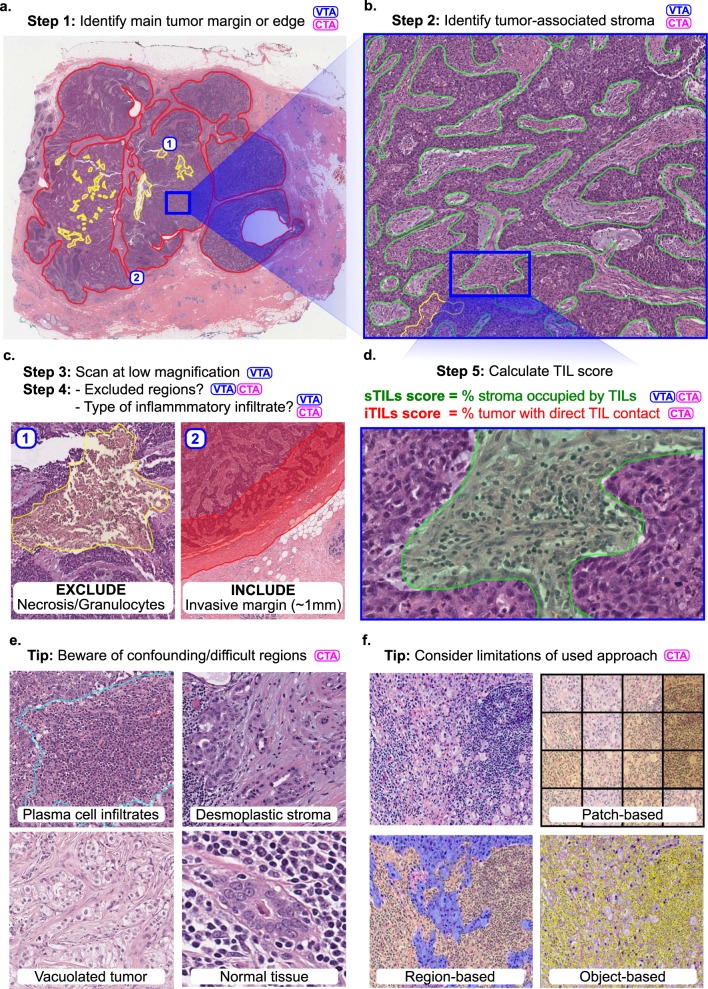
Fig. 1. Outline of the visual (VTA) and computational (CTA) procedure for scoring TILs in breast carcinomas.
TIL scoring is a complex procedure, and breast carcinomas are used as an example. Specific guidelines for scoring different tumors are provided in the references. Steps involved in VTA and/or CTA are tagged with these abbreviations. CTA according to TIL-WG guidelines involves TIL scoring in different tissue compartments. a Invasive edge is determined (red) and key confounding regions like necrosis (yellow) are delineated. b Within the central tumor, tumor-associated stroma is determined (green). Other considerations and steps are involved depending on histologic subtype, slide quality, and clinical context. c Determination of regions for inclusion or exclusion in the analysis in accordance with published guidelines. d Final score is estimated (visually) or calculated (computationally). In breast carcinomas, stromal TIL score (sTIL) is used clinically. Intratumoral TIL score (iTIL) is subject to more VTA variability, which has hampered the generation of evidence demonstrating prognostic value; perhaps CTA of iTILs will prove less variable and, consequently, prognostic. e The necessity of diverse pathologist annotations for robust analytical validation of computational models. Desmoplastic stroma may be misclassified as tumor regions; Vacuolated tumor may be misclassified as stroma; intermixed normal acini or ducts, DCIS/LCIS, and blood vessels may be misclassified as tumor; plasma cells are sometimes misclassified as carcinoma cells. Note that while the term “TILs” includes lymphocytes, plasma cells and other small mononuclear infiltrates, lumping these categories may not be optimal from an algorithm design perspective; plasma cells tend to be morphologically different from lymphocytes in nuclear texture, size, and visible cytoplasm. f Various computational approaches may be used for computational scoring. The more granular the algorithm is, the more accurate/useful it is likely to be, but—as a trade-off—the more it relies on exhaustive manual annotations from pathologists. The least granular approach is patch classification, followed by region delineation (segmentation), then object detection (individual TILs). A robust computational scoring algorithm likely utilizes a combination of these (and related) approaches.