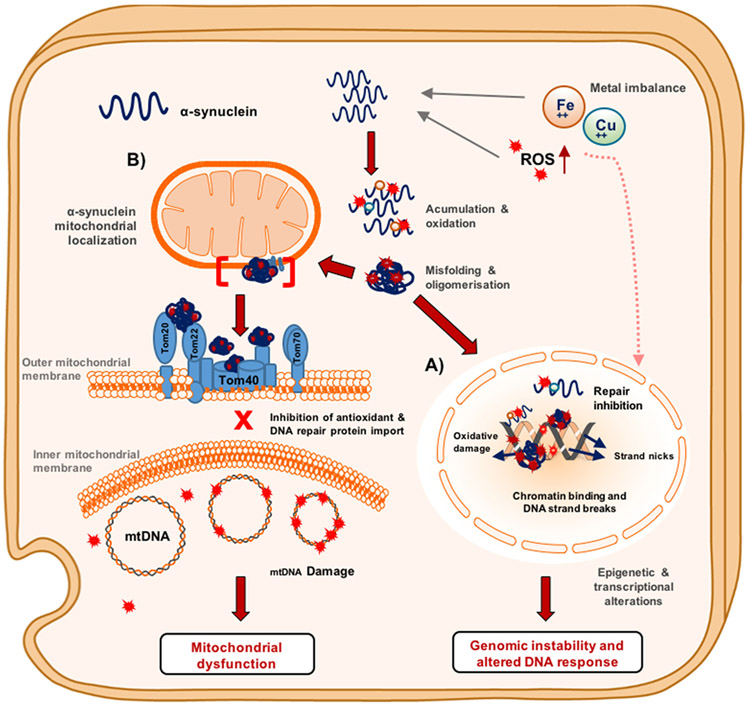
Figure 6. Multi-faceted toxicity of α-synuclein in dopaminergic neurons.
Schematic representation of the reactive oxygen ROS accumulation and metal imbalance that promote α-synuclein DNA nicking activity and mitochondrial dysfunction in dopaminergic neurons. (A) Nucleus. Metal ions can contribute to α-synuclein oxidation by binding to the oxidation prone residues, methionine, serine, and tyrosine, which facilitates DNA nicking activity when α-synuclein is bound to chromatin. Chromatin-bound oxidized α-synuclein can lead to DNA repair inhibition and DNA strand breaks that result in genomic instability. Pro-oxidant metals and ROS can induce the inactivation of DNA repair proteins. (B) Mitochondria. Oxidized or oligomeric α-synuclein can induce mitochondrial dysfunction by binding to TOMM complex proteins, which inhibits the translocation of important antioxidant proteins and potential DNA repair machinery. In the absence of these repair machineries, ROS can induce mtDNA strands breaks and somatic point mutations. These defects can result in deficient oxidative phosphorylation complex activity that leads to mitochondrial dyshomeostasis.