care unit, q.i.d. four times a day, RMF remifentanil, RSV respiratory syncytial virus, t.i.d. three times a day, VAD ventricular assist device
The following neurally related variables were determined in order to study the neural drive to mechanical trigger: ventilator trigger delay (Td), the time from the onset of neural inspiration to that of inspiratory flow; Td normalized to nTi (Td/nTi); EAdi change during trigger delay (ΔEAdiTd); ΔEAdiTd normalized to maximum inspiratory EAdi difference (ΔEAdinTi = maxEAdi − minEAdi) (ΔEAdiTd/ΔEAdinTi); EAdi-time-product (EAdiTP), or area under the EAdi-time curve, during the trigger delay (EAdiTPTd) normalized to that during the neural inspiratory time (EAdiTPnTi) interval (EAdiTPTd/EAdiTPnTi). Figure 1 and Fig. E1 in the ESM show a graphic representation of these breath variables.
Fig. 1.
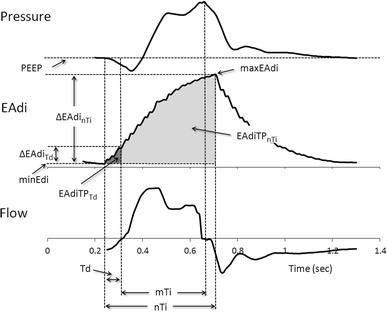
Definition of breath variables. Breath with trigger delay (Td): the onset of neural inspiration preceded that of inspiratory flow. EAdi Electrical activity of the diaphragm in microvolts, ΔEAdi nTi maximum inspiratory EAdi difference, ΔEAdi Td EAdi change during trigger delay, EAdiTP nTi EAdi–time-product during the neural inspiratory time, EAdiTP Td EAdi-time-product during the trigger delay, maxEAdi maximum EAdi per breath, minEAdi initial baseline inspiratory EAdi per breath, mTi mechanical inspiratory time, nTi neural inspiratory time, PEEP positive end expiratory pressure (in cmH2O)
The coefficient of variation (CV) for the following breath-related variables were determined: the maximum EAdi (CVmaxEAdi); peak inspiratory pressure (CVPmax); V t (CVVt); Ti (CVTi); V t/mTi (CVVt/mTi); RR (CVRR).
Statistical analysis
Data are expressed as median (Mdn) and interquartile range (IQR), unless otherwise stated. Patient data from the four study periods were compared using the Friedman test for multiple related samples, where p < 0.05 was considered to be significant. Post hoc analysis with the Wilcoxon signed-rank test was conducted, with a Bonferroni correction applied (new p = p/running tests = 0.05/4), resulting in a significant level set at p < 0.0125. As a measure of effect size, we used the Pearson’s correlation coefficient, r, computed from Wilcoxon Z values (r = Z/√N) [15]: small r < |0.3|, medium r = |0.3| to |0.5| and large r > |0.5|. Confidence limits for effect size were calculated according to Hedges and Olkin [16]. The CV was calculated as standard deviation (SD)/mean. Statistics were computed using SPSS ver. 15.0 for Windows (SPSS, Chicago, IL).
Results
Patient demographics, general ventilator settings and ventilatory variables are listed in Tables 1 and 2.
Table 2.
Ventilator settings and ventilatory variables
| Ventilator settings/ventilatory variablesa | Ventilation periodsb | p | |||
|---|---|---|---|---|---|
| 1-PSb | 2-PSopt | 3-NAVA | 4-PSopt | ||
| Ventilator settings | |||||
| PS (cmH2O) | 12.4 (1.4) | 12.6 (1.6) | 13.0 (1.6) | 0.51 | |
| PEEP (cmH2O) | 5.8 (0.5) | 5.8 (0.6) | 5.8 (0.6) | 6.0 (0.6) | 0.30 |
| Flow trigger | 7.5 (2.3) | 7.3 (1.7) | 7.3 (1.7) | 7.3 (1.7) | 0.80 |
| NAVA level | 1.41 (0.23) | ||||
| NAVA trigger (μV) | 0.46 (0.12) | ||||
| FiO2 | 0.50 (0.4) | 0.51 (0.4) | 0.51 (0.4) | 0.51 (0.4) | 0.39 |
| Ventilatory variables | |||||
| V t (kg PBW) | 5.5 (4.2–6.8) | 5.8 (4.6–7.0) | 5.6 (3.4–7.4) | 5.4 (4.2–6.5) | 0.13 |
| maxEAdi (μV) | 11.5 (7.1–19.7) | 14.3 (9.2–22.0) | 11.5 (6.7–17.3) | 13.6 (8.3–20.7) | 0.55 |
| P max (cmH2O) | 19.6 (14.6–20.5) | 19.2 (15.2–21.6) | 18.0 (15.5–24.5) | 19.6 (14.6–21.0) | 0.85 |
| RR (bpm) | 44.4 (25.4–53.5) | 40.4 (29.3–51.9) | 37.1 (26.3–55.7) | 40.3 (30.2–53.2) | 0.92 |
| SpaO2 (%) | 97.5 (95.5–99.5) | 97.0 (95–99.0) | 98.0 (97.0–99.5) | 95.5 (94.5–97.5) | 0.02 |
| etCO2 (mmHg) | 32.8 (30.5–41.2) | 33.8 (31.6–40.9) | 34.2 (30.0–39.5) | 33.8 (30.0–41.2) | 0.93 |
etCO 2 End tidal CO2 level, maxEAdi highest electrical activity of the diaphragm for the breath, NAVA neurally adjusted ventilatory assist, PEEP positive end expiratory pressure, P max maximum airway pressure, PS b baseline pressure support, PS opt optimized PS, RR respiratory rate, V t tidal volume, SpaO 2 oxygen saturation by pulsioxymetry
aVentilator setting data are given as the mean, with 1 standard deviation (SD) given in parenthesis; Ventilatory variables data are given as the median, with the 25–75 interquartile range (IQR) given in parenthesis
bFour sequential 10-min periods of data were recorded after 20 min of ventilatory stabilization (wash-out): 1-PSb, baseline pressure support (PS) with the ventilator settings determined by the attending physician; 2-PSopt, PS after optimization; 3-NAVA, NAVA level set so that maximum inspiratory pressure (P max) equaled P max in PS; 4-PSopt, same settings as in 2-PSopt. Post hoc analysis did not show significant differences between periods
Major asynchronies
The asynchrony index (AI) was significantly lower during NAVA as a result of a decrease in the number of auto-triggered and non-triggered breaths (Fig. 2). There were no differences in the number of double triggered events in PS after optimization and NAVA. We found that 69 % of the double triggered events were associated with an EAdi wave (in at least one of the double triggered breaths) where maxEAdi was ≥95th percentile of the maxEAdi for the patient and period. In addition, in 44 % of the double triggered events, minEAdi was ≥95th percentile of minEAdi for the patient and period.
Fig. 2.
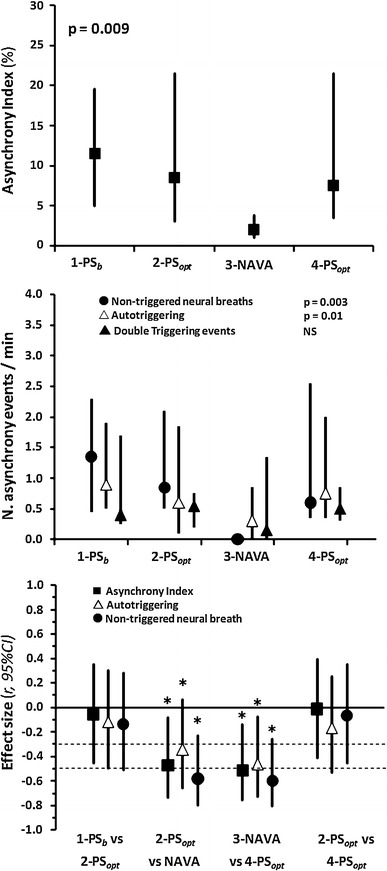
Major asynchrony events. 1-PS b Baseline pressure support (PS) with ventilator settings determined by the attending physician, 2-PS opt pressure support after optimization, 3-NAVA level set so that maximum airway pressure equaled maximum airway pressure during 1-PSb and 2-PSopt, 4-PS opt same pressure support settings as in 2-PSopt. N number of asynchrony events per minute. Values in the two upper figures are presented as the median with 25–75 % interquartile range (IQR). Values in the effect size figure are given as Pearson’s correlation coefficient, r, with the 95 % confident interval (CI). Horizontal dotted lines depict the limits for small, mild and large effect size, respectively.*p < 0.0125
Breath-to-breath variability
Neural breathing pattern variables, maxEAdi and nTi, showed a high variability during all four periods. The CVs of the mechanical breathing pattern variables (P max, mTi, V t, V t/mTi, RR) were significantly higher in NAVA (Fig. 3).
Fig. 3.
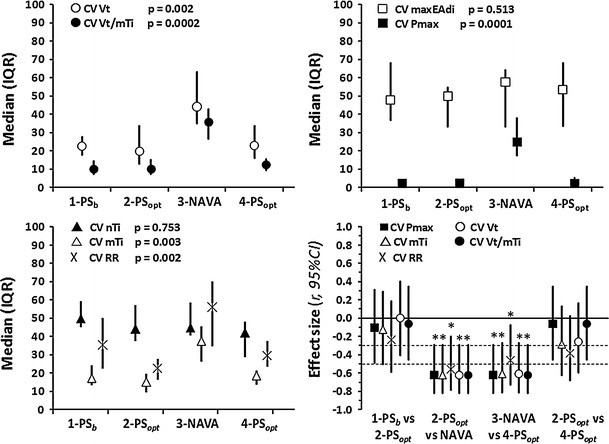
Breath-to-breath breathing variability. CV Coefficient of variation, P max maximum airway pressure per breath, b baseline, opt optimized, RR respiratory rate, V t tidal volume, V t/mTi mean inspiratory flow; for other abbreviations, see Figs. 1 and 2. Values are presented as the median with 25–75 % IQR, except for the effect size figure (bottom right corner) where values are given as Pearson’s correlation coefficient, r, with the 95 % CI; horizontal dotted lines depict the limits for small, mild and large effect size, respectively. *p < 0.0125
Neural drive to trigger
The onset of inspiratory flow preceded that of neural inspiration (neural delay) in 12.1 % (SD 2.9 %) of breaths, with no significant differences among periods. Of the breaths with neural delay, 31 % in 1-PSb, 30 % in 2-PSopt, 11 % in 3-NAVA and 36 % in 4-PSopt showed a delay longer than 150 ms.
There was a significant reduction in Td (ms), ΔEAdiTd (μV) and EAdiTPTd (μV s) (see Fig. E2 of the ESM) and in their nTi normalized variables Td/nTi, ΔEAdiTd/ΔEAdinTi and EAdiTPTd/EAdiTPnTi during NAVA versus the PSopt periods (Fig. 4). No significant changes were observed between 1-PSb and 2-PSopt. In NAVA mode, NAVA triggering accounted on average for 66 % (Mdn 81 %, IQR 33–89 %; p = 0.026, r = −0.40, 95 % CI 0.00−0.69) of the triggered breaths.
Fig. 4.
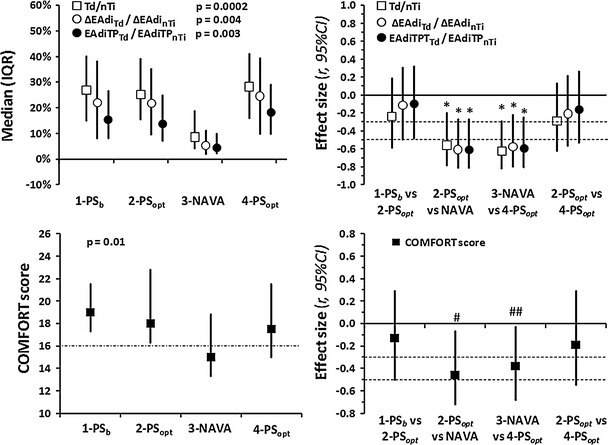
Neural drive to triggering and COMFORT score. ΔEAdi Td/ΔEAdi nTi EAdi change during the triggering delay as a percentage of maximum inspiratory EAdi difference, EAdiTP Td/EAdiTP nTi EAdi–time-product during the triggering delay as a percentage of the EAdi–time-product during neural inspiratory time, Td/nTi triggering delay as a percentage of neural inspiratory time (see Fig. 1). Values in two left figures are given as the median with 25–75 % IQR. COMFORT score dash–dotted line delimits light (above) from deep (below) sedation level. Values in the two right figures are given as Pearson’s correlation coefficient, r, with 95 % CI; horizontal dotted lines depict the limits for small, mild and large effect size, respectively.*p < 0.0125. # p = 0.0125. ## p = 0.050
Psychological distress and sedation assessment
Sedative dose was the same throughout the study for each patient. However, the COMFORT scale did significantly change over the four periods. There was a difference in COMFORT score between NAVA and optimized PS (Fig. 4). No significant changes were observed between 1-PSb and 2-PSopt.
Discussion
The most important findings of this study can be summarized as follows: In children, (1) NAVA decreases the frequency of major asynchronous events; (2) NAVA improves inspiratory trigger synchronization; (3) NAVA decreases the neural drive required to trigger the ventilator; (4) EAdi activity exhibits a high intra-patient breath-to-breath variability both in magnitude (maxEAdi) and time (nTi) during both NAVA and PS; (5) NAVA increases breath-to-breath variability of the following mechanical variables; P max, mTi, V T/Ti and RR; (6) NAVA results in a decrease in psychological distress as compared to optimized PS.
Major asynchronies
We did not find differences in the AI between PSb and PSopt, indicating that ventilator setting were already optimized in PSb or that the efficacy of the pneumatic adjuncts of current ventilators for improving patient–ventilator synchronization in PS (trigger, cycling-off and rise time) is limited in children.
The AI decreased notably during NAVA. Similar reductions in AI during NAVA versus PS have been reported in adult patients [17]. Adult studies indicate that this improvement in AI is clinically relevant since an AI >10 % is associated with a longer duration of mechanical ventilation and longer length of ICU stay [13, 18].
Auto-triggered and non-triggered breaths were primarily responsible for the differences between NAVA and PS. As in adults with acute respiratory failure [11], all neural breaths were triggered during NAVA. The decrease in auto-triggering during NAVA can be partially attributed to the difficulties in distinguishing artifacts (low EAdi activity that results in triggering) from auto-triggering, since in NAVA auto-triggering is not followed by an airway pressure wave. Although the results of our study related to double triggering need additional study, our results suggest that most double triggers were caused by sighs or periodic neural respiratory hyperactivity.
Breath-to-breath variability
The high CV for maxEAdi and nTi in both NAVA and PS confirm the existence of neural breath-to-breath variability of the breathing of children undergoing mechanical ventilation. However, only NAVA was able to improve the match between mechanical (V t, mTi, V t/mTi, RR and P max) and neural variations in breathing (maxEAdi and nTi) (Fig. 3).
In their study on acutely ill mechanically ventilated adult patients, Schmidt et al. [9] recently found that switching from PS to NAVA followed by increasing the NAVA level was associated with an increased variability in breathing and a greater complexity of the ventilatory flow signal. In addition, they also found that the breath-to-breath variability and complexity of EAdi was high in PS and not influenced by NAVA.
Breath-to-breath variations in the pattern of breathing are an inherent feature of respiration [19]. In awake healthy humans, breath-to-breath variability decreases in response to mechanical loading [20]. Brack et al. [3] found that in patients with restrictive lung disease, the CV for V t was more than fivefold smaller than that in healthy subjects, probably as a strategy to avoid dyspnea. We found that the CV for V t and V t/mTi were on average two- to threefold higher in NAVA than in PS. In addition, during PS, the ventilator was unable to generate mechanical breaths (P max, mTi, V t, V t/mTi and RR) that matched neural breath-to-breath variations (maxEAdi and nTi) (Fig. 3), suggesting that loss of breath-to-breath variability is a form of asynchrony that may contribute to dyspnea [21].
Neural drive to trigger the ventilator
Most patients exhibited a low rate of neural delay of <12 %; nevertheless, in patient 12 during PS and NAVA, the onset of neural inspiration was clearly delayed in 36 % of the breaths (median 130 ms, IQR 70–240 ms). As shown by Corda et al. [22], external intercostal muscles are normally stimulated before the diaphragm as an initial stabilization of the chest wall to make diaphragmatic contraction more efficient. We hypothesized that neural delay was due to intercostal muscle activity which generated inspiratory flow before diaphragmatic activation and that this was exaggerated in patient 12 (with recent median sternotomy) in order to stabilize his chest wall [23].
The neural beginning of inspiration preceded mechanical inspiration in the remaining 90 % of breaths regardless of mode. In adults, the Td was half both in PS and in NAVA, possibly because the RR was also half [17]. Indeed, the Td percentage of the inspiratory time in adults is similar to that in children. In 50 % of the breaths in PS, Td was >25 % of nTi, which is threefold more than in NAVA (Fig. 4). These differences can be explained by the fact that the NAVA trigger accounts for most of the triggered breaths during NAVA, as has been previously shown [6, 24].
It has been shown that the EAdi signal strength is related to diaphragm activation and represents total diaphragm activity [25, 26]. We measured the neural drive to trigger using two variables, ∆EAdiTd and EAdiTPTd. According to the concept of neuromechanical coupling of the diaphragm as the transformation of electrical activity (EAdi) into pressure generation [25, 26], ∆EAdiTd is a direct measure of neural drive, and EAdiTPTd would represent an index of neural work to trigger. We normalized these neural drive indices to the respective neural inspiratory drive variables, ∆EAdinTi and EAdiTPnTi, EAdiTPTd and EAdiTPnTi, of a breath. These two indices take into account both the magnitude and the duration of neural stimulus and thereby provide a good picture of the intensity of the neural drive. We were unable to compare these with data from the literature because as far as we know this is the first time they have been measured in children.
The neural drive indices (∆EAdiTd and EAdiTPTd) were significantly lower in NAVA, indicating that a lower diaphragm activation was necessary for triggering in NAVA than in PS (Fig. 4). The neural trigger in NAVA can explain the results. Indeed, median ∆EAdiTd was equal to the mean NAVA trigger set.
The amount of assist delivered during NAVA is set by a proportionality factor, the “NAVA level”, which defines the magnitude of pressure delivered for a given EAdi [27]. Since EAdi change to trigger occurs before pressure is delivered, the improvement in neural drive to trigger in NAVA is independent of the NAVA level. Therefore, NAVA decreases respiratory muscle load in two ways—by improving triggering and through the set NAVA level.
Our data can at least indirectly be compared to those of Clement et al. [28] who evaluated trigger delay and ventilator response during NAVA compared to PS in a mechanical lung model. These authors observed a decrease in trigger delay, ventilator response time and the pressure–time product during ventilator activation. These finding are consistent with the improvement in neural drive we observed in our patients.
Psychological distress and sedation assessment
The COMFORT score defines two levels of sedation: deep sedation, indicated by a score between 8 and 16, and light sedation, evidenced with a score ranging from 17 to 26 [29].
In our study children transitioned from a light to deeper sedation when moved from PSopt to NAVA and then reverted to light sedation when moved again to PSopt (Fig. 4). Because of the small number of patients studied, we cannot fully disregard the possibility that the differences in sedation level between NAVA and PSopt periods were random. Nevertheless, we found a difference in COMFORT score between NAVA and optimized PS that may have important implications for sedation in mechanically ventilated children.
The observed decrease in psychological distress during NAVA did not seem to be associated with the level of ventilatory support since it was similar in each patient for the length of the study (Table 2) and since each child received the same sedative and dose during the study.
No studies have been published that have directly examined the sedation needs of children being mechanically ventilated using NAVA mode compared to traditional modes. However, there is evidence of an association between sedation and asynchrony. In our study, children tended to go into a deeper sedation state while in NAVA, the period with the lowest AI and ineffective triggering, suggesting a lower level of psychological distress as a result of the improvement in synchrony. In addition, the neural drive associated with the mechanical load to trigger the ventilator was threefold lower in NAVA than in PS, and the variability of breathing increased in NAVA, matching neural breath-to-breath variations. It seems reasonable to believe that this improvement in synchrony, neural drive and respiratory variability during NAVA would have decreased the feeling of dyspnea, therefore impacting sedation level. The binomial effect size display (BESD) [29] interpretation of the Pearson coefficient may better illustrate the relevance of our findings: according to our results 76 % (95 % CI 53–86 %) of the children in NAVA versus 27 % (14–46 %) of those in optimized PS would need a lower dose of sedatives to reach an optimum light level of sedation, as evidenced by the COMFORT score, during mechanical ventilation.
Reducing the dose of sedatives in adults and children has been shown to decrease the duration of mechanical ventilation and the length of stay in ICUs [30, 31]. Additionally, as stated previously, a reduction of the asynchrony index can reduce the duration of mechanical ventilation and length of ICU stay [13, 18]. Accordingly, our results indicate that through the decrease of ventilatory asynchrony and neural drive to trigger and the improvement of breath-to-breath mechanical variability, NAVA in children with asynchronies decreases psychological distress and may result in a notable reduction in the dose of sedative drugs, thereby potentially decreasing the duration of mechanical ventilation and the length of PICU stay.
Our study has a number of limitations. The main limitation is that our study is not randomized. The small number of patients and the case mix may not represent the global pediatric population. There is a high heterogeneity in terms of age and reason for mechanical ventilation. Patients were studied at different moments of their disease course (some in a more acute phase, others during recovery), which may have affected the “psychological distress” (and so the COMFORT score) as well as mechanical results. Although sedation was maintained constant during the study for each patient, there were intra-patient variables beyond our control (sleep, hungry, secretions, mother’s presence) that could have affected the homogeneity among phases. We did not measure esophageal pressure, so we cannot be sure about the minimal patient EAdi activity required to recruit diaphragmatic muscle units and distinguish true neural breaths from artifacts.
Conclusion
This study shows that NAVA as compared to optimized PS results in improved synchrony, reduced ventilatory drive, increased breath-to-breath mechanical variability and increased patient comfort. Our results indicate that, during mechanical ventilation, the use of NAVA in children with asynchronies would permit a reduction in sedation and therefore may decrease the duration of mechanical ventilation and the length of PICU stay.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
This study was performed at the Pediatric ICU of the Hospital Universitario Materno-Infantil La Paz. Madrid. Spain. The study was supported, in part, by Maquet Critical Care AB (Solna, Sweden) and by the Hospital Universitario La Paz (Madrid, Spain).
Footnotes
ClinicalTrials.gov Identifier: NCT01159106.
References
- 1.Manning HL, Schwartzstein RM. Pathophysiology of dyspnea. N Engl J Med. 1995;333:1547–1553. doi: 10.1056/NEJM199512073332307. [DOI] [PubMed] [Google Scholar]
- 2.Brack T, Jubran A, Tobin MJ. Effect of elastic loading on variational activity of breathing. Am J Respir Crit Care Med. 1997;155:1341–1348. doi: 10.1164/ajrccm.155.4.9105077. [DOI] [PubMed] [Google Scholar]
- 3.Brack T, Jubran A, Tobin MJ. Dyspnea and decreased variability of breathing in patients with restrictive lung disease. Am J Respir Crit Care Med. 2002;165:1260–1264. doi: 10.1164/rccm.2201018. [DOI] [PubMed] [Google Scholar]
- 4.Beck J, Tucci M, Emeriaud G, Lacroix J, Sinderby C. Prolonged neural expiratory time induced by mechanical ventilation in infants. Pediatr Res. 2004;55:747–754. doi: 10.1203/01.PDR.0000119368.21770.33. [DOI] [PubMed] [Google Scholar]
- 5.Beck J, Reilly M, Grasselli G, Mirabella L, Slutsky AS, Dunn MS, Sinderby C. Patient-ventilator interaction during neurally adjusted ventilatory assist in low birth weight infants. Pediatr Res. 2009;65:663–668. doi: 10.1203/PDR.0b013e31819e72ab. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Breatnach C, Conlon NP, Stack M, Healy M, O’Hare BP. A prospective crossover comparison of neurally adjusted ventilatory assist and pressure-support ventilation in a pediatric and neonatal intensive care unit population. Pediatr Crit Care Med. 2010;11:7–11. doi: 10.1097/PCC.0b013e3181b0630f. [DOI] [PubMed] [Google Scholar]
- 7.Spahija J, de Marchie M, Albert M, Bellemare P, Delisle S, Beck J, Sinderby C. Patient-ventilator interaction during pressure support ventilation and neurally adjusted ventilatory assist. Crit Care Med. 2010;38:518–526. doi: 10.1097/CCM.0b013e3181cb0d7b. [DOI] [PubMed] [Google Scholar]
- 8.Kacmarek RM. Proportional assist ventilation and neurally adjusted ventilatory assist. Respir Care. 2011;56:140–148. doi: 10.4187/respcare.01021. [DOI] [PubMed] [Google Scholar]
- 9.Schmidt M, Demoule A, Cracco C, Gharbi A, Fiamma MN, Straus C, Duguet A, Gottfried SB, Similowski T. Neurally adjusted ventilatory assist increases respiratory variability and complexity in acute respiratory failure. Anesthesiology. 2010;112:670–681. doi: 10.1097/ALN.0b013e3181cea375. [DOI] [PubMed] [Google Scholar]
- 10.Coisel Y, Chanques G, Jung B, Constantin JM, Capdevila X, Matecki S, Grasso S, Jaber S. Neurally adjusted ventilatory assist in critically ill postoperative patients: a crossover randomized study. Anesthesiology. 2010;113:925–935. doi: 10.1097/ALN.0b013e3181ee2ef1. [DOI] [PubMed] [Google Scholar]
- 11.Colombo D, Cammarota G, Bergamaschi V, De LM, Corte FD, Navalesi P. Physiologic response to varying levels of pressure support and neurally adjusted ventilatory assist in patients with acute respiratory failure. Intensive Care Med. 2008;34:2010–2018. doi: 10.1007/s00134-008-1208-3. [DOI] [PubMed] [Google Scholar]
- 12.Stufken J (1996) Optimal crossover designs, In: Ghosh S, Rao CR (eds) Handbook of statistics 13. Design and analysis of experiments, 13 edn. Elsevier/North-Holland, Linn, pp 63–69
- 13.Thille AW, Rodriguez P, Cabello B, Lellouche F, Brochard L. Patient-ventilator asynchrony during assisted mechanical ventilation. Intensive Care Med. 2006;32:1515–1522. doi: 10.1007/s00134-006-0301-8. [DOI] [PubMed] [Google Scholar]
- 14.Ambuel B, Hamlett KW, Marx CM, Blumer JL. Assessing distress in pediatric intensive care environments: the COMFORT scale. J Pediatr Psychol. 1992;17:95–109. doi: 10.1093/jpepsy/17.1.95. [DOI] [PubMed] [Google Scholar]
- 15.Field A. Discovering statistic using SPSS. 2. London: SAGE Publ; 2005. pp. 563–567. [Google Scholar]
- 16.Hedges L, Olkin I. Statistical methods for meta-analysis. New York: Academic Press; 1985. pp. 85–91. [Google Scholar]
- 17.Piquilloud L, Vignaux L, Bialais E, Roeseler J, Sottiaux T, Laterre PF, Jolliet P, Tassaux D. Neurally adjusted ventilatory assist improves patient ventilator interaction. Intensive Care Med. 2011;37:263–271. doi: 10.1007/s00134-010-2052-9. [DOI] [PubMed] [Google Scholar]
- 18.Wit M, Miller KB, Green DA, Ostman HE, Gennings C, Epstein SK. Ineffective triggering predicts increased duration of mechanical ventilation. Crit Care Med. 2009;37:2740–2745. doi: 10.1097/ccm.0b013e3181a98a05. [DOI] [PubMed] [Google Scholar]
- 19.Bruce EN. Temporal variations in the pattern of breathing. J Appl Physiol. 1996;80:1079–1087. doi: 10.1063/1.362844. [DOI] [PubMed] [Google Scholar]
- 20.Brack T, Jubran A, Tobin M. Effect of resistive loading on variational activity of breathing. Am J Respir Crit Care Med. 1998;157:1756–1763. doi: 10.1164/ajrccm.157.6.9704114. [DOI] [PubMed] [Google Scholar]
- 21.Wysocki M, Richard JC, Meshaka P. Noninvasive proportional assist ventilation compared with noninvasive pressure support ventilation in hypercapnic acute respiratory failure. Crit Care Med. 2002;30:323–329. doi: 10.1097/00003246-200202000-00010. [DOI] [PubMed] [Google Scholar]
- 22.Corda M, Eklund G, Euler Von. External intercostal and phrenic alpha-motor responses to changes in respiratory load. Acta Physiol Scand. 1965;63:391–400. doi: 10.1111/j.1748-1716.1965.tb04079.x. [DOI] [PubMed] [Google Scholar]
- 23.Whitelaw WA, Feroah T. Patterns of intercostal muscle activity in humans. J Appl Physiol. 1989;67:2087–2094. doi: 10.1152/jappl.1989.67.5.2087. [DOI] [PubMed] [Google Scholar]
- 24.Bengtsson JA, Edberg KE. Neurally adjusted ventilatory assist in children: an observational study. Pediatr Crit Care Med. 2010;11:253–257. doi: 10.1097/PCC.0b013e3181b0655e. [DOI] [PubMed] [Google Scholar]
- 25.Sinderby C, Beck J, Spahija J, Weinberg J, Grassino A. Voluntary activation of the human diaphragm in health and disease. J Appl Physiol. 1998;85:2146–2158. doi: 10.1152/jappl.1998.85.6.2146. [DOI] [PubMed] [Google Scholar]
- 26.Beck J, Gottfried SB, Navalesi P, Skrobik Y, Comtois N, Rossini M, Sinderby C. Electrical activity of the diaphragm during pressure support ventilation in acute respiratory failure. Am J Respir Crit Care Med. 2001;164:419–424. doi: 10.1164/ajrccm.164.3.2009018. [DOI] [PubMed] [Google Scholar]
- 27.Sinderby C, Beck J, Spahija J, de Marchie M, Lacroix J, Navalesi P, Slutsky AS. Inspiratory muscle unloading by neurally adjusted ventilatory assist during maximal inspiratory efforts in healthy subjects. Chest. 2007;131:711–717. doi: 10.1378/chest.06-1909. [DOI] [PubMed] [Google Scholar]
- 28.Clement KC, Thurman TL, Holt SJ, Heulitt MJ. Neurally triggered breaths reduce trigger delay and improve ventilator response times in ventilated infants with bronchiolitis. Intensive Care Med. 2011;37:1826–1832. doi: 10.1007/s00134-011-2352-8. [DOI] [PubMed] [Google Scholar]
- 29.Randolf JJ, Edmondson RS. Using the binomial effect size display (BESD) to present the magnitude of effect sizes to the evaluation audience. Prac Assess Res Eval. 2005;10:1–7. [Google Scholar]
- 30.Jin HS, Yum MS, Kim SL, Shin HY, Lee EH, Ha EJ, Hong SJ, Park SJ. The efficacy of the COMFORT scale in assessing optimal sedation in critically ill children requiring mechanical ventilation. J Korean Med Sci. 2007;22:693–697. doi: 10.3346/jkms.2007.22.4.693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kress JP, Pohlman AS, O’Connor MF, Hall JB. Daily interruption of sedative infusions in critically ill patients undergoing mechanical ventilation. N Engl J Med. 2000;342:1471–1477. doi: 10.1056/NEJM200005183422002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


