Abstract
The ideal laboratory test to detect Neisseria gonorrhoeae (Ng) should be sensitive, specific, easy to use, rapid, and affordable and should provide information about susceptibility to antimicrobial drugs. Currently, such a test is not available and presumably will not be in the near future. Thus, diagnosis of gonococcal infections presently includes application of different techniques to address these requirements. Microscopy may produce rapid results but lacks sensitivity in many cases (except symptomatic urogenital infections in males). Highest sensitivity to detect Ng was shown for nucleic acid amplification technologies (NAATs), which, however, are less specific than culture. In addition, comprehensive analysis of antibiotic resistance is accomplished only by in vitro antimicrobial susceptibility testing of cultured isolates. As a light at the end of the tunnel, new developments of molecular techniques and microfluidic systems represent promising opportunities to design point-of-care tests for rapid detection of Ng with high sensitivity and specificity, and there is reason to hope that such tests may also provide antimicrobial resistance data in the future.
Keywords: gonorrhea, diagnostic, microscopy, culture, antimicrobial resistance, NAAT, point-of-care test, microfluidic
1. Introduction
Neisseria gonorrhoeae (Ng) infections belong to the most frequent sexually transmitted infections with worldwide around 87 million new infections per year according to WHO estimations [1]. Of these, about 4 million occur in Europe, North America, Australia, and New Zeeland. The vast majority (>80 million) of gonococcal infections are in low- and middle-income countries of Asia, Africa, Latin America, and the Caribbean [1], but increasing incidence is reported in Europe and the USA [2,3]. In Europe, men having sex with men (MSM) accounted for almost half of the reported cases [3]. Transmission of the bacteria is by direct mucosal contact and may lead to infections at the urethra, endocervix, rectum, pharynx, or conjunctiva. While 90% of male urethral infections present with discharge or dysuria, less than 50% of female urethral and cervical infections are symptomatic, and most rectal infections and almost all pharyngeal infections are asymptomatic [4,5,6]. By transluminal dissemination, starting from the urethral or endocervical mucosa, Ng may cause ascending infections resulting in epididymo-orchitis, salpingitis, and pelvic inflammatory disease (PID) [7,8,9]. In rare cases, the bacteria may spread systemically resulting in severe complications like fever/septicemia, arthritis, tenosynovitis, endocarditis, or vasculitis [10]. In addition, gonococcal infection in pregnancy is associated with adverse pregnancy outcomes, like low birth weight infants, small for gestational age infants, and transmission to newborns that may result in conjunctivitis (ophthalmia neonatorum) and oropharyngeal infections [11,12].
Due to the variety of symptoms that are largely not specific for gonorrhea, timely and accurate laboratory testing of symptomatic patients is required including resistance testing of Ng-positive cases for targeted antimicrobial treatment. In addition, screening of key populations to identify and treat asymptomatic infections is no less important to reduce transmission of infection and to control spreading of antibiotic resistance. Hence, the following conditions indicate laboratory testing of gonococcal infection: diagnostic evaluation of clinical symptoms, treatment monitoring, sexual partner(s) diagnosed with Ng, other diagnosed STI, new sexual partners or frequently changed sexual partners, and sexual abuse. In the absence of laboratory diagnostics in resource-poor settings, Ng infection is usually identified by clinical manifestations combined with medical history and a typical incubation period (2–8 days). However, even classical clinical symptoms like male purulent discharge and urethritis or purulent vaginal discharge or proctitis are not sufficient evidence of gonorrhea, as various other pathogens can cause very similar or identical images. The syndromic approach may possibly suffice for urethral discharge in men but has poor sensitivity and specificity to detect infections in women and non-urethral infection in men, potentially resulting in inadequate treatment with the risk of inducing resistance. It is therefore essential to make laboratory testing available to resource-limited settings.
Laboratory diagnosis of gonococcal infection is established by direct detection of the pathogen in urogenital, anorectal, pharyngeal, or conjunctival swab specimens or first-catch urine. Presently, several different techniques are available for Ng detection, of which culture and nucleic acid amplification technologies (NAATs) are best suited [13]. Microscopy of stained urogenital specimens can also be used in certain cases. DNA probe assays, antigen tests, and serology to detect antibodies against Ng are not recommended for laboratory testing due to insufficient sensitivity and specificity [13].
During the last decades, diagnostic procedures have been improved continuously, resulting in a better management of individual patients. There are, however, some public health issues to be considered in this context.
Improvements of Ng testing resulted in increased detection rates that may have influenced epidemiologic data (i.e., higher detection rates do not necessarily indicate an increase in transmitted infections but may just reflect more sensitive and more frequent testing). For instance, introduction of NAATs in routine diagnostic testing have shown pharyngeal and rectal infection to be much more prevalent than previously assumed [14].
Since rectal and pharyngeal infections, as well as cervical infections in women, are frequently asymptomatic and will be missed by symptom-based examinations, laboratory testing should consider inclusion of both urogenital, anorectal, and pharyngeal samples, depending on sexual behavior, to identify infected individuals with higher sensitivity [5,15,16].
NAAT-based treatment monitoring has improved identification of treatment failures that particularly relate to pharyngeal infections [17]. Considering the presence of non-gonococcal Neisseria species at the pharyngeal mucosa that may transfer resistance to Ng [18,19], the pharynx has been suggested an important site for resistance development. Currently, the frequency and impact of genetic exchange in the pharynx is not known exactly but is important to be clarified, as it would strongly support pharyngeal screening and clearance of pharyngeal infections to be essential.
The objective of this review article is to summarize current diagnostic procedures for Ng detection according to recommendations of several guidelines and to review recent advances and novel developments that may potentially improve Ng diagnostic testing, based on publications primarily of the last 5 years identified by a PubMed literature search.
2. Microscopy
Direct microscopy is suitable in defined settings for the detection of Ng as a point-of-care test. Depending on the clinical picture, direct microscopy may be a valid diagnostic tool in settings with more modest resources.
For direct microscopy, two different staining methods are used: methylene blue staining and Gram´s staining. To prepare a staining preparation, the secretion is spread out in a thin layer on a microscope slide and is heated for fixation. For methylene blue staining, the slide is coated with 1% aqueous methylene blue solution or immersed in a cuvette. After a short exposure (15 s), the preparation is rinsed with water and dried between groundwood filter paper. At methylene blue staining, all bacteria turn blue. It should only be used as a diagnostic criterion for uncomplicated male urethritis in combination with typical clinical symptoms. In women’s gonorrhea and all other manifestations of disease, Gram´s staining is required [20,21]. Complete staining sets are commercially available. It allows the differentiation into Gram-negative (red) and Gram-positive (blue-violet) bacteria following a stepwise procedure of staining.
At high magnification (1000×), the leukocyte-rich sites are searched and examined with oil immersion. The typical pattern for gonococci is the paired (diplococci), piled, intraleukocytic storage. Diplococci are perpendicular to each other, have the same size and bean or kidney shape. In Gram´s staining, Ng shows as Gram-negative diplococci, often intracellular in polymorphonuclear leukocytes with typical morphology (Figure 1). However, detection of extracellular diplococci, especially in connection with a typical clinical picture, also indicates the presence of Ng. The detection is doubtful if atypical Gram-negative or Gram-labile diplococci are present. In symptomatic male urethritis, the sensitivity of Gram´s staining is up to 95% and highly specific (97%) for an experienced examiner. In endocervical samples, the sensitivity decreases to 40–60% [21,22]. In asymptomatic patients and in pharyngeal and rectal smears, the sensitivity is extremely low, and the method is not recommended [23]. Other bacteria, especially other Neisseria species, which have a similar morphology, compromise the microscopic result in extragenital specimens. In asymptomatic patients, the load of gonococci to be detected is usually too low.
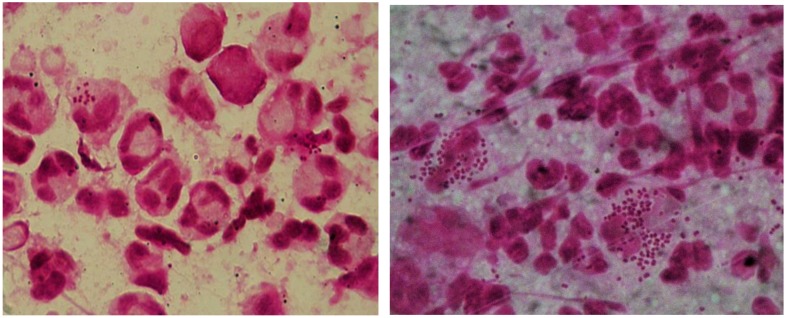
Figure 1.
Gram stain from a male urethral swab, depiction of polymorphonuclear leukocytes (PMNL) with Gram-negative intracellular diplococci. The left and right picture represent two different microscopic slides prepared from the same swab.
3. Culture
Antimicrobial resistance in Ng is a severe problem worldwide, and reliable results are indispensable preconditions for a successful therapy. Bacterial culture is sensitive and highly specific. In urogenital specimens, sensitivity may reach 85–95% under optimal conditions [23,24], and specificity is up to 100% when species identification is performed, as shown below. Up to now, it is the only method which allows complete antimicrobial susceptibility testing.
As an example, culture of Ng from a male urethral swab is shown in Figure 2. Cultivation does not succeed equally well from every sample material. Smear materials from the urethra and cervix are favorable. Bacterial culture from conjunctival, rectal, and oropharyngeal samples require optimal growth conditions, are time consuming, and often frustrating, especially in the case of throat swabs. Vaginal swab specimens and urine are rarely successfully cultivated [23,25,26].
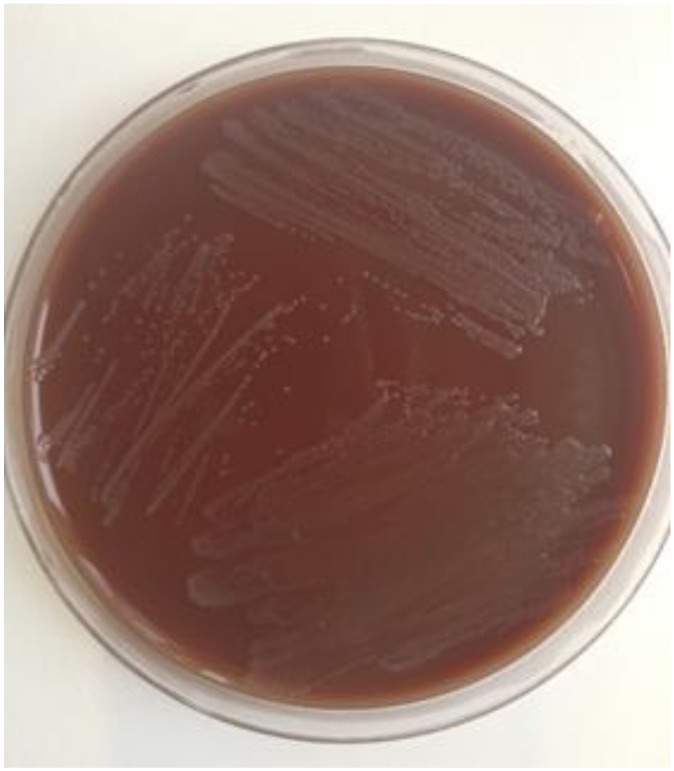
Figure 2.
Culture of Neisseria gonorrhoeae (Ng) from a male urethral swab.
Gonococci are very demanding and fastidious pathogens. They do not tolerate dehydration and should be inoculated immediately after swab collection onto culture media (nutritious selective culture medium and non-selective culture medium) [27]. Culture plates must be incubated at 35–37 °C and high humidity (70–80%), pH 6.75–7.5, and in a 4–6% CO2-enriched atmosphere. After 18–24 (–48) h, small, shiny gray colonies appear, whereby colony growth variations are possible [23]. After cultivation, identification of Ng is assessed by combining several detection methods. A presumptive identification is performed by microscopic Gram´s staining preparation and positive cytochrome oxidase reaction. To confirm the identification and distinguish between other Neisseria species like Neisseria meningitidis and apathogenic Neisseria spp. especially in extragenital sites, biochemical tests, immunological test, spectrometric test, or molecular test are applied. In biochemical identification tests (e.g., API-NH, bioMerieux), various enzymatic reactions and metabolic reactions are displayed by color change. The determined numerical profile allows the identification of the pathogen [28]. Alternative coagglutination tests with gonococcal-specific monoclonal antibodies (e.g., Phadebact Monoclonal GC test, MKL diagnostics; Gonocheck II TCS Biosciences Ltd.) can be performed [28,29].
Furthermore, mass spectrometric identification of Ng as a culture-based detection method can be used. MALDI-TOF (matrix-assisted laser desorption ionization time-of-flight) mass spectrometry identifies bacteria that can be taken directly from the agar plate. The result is a spectral fingerprint that can be assigned to the respective microorganism. The method has become established for the detection of Ng [30], with a reported positive predictive value of 99.3% [31,32]. However, results should be interpreted with caution for Ng and commensal Neisseria species when isolated from extragenital and oropharyngeal samples [33]. Molecular confirmation of Ng can also be performed using NAATs (see Section 5). Gonococcal typing and genome sequencing are mostly reserved for scientific, epidemiological, and forensic questions.
4. Antimicrobial Susceptibility Testing
Antimicrobial susceptibility testing is one of the most important procedures while processing Ng. This allows a reliable statement about the possible effectiveness of an antimicrobial therapeutic agent. Usually, the testing is performed as an indication of the minimum inhibitory concentration (MIC) of an antimicrobial agent in μg/ml or mg/l that inhibits growth. Various breakpoint standards are available for the assessment of susceptibility. It should therefore always be clear according to which standard the assessment is made. Currently, the following two standards are mainly used: CLSI (Clinical and Laboratory Standards Institute) (https://clsi.org/) or EUCAST (European Committee for Antimicrobial Susceptibility Testing) (http://www.eucast.org/clinical_breakpoints/). In addition, national antibiotic sensitivity test committees exist and should be consulted. MIC breakpoints were divided into three categories: “susceptible”, “intermediate”, and “resistant”. Since 2019, EUCAST has been introducing a new classification split into “susceptible“, “susceptible with increased exposure“, and “resistant“. In comparison, the breakpoints for Ng of the annually revised and freely accessible recommendations of EUCAST are slightly lower than the ones recommended by CLSI. EUCAST provides no information to zone diameter breakpoints and hence no information on disc diffusion testing. Currently, the main difference for Ng breakpoints between the two standards exists for azithromycin. CLSI provides no breakpoints for azithromycin. EUCAST changed from defined breakpoints to an epidemiological cut-off (ECOFF) in 2019. The ECOFF of 1 mg/L applies now to report acquired resistance.
Before performing susceptibility testing, it is important to select a test panel that is appropriate for the pathogen, the expected resistance, and the possible therapy options. There are three main test options for determining susceptibility: agar dilution method, MIC gradient strip test method, and disc diffusions assay. Regardless of the test method, strict and constant quality assurance, the use of WHO control strains, and intra-laboratory and external quality control assessments are required [34,35].
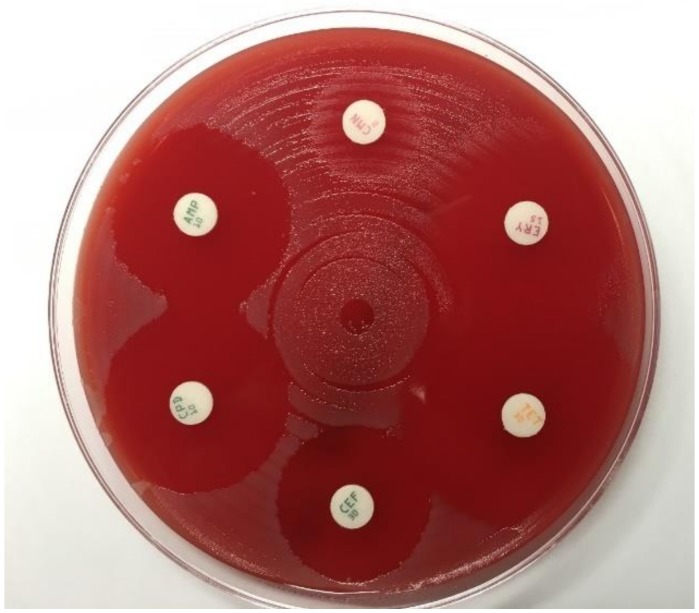
A limited qualitative estimation of antimicrobial susceptibility can be obtained by using a disc diffusion assay [36]. Thereby, discs containing defined antibiotic concentrations are placed on the agar surface. The antibiotic agent diffuses into the culture medium and inhibits growth. After incubation, the inhibition zone diameters are measured in mm. The growth inhibition zone is considered an approximation of the susceptibility (Figure 3). Several disc diffusion methods are described, and the method is only recommended when MIC determination cannot be performed, e.g., due to limited resources [37,38,39]. Therapeutically relevant disc diffusion results should be supplemented by confirmation tests using other methods.
Figure 3.
Disc diffusion assay (exemplary, no presentation of Ng testing). Photo by Andreas Gross (MVZ Laboratory Krone GbR, Bad Salzuflen, Germany).
The agar dilution method is the WHO-recommended gold standard method for antimicrobial susceptibility testing in Ng. A defined series of concentrations of an antibiotic substance is incorporated into nutrient medium. The corresponding growth inhibition is read off in relation to the rising concentration of the antibiotic. The lowest concentration that inhibits growth gives the MIC value in μg/ml (mg/l). However, this method is complex and predominantly suitable for a large number of tests [23]. A similar approach with broth microdilution has been developed but not established as a standard testing method so far [40].
Therefore, the standardized and quality-assured MIC gradient stripe test method (Etest), which correlates with the agar dilution method, is currently the preferred method [23,41,42]. MIC gradient stripe tests are plastic test strips with a predefined concentration gradient for a single antibiotic (Figure 4). The antibiotic agent diffuses into the culture medium, and the elliptical growth inhibition can be read off after incubation by using the printed MIC scale in μg/mL (mg/L). In 2018, various manufacturers of MIC strip test (MIC Test Strips—Liofilchem, M.I.C.Evaluator—Oxoid, and Ezy MIC Strip—HiMedia) were compared and evaluated compared to the reference Etest by bioMérieux for Ng testing. None of the tests met the high standards of Etest. M.I.C.Evaluator strips did not offer the antibiotic panel relevant for Ng testing. Ezy MIC strips showed inconsistent accuracy and quality. Liofilchem MIC Test Strips proofed relatively accurately but were still not fully comparable to Etest (bioMerieux) results [42,43]. In particular, testing of azithomycin is difficult, and test results can vary considerably [44].
Figure 4.
Minimum inhibitory concentration (MIC) gradient strip test method.
Some Ng strains carry a plasmid that encodes a high-level resistance to penicillin. This can be rapidly detected by color change using the Nitrocefin test. However, a negative ß-lactamase test does not exclude low-level penicillin resistance [45].
Recent studies show that early transcriptional changes upon exposure to antibiotics may be used to assess antimicrobial resistance (AMR). For Ng, transcripts have been identified that are differentially regulated in strains susceptible or resistant to ceftriaxone, azithromycin, or ciprofloxacin [46,47,48]. Quantification of these antibiotic-responsive transcripts results in a signature indicating susceptibility or resistance that may be used as a diagnostic tool in the future. In contrast to DNA-based resistance, test levels of antibiotic-responsive transcripts are independent of genetic mechanisms of resistance, but variance of gene expression may also result from genetic distance effects. Expression levels of porB and rpmB transcripts have been described to diagnose ciprofloxacin resistance in Ng isolates [46], but when testing a genetically more diverse panel of isolates’ expression levels of the two markers, they were no longer able to differentiate between susceptible and resistant strains [47]. The technique has great diagnostic potential, but large numbers of diverse Ng isolates from all over the world need to be tested to confirm that specific transcript levels indicate susceptibility or resistance to specific drugs.
5. NAATs
NAATs are the most sensitive techniques to detect Ng. Sensitivity and specificity of Ng NAATs is generally >95% and >99% in swabs and male first-catch urine (FCU) [4,13]. Currently available commercial tests are based on polymerase chain reaction (PCR) or isothermal transcription-mediated amplification. A list of FDA-approved commercial NAATs as of December 2019 is shown in Table 1. Superiority of NAATs over culture has been demonstrated in a number of studies [13,49,50,51,52,53,54]. The higher sensitivity of NAAT is partly due to the independence of viable bacteria and applies especially to extragenital specimens [13,14]. Another advantage compared to culture relates to the utility of diverse specimen types that are easier to handle, as no viable bacteria are required [50]. Moreover, NAATs are easier to perform and faster than culture with less hands-on time and the capability of automation allowing high throughput testing [55,56,57]. In addition, many commercial NAATs were designed to detect both Ng and Chlamydia trachomatis (Ct) in a single reaction [13].
Table 1.
Current FDA-approved nucleic acid amplification technologies (NAATs) for detection of Neisseria gonorrhoeae.
| Assay (Company) | Ng Targets | Cleared Specimen Types |
|---|---|---|
| Abbott RealTime CT/NG (Abbott) |
Opa gene | Women: urine, swabs (vaginal, endocervical) Men: urine, urethral swab |
| cobas CT/NG (Roche) |
Two different targets in the DR 9 region | Women: urine, swabs (vaginal, endocervical) Men: urine |
| APTIMA Combo 2 Assay (Hologic) |
16S-rRNA | urine swabs (vaginal, endocervical, urethral, rectal, pharyngeal) |
| BD MAX GC BD MAX CT/GC BD MAX CT/GC/TV |
OpcA gene | urine (20-60mL of first morning urine recommended), swabs (vaginal endocervical) |
| BD ProbeTec Neisseria gonorrhoeae (GC) Qx Amplified DNA Assay | Pilin-gene inverting protein homologue | Women: urine, swabs (vaginal, endocervical) Men: urine, urethral swab |
| BDProbeTec ET Chlamydia trachomatis and Neisseria gonorrhoeae Amplified DNA Assays | Pilin-gene inverting protein homologue | Women: urine, endocervical swab Men: urine, urethral swab |
| Xpert CT/NG (Cepheid) |
Two distinct chromosomal targets | urine swabs (vaginal, endocervical, rectal, pharyngeal) |
| binx io CT/NG Assay (binx health) |
Not specified | vaginal swabs |
Notes: As of December 2019.
Although sensitivity of NAATs is superior to other detection methods, it should be considered that diagnostic accuracy may be affected by genetic variations and the genomic plasticity of Neisseria. Loss or modification of target regions were shown to reduce sensitivity [58,59], whereas specificity may be diminished by cross-reactive non-pathogenic Neisseria species as well as horizontal transfer of Ng gene sequences to commensal Neisseria [60,61]. Another disadvantage of NAAT-based detection is the lack of information about AMR that still requires isolation of the bacteria by culture and subsequent susceptibility testing. Many commercial NAATs use specific specimen collection kits inappropriate for bacterial culture. However, NAATs may also work properly with nylon flocked swabs in ESwab collection kits [62], from which Ng culture succeeded in up to 70% after storage at 4 °C for one day [63], allowing a deferred culture strategy depending on antecedent NAAT results.
To detect male urethral gonococcal infection, first-catch urine (FCU, the first 10–20 mL of micturition) and urethral swabs are equally good, whereas in females, vaginal or endocervical swabs are more sensitive than FCU [13]. FCU is typically self-collected, but vaginal swabs and even meatal swabs may also be collected by the patients themselves and were shown to attain reliable NAAT results [64,65,66]. Accordingly, guidelines recommend NAAT testing of FCU in men and self-collected vaginal swabs in women, respectively, for laboratory diagnosis of urethral/cervical Ng infection [4,13,67].
NAATs are also the most sensitive tests to diagnose extragenital Ng infection [14,68,69] and are therefore recommended for laboratory diagnosis of rectal or pharyngeal infection [13,70]. The relevance of testing extragenital sites is mainly based on two findings: (i) additional testing of pharyngeal and rectal swabs was shown to increase the number of infected individuals [5,16]; (ii) patients can be infected at multiple sites [71], and pharyngeal infections are more difficult to treat [72].
Positive NAAT results obtained with extragenital specimens should be confirmed by detection of an alternative target to exclude false positive results due to cross reactivity with commensal Neisseria [73]. Confirmatory testing should also be considered in populations with low prevalence (i.e., screening of low-risk populations) with NAAT positive predictive values (PPV) less than 90% [4,74,75]. For routine diagnostic testing, a second test for confirmation seems impractical but may be bypassed using dual target assays including two Ng target regions that both need to be amplified for a positive test result [76,77].
Until recently, no NAATs were FDA-cleared for testing extragenital specimens. Thus, compliance with Clinical Laboratory Improvement Amendments (CLIA) for test modifications or equivalent regulatory standards for quality assurance were required. In April 2019, however, two Ct/Ng NAATs received FDA approval for testing rectal and pharyngeal specimens (Table 1).
The panel of pathogens included in NAATs for STI testing was recently extended further. Next to Ct and Ng, Mycoplsma genitalium (Mg) and Trichomonas vaginalis (Tv) were incorporated in two commercial assays [57,78]. Performance of BD Max CT/GC/TV assay to detect the three organisms in urine, endocervical, and vaginal swabs from 1990 female and 840 male subjects was consistent with comparator assays for Ct/Ng or Tv with a sensitivity >95.5% and specificity >98.6% for Ng in all specimen types [57]. Similarly, by analyzing 441 urine specimens with the BioRad real-time Dx CT/NG/MG assay, highly concordant results to comparator assays for Ct/Ng or Mg were described that resulted in a calculated sensitivity and specificity of 92% and 100%, respectively, for Ng detection [78].
Some multiplex NAATs allow testing an even more extensive panel of STI pathogens. Selected commercial multiplex NAATs are shown in Table 2. These assays differ with respect to amplification techniques and detection of amplified products, as well as the time to result and the STI panel included. Performance evaluations were published for some of these tests and generally showed good agreement with diagnostic assays for single targets. For instance, the AmpliSense Multiprime FRT real-time PCR assay for Ct, Ng, Tv, and Mg was evaluated by comparing it with APTIMA tests for Ct, Ng, Tv, and Mg on 209 vaginal swabs and 498 female FCU and 554 male FCU [79]. The authors reported excellent sensitivity and specificity for all pathogens except Mg, which lacks sufficient sensitivity. For Ng, AmpliSens and APTIMA AC2 results were 100% concordant. The STI FilmArray was applied to 295 clinical specimens and results compared to standard testing [80]. For Ct and Ng, Roche Amplicor was used as a comparator test and results for Ct and Ng were 98% and 97% concordant [80]. The STI multiplex assay from Seegene (Anyplex II) uses real-time amplification with multiple primers for seven different STI pathogens. Amplified products were specified by a combination of fluorescence labels and melting point analysis of specially designed oligonucleotides used as hybridization probes. Anyplex II test results largely correspond to results of other diagnostic tests for individual pathogens [81,82]. In a French study, Ng test results of 213 specimens obtained with Abbott CT/NG and Anyplex II were 97.2% concordant. Using Abbott CT/NG as a reference test, sensitivity and specificity of Anyplex II were 90% and 98.4%. However, further analysis of samples with discrepant results confirmed one of three Anyplex-positive/Abbott-negative as positive and one of three Anyplex-negative/Abbott-positive test results as negative, indicating sensitivity and specificity of Anyplex II may be even higher [82].
Table 2.
Multiplex PCR assays for STI testing.
| Assay (Company) |
Method of Amplification and Detection | Time to Result | Detected Pathogens |
|---|---|---|---|
| FTD STD 9 (FastTrack Diagnostics) |
Real-time PCR Fluorescence |
3–4 h | Ct, Ng, Tv, Mg, Uu, Up, Gv, HSV1, HSV2 |
| Anyplex II STI-7 (Seegene) |
Real-time PCR fluorescence and melting curve |
4–5 h | Ct, Ng, Tv, Mg, Uu, Up, Mh |
| Amplisense (Interlab Service) |
Real-time PCR Fluorescence |
3–4 h | Ct, Ng, Tv, Mg |
| FilmArray STI (BioMerieux) |
Nested PCR Fluorescence |
1 h | Ct, Ng, Tp, Tv, Mg, HSV1, HSV2, Uu, Hd |
| Easy Screen (Genetic Signatures) |
3-base real-time PCR (Bisufit-PCR) melting curve | 3 h | Ct, LGV, Ng, Mg, Tv, Uu, Up, Mh, GBS, Candida, Gv, HSV 1, HSV 2, VZV, Tp |
| STI Multiplex Aray (Randox Laboratories) |
Real-time PCR Fluorescence |
30 min | Ct, Ng, Mg, Tv, Uu, Mh, Hd, Tp, HSV 1, HSV 2 |
Ct: Chlamydia trachomatis, Ng: Neisseria gonorrhoeae, Tv: Trichomonas vaginalis, Mg: Mycoplasma genitalium, Uu: Ureaplasma urealyticum, Up: Ureaplasma parvum, Gv: Gardnerella vaginalis, HSV: Herpes simplex virus, Mh: Mycoplasma hominis, Hd: Hemophilus ducreyi, LGV: Lymphogranuloma venereum, GBS: Group B streptococci, VZV: Varicella zoster virus, Tp: Treponema pallidum.
In summary, several studies have shown high clinical sensitivity and specificity of STI multiplex NAATs not inferior to standard NAAT tests for single pathogens or duplex assays. These assays may potentially improve STI diagnostics due the higher content of information. However, depending on the test panel, a controversial discussion about the relevance of test results arose, especially considering microbial agents with low pathogenic potential (like Ureaplasma parvum or Mycoplasma hominis).
6. Rapid Tests and Point-of-Care Tests (POCT)
Although NAATs are considered the primary tests to detect Ng, their use in low- and middle-income countries is greatly limited due to relatively high costs. Management of patients in areas with limited access to laboratory testing is based mainly on clinical symptoms (syndromic-guided management) that, however, may result in inappropriate treatment, potentially increasing the risk of AMR development and spreading. For these regions, low-priced rapid diagnostic tests for Ng that can be performed independent from expensive laboratory equipment at the point of care are considered an important approach for confirmation of diagnosis and subsequent targeted treatment. There is also a demand for rapid diagnostic tests in countries with well-developed health care systems. Testing in a central laboratory is associated with increased turnaround times due to sample transport and reporting of test results that requires a follow-up visit of patients in order to start treatment in case of positive test results. Rapid diagnostic tests (RDTs) that can be performed at the point of care may produce test results within a timeframe the patient is willing to wait, allowing initiation of antibiotic treatment and instigation of partner notification at the same visit. Thus, early diagnosis and treatment of Ng infection by RDTs may potentially reduce ongoing transmission.
In principle, microscopy can be considered an Ng RDT that may be performed at the point of care, given the availability of a microscope. However, microscopic evaluation requires skilled investigators and lacks sensitivity in asymptomatic infections as well as in anorectal and pharyngeal specimens. Due to the presence of non-pathogenic Neisseria, specificity is also impaired when analyzing rectal or pharyngeal samples [83].
Other Ng RDTs are based on antigen-detection by immunochromatography (lateral flow assays) or optical immunoassays. Several immunochromatographic Ng RDTs were evaluated in clinical studies and consistently lack sufficient diagnostic performance with a sensitivity between 12.5% and 94% and a specificity between 89% and 99.8% [84,85]. Sensitivity may be even lower, as some studies used culture or an outdated PCR test with suboptimal sensitivity as the reference method [86,87,88]. In addition, the PPV of 97% reported in a Japanese study results from analysis of a specimen collection with >50% gonorrhea prevalence that usually does not reflect real situations. In another study, 100% sensitivity and 93% specificity was reported for the Biostar Optical immunoassay; this study, however, included only 5 Ng-positive urine specimens [89]. In conclusion, due to insufficient sensitivity and/or specificity, Ng RDTs depending on antigen detection are unsuitable to detect Ng infection [84].
In contrast, molecular PCR-based rapid tests were shown to have a much better diagnostic performance, comparable to that of reference laboratory NAATs. The GeneXpert CT/NG assay simultaneously detects Ng and Ct in a closed system and can be used at the point of care. However, the test does not fulfill the classic ASSURED criteria [90] for POCT (i.e., affordable, sensitive, specific, user-friendly, rapid and robust, equipment-free, and deliverable), as it is high-priced, needs electricity, and takes approximately 90 min. Therefore, the GeneXpert assay is usually referred to as a near-patient test rather than a POCT. By analyzing FCU from males, as well as vaginal and cervical swabs, the assay was able to detect Ng and Ct with high sensitivity (98–100%) and specificity (99.9–100%) that did not differ from reference laboratory NAATs [91]. The significantly higher clinical sensitivity of Xpert results from the low detection limit of 10 Ng genome copies [92], which is much lower than for antigen-based assays. The excellent performance was recently confirmed in another evaluation study on vaginal swabs from young South African women [93]. The high specificity results from amplification of two highly specific chromosomal targets that are both required for a positive test result. Therefore, the assay was also evaluated for anorectal and pharyngeal swabs, usually containing numerous commensal Neisseria species. Whereas no significant differences were found by comparing GeneXpert CT/NG with Aptima Combo2 in self-collected rectal swabs [94], sensitivity of GeneXpert was lower when analyzing male pharyngeal samples [95].
Main disadvantages of the GeneXpert system are the relatively high costs and the test duration of about 90 min that may compromise immediate antibiotic treatment in case of positive results if patients are not willing to wait that long and have to return for treatment initiation. Costs of the Xpert assay may vary among high-income countries but will be unacceptably high for resource-poor settings. Some other commercial PCR-based POCTs or near-patient tests to detect Ng are now available. Truelab Realtime micro PCR system (Molbio) and Randox STI multiplex array that runs on the Bosch Vivalytic platform are both faster than GeneXpert and take only 50 and 30 min, respectively, but so far, evaluation of both tests has not been published in peer-reviewed papers [96]. The binx io platform (binx health, formerly Atlas Genetics) is based on PCR and electrochemical detection of amplified products. A duplex assay for Ct and Ng provides results in about 30 min and recently received FDA approval (Table 1). The assay was evaluated in a multicenter study with more than 1500 symptomatic and asymptomatic patients (ClinicalTrials.gov Identifier: NCT03071510). Performance data have not yet been published in a peer-reviewed paper, but according to the company, sensitivity and specificity for Ng were 100% and 99.9%, respectively (https://mybinxhealth.com/news/binx-health-receives-fda-510k-clearance-for-rapid-point-of-care-platform-for-womens-health/).
Other NAATs that use isothermal amplification, instead of thermal cycling as in PCR, may reduce both costs and the time to result. Horst et al. recently described a paperfluidic device, integrating swab sample lysis, nucleic acid extraction, DNA amplification by isothermal thermophilic helicase-dependent amplification, and visual detection of amplified products on lateral flow strips. Using this device, Ng has been detected with 95% sensitivity and 100% specificity in a proof-of-concept study on 40 urethral and vaginal swab swabs [97]. The turnaround time of this low-cost assay is 80 min and may, according to the authors, be reduced to 60 min, making the assay particularly useful in settings with limited resources. Using recombinase polymerase amplification (RPA) Ct/Ng infection can be detected even faster in approximately 15 minutes [98]. A prototype version of this assay (TwistDx RPA assay) runs on the battery-powered Alere i instrument (Alere, Waltham, MA, USA), independent of electricity from the socket. The preliminary evaluation of diagnostic accuracy to detect Ct and Ng showed sensitivity, specificity, PPV, and NPV > 94% for male FCU, female FCU, and self-collected vaginal swabs [98]. Several other novel developments combining NAAT with microfluidics and nanotechnology that link high sensitivity with rapidity are in the pipeline and represent promising strategies towards a reliable and inexpensive POC testing [96]. These assays are performed on small devices using various microfluidic platforms that direct fluids through channels and reaction chambers for sample preparation and target detection by mechanical and electrokinetic mechanisms. Due to the integration of analytical steps on miniaturized devices, they were also named lab-on-a-chip systems. For example, mCHIP is a portable microfluidic diagnostic device for the detection of antigens and antibodies [99]. It was arranged for diagnosing HIV and syphilis but may also be used for detection of Ng and Ct antigens. The Vivalytic Analyzer (Bosch) also uses microfluidic techniques for fully automated qualitative and quantitative PCR analysis. The instrument represents an open system able to process diagnostic tests from different manufacturers. Most NAAT-based rapid tests require specific instruments and current supply and are probably unaffordable for low-income countries. However, recently a low-cost, portable analyzer for NAAT-based POCT was described [100] that appears attractive for settings with limited resources.
7. Future Demands
Among the methods for Ng detection, NAATs are the most sensitive, but so far, the vast majority of commercially available NAATs does not provide any information about resistance against antimicrobial compounds. Due to the higher sensitivity and easier workflow, as well as more rapid, automated, and high throughput testing, laboratories are increasingly using NAATs instead of culture, leading to a reduction of phenotypic AMR data. In the absence of resistance data, patients were treated empirically, which in the past has led to development of resistance to virtually all antibiotics used for Ng, especially in case of monotherapy.
The emergence of AMR strongly impairs the efficacy of Ng treatment and represents a significant clinical and public health challenge. Thus, bacterial culture should be attempted whenever possible. When using NAAT as a primary diagnostic test, cultivation of Ng subsequent to positive NAAT results frequently fails due to the limited viability of Ng. Although deferred culture of the bacteria has been improved using novel flocked swabs for collection of clinical specimens, the probability of successful culture declines with increasing storage time at 4 ℃ (to 69% after one day and 56% after 2 days) [63], indicating the need of further improvement of specimen collection and culture methods. On the other hand, predictions of AMR might be derived from genotypic data obtained in the context of NAAT analysis, ideally using a rapid molecular POCT.
Molecular alterations causing resistance to antimicrobials used to treat Ng were well characterized [101,102], and a number of real-time PCR tests detecting resistance determinants have been published in the last years (see [102] for a review). The assays were designed to detect single resistance mutations or multiple mutations associated with resistance to particular drugs or to a class of antibiotic compounds, like quinolones, macrolides, or beta lactam antibiotics [103,104,105,106,107,108,109]. To our knowledge, there is currently only one commercial assay for genotypic resistance testing of Ng (SpeeDx ResistancePlus GC for quinolone susceptibility). Some other molecular techniques were published that use a MASSarray iPLEX platform, multiplex bead arrays, and multiplex PCR with high-resolution melting analysis or based on mismatch amplification, allowing a more comprehensive analysis of resistance to multiple antimicrobials [110,111,112,113].
Generally, these assays were shown to accurately differentiate between wild type and mutation in both clinical samples and isolated bacteria but differ with respect to performance characteristics. Clinical sensitivity and specificity of some assays is limited, mainly by low Ng load and cross-reactive species [102]. Furthermore, prediction of resistance to antimicrobial drugs based on detection of individual resistance determinants is difficult, as development of resistance requires accumulation of several determinants in most cases. In order to standardize interpretation of genetic alterations associated with antibiotic resistance, the Public Health Agency of Canada has developed a web-based system (NG-STAR) for classification of seven genes associated with resistance to three classes of antibiotics (cephalosporins, macrolides, and fluoroquinolones). Currently, prediction of resistance based on genetic changes is most accurate for fluoroquinolones [104,114,115]. The primary mutations associated with ciprofloxacin resistance are located in the quinolone resistance determining region (QRDR) of gyrA, encoding subunit A of DNA gyrase. Additional mutations in parC encoding a subunit of topoisomerase IV are required for high-level resistance [101]. In regions with less prevalent ciprofloxacin resistance (i.e., outside Asia) implementation of genotypic ciprofloxacin resistance testing appears useful, as shown in an American study describing a significant decline of ceftriaxone use when guiding treatment by a PCR-based assay for ciprofloxacin resistance [116].
However, resistance to other antimicrobials used in Ng treatment is more complex, as multiple mechanisms contribute to the development of resistance. For instance, resistance to beta lactam antibiotics includes expression of beta lactamases, altered PBPs (point mutation and mosaic variants), increased discharge by efflux pumps, and reduced uptake by porins [102]. Consequently, in these cases, the presence of individual AMR determinants is insufficient to predict phenotypic resistance, but on the other hand, exclusion of any resistance determinant may indicate susceptibility with high probability [104].
In addition to determining the level of resistance analogous to MIC values in bacterial culture, genotypic resistance testing of Ng faces several other challenges, like cross-reactivity, especially in extragenital samples [103,106]; mixed infections of susceptible and resistant strains [117]; and the necessity of internal controls to exclude false negative results due to low gonococcal concentrations [118]. In addition, genetic assays are able to detect only known but not new AMR determinants that frequently develop in Ng, as we have seen in the past. Thus, any putative commercial genotypic test needs to be modified accordingly in a reasonable period without time-consuming and costly clinical validation.
At present, application of genotypic resistance testing appears to be useful primarily for surveillance of gonococcal resistance. To guide individual therapy of patients, further improvements are required to achieve high diagnostic accuracy and a high predictive value of AMR. This may be achieved in the future by applying whole genome sequencing [47,119,120] and deep learning systems for evaluation of sequencing data that, based on large sets of correlated genotypic and phenotypic data, may provide quantitative information about resistance to particular drugs, similar to the algorithms used in guiding antiretroviral therapy, for instance.
Acknowledgments
We kindly thank Dipl. Biol. Andreas Gross (MVZ Laboratory Krone GbR, Bad Salzuflen, Germany) for the permission to use the image in Figure 1c.
Author Contributions
All authors have read and agree to the published version of the manuscript. S.B. and T.M. both contributed to conceptualization, methodology of literature research, and writing (original draft preparation and review).
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Rowley J., Vander Hoorn S., Korenromp E., Low N., Unemo M., Abu-Raddad L.J., Chico R. Chlamydia, gonorrhoea, trichomoniasis and syphilis: Global prevalence and incidence estimates, 2016. Bull World Heal. Organ. 2019;97:548–562. doi: 10.2471/BLT.18.228486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.European Centre for Disease Prevention and Control . Gonorrhoea—Annual Epidemiological Report for 2017. European Centre for Disease Prevention and Control; Stockholm, Sweden: 2019. [Google Scholar]
- 3.Department of Health and Human Services . Sexually Transmitted Disease Surveillance 2018. Centers for Disease Control and Prevention; Atlanta, GE, USA: 2019. [DOI] [Google Scholar]
- 4.Fifer H., Saunders J., Soni S., Sadiq T., FitzGerald M. British Association for Sexual Health and HIV Web Site; 2019. [(accessed on 30 January 2020)]. National Guideline for the Management of Infection with Neisseria gonorrhoeae. Available online: https://www.bashhguidelines.org/media/1208/gc-2019.pdf. [DOI] [PubMed] [Google Scholar]
- 5.Dudareva-Vizule S., Haar K., Sailer A., Wisplinghoff H., Wisplinghoff F., Marcus U., PARIS Study Group Prevalence of pharyngeal and rectal Chlamydia trachomatis and Neisseria gonorrhoeae infections among men who have sex with men in Germany. Sex Transm. Infect. 2014;90:46–51. doi: 10.1136/sextrans-2012-050929. [DOI] [PubMed] [Google Scholar]
- 6.Buder S., Dudareva S., Jansen K., Loenenbach A., Nikisins S., Sailer A., Guhl E., Kohl P.K., Bremer V., GORENET Study Group Antimicrobial resistance of Neisseria gonorrhoeae in Germany: Low levels of cephalosporin resistance, but high azithromycin resistance. BMC Infect. Dis. 2018;18:44. doi: 10.1186/s12879-018-2944-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McConaghy J.R., Panchal B. Epididymitis: An Overview. Am. Fam. Physician. 2016;94:723–726. [PubMed] [Google Scholar]
- 8.Reekie J., Donovan B., Guy R., Hocking J.S., Kaldor J.M., Mak D., Preen D., Ward J., Liu B. Risk of Pelvic Inflammatory Disease in Relation to Chlamydia and Gonorrhea Testing, Repeat Testing, and Positivity: A Population-Based Cohort Study. Clin. Infect. Dis. 2018;66:437–443. doi: 10.1093/cid/ciz145. [DOI] [PubMed] [Google Scholar]
- 9.Brunham R.C., Gottlieb S.L., Paavonen J. Pelvic inflammatory disease. N. Engl. J. Med. 2015;372:2039–2048. doi: 10.1056/NEJMra1411426. [DOI] [PubMed] [Google Scholar]
- 10.Belkacem A., Caumes E., Ouanich J., Jarlier V., Dellion S., Cazenave B., Goursaud R., Lacassin F., Breuil J., Patey O., et al. Changing patterns of disseminated gonococcal infection in France: Cross-sectional data 2009–2011. Sex Transm. Infect. 2013;89:613–615. doi: 10.1136/sextrans-2013-051119. [DOI] [PubMed] [Google Scholar]
- 11.Heumann C.L., Quilter L.A., Eastment M.C., Heffron R., Hawes S.E. Adverse Birth Outcomes and Maternal Neisseria gonorrhoeae Infection: A Population-Based Cohort Study in Washington State. Sex Transm. Dis. 2017;44:266–271. doi: 10.1097/OLQ.0000000000000592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alexander E.R. Gonorrhea in the newborn. Ann. N. Y. Acad. Sci. 1988;549:180–186. doi: 10.1111/j.1749-6632.1988.tb23970.x. [DOI] [PubMed] [Google Scholar]
- 13.Papp J.R., Schachter J., Gaydos C.A., van der Pol B. Recommendations for the laboratory-based detection of Chlamydia trachomatis and Neisseria gonorrhoeae—2014. MMWR Recomm. Rep. 2014;63:1–19. [PMC free article] [PubMed] [Google Scholar]
- 14.Cornelisse V.J., Chow E.P., Huffam S., Fairley C.K., Bissessor M., De Petra V., Howden B.P., Denham I., Bradshaw C.S., Williamson D., et al. Increased Detection of Pharyngeal and Rectal Gonorrhea in Men Who Have Sex With Men After Transition From Culture To Nucleic Acid Amplification Testing. Sex Transm. Dis. 2017;44:114–117. doi: 10.1097/OLQ.0000000000000553. [DOI] [PubMed] [Google Scholar]
- 15.Kent C.K., Chaw J.K., Wong W., Liska S., Gibson S., Hubbard G., Klausner J.D. Prevalence of rectal, urethral, and pharyngeal Chlamydia and gonorrhea detected in 2 clinical settings among men who have sex with men: San Francisco, California, 2003. Clin. Infect. Dis. 2005;41:67–74. doi: 10.1086/430704. [DOI] [PubMed] [Google Scholar]
- 16.Weston E.J., Kirkcaldy R.D., Stenger M., Llata E., Hoots B., Torrone E.A. Narrative Review: Assessment of Neisseria gonorrhoeae Infections Among Men Who Have Sex With Men in National and Sentinel Surveillance Systems in the United States. Sex Transm. Dis. 2018;45:243–249. doi: 10.1097/OLQ.0000000000000740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Allen V.G., Mitterni L., Seah C., Rebbapragada A., Martin I.E., Lee C., Siebert H., Towns L., Melano R.G., Low D.E. Neisseria gonorrhoeae treatment failure and susceptibility to cefixime in Toronto, Canada. JAMA. 2013;309:163–170. doi: 10.1001/jama.2012.176575. [DOI] [PubMed] [Google Scholar]
- 18.Spratt B.G., Bowler L.D., Zhang Q.Y., Zhou J., Smith J.M. Role of interspecies transfer of chromosomal genes in the evolution of penicillin resistance in pathogenic and commensal Neisseria species. J. Mol. Evol. 1992;34:115–125. doi: 10.1007/bf00182388. [DOI] [PubMed] [Google Scholar]
- 19.Wadsworth C.B., Arnold B.J., Sater M.R.A., Grad Y.H. Azithromycin Resistance through Interspecific Acquisition of an Epistasis-Dependent Efflux Pump Component and Transcriptional Regulator in Neisseria gonorrhoeae. MBio. 2018;9 doi: 10.1128/mBio.01419-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Unemo M., Seifert H.S., Hook E.W., III, Hawkes S., Ndowa F., Dillon J.R. Gonorrhoea. Nat. Rev. Dis. Primers. 2019;5:79. doi: 10.1038/s41572-019-0128-6. [DOI] [PubMed] [Google Scholar]
- 21.Thorley N., Radcliffe K. The performance and clinical utility of cervical microscopy for the diagnosis of gonorrhoea in women in the era of the NAAT. Int. J. STD AIDS. 2015;26:656–660. doi: 10.1177/0956462414551233. [DOI] [PubMed] [Google Scholar]
- 22.Mensforth S., Thorley N., Radcliffe K. Auditing the use and assessing the clinical utility of microscopy as a point-of-care test for Neisseria gonorrhoeae in a Sexual Health clinic. Int. J. STD AIDS. 2018;29:157–163. doi: 10.1177/0956462417721062. [DOI] [PubMed] [Google Scholar]
- 23.Unemo M., Ison C. Gonorrhea. In: Unemo M., Ballard R., Ison C., Lewis D., Ndowna F., Peeling R., editors. Laboratory Diagnosis of Sexually Transmitted Infections, Including Human Immunodeficiency Virus. WHO; Geneva, Switzerland: 2013. [(accessed on 30 January 2020)]. Available online: https://www.who.int/reproductivehealth/publications/rtis/9789241505840/en/ [Google Scholar]
- 24.Bignell C., Ison C.A., Jungmann E. Gonorrhoea. Sex Transm. Infect. 2006;82(Suppl. 4):iv6–iv9. doi: 10.1136/sti.2006.023036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ota K.V., Tamari I.E., Smieja M., Jamieson F., Jones K.E., Towns L., Juzkiw J., Richardson S.E. Detection of Neisseria gonorrhoeae and Chlamydia trachomatis in pharyngeal and rectal specimens using the BD Probetec ET system, the Gen-Probe Aptima Combo 2 assay and culture. Sex Transm. Infect. 2009;85:182–186. doi: 10.1136/sti.2008.034140. [DOI] [PubMed] [Google Scholar]
- 26.Alexander S. The challenges of detecting gonorrhoea and Chlamydia in rectal and pharyngeal sites: Could we, should we, be doing more? Sex Transm. Infect. 2009;85:159–160. doi: 10.1136/sti.2008.035469. [DOI] [PubMed] [Google Scholar]
- 27.Visser M., van Westreenen M., van Bergen J., van Benthem B.H.B. Low gonorrhoea antimicrobial resistance and culture positivity rates in general practice: A pilot study. Sex Transm. Infect. 2019 doi: 10.1136/sextrans-2019-054006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Alexander S., Ison C. Evaluation of commercial kits for the identification of Neisseria gonorrhoeae. J. Med. Microbiol. 2005;54:827–831. doi: 10.1099/jmm.0.46108-0. [DOI] [PubMed] [Google Scholar]
- 29.Dillon J.R., Carballo M., Pauzé M. Evaluation of eight methods for identification of pathogenic Neisseria species: Neisseria-Kwik, RIM-N, Gonobio-Test, Minitek, Gonochek II, GonoGen, Phadebact Monoclonal GC OMNI Test, and Syva MicroTrak Test. J. Clin. Microbiol. 1988;26:493–497. doi: 10.1128/jcm.26.3.493-497.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Carannante A., De Carolis E., Vacca P., Vella A., Vocale C., De Francesco M.A., Cusini M., Del Re S., Dal Conte I., Cristaudo A., et al. Evaluation of matrix-assisted laser desorption ionization-time of flight mass spectrometry (MALDI-TOF MS) for identification and clustering of Neisseria gonorrhoeae. BMC Microbiol. 2015;15:142. doi: 10.1186/s12866-015-0480-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Buchanan R., Ball D., Dolphin H., Dave J. Matrix-assisted laser desorption-ionization time-of-flight mass spectrometry for the identification of Neisseria gonorrhoeae. Clin. Microbiol. Infect. 2016;22:815. doi: 10.1016/j.cmi.2016.06.010. [DOI] [PubMed] [Google Scholar]
- 32.Schweitzer V.A., van Dam A.P., Hananta I.P., Schuurman R., Kusters J.G., Rentenaar R.J. Identification of Neisseria gonorrhoeae by the Bruker Biotyper Matrix-Assisted Laser Desorption Ionization-Time of Flight Mass Spectrometry System Is Improved by a Database Extension. J. Clin. Microbiol. 2016;54:1130–1132. doi: 10.1128/JCM.00016-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Morel F., Jacquier H., Desroches M., Fihman V., Kumanski S., Cambau E., Decousser J.W., Berçot B. Use of Andromas and Bruker MALDI-TOF MS in the identification of Neisseria. Eur. J. Clin. Microbiol. Infect. Dis. 2018;37:2273–2277. doi: 10.1007/s10096-018-3368-6. [DOI] [PubMed] [Google Scholar]
- 34.Unemo M., Fasth O., Fredlund H., Limnios A., Tapsall J. Phenotypic and genetic characterization of the 2008 WHO Neisseria gonorrhoeae reference strain panel intended for global quality assurance and quality control of gonococcal antimicrobial resistance surveillance for public health purposes. J. Antimicrob. Chemother. 2009;63:1142–1151. doi: 10.1093/jac/dkp098. [DOI] [PubMed] [Google Scholar]
- 35.Unemo M., Golparian D., Sánchez-Busó L., Grad Y., Jacobsson S., Ohnishi M., Lahra M.M., Limnios A., Sikora A.E., Wi T., et al. The novel 2016 WHO Neisseria gonorrhoeae reference strains for global quality assurance of laboratory investigations: Phenotypic, genetic and reference genome characterization. J. Antimicrob. Chemother. 2016;71:3096–3108. doi: 10.1093/jac/dkw288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu H., Taylor T.H., Jr., Pettus K., Johnson S., Papp J.R., Trees D. Comparing the disk-diffusion and agar dilution tests for Neisseria gonorrhoeae antimicrobial susceptibility testing. Antimicrob. Resist. Infect. Control. 2016;5:46. doi: 10.1186/s13756-016-0148-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Singh V., Bala M., Kakran M., Ramesh V. Comparative assessment of CDS, CLSI disc diffusion and Etest techniques for antimicrobial susceptibility testing of Neisseria gonorrhoeae: A 6-year study. BMJ Open. 2012;2 doi: 10.1136/bmjopen-2012-000969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mal P.B., Jabeen K., Farooqi J., Unemo M., Khan E. Antimicrobial susceptibility testing of Neisseria gonorrhoeae isolates in Pakistan by Etest compared to Calibrated Dichotomous Sensitivity and Clinical Laboratory Standards Institute disc diffusion techniques. BMC Microbiol. 2016;16:236. doi: 10.1186/s12866-016-0707-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Enriquez R.P., Goire N., Kundu R., Gatus B.J., Lahra M.M. A comparison of agar dilution with the Calibrated Dichotomous Sensitivity (CDS) and Etest methods for determining the minimum inhibitory concentration of ceftriaxone against Neisseria gonorrhoeae. Diagn. Microbiol. Infect. Dis. 2016;86:40–43. doi: 10.1016/j.diagmicrobio.2016.04.016. [DOI] [PubMed] [Google Scholar]
- 40.Foerster S., Desilvestro V., Hathaway L.J., Althaus C.L., Unemo M. A new rapid resazurin-based microdilution assay for antimicrobial susceptibility testing of Neisseria gonorrhoeae. J. Antimicrob. Chemother. 2017;72:1961–1968. doi: 10.1093/jac/dkx113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu H., Taylor T.H., Jr., Pettus K., Trees D. Assessment of Etest as an alternative to agar dilution for antimicrobial susceptibility testing of Neisseria gonorrhoeae. J. Clin. Microbiol. 2014;52:1435–1440. doi: 10.1128/JCM.02131-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Papp J.R., Rowlinson M.C., O’Connor N.P., Wholehan J., Razeq J.H., Glennen A., Ware D., Iwen P.C., Lee L.V., Hagan C. Accuracy and reproducibility of the Etest to detect drug-resistant Neisseria gonorrhoeae to contemporary treatment. J. Med. Microbiol. 2018;67:68–73. doi: 10.1099/jmm.0.000651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jönsson A., Jacobsson S., Foerster S., Cole M.J., Unemo M. Performance characteristics of newer MIC gradient strip tests compared with the Etest for antimicrobial susceptibility testing of Neisseria gonorrhoeae. APMIS. 2018;126:822–827. doi: 10.1111/apm.12887. [DOI] [PubMed] [Google Scholar]
- 44.McAuliffe G.N., Smith M., Cooper G., Forster R.F., Roberts S.A. Variability in Azithromycin Susceptibility Results for Neisseria gonorrhoeae Obtained Using Gradient MIC Strip and Agar Dilution Techniques. J. Clin. Microbiol. 2019;57:e01353–19. doi: 10.1128/JCM.01353-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tapsall J.W., Phillips E.A., Morris L.M. Chromosomally mediated intrinsic resistance to penicillin in penicillinase producing strains of Neisseria gonorrhoeae isolated in Sydney: Guide to treatment with Augmentin. Genitourin. Med. 1987;63:305–308. doi: 10.1136/sti.63.5.305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Khazaei T., Barlow J.T., Schoepp N.G., Ismagilov R.F. RNA markers enable phenotypic test of antibiotic susceptibility in Neisseria gonorrhoeae after 10 minutes of ciprofloxacin exposure. Sci. Rep. 2018;8:11606. doi: 10.1038/s41598-018-29707-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wadsworth C.B., Sater M.R.A., Bhattacharyya R.P., Grad Y.H. Impact of Species Diversity on the Design of RNA-Based Diagnostics for Antibiotic Resistance in Neisseria gonorrhoeae. Antimicrob. Agents Chemother. 2019;63 doi: 10.1128/AAC.00549-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Zhao Y.H., Qin X.L., Yang J.Y., Liao Y.W., Wu X.Z., Zheng H.P. Identification and expression analysis of ceftriaxone resistance-related genes in Neisseria gonorrhoeae integrating RNA-Seq data and qRT-PCR validation. J. Glob. Antimicrob. Resist. 2019;16:202–209. doi: 10.1016/j.jgar.2018.10.008. [DOI] [PubMed] [Google Scholar]
- 49.Martin D.H., Cammarata C., van der Pol B., Jones R.B., Quinn T.C., Gaydos C.A., Crotchfelt K., Schachter J., Moncada J., Jungkind D., et al. Multicenter evaluation of AMPLICOR and automated cobas AMPLICOR CTT/NG tests for Neisseria gonorrhoeae. J. Clin. Microbiol. 2000;38:3544–3549. doi: 10.1128/jcm.38.10.3544-3549.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Cook R.L., Hutchison S.L., Ostergaard L., Braithwaite R.S., Ness R.B. Systematic review: Noninvasive testing for Chlamydia trachomatis and Neisseria gonorrhoeae. Ann. Intern. Med. 2005;142:914–925. doi: 10.7326/0003-4819-142-11-200506070-00010. [DOI] [PubMed] [Google Scholar]
- 51.Van Dyck E., Ieven M., Pattyn S., Van Damme L., Laga M. Detection of Chlamydia trachomatis and Neisseria gonorrhoeae by enzyme immunoassay, culture, and three nucleic acid amplification tests. J. Clin. Microbiol. 2001;39:1751–1756. doi: 10.1128/JCM.39.5.1751-1756.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Serra-Pladevall J., Caballero E., Roig G., Juvé R., Barbera M.J., Andreu A. Comparison between conventional culture and NAATs for the microbiological diagnosis in gonococcal infection. Diagn. Microbiol. Infect. Dis. 2015;83:341–343. doi: 10.1016/j.diagmicrobio.2015.08.005. [DOI] [PubMed] [Google Scholar]
- 53.Bromhead C., Miller A., Jones M., Whiley D. Comparison of the cobas 4800 CT/NG test with culture for detecting Neisseria gonorrhoeae in genital and nongenital specimens in a low-prevalence population in New Zealand. J. Clin. Microbiol. 2013;51:1505–1509. doi: 10.1128/JCM.03223-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Van der Pol B., Hook E.W., III, Williams J.A., Smith B., Taylor S.N. Performance of the BD CTQx and GCQx Amplified Assays on the BD Viper LT Compared With the BD Viper XTR System. Sex Transm. Dis. 2015;42:521–523. doi: 10.1097/OLQ.0000000000000313. [DOI] [PubMed] [Google Scholar]
- 55.Cheng A., Kirby J.E. Evaluation of the Hologic gen-probe PANTHER, APTIMA Combo 2 assay in a tertiary care teaching hospital. Am. J. Clin. Pathol. 2014;141:397–403. doi: 10.1309/AJCPFQ25SQVZAWHZ. [DOI] [PubMed] [Google Scholar]
- 56.Marlowe E.M., Hardy D., Krevolin M., Gohl P., Bertram A., Arcenas R., Seiverth B., Schneider T., Liesenfeld O. High-throughput testing of urogenital and extragenital specimens for detection of Chlamydia trachomatis and Neisseria gonorrhoeae with Cobas CT/NG. Eur. J. Microbiol. Immunol. Bp. 2017;7:176–186. doi: 10.1556/1886.2017.00018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Van der Pol B., Williams J.A., Fuller D., Taylor S.N., Hook E.W., III Combined Testing for Chlamydia, gonorrhea, and Trichomonas by use of the BD Max CT/GC/TV Assay with genitourinary specimen types. J. Clin. Microbiol. 2017;55:155–164. doi: 10.1128/JCM.01766-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Whiley D.M., Limnios A., Moon N.J., Gehrig N., Goire N., Hogan T., Lam A., Jacob K., Lambert S.B., Nissen M.D., et al. False-negative results using Neisseria gonorrhoeae porA pseudogene PCR—A clinical gonococcal isolate with an N. meningitidis porA sequence, Australia, March 2011. Eurosurveillance. 2011;16:19874. [PubMed] [Google Scholar]
- 59.Ison C.A., Golparian D., Saunders P., Chisholm S., Unemo M. Evolution of Neisseria gonorrhoeae is a continuing challenge for molecular detection of gonorrhoea: False negative gonococcal porA mutants are spreading internationally. Sex Transm. Infect. 2013;89:197–201. doi: 10.1136/sextrans-2012-050829. [DOI] [PubMed] [Google Scholar]
- 60.Upton A., Bromhead C., Whiley D.M. Neisseria gonorrhoeae false-positive result obtained from a pharyngeal swab by using the Roche cobas 4800 CT/NG assay in New Zealand in 2012. J. Clin. Microbiol. 2013;51:1609–1610. doi: 10.1128/JCM.00485-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Frosch M., Meyer T.F. Transformation-mediated exchange of virulence determinants by co-cultivation of pathogenic Neisseriae. FEMS Microbiol. Lett. 1992;100:345–349. doi: 10.1111/j.1574-6968.1992.tb14062.x. [DOI] [PubMed] [Google Scholar]
- 62.Li J., Jang D., Gilchrist J., Smieja M., Ewert R., MacRitchie C., Chernesky M. Comparison of flocked and aptima swabs and two specimen transport media in the aptima combo 2 assay. J. Clin. Microbiol. 2014;52:3808–3809. doi: 10.1128/JCM.01614-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wind C.M., de Vries H.J., Schim van der Loeff M.F., Unemo M., van Dam A.P. Successful Combination of Nucleic Acid Amplification Test Diagnostics and Targeted Deferred Neisseria gonorrhoeae Culture. J. Clin. Microbiol. 2015;53:1884–1990. doi: 10.1128/JCM.00369-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Schachter J., Chernesky M.A., Willis D.E., Fine P.M., Martin D.H., Fuller D., Jordan J.A., Janda W., Hook E.W., III Vaginal swabs are the specimens of choice when screening for Chlamydia trachomatis and Neisseria gonorrhoeae: Results from a multicenter evaluation of the APTIMA assays for both infections. Sex Transm. Dis. 2005;32:725–728. doi: 10.1097/01.olq.0000190092.59482.96. [DOI] [PubMed] [Google Scholar]
- 65.Stewart C.M., Schoeman S.A., Booth R.A., Smith S.D., Wilcox M.H., Wilson J.D. Assessment of self taken swabs versus clinician taken swab cultures for diagnosing gonorrhoea in women: Single centre, diagnostic accuracy study. BMJ Clin. Res. Ed. 2012;345:e8107. doi: 10.1136/bmj.e8107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Dize L., Agreda P., Quinn N., Barnes M.R., Hsieh Y.H., Gaydos C.A. Comparison of self-obtained penile-meatal swabs to urine for the detection of C. trachomatis, N. gonorrhoeae and T. vaginalis. Sex Transm. Infect. 2013;89:305–307. doi: 10.1136/sextrans-2012-050686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.AWMF Leitlinie: Sexuell Übertragbare Infektionen (STI)—Beratung, Diagnostik, Therapie, 059-006. [(accessed on 30 January 2020)];2019 Available online: https://www.awmf.org/uploads/tx_szleitlinien/059-006l_S2k_Sexuell-uebertragbare-Infektionen-Beratung-Diagnostik-Therapie-STI_2019-09.pdf.
- 68.Page-Shafer K., Graves A., Kent C., Balls J.E., Zapitz V.M., Klausner J.D. Increased sensitivity of DNA amplification testing for the detection of pharyngeal gonorrhea in men who have sex with men. Clin. Infect. Dis. 2002;34:173–176. doi: 10.1086/338236. [DOI] [PubMed] [Google Scholar]
- 69.Schachter J., Moncada J., Liska S., Shayevich C., Klausner J.D. Nucleic acid amplification tests in the diagnosis of chlamydial and gonococcal infections of the oropharynx and rectum in men who have sex with men. Sex Transm. Dis. 2008;35:637–642. doi: 10.1097/OLQ.0b013e31817bdd7e. [DOI] [PubMed] [Google Scholar]
- 70.Hughes G., Ison C., Field N. Guidance for the Detection of Gonorrhoea in England. Public Health England; London, UK: 2014. [(accessed on 30 January 2020)]. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/769003/170215_Gonorrhoea_testing_guidance_REVISED__2_.pdf. [Google Scholar]
- 71.Benn P.D., Rooney G., Carder C., Brown M., Stevenson S.R., Copas A., Robinson A.J., Ridgway G.L. Chlamydia trachomatis and Neisseria gonorrhoeae infection and the sexual behaviour of men who have sex with men. Sex Transm. Infect. 2007;83:106–112. doi: 10.1136/sti.2006.021329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fifer H., Natarajan U., Jones L., Alexander S., Hughes G., Golparian D., Unemo M. Failure of Dual Antimicrobial Therapy in Treatment of Gonorrhea. N. Engl. J. Med. 2016;374:2504–2506. doi: 10.1056/NEJMc1512757. [DOI] [PubMed] [Google Scholar]
- 73.Palmer H.M., Mallinson H., Wood R.L., Herring A.J. Evaluation of the specificities of five DNA amplification methods for the detection of Neisseria gonorrhoeae. J. Clin. Microbiol. 2003;41:835–837. doi: 10.1128/jcm.41.2.835-837.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Field N., Clifton S., Alexander S., Ison C.A., Hughes G., Beddows S., Tanton C., Soldan K., Coelho da Silva F., Mercer C.H., et al. Confirmatory assays are essential when using molecular testing for Neisseria gonorrhoeae in low-prevalence settings: Insights from the third National Survey of Sexual Attitudes and Lifestyles (Natsal-3) Sex Transm. Infect. 2015;91:338–341. doi: 10.1136/sextrans-2014-051850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Chow E.P., Fehler G., Read T.R., Tabrizi S.N., Hocking J.S., Denham I., Bradshaw C.S., Chen M.Y., Fairley C.K. Gonorrhoea notifications and nucleic acid amplification testing in a very low-prevalence Australian female population. Med. J. Aust. 2015;202:321–323. doi: 10.5694/mja14.00780. [DOI] [PubMed] [Google Scholar]
- 76.Tabrizi S.N., Unemo M., Golparian D., Twin J., Limnios A.E., Lahra M., Guy R., TTANGO Investigators Analytical evaluation of GeneXpert CT/NG, the first genetic point-of-care assay for simultaneous detection of Neisseria gonorrhoeae and Chlamydia trachomatis. J. Clin. Microbiol. 2013;51:1945–1947. doi: 10.1128/JCM.00806-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Perry M.D., Jones R.N., Corden S.A. Is confirmatory testing of Roche cobas 4800 CT/NG test Neisseria gonorrhoeae positive samples required? Comparison of the Roche cobas 4800 CT/NG test with an opa/pap duplex assay for the detection of N. gonorrhoeae. Sex Transm. Infect. 2014;90:303–308. doi: 10.1136/sextrans-2013-051410. [DOI] [PubMed] [Google Scholar]
- 78.Ursi D., Crucitti T., Smet H., Ieven M. Evaluation of the Bio-Rad Dx CT/NG/MG® assay for simultaneous detection of Chlamydia trachomatis, Neisseria gonorrhoeae and Mycoplasma genitalium in urine. Eur. J. Clin. Microbiol. Infect. Dis. 2016;35:1159–1163. doi: 10.1007/s10096-016-2646-4. [DOI] [PubMed] [Google Scholar]
- 79.Rumyantseva T., Golparian D., Nilsson C.S., Johansson E., Falk M., Fredlund H., Van Dam A., Guschin A., Unemo M. Evaluation of the new AmpliSens multiplex real-time PCR assay for simultaneous detection of Neisseria gonorrhoeae, Chlamydia trachomatis, Mycoplasma genitalium and Trichomonas vaginalis. APMIS. 2015;123:879–886. doi: 10.1111/apm.12430. [DOI] [PubMed] [Google Scholar]
- 80.Kriesel J.D., Bhatia A.S., Barrus C., Vaughn M., Gardner J., Crisp R.J. Multiplex PCR testing for nine different sexually transmitted infections. Int. J. STD. AIDS. 2016;27:1275–1282. doi: 10.1177/0956462415615775. [DOI] [PubMed] [Google Scholar]
- 81.Choe H.S., Lee D.S., Lee S.J., Hong S.H., Park D.C., Lee M.K., Kim T.H., Cho Y.H. Performance of Anyplex™ II multiplex real-time PCR for the diagnosis of seven sexually transmitted infections: Comparison with currently available methods. Int. J. Infect. Dis. 2013;17:e1134–40. doi: 10.1016/j.ijid.2013.07.011. [DOI] [PubMed] [Google Scholar]
- 82.Berçot B., Amarsy R., Goubard A., Aparicio C., Loeung H.U., Segouin C., Gueret H., Jacquier F., Meunier F., Mougari E., et al. Assessment of coinfection of sexually transmitted pathogen microbes by use of the anyplex II STI-7 molecular kit. J. Clin. Microbiol. 2015;53:991–993. doi: 10.1128/JCM.03370-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Bignell C., Unemo M. European STI Guidelines Editorial Board. 2012 European guideline on the diagnosis and treatment of gonorrhoea in adults. Int. J. STD. AIDS. 2013;24:85–92. doi: 10.1177/0956462412472837. [DOI] [PubMed] [Google Scholar]
- 84.Guy R.J., Causer L.M., Klausner J.D., Unemo M., Toskin I., Azzini A.M., Peeling R.W. Performance and operational characteristics of point-of-care tests for the diagnosis of urogenital gonococcal infections. Sex Transm. Infect. 2017;93:S16–S21. doi: 10.1136/sextrans-2017-053192. [DOI] [PubMed] [Google Scholar]
- 85.Abbai N.S., Moodley P., Reddy T., Zondi T.G., Rambaran S., Naidoo K., Ramjee G. Clinical evaluation of the OneStep Gonorrhea RapiCard InstaTest for detection of Neisseria gonorrhoeae in symptomatic patients from KwaZulu-Natal, South Africa. J. Clin. Microbiol. 2015;53:1348–1350. doi: 10.1128/JCM.03603-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Benzaken A.S., Galban E.G., Antunes W., Dutra J.C., Peeling R.W., Mabey D., Salama A. Diagnosis of gonococcal infection in high risk women using a rapid test. Sex Transm. Infect. 2006;82(Suppl. 5):v26–v28. doi: 10.1136/sti.2006.022566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Suzuki K., Matsumoto T., Murakami H., Tateda K., Ishii N., Yamaguchi K. Evaluation of a rapid antigen detection test for Neisseria gonorrhoeae in urine sediment for diagnosis of gonococcal urethritis in males. J. Infect. Chemother. 2004;10:208–211. doi: 10.1007/s10156-004-0322-6. [DOI] [PubMed] [Google Scholar]
- 88.Nuñez-Forero L., Moyano-Ariza L., Gaitán-Duarte H., Ángel-Müller E., Ruiz-Parra A., González P., Rodríguez A., Tolosa J.E. Diagnostic accuracy of rapid tests for sexually transmitted infections in symptomatic women. Sex Transm. Infect. 2016;92:24–28. doi: 10.1136/sextrans-2014-051891. [DOI] [PubMed] [Google Scholar]
- 89.Samarawickrama A., Cheserem E., Graver M., Wade J., Alexander S., Ison C. Pilot study of use of the BioStar Optical ImmunoAssay GC point-of-care test for diagnosing gonorrhoea in men attending a genitourinary medicine clinic. J. Med. Microbiol. 2014;63:1111–1112. doi: 10.1099/jmm.0.071852-0. [DOI] [PubMed] [Google Scholar]
- 90.Peeling R.W., Holmes K.K., Mabey D., Ronald A. Rapid tests for sexually transmitted infections (STIs): The way forward. Sex Transm. Infect. 2006;82:v1–v6. doi: 10.1136/sti.2006.024265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Gaydos C.A., van der Pol B., Jett-Goheen M., Barnes M., Quinn N., Clark C., Daniel G.E., Dixon P.B., Hook E.W., III, CT/NG Study Group Performance of the Cepheid CT/ NG Xpert Rapid PCR Test for Detection of Chlamydia trachomatis and Neisseria gonorrhoeae. J. Clin. Microbiol. 2013;51:1666–1672. doi: 10.1128/JCM.03461-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gaydos C.A. Review of use of a new rapid real-time PCR, the Cepheid GeneXpert® (Xpert) CT/NG assay, for Chlamydia trachomatis and Neisseria gonorrhoeae: Results for patients while in a clinical setting. Expert. Rev. Mol. Diagn. 2014;14:135–137. doi: 10.1586/14737159.2014.871495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Garrett N., Mitchev N., Osman F., Naidoo J., Dorward J., Singh R., Ngobese H., Rompalo A., Mlisana K., Mindel A. Diagnostic accuracy of the Xpert CT/NG and OSOM Trichomonas Rapid assays for point-of-care STI testing among young women in South Africa: A cross-sectional study. BMJ Open. 2019;9:e026888. doi: 10.1136/bmjopen-2018-026888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Dize L., Silver B., Gaydos C. Comparison of the Cepheid GeneXpert CT/NG assay to the Hologic Aptima Combo2 assay for the detection of Chlamydia trachomatis and Neisseria gonorrhoeae in self-collected rectal swabs. Diagn. Microbiol. Infect. Dis. 2018;90:83–84. doi: 10.1016/j.diagmicrobio.2017.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Geiger R., Smith D.M., Little S.J., Mehta S.R. Validation of the GeneXpert(R) CT/NG Assay for use with Male Pharyngeal and Rectal Swabs. Austin. J. HIV. AIDS Res. 2016;3:1021. [PMC free article] [PubMed] [Google Scholar]
- 96.Murtagh M.M. WHO; 2019. [(accessed on 30 January 2020)]. The Point-of-Care Diagnostic Landscape for Sexually Transmitted Infections (STIs) Available online: https://www.who.int/reproductivehealth/topics/rtis/Diagnostic-Landscape-for-STIs-2019.pdf. [Google Scholar]
- 97.Horst A.L., Rosenbohm J.M., Kolluri N., Hardick J., Gaydos C.A., Cabodi M., Klapperich C.M., Linnes J.C. A paperfluidic platform to detect Neisseria gonorrhoeae in clinical samples. Biomed. Microdevices. 2018;20:35. doi: 10.1007/s10544-018-0280-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Harding-Esch E.M., Fuller S.S., Chow S.L., Nori A.V., Harrison M.A., Parker M., Piepenburg O., Forrest M.S., Brooks D.G., Patel R., et al. Diagnostic accuracy of a prototype rapid chlamydia and gonorrhoea recombinase polymerase amplification assay: A multicentre cross-sectional preclinical evaluation. Clin. Microbiol. Infect. 2019;25:380.e1–380.e7. doi: 10.1016/j.cmi.2018.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Chin C.D., Laksanasopin T., Cheung Y.K., Steinmiller D., Linder V., Parsa H., Wang J., Moore H., Rouse R., Umviligihozo G., et al. Microfluidics-based diagnostics of infectious diseases in the developing world. Nat. Med. 2011;17:1015–1019. doi: 10.1038/nm.2408. [DOI] [PubMed] [Google Scholar]
- 100.Tsaloglou M.N., Nemiroski A., Camci-Unal G., Christodouleas D.C., Murray L.P., Connelly J.T., Whitesides G.M. Handheld isothermal amplification and electrochemical detection of DNA in resource-limited settings. Anal. Biochem. 2018;543:116–121. doi: 10.1016/j.ab.2017.11.025. [DOI] [PubMed] [Google Scholar]
- 101.Unemo M., Shafer W.M. Antimicrobial resistance in Neisseria gonorrhoeae in the 21st century: Past, evolution, and future. Clin. Microbiol. Rev. 2014;27:587–613. doi: 10.1128/CMR.00010-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Donà V., Low N., Golparian D., Unemo M. Recent advances in the development and use of molecular tests to predict antimicrobial resistance in Neisseria gonorrhoeae. Expert. Rev. Mol. Diagn. 2017;17:845–859. doi: 10.1080/14737159.2017.1360137. [DOI] [PubMed] [Google Scholar]
- 103.Peterson S.W., Martin I., Demczuk W., Bharat A., Hoang L., Wylie J., Allen V., Lefebvre B., Tyrrell G., Horsman G., et al. Molecular assay for detection of ciprofloxacin resistance in Neisseria gonorrhoeae isolates from cultures and clinical nucleic acid amplification test specimens. J. Clin. Microbiol. 2015;53:3606–3608. doi: 10.1128/JCM.01632-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Pond M.J., Hall C.L., Miari V.F., Cole M., Laing K.G., Jagatia H., Harding-Esch E., Monahan I.M., Planche T., Hinds J., et al. Accurate detection of Neisseria gonorrhoeae ciprofloxacin susceptibility directly from genital and extragenital clinical samples: Towards genotype-guided antimicrobial therapy. J. Antimicrob. Chemother. 2016;71:897–902. doi: 10.1093/jac/dkv432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Siedner M.J., Pandori M., Castro L., Barry P., Whittington W.L., Liska S., Klausner J.D. Real-time PCR assay for detection of quinolone- resistant Neisseria gonorrhoeae in urine samples. J. Clin. Microbiol. 2007;45:1250–1254. doi: 10.1128/JCM.01909-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Trembizki E., Buckley C., Donovan B., Chen M., Guy R., Kaldor J., Lahra M.M., Regan D.G., Smith H., Ward J., et al. Direct real-time PCR-based detection of Neisseria gonorrhoeae 23S rRNA mutations associated with azithromycin resistance. J. Antimicrob. Chemother. 2015;70:3244–3249. doi: 10.1093/jac/dkv274. [DOI] [PubMed] [Google Scholar]
- 107.Ochiai S., Ishiko H., Yasuda M., Deguchi T. Rapid detection of the mosaic structure of the Neisseria gonorrhoeae penA gene, which is associated with decreased susceptibilities to oral cephalosporins. J. Clin. Microbiol. 2008;46:1804–1810. doi: 10.1128/JCM.01800-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Goire N., Freeman K., Lambert S.B., Nimmo G.R., Limnios A.E., Lahra M.M., Nissen M.D., Sloots T.P., Whiley D.M. The influence of target population on nonculture-based detection of markers of Neisseria gonorrhoeae antimicrobial resistance. Sex Health. 2012;9:422–429. doi: 10.1071/SH12026. [DOI] [PubMed] [Google Scholar]
- 109.Peterson S.W., Martin I., Demczuk W., Bharat A., Hoang L., Wylie J., Lefebvre B., Tyrrell G., Horsman G., Haldane D., et al. Molecular assay for detection of genetic markers associated with decreased susceptibility to cephalosporins in Neisseria gonorrhoeae. J. Clin. Microbiol. 2015;53:2042–2048. doi: 10.1128/JCM.00493-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Trembizki E., Wand H., Donovan B., Chen M., Fairley C.K., Freeman K., Guy R., Kaldor J.M., Lahra M.M., Lawrence A., et al. The molecular epidemiology and antimicrobial resistance of Neisseria gonorrhoeae in Australia: A nationwide cross-sectional study, 2012. Clin. Infect. Dis. 2016;63:1591–1598. doi: 10.1093/cid/ciw648. [DOI] [PubMed] [Google Scholar]
- 111.Dona V., Kasraian S., Lupo A., Guilarte Y.N., Hauser C., Furrer H., Unemo M., Low N., Endimiani A. Multiplex real-time PCR assay with high-resolution melting analysis for characterization of antimicrobial resistance in Neisseria gonorrhoeae. J. Clin. Microbiol. 2016;54:2074–2081. doi: 10.1128/JCM.03354-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Balashov S., Mordechai E., Adelson M.E., Gygax S.E. Multiplex bead suspension array for screening Neisseria gonorrhoeae antibiotic resistance genetic determinants in noncultured clinical samples. J. Mol. Diagn. 2013;15:116–129. doi: 10.1016/j.jmoldx.20.12.08.005. [DOI] [PubMed] [Google Scholar]
- 113.Donà V., Smid J.H., Kasraian S., Egli-Gany D., Dost F., Imeri F., Unemo M., Low N., Endimiani A. Mismatch Amplification Mutation Assay-Based Real-Time PCR for Rapid Detection of Neisseria gonorrhoeae and Antimicrobial Resistance Determinants in Clinical Specimens. J. Clin. Microbiol. 2018;56:e00365–18. doi: 10.1128/JCM.00365-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Low N., Unemo M. Molecular tests for the detection of antimicrobial resistant Neisseria gonorrhoeae: When, where, and how to use? Curr. Opin. Infect. Dis. 2016;29:45–51. doi: 10.1097/QCO.0000000000000230. [DOI] [PubMed] [Google Scholar]
- 115.Goire N., Lahra M.M., Chen M., Donovan B., Fairley C.K., Guy R., Kaldor J., Regan D., Ward J., Nissen M.D., et al. Molecular approaches to enhance surveillance of gonococcal antimicrobial resistance. Nat. Rev. Microbiol. 2014;12:223–229. doi: 10.1038/nrmicro3217. [DOI] [PubMed] [Google Scholar]
- 116.Allan-Blitz L.T., Klausner J.D. Codon 91 Gyrase A testing is necessary and sufficient to predict ciprofloxacin susceptibility in Neisseria gonorrhoeae. J. Infect. Dis. 2017;215:491. doi: 10.1093/infdis/jiw551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Goire N., Kundu R., Trembizki E., Buckley C., Hogan T.R., Lewis D.A., Branley J.M., Whiley D.M., Lahra M.M. Mixed gonococcal infections in a high-risk population, Sydney, Australia 2015: Implications for antimicrobial resistance surveillance? J. Antimicrob. Chemother. 2017;72:407–409. doi: 10.1093/jac/dkw406. [DOI] [PubMed] [Google Scholar]
- 118.Zhao L., Zhao S. TaqMan real-time quantitative PCR assay for detection of fluoroquinolone-resistant Neisseria gonorrhoeae. Curr. Microbiol. 2012;65:692–695. doi: 10.1007/s00284-012-0212-6. [DOI] [PubMed] [Google Scholar]
- 119.Golparian D., Donà V., Sánchez-Busó L., Foerster S., Harris S., Endimiani A., Low N., Unemo M. Antimicrobial resistance prediction and phylogenetic analysis of Neisseria gonorrhoeae isolates using the Oxford Nanopore MinION sequencer. Sci. Rep. 2018;8:17596. doi: 10.1038/s41598-018-35750-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Eyre D.W., Golparian D., Unemo M. Prediction of Minimum Inhibitory Concentrations of Antimicrobials for Neisseria gonorrhoeae Using Whole-Genome Sequencing. Methods Mol. Biol. 2019;1997:59–76. doi: 10.1007/978-1-4939-9496-0_4. [DOI] [PubMed] [Google Scholar]