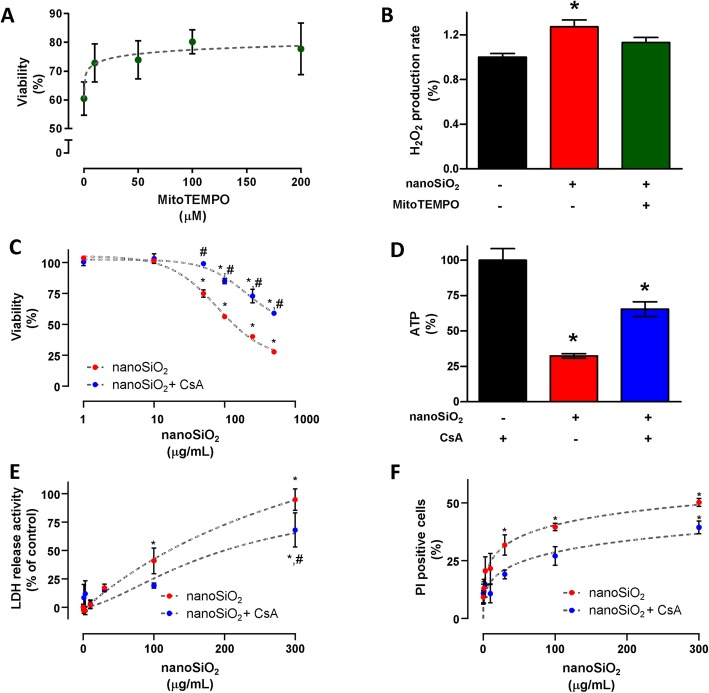
Fig. 5.
The toxicity mechanism of nanoSiO2 in cardiac cells is driven by reactive oxygen species and the opening of the mPTP. a MitoTEMPO dose-dependent cellular death prevention with 200 μg/mL of nanoSiO2 administration in H9c2 cells. b H2O2 production after nanoSiO2 administration (200 μg/mL) in presence or absence of MitoTEMPO (100 μM) in H9c2 cells. c Cellular viability in ventricular myocytes after nanoSiO2 administration in the absence or presence of CsA (0.5 μM). d ATP production in cardiomyocytes after nanoSiO2 administration (100 μg/mL) in absence or presence of CsA (0.5 μM). For human cardiomyocytes: (e) LDH release activity, (f) PI positive cells. MitoTEMPO or CsA were applied 30 min prior to nanoSiO2 administration. nanoSiO2 was incubated during 24 h. Values are percentage of control and represent mean ± SEM. *p ≤ 0.05 vs control, #p ≤ 0.05 vs CsA