Analyses of contact-tracing data on the spread of infectious disease, combined with mathematical models, show that control measures require better knowledge of variability in individual infectiousness.
Coughs and sneezes...
From Typhoid Mary to SARS, it has long been known that some people spread disease more than others. But for diseases transmitted via casual contact, contagiousness arises from a plethora of social and physiological factors, so epidemiologists have tended to rely on population averages to assess a disease's potential to spread. A new analysis of outbreak data shows that individual differences in infectiousness exert powerful influences on the epidemiology of ten deadly diseases. SARS and measles (and perhaps avian influenza) show strong tendencies towards ‘superspreading events’ that can ignite explosive epidemics — but this same volatility makes outbreaks more likely to fizzle out. Smallpox and pneumonic plague, two potential bioterrorism agents, show steadier growth but still differ markedly from the traditional average-based view. These findings are relevant to how emerging diseases are detected and controlled.
The SARS epidemic was notable for the existence of ‘superspreaders’ who infected dozens of people, whereas other infectious individuals infected few or none. Were SARS superspreaders anomalies, or are superspreaders characteristic of most infectious diseases? What effects does heterogeneity in infectiousness have on disease emergence and control? On page 355 of this issue, Lloyd-Smith et al.1 provide insight into such questions, and more.

K. HESSE/GETTY IMAGES
The first question any ecologist asks about an invasive species is: what is the invader's intrinsic capacity for population increase? To answer this, the species' basic reproductive number, R0, is measured by the average number of offspring per capita that survive to reproductive age. For a directly transmitted infectious disease, be it polio, smallpox, SARS, HIV/AIDS or some newly emerging pathogen, R0 is the average number of infections produced by an infected individual in a susceptible population2. If R0 is less than one, a self-sustaining epidemic is not possible (at least without further pathogen evolution). If R0 exceeds one, then although early stochastic fluctuations may extinguish the invader, an epidemic is possible. If R0 is large, an epidemic is virtually certain.
Initial work in this area largely treated individuals in populations as having an equal chance of transmitting disease — that is, as being homogeneous — and ignored stochastic fluctuations in transmission capability. However, studies of gonorrhoea3, and of HIV/AIDS4, could not explain epidemiological patterns without acknowledging heterogeneities in patterns of sexual-partner acquisition, including the disproportionate influence of superspreaders. Similarly, knowledge of heterogeneous parasite burdens is fundamental to accurate modelling of helminthic diseases5,6. Explanations of epidemiological patterns of malaria also depend on understanding heterogeneous biting by the mosquito vector7.
These observations led to the proposal of the 20/80 rule2,8, which suggests that roughly 20% of the most infectious individuals are responsible for 80% of the transmission (Fig. 1, overleaf). This rule has been applied mainly to helminthic and sexually transmitted diseases7; for other directly transmitted diseases, such as smallpox or influenza, heterogeneity in infectiousness has been neglected. The superspreading that seemed to fuel the 2003 SARS epidemic was largely treated as anomalous in most models, but it highlighted the need for a reassessment of heterogeneous infectiousness9.
Figure 1. Heterogeneity in infectiousness for a range of diseases.
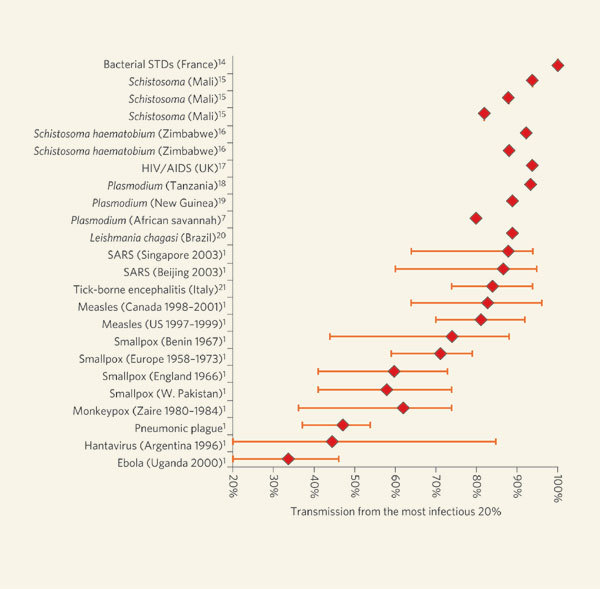
The measure used is the 20/XX index, which quantifies the proportion of the transmission (XX%) that results from the most infectious 20% of the population. Confidence intervals are included where available. The interval for tick-borne encephalitis (Italy)21 indicates possible values depending on assumptions made about host susceptibility. STDs, sexually transmitted diseases; HIV, human immunodeficiency virus; AIDS, acquired immunodeficiency syndrome; SARS, severe acute respiratory syndrome.
Lloyd-Smith et al.1 address this point by posing infectiousness as a continuous variable, and formulate an unambiguous and universally applicable definition of superspreaders as those who transmit more infection than is predicted by a homogeneous ‘null model’. The authors analyse data from eight human infections, including SARS, measles, smallpox, monkeypox and pneumonic plague, to show that superspreading occurs across the board, although to a greater or lesser extent depending on the disease. Heterogeneity is greatest for SARS and least for Ebola haemorrhagic fever.
Analysis of the epidemiological dynamics shows that, for a given R0, both the probability that an epidemic will take off, and the subsequent course of the epidemic, are affected by such heterogeneity. These results may be appreciated intuitively. For a given value of R0, high heterogeneity in infectiousness implies that relatively few individuals are responsible for most of the transmission — or conversely, that many individuals do not transmit at all. In turn, such small numbers tend to generate pronounced stochastic fluctuations in the initial stages of the epidemic. Consequently, a heterogeneously infectious emerging disease will be less likely to generate an epidemic, but if sustained, the resulting epidemic is more likely to be explosive. Thus, it is dangerous to underestimate a disease on the basis of frequent ‘failed’ attempts, as exemplified by bird flu.
The authors highlight the practical implications of their work. Control efforts should aim to identify the highly infectious superspreaders, and target vaccination or other interventions at them. In this way, the outbreak may be halted sooner, and with fewer people treated, than if efforts are directed at random individuals. Furthermore, Lloyd-Smith et al. distinguish between individual-specific and population-wide control measures (for example isolating individual patients as opposed to advising an entire population to reduce the behaviours associated with transmission). They show that individual-specific strategies are more likely to exterminate an emerging disease than population-wide interventions, because the former increase heterogeneity in infectiousness.
Comparisons can be drawn between these new findings1 and work on heterogeneities in contact patterns among individuals (for example in SARS10, or sexually transmitted diseases such as HIV/AIDS4) and among groups (foot-and-mouth disease, for instance11). Earlier research on contact patterns led to a generalized theorem (ref. 2; equation 12.23), which was based on the following reasoning. We can estimate the proportion, p*, to be vaccinated or otherwise treated in order to eradicate infection in a homogeneous population (p*=1−1/R0). However, if we take advantage of the heterogeneity, and target the more infectious or more sexually active individuals (depending on the disease), then we can achieve our aim by treating a smaller proportion than is estimated by p*, as echoed in the results of Lloyd-Smith and colleagues. This general result, first discovered in the epidemiological literature in the mid-1980s, also applies to the structure of information-technology networks in relation to targeted versus random ‘viral’ attacks12.
There are also differences between the work of Lloyd-Smith et al. and work on heterogeneities in contact patterns. Given that contact rates govern the likelihood both of becoming infected and of passing on infection, models based on heterogeneous contact rates have assumed perfect correlation between infectiousness and susceptibility. Consider HIV/AIDS, where in the simplest case R0=βDc, with β being the transmission probability (a measure of the infectiousness of an infected individual), D the duration of the infectiousness, and c the average rate at which new sexual partners are acquired. Heterogeneities among individuals with respect to β or in D do not directly affect R0 as such: the quantities β and D enter the dynamic equations linearly, and the appropriate values for estimating R0 are just the simple averages.
By contrast, the distribution of partner-acquisition rates enters nonlinearly; those with more partners are more likely to acquire infection by virtue of their higher activity, and they are also more likely to transmit infection. Consequently, the epidemiologically appropriate ‘average partner-acquisition rate’, c, is not the mean of the distribution, but rather the mean-square divided by the mean. An incorrect result is obtained for R0 when the average of the partner-acquisition or contact distribution is used. (This observation, incidentally, helps explain how large geographical variation in HIV incidence can arise from differences in the tails of such distributions.) In contrast, Lloyd-Smith et al. evaluate heterogeneity in overall R0 (integrating all contributing factors), and assume that infectiousness is not correlated with susceptibility. As they note, the reality probably lies somewhere in between, with some intermediate level of correlation between infectiousness and susceptibility.
Although there is a considerable advantage in targeted control measures if highly infectious individuals can be identified before they have transmitted infection1,8, this is easier said than done. In the case of sexually transmitted diseases and contact patterns, however, Cohen et al.13 formulated a seemingly paradoxical method for achieving this aim, without directly identifying the active individuals. This procedure is based on the realization that one's contacts will on average be more highly connected within a contact network than oneself, simply by virtue of being a contact. Thus, highly connected individuals can be identified for intervention by first picking individuals at random, and then selecting randomly among their acquaintances. In this way, highly connected individuals are identified with minimal effort. Moreover, this procedure can be carried out either before or after an outbreak.
Among the next steps to be taken are further parametrization of heterogeneity for different diseases and for the same disease in different settings, and determination of the characteristics of emerging diseases that are likely to exhibit the most pronounced heterogeneity. Heterogeneous infectiousness, and its extreme manifestation of superspreading, are likely to be general properties of disease transmission in populations. The ambitious aim of controlling disease emergence will require a better understanding of those properties, which is most likely to be achieved through the combination of data analysis and epidemiological theory exemplified by Lloyd-Smith and colleagues' study.
Contributor Information
Alison P. Galvani, Email: alison.galvani@yale.edu
Robert M. May, Email: robert.may@zoo.ox.ac.uk
References
- 1.Lloyd-Smith JO, Schreiber SJ, Kopp PE, Getz WM. Nature. 2005;438:355–359. doi: 10.1038/nature04153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Anderson RM, May RM. Infectious Diseases of Humans: Dynamics and Control. 1991. [Google Scholar]
- 3.Hethcote HW, Yorke JA. Lect. Notes Biomath. 1984;56:1–105. doi: 10.1007/978-3-662-07544-9_1. [DOI] [Google Scholar]
- 4.May RM, Anderson RM. Nature. 1987;326:137–142. doi: 10.1038/326137a0. [DOI] [PubMed] [Google Scholar]
- 5.Grenfell BT, Wilson K, Isham VS, Boyd HEG, Dietz K. Parasitology. 1995;111:S135–S151. doi: 10.1017/S0031182000075867. [DOI] [PubMed] [Google Scholar]
- 6.Galvani AP. J. Parasitol. 2003;89:232–241. doi: 10.1645/0022-3395(2003)089[0232:IAHAAO]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 7.Smith, D. L., Dushoff, J., Snow, R. W. & Hay, S. I. Nature (in the press); doi:10.1038/nature04024. [DOI] [PMC free article] [PubMed]
- 8.Woolhouse MEJ. Proc. Natl Acad. Sci. USA. 1997;94:338–342. doi: 10.1073/pnas.94.1.338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bauch CT, Lloyd-Smith JO, Coffee M, Galvani AP. Epidemiology. 2005;16:791–801. doi: 10.1097/01.ede.0000181633.80269.4c. [DOI] [PubMed] [Google Scholar]
- 10.McLean AR, May RM, Pattison J, Weiss RA. SARS: A Case Study in Emerging Infections. 2005. [Google Scholar]
- 11.Haydon DT, et al. Proc. Biol. Sci. 2003;270:121–127. doi: 10.1098/rspb.2002.2191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Albert R, Jeong H, Barabasi AL. Nature. 2000;406:378–382. doi: 10.1038/35019019. [DOI] [PubMed] [Google Scholar]
- 13.Cohen R, Havlin S, Ben-Avraham D. Phys. Rev. Lett. 2003;91:2479011–2479014. doi: 10.1103/PhysRevLett.91.247901. [DOI] [PubMed] [Google Scholar]
- 14.Spira A, Bajos N. Sexual Behavior and AIDS. 1994. [Google Scholar]
- 15.Etard JF, Audibert M, Dabo A. Am. J. Trop. Med. Hyg. 1995;52:549–558. doi: 10.4269/ajtmh.1995.52.549. [DOI] [PubMed] [Google Scholar]
- 16.Chandiwana SK, Woolhouse ME. Parasitology. 1991;103:363–370. doi: 10.1017/S0031182000059874. [DOI] [PubMed] [Google Scholar]
- 17.Johnson AM, Wadsworth J, Wellings K, Field J. Sexual Attitudes and Lifestyles. 1994. [PubMed] [Google Scholar]
- 18.Smith T, Charlwood JD, Takken W, Tanner M, Spiegelhalter DJ. Acta Trop. 1995;59:1–18. doi: 10.1016/0001-706X(94)00082-C. [DOI] [PubMed] [Google Scholar]
- 19.Hii JL, et al. J. Med. Entomol. 1997;34:193–205. doi: 10.1093/jmedent/34.2.193. [DOI] [PubMed] [Google Scholar]
- 20.Quinnell RJ, Dye C. Med. Vet. Entomol. 1994;8:219–224. doi: 10.1111/j.1365-2915.1994.tb00502.x. [DOI] [PubMed] [Google Scholar]
- 21.Perkins SE, Cattadori IM, Tagliapietra V, Rizzoli AP, Hudson PJ. Int. J. Parasitol. 2003;33:909–917. doi: 10.1016/S0020-7519(03)00128-0. [DOI] [PubMed] [Google Scholar]


