Abstract
Background
Noncardiac organ failure has been associated with worse outcomes among a cardiac intensive care unit (CICU) population.
Hypothesis
We hypothesized that early organ failure based on the sequential organ failure assessment (SOFA) score would be associated with mortality in CICU patients.
Methods
Adult CICU patients from 2007 to 2015 were reviewed. Organ failure was defined as any SOFA organ subscore ≥3 on the first CICU day. Organ failure was evaluated as a predictor of hospital mortality and postdischarge survival after adjustment for illness severity and comorbidities.
Results
We included 10 004 patients with a mean age of 67 ± 15 years (37% female). Admission diagnoses included acute coronary syndrome in 43%, heart failure in 46%, cardiac arrest in 12%, and cardiogenic shock in 11%. Organ failure was present in 31%, including multiorgan failure in 12%. Hospital mortality was higher in patients with organ failure (22% vs 3%, adjusted OR 3.0, 95% CI 2.5‐3.7, P < .001). After adjustment, each failing organ system predicted twofold higher odds of hospital mortality (adjusted OR 1.9, 95% CI 1.1‐2.1, P < .001). Mortality risk was highest with cardiovascular, coagulation and liver failure. Among hospital survivors, organ failure was associated with higher adjusted postdischarge mortality risk (P < .001); multiorgan failure did not confer added long‐term mortality risk.
Conclusions
Early noncardiovascular organ failure, especially multiorgan failure, is associated with increased hospital mortality in CICU patients, and this risk continues after hospital discharge, emphasizing the need to promote early recognition of organ failure in CICU patients.
Keywords: cardiac critical care, cardiac intensive care unit, mortality, organ failure, sequential organ failure assessment (SOFA) score
Abbreviations
- APACHE
acute physiology and chronic health evaluation
- BMI
body mass index
- CCI
Charlson comorbidity index
- CI
confidence interval
- CICU
cardiac intensive care unit
- IABP
intra‐aortic balloon pump
- LOS
length of stay
- OASIS
Oxford Acute Severity of Illness Score
- OR
odds ratio
- PAC
pulmonary artery catheter
- PCI
percutaneous coronary intervention
- SOFA
sequential organ failure assessment
1. INTRODUCTION
The contemporary cardiac intensive care unit (CICU) serves an increasingly complex patient population with acute and chronic multiorgan dysfunction and superimposed cardiac pathology.1, 2, 3, 4, 5 Acute cardiovascular diseases remain an important focus of CICU care, yet noncardiovascular disease processes including extracardiac organ failure are becoming more prevalent in CICU patients.1, 4, 5 Organ failure, particularly multiorgan failure, is a final common pathway of critical illness and an important driver of mortality among CICU patients. In particular, acute kidney injury, and acute respiratory failure are increasingly prevalent and have been associated with higher mortality in CICU populations, emphasizing the importance of early recognition and management of these concomitant acute noncardiovascular illnesses.1, 4
Organ failure can be quantified using the sequential organ failure assessment (SOFA) score, an illness severity score initially described in patients with sepsis.6, 7, 8 The SOFA score is calculated based on a four‐point functional assessment for each of six organ systems (central nervous, cardiovascular, respiratory, renal, liver, and coagulation); individual SOFA organ subscores ≥3 reflect failure of that organ system.6, 7, 8, 9, 10, 11, 12, 13 The number and pattern of failing organ systems are associated with mortality in critically ill patients (especially among patients with sepsis).10, 14 In recent studies, the SOFA score has been examined for prediction of short‐term mortality in CICU populations.9, 13, 15, 16 The SOFA score on the first CICU day has very good discrimination for hospital mortality in our CICU population, and even minimal organ dysfunction (total SOFA score ≥ 2 on the first CICU day) was associated with higher in‐hospital and postdischarge mortality.13 If simple SOFA‐based definitions of organ failure provide useful mortality risk stratification among CICU patients, this could justify their more frequent use.
To the best of our knowledge, no prior studies have explored the epidemiology or prognostic value of early organ failure in CICU patients. The purpose of this study was to evaluate the short‐ and long‐term mortality associated with organ failure on the first CICU day among unselected CICU patients.
2. METHODS
This study (IRB # 16‐000722) was approved by the Mayo Clinic Institutional Review Board under an exception from informed consent as posing minimal risk to patients. This was a historical cohort analysis of patients admitted to the CICU at the Mayo Clinic Hospital, St. Mary's Campus, a tertiary‐care hospital in Rochester, Minnesota.13, 17 This CICU is a closed 16‐bed ICU which cares for critically ill patients with primary cardiac diseases or significant cardiac comorbidities, but does not admit postoperative cardiac surgery patients. Unique adult patients ≥18 years old admitted to the CICU between January 1, 2007, and December 31, 2015, were included using data from the first CICU admission for each patient.18 Patients admitted prior to January 1, 2007, patients still hospitalized on December 31, 2015, and patients who did not provide Minnesota Research Authorization under Minnesota state law were excluded.
Demographic, clinical, and laboratory data and information relating to therapeutic interventions and procedures were collected. SOFA scores and individual organ subscores were electronically generated using the worst physiologic values from the first 24 hours of CICU admission.11, 12, 13, 16 Consistent with the established SOFA‐based definition, organ failure was defined as a SOFA organ subscore ≥ 3 for that organ system (Supplemental Table S1).10 The acute physiology and chronic health evaluation (APACHE) III score, APACHE IV predicted hospital mortality, and Oxford Acute Severity of Illness Score (OASIS) were generated using data from the first 24 hours of CICU admission.17, 19, 20, 21, 22 Missing data for calculating the SOFA, APACHE III/IV, and OASIS scores were imputed as normal (score of 0), as is customary for prognostic scoring systems.11, 12, 20 The Charlson comorbidity index (CCI) and individual comorbidities were determined using a previously validated electronic algorithm.23 Admission diagnoses were defined as all International Classification of Diseases (ICD)‐9 diagnosis codes recorded on the day of CICU admission and the day before or after CICU admission; these admission diagnoses were not mutually exclusive, and the primary admission diagnosis could not be determined.
The primary endpoint was all‐cause hospital mortality; the secondary endpoints were all‐cause CICU mortality and postdischarge mortality during long‐term follow‐up. Mortality data were extracted electronically from Mayo Clinic databases, the state of Minnesota electronic death certificates, and the Rochester Epidemiology Project database on February 1, 2018, using previously described methodology.24 Categorical variables are reported as number (%), and the chi‐squared test was used to compare groups. Continuous variables are reported as mean ± SD; Student's t test was used to compare two groups, and analysis of variance was used to compare more than two groups. Logistic regression was performed to assess organ failure as a predictor of hospital mortality before and after adjusting for age, gender, race, APACHE IV predicted hospital mortality, CCI, and admission diagnoses of ACS and heart failure (HF). Kaplan‐Meier analysis was performed to determine postdischarge survival in patients discharged alive from the hospital; groups were compared using the log‐rank test. Cox proportional‐hazards models were used to assess the association of organ failure with postdischarge survival, adjusting for age, gender, race, APACHE IV predicted hospital mortality, CCI, and admission diagnoses of ACS and HF. P values <.05 were considered statistically significant. Statistical analyses were performed using JMP version 13.0 Pro (SAS Institute, Cary, North Carolina).
3. RESULTS
3.1. Study population
We screened 12 904 adult admissions to the CICU during the study period; 2900 patients were excluded (1877 readmissions, 755 patients without Minnesota Research Authorization and 268 patients admitted before or still hospitalized after the study period), yielding an overall study population of 10 004 patients.13 Day 1 SOFA score data were available for 9989 (99.9%) patients. Day 1 SOFA organ subscore data were available for >90% of patients for the cardiovascular, coagulation, central nervous system, and renal subscores, but fewer than one‐third of patients had available data to calculate the liver or respiratory subscores (Supplemental Table S1). Patients who did not have data available to calculate an individual SOFA subscore were considered not to have failure of that organ system. The mean age was 67 ± 15 years, and 3746 (37%) patients were female. Admission diagnoses included acute coronary syndrome (ACS) in 4267 (43.1%) patients, HF in 4564 (46.1%) patients, cardiogenic shock in 1078 (10.9%) patients, cardiac arrest in 1193 (12.0%) patients, and sepsis in 605 (6.1%) patients.
3.2. Prevalence of organ failure
Early organ failure (any SOFA organ subscore ≥ 3 on the first CICU day) was present in 3102 (31.0%) patients, with a mean of 1.6 failing organ systems. The most common failing organ systems were respiratory and cardiovascular, followed by renal (Supplemental Table S1); coagulation and liver failure were uncommon (fewer than 1% of patients). The majority of patients with organ failure had single‐organ failure (60.3%) or two‐organ failure (25.3%), while only 14.3% had failure of >2 organs. The majority of patients with central nervous system (91.3%) or cardiovascular (74.9%) failure had multiorgan failure. Noncardiovascular organ failure was present in 2820 (28.2%) patients, including 2029 (20.3%) with noncardiovascular single organ failure and 791 (7.9%) with noncardiovascular multiorgan failure.
Patients with single‐organ and multiorgan failure were older and expectedly had greater overall illness severity, higher rates of comorbidities, different rates of therapeutic interventions, and longer LOS than patients without organ failure (Table 1). The majority of patients with multiorgan failure received catecholamines and mechanical ventilation on the first CICU day. Patients with organ failure (especially multiorgan failure) more frequently had admission diagnoses of cardiogenic shock, cardiac arrest, and sepsis; the prevalence of organ failure was likewise higher in patients with these admission diagnoses (Supplemental Figure S1).
Table 1.
Baseline characteristics and provided therapies for patients without early organ failure and with one or more than one failing organ system on the first CICU day. Data represented as number (% of total) for categorical variables and mean ± SD for continuous variables. P value is for chi‐squared test (categorical variables) or analysis of variance (continuous variables) between the three groups
| No organ failure N = 6902 (69.0%) | Single organ failure N = 1872 (18.7%) | Multiorgan failure N = 1230 (12.3%) | P value | |
|---|---|---|---|---|
| Baseline characteristics | ||||
| Age | 66.8 ± 15.3 | 68.9 ± 15.3 | 68.9 ± 14.2 | <.0001 |
| Female | 2514 (36.5%) | 765 (40.9%) | 459 (37.3%) | .0025 |
| BMI | 29.3 ± 6.8 | 29.8 ± 7.4 | 30.1 ± 7.9 | .0002 |
| Comorbidities | ||||
| CCI | 2.1 ± 2.5 | 3.0 ± 2.8 | 2.8 ± 2.8 | <.0001 |
| Myocardial infarction | 1345 (19.6%) | 406 (21.8%) | 224 (18.2%) | .0383 |
| Chronic heart failure | 151 (16.7%) | 518 (27.8%) | 284 (23.1%) | <.0001 |
| Stroke | 803 (11.7%) | 269 (14.4%) | 157 (12.8%) | .0059 |
| Chronic kidney disease | 1133 (16.5%) | 563 (30.2%) | 331 (27.0%) | <.0001 |
| Diabetes mellitus | 1792 (26.1%) | 621 (33.3%) | 417 (34.0%) | <.0001 |
| Cancer | 1404 (20.4%) | 444 (23.8%) | 282 (23.0%) | .0027 |
| Lung disease | 1221 (17.8%) | 455 (24.4%) | 267 (21.7%) | <.0001 |
| Prior dialysis | 165 (2.4%) | 243 (13.0%) | 162 (13.2%) | <.0001 |
| Admission diagnoses | ||||
| Acute coronary syndrome | 3036 (44.4%) | 692 (37.4%) | 539 (44.3%) | <.0001 |
| Heart failure | 2678 (39.2%) | 1106 (59.7%) | 780 (64.1%) | <.0001 |
| Cardiac arrest | 412 (6.0%) | 247 (13.3%) | 534 (43.9%) | <.0001 |
| Cardiogenic shock | 256 (3.8%) | 265 (14.3%) | 557 (45.8%) | <.0001 |
| Sepsis | 181 (2.6%) | 175 (9.4%) | 249 (20.5%) | <.0001 |
| Therapeutics in CICU | ||||
| Inpatient PCI | 2587 (37.6%) | 488 (26.1%) | 348 (28.3%) | <.0001 |
| Invasive ventilation day 1 | 59 (0.9%) | 421 (22.5%) | 917 (74.6%) | <.0001 |
| Catecholamines day 1 | 363 (5.3%) | 479 (25.6%) | 928 (75.4%) | <.0001 |
| >1 catecholamine day 1 | 20 (0.3%) | 93 (5.0%) | 463 (37.6%) | <.0001 |
| IABP in CICU | 393 (5.7%) | 191 (10.2%) | 281 (22.8%) | <.0001 |
| PAC in CICU | 372 (5.4%) | 140 (7.5%) | 209 (17.0%) | <.0001 |
| Blood transfusion in CICU | 499 (7.2%) | 316 (16.9%) | 358 (29.1%) | <.0001 |
| Dialysis in CICU | 211 (3.1%) | 113 (6.0%) | 162 (13.2%) | <.0001 |
| Severity of illness and LOS | ||||
| APACHE‐III score | 51.6 ± 16.9 | 70.0 ± 19.2 | 100.0 ± 30.5 | <.0001 |
| APACHE‐IV mortality (%) | 9.2 ± 9.1 | 22.0 ± 17.4 | 52.3 ± 26.7 | <.0001 |
| OASIS | 21.3 ± 6.9 | 29.4 ± 9.0 | 41.7 ± 9.9 | <.0001 |
| Day 1 total SOFA score | 1.9 ± 1.3 | 5.4 ± 1.6 | 10.1 ± 2.7 | <.0001 |
| CICU LOS | 2.1 ± 4.9 | 2.9 ± 3.3 | 3.8 ± 3.9 | <.0001 |
| Hospital LOS | 6.8 ± 13.1 | 10.1 ± 12.5 | 10.8 ± 13.6 | <.0001 |
| Failing organ systems | ||||
| Cardiovascular failure | N/A | 282 (15.1%) | 842 (68.5%) | <.0001 |
| Central nervous system failure | N/A | 54 (2.9%) | 566 (46.0%) | <.0001 |
| Coagulation failure | N/A | 40 (2.1%) | 51 (4.2%) | <.0001 |
| Liver failure | N/A | 16 (0.8%) | 29 (2.4%) | <.0001 |
| Renal failure | N/A | 650 (34.7%) | 392 (31.9%) | <.0001 |
| Respiratory failure | N/A | 830 (44.3%) | 1121 (91.1%) | <.0001 |
Abbreviations: APACHE, Acute Physiology and Chronic Health Evaluation; BMI, body mass index; CICU, cardiac intensive care unit; IABP, intra‐aortic balloon pump; LOS, length of stay; OASIS, Oxford Acute Severity of Illness Score; PAC, pulmonary artery catheter; PCI, percutaneous coronary intervention; SOFA, Sequential Organ Failure Assessment.
3.3. Mortality and organ failure
All‐cause hospital mortality occurred in 908 (9.1%) patients, including CICU mortality in 570 (5.7%) patients. Patients with organ failure had higher hospital mortality (22.3% vs 3.1%, P < .001), accounting for 76% of hospital deaths (Figure 1A). Patients with multiorgan failure had even higher hospital mortality (36.9% vs 5.2%, P < .001), accounting for 50% of all hospital deaths. There was a stepwise increase in CICU and hospital mortality as a function of the increasing number of failing organ systems (Figure 1B). Hospital mortality occurred in 23.0% of patients with any noncardiovascular organ failure and 40.8% of patients with noncardiovascular multiorgan failure, with a similar stepwise increase in CICU and hospital mortality as a function of increasing number of failing noncardiovascular organ systems.
Figure 1.

A, Cardiac intensive care unit (CICU) and hospital mortality as a function of the number of early failing organ systems. B, CICU and hospital mortality in patients with failure of each organ system
After multivariate adjustment (Table 2), the presence of organ failure was associated with more than threefold higher hospital mortality (adjusted OR 3.02, 95% CI 2.47‐3.68, P < .001) and each failing organ system was associated with a twofold higher risk of hospital mortality (adjusted OR 1.93, 95% CI 1.74‐2.14, P < .001). Hospital mortality among patients with single and multiorgan failure varied as a function of admission diagnosis (Figure 2). Patients with multiorgan failure had high hospital mortality regardless of admission diagnosis, but mortality was higher among patients with cardiogenic shock, cardiac arrest, and sepsis in the absence of multiorgan failure.
Table 2.
Logistic regression for prediction of hospital mortality before (unadjusted) and after adjusting for age, gender, race, CCI, APACHE‐III score, and admission diagnoses of ACS and HF. OR and 95% CI values are compared to patients without organ failure. *All P < .0001 except adjusted OR for central nervous system failure (P = .80)
| Unadjusted OR (95% CI) | Adjusted OR (95% CI) | |
|---|---|---|
| Any organ failure | 8.888 (7.576‐10.427) | 3.015 (2.473‐3.677) |
| Single organ failure | 4.509 (3.721‐5.463) | 2.675 (2.171‐3.294) |
| Multiorgan failure vs single‐organ failure |
18.110 (15.153‐21.643) 4.017 (3.360‐4.802) |
4.590 (3.536‐5.957) 1.716 (1.373‐3.145) |
| Each failing organ system | 3.109 (2.907‐3.324) | 1.927 (1.738‐2.137) |
| Cardiovascular failure | 8.629 (7.416‐10.040) | 2.704 (2.229‐3.280) |
| Central nervous system failure | 8.522 (7.123‐10.197) | 0.965 (0.746‐1.250)* |
| Coagulation failure | 4.562 (2.907‐7.158) | 2.907 (1.728‐4.888) |
| Liver failure | 6.793 (3.727‐12.383) | 3.983 (2.014‐7.874) |
| Renal failure | 4.624 (3.941‐5.427) | 1.968 (1.623‐2.386) |
| Respiratory failure | 6.735 (5.840‐7.768) | 1.714 (1.403‐2.094) |
| Unadjusted | Adjusted | |||
|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | |
| Any organ failure | 8.888 (7.576‐10.427) | <.0001 | 3.015 (2.473‐3.677) | <.0001 |
| Single organ failure | 4.509 (3.721–5.463) | <.0001 | 2.675 (2.171–3.294) | <.0001 |
| Multiorgan failure vs single‐organ failure |
18.110 (15.153‐21.643) 4.017 (3.360‐4.802) |
<.0001 <.0001 |
4.590 (3.536‐5.957) 1.716 (1.373‐3.145) |
<.0001 <.0001 |
| Each failing organ system | 3.109 (2.907‐3.324) | <.0001 | 1.927 (1.738‐2.137) | <.0001 |
| Cardiovascular failure | 8.629 (7.416‐10.040) | <.0001 | 2.704 (2.229‐3.280) | <.0001 |
| Central nervous system failure | 8.522 (7.123–10.197) | <.0001 | 0.965 (0.746–1.250) | .7896 |
| Coagulation failure | 4.562 (2.907–7.158) | <.0001 | 2.907 (1.728–4.888) | <.0001 |
| Liver failure | 6.793 (3.727–12.383) | <.0001 | 3.983 (2.014–7.874) | <.0001 |
| Renal failure | 4.624 (3.941–5.427) | <.0001 | 1.968 (1.623–2.386) | <.0001 |
| Respiratory failure | 6.735 (5.840–7.768) | <.0001 | 1.714 (1.403–2.094) | <.0001 |
Abbreviations: ACS, acute coronary syndrome; APACHE, Acute Physiology and Chronic Health Evaluation; CCI, Charlson comorbidity index; CI, confidence interval; HF, heart failure; OR, odds ratio.
Figure 2.
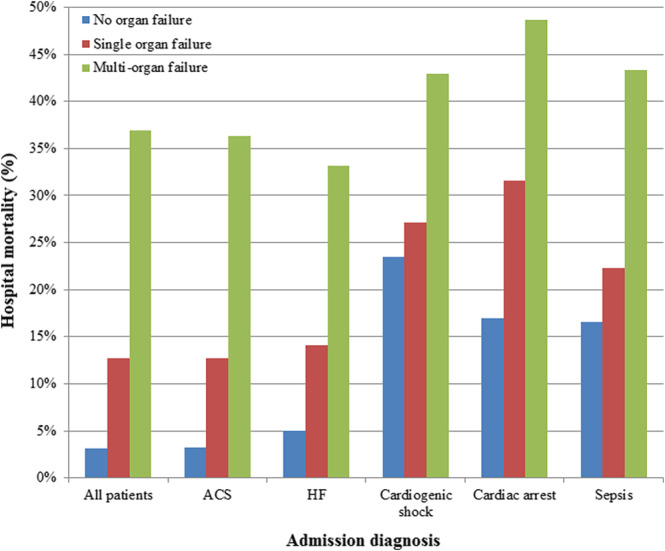
Hospital mortality in patients with single‐organ and multiorgan failure as a function of admission diagnosis
CICU and hospital mortality risk in patients with organ failure varied with the organ system involved (Figure 1), being highest for liver, central nervous system, and cardiovascular failure. Unadjusted OR values for hospital mortality were highest for central nervous system and cardiovascular failure (Table 2), while adjusted OR values were highest for liver and coagulation failure, followed by cardiovascular failure (Table 2). Hospital mortality varied among patients with each single‐organ failure and various combinations of two‐organ failure (Supplemental Figures S2 and S3); P < .001 between groups.
3.4. Postdischarge outcomes
Postdischarge mortality occurred in 3623 (39.8%) of the 9096 hospital survivors during a mean follow‐up of 3.4 ± 2.9 years; 1284 (14.1%) hospital survivors had follow‐up less than 1 year. Postdischarge survival was lower among the hospital survivors with single or multiorgan failure compared to those without organ failure (Figure 3; P < .001 by log‐rank), but was similar between patients with single‐ or multiorgan failure (P > .1 by log‐rank). Using Cox proportional‐hazards models, hospital survivors with organ failure (adjusted RR 1.23, 95% CI 1.13‐1.34, P < .001) had an increased risk of postdischarge mortality. Hospital survivors with organ failure were less likely to be discharged to home (59.0% vs 80.5%, P < .001) and more likely to be discharged to a skilled nursing facility (32.4% vs 15.4%, P < .001) (Supplemental Figure S4).
Figure 3.

Kaplan‐Meier survival curves for hospital survivors with 1 (green) or ≥2 (blue) organ failures compared to patients without organ failure (red). P < .001 by log‐rank for no organ failure vs either single or multiorgan failure; P > .1 by log‐rank for single vs multiorgan failure
4. DISCUSSION
We report the association of early organ failure with hospital and postdischarge mortality in a contemporary CICU population using SOFA‐based organ failure definitions. Organ failure was present in nearly one‐third of patients, the majority of which was noncardiovascular organ failure. Early organ failure was associated with increased mortality at all time points, with a stepwise increase in hospital mortality as the number of failing organ systems increased. Organ failure remained associated with higher hospital mortality after adjustment for the APACHE IV predicted mortality, implying that early organ failure is associated with adverse outcomes beyond standard measures of illness severity. Patients admitted with cardiac arrest, cardiogenic shock, or sepsis had a higher prevalence of organ failure, and the strength of the association between organ failure and hospital mortality differed depending on the admission diagnosis. The hospital mortality risk varied with specific single organ and multiorgan failures, emphasizing the association between particular patterns of organ failure and outcomes in this CICU population. Among hospital survivors, the presence of early organ failure was associated with decreased postdischarge survival, although unexpectedly the presence of multiorgan failure was not worse than single‐organ failure. These results emphasize the importance of organ failure, especially noncardiovascular organ failure, as a contributor to adverse outcomes among CICU patients and validate the utility of simple SOFA‐based organ failure definitions for risk stratification in this population.
In a recent CICU study, acute respiratory failure and acute kidney injury (based on ICD‐9 discharge diagnosis codes) were both independent predictors of hospital mortality after adjusting for illness severity using the OASIS.4 Both respiratory failure and renal failure were associated with hospital mortality after adjusting for illness severity in this study, but the risk was lower than with cardiovascular failure. The total SOFA score and individual SOFA subscores on the first CICU day can predict hospital mortality in CICU patients as in general ICU populations.9, 13, 15, 16 Patients with SOFA scores <2 on the first CICU day (who are free from organ failure) have a very low risk of mortality during short‐ and long‐term follow‐up.13 The present study expands on our prior work by elucidating the association between the number and pattern of organ failures with hospital and postdischarge mortality.13, 16 Unexpectedly, postdischarge mortality did not differ for patients with multiorgan failure compared to single‐organ failure, potentially reflecting survivor bias given the high hospital mortality rate in this group.
We used the same SOFA‐based organ failure definition as in the seminal study by Moreno et al that established the relationship between organ failure and mortality in critically ill patients.10 As in our unadjusted analyses, cardiovascular and central nervous system failure were of primary importance for predicting mortality in the study by Moreno et al.10 Notably, the SOFA score was initially developed based on variables that are relevant for patients with sepsis, as opposed to CICU patients.6, 7, 8, 9 We observed a stepwise increase in hospital mortality as a function of increasing number of organ failures, similar to a recent study of nearly 1 million patients with sepsis by Shankar‐Hari et al.14 As in our study, the mortality risk varied with individual organ failures and specific combinations of organ failure.14 A stepwise increase in hospital mortality associated with an increasing number of acute organ failures was observed among patients with cardiogenic shock due to acute myocardial infarction in the Nationwide Inpatient Sample using ICD‐9 discharge diagnoses to define organ failure.25 We replicated these results in our broader CICU population using a more objective SOFA‐based definition of organ failure near the time of CICU admission.
In prior studies, the cardiovascular, central nervous system and renal SOFA subscores had the greatest discrimination for hospital mortality.10, 13, 16, 26, 27 Similarly, cardiovascular and central nervous system failure had the strongest association with mortality in our unadjusted analyses, followed by respiratory and liver failure. Studies in general critically ill patients have demonstrated that the central nervous system SOFA subscore (ie, the Glasgow Coma Scale) carries the greatest prognostic value for short‐term mortality.26, 27 Toma et al demonstrated that the central nervous system and cardiovascular failure SOFA organ subscores were most important for predicting mortality.27 In our study, cardiovascular failure remained a strong predictor of mortality even after adjustment, emphasizing the importance of shock as a determinant of outcomes beyond standard measures of illness severity. We observed an important discrepancy in the associations between central nervous system failure and mortality when comparing adjusted and unadjusted analyses: central nervous system failure had the highest unadjusted OR for hospital mortality, yet was not significantly associated with mortality after adjustment for APACHE‐IV. Rather than excluding a meaningful association between central nervous system failure and mortality in this population, we believe that this paradoxical finding reflects the ability of the APACHE score to effectively account for central nervous system failure.19, 22 The least frequent organ failures (coagulation and liver) had the strongest adjusted association with mortality, differing from our prior study which concluded that the coagulation and liver SOFA subscores contributed the least to mortality risk stratification.16
The association between liver and coagulation system failure and mortality has not been explored in prior studies of CICU patients, and our study highlights the importance of noncardiovascular organ failure among CICU patients. The strong association between coagulation and liver failure and mortality in CICU patients is presumably multifactorial. Coagulation and liver failure were uncommon, suggesting that patients with these specific organ failures were outliers who likely differed from other patients in meaningful ways that contributed to worse outcomes. Notably, the APACHE scoring systems do not incorporate platelet count, although they do include bilirubin, and therefore might not adequately account for the mortality risk conferred by the extreme abnormalities in these variables that characterize liver and coagulation failure.19, 22 Finally, it is probable that the underlying disease processes producing liver and coagulation failure were themselves associated with adverse outcomes. Sepsis was approximately twice as prevalent in patients with liver and coagulation failure, and the liver and coagulation SOFA subscores were most strongly predictive of mortality in a recent study of patients with sepsis.28
As with all retrospective single‐center cohort studies, this study has a number of limitations despite its large sample size, including the potential for unmeasured confounding and bias due to local practice styles. Our patient population may be distinct from other centers, as reflected by a lower hospital mortality rate and a lower rate of acute coronary syndromes.2, 3, 4, 15 Data for calculating each individual SOFA subscore were not present for all patients, and data for the respiratory and liver subscores were available in fewer than one‐third of patients. Although these missing data could have potentially influenced our results, our clinical experience is that patients with or at risk for significant organ dysfunction would usually have had laboratory testing to allow calculation of these individual SOFA subscores, minimizing this effect. SOFA‐based organ failure definitions cannot distinguish acute from chronic organ failure and do not consider other potential markers of organ dysfunction.7 This study did not evaluate the development of organ failure after the first CICU day. To adjust for illness severity and comorbidities, we used the APACHE IV predicted mortality, which shares some variables with the SOFA score and could have influenced our adjusted mortality results particularly as it relates to central nervous system failure.6, 7, 8, 17, 19, 22 We could not determine the primary admission diagnosis or specific indication for CICU admission. Postdischarge mortality was adjudicated using electronic review of health records for notification of patient death, which may be less accurate than methods such as the Social Security Death Index.
5. CONCLUSIONS
Early organ failure based on the SOFA score is common in CICU patients and associated with higher hospital and postdischarge mortality. The number and pattern of failing organs are associated with short‐term mortality. Our findings emphasize the importance of organ failure as a predictor of mortality in CICU patients and highlight the persistent long‐term mortality hazard faced by patients who develop organ failure during their early CICU course. Patients with noncardiovascular organ failure, particularly less‐frequent organ failure such as liver and coagulation, fared the worst. These simple and clinically relevant SOFA‐based definitions of organ failure can be easily applied at the bedside by CICU practitioners and may be useful endpoints for future clinical trials in CICU populations.
CONFLICT OF INTEREST
The authors declare that there are no relevant financial disclosures or conflicts of interest related to this work.
Supporting information
Appendix S1 Supporting Information
ACKNOWLEDGMENTS
The authors would like to acknowledge the dedicated physicians and nurses who care for our CICU patients every day. This research did not receive any specific grant from funding agencies in the public, commercial, or not‐for‐profit sectors.
Jentzer JC, Wiley B, Bennett C, et al. Early noncardiovascular organ failure and mortality in the cardiac intensive care unit. Clinical Cardiology. 2020;43:516–523. 10.1002/clc.23339
REFERENCES
- 1. Jentzer JC, van Diepen S, Barsness GW, et al. Changes in comorbidities, diagnoses, therapies and outcomes in a contemporary cardiac intensive care unit population. Am Heart J. 2019;215:12‐19. [DOI] [PubMed] [Google Scholar]
- 2. Katz JN, Shah BR, Volz EM, et al. Evolution of the coronary care unit: clinical characteristics and temporal trends in healthcare delivery and outcomes. Crit Care Med. 2010;38(2):375‐381. [DOI] [PubMed] [Google Scholar]
- 3. Casella G, Cassin M, Chiarella F, et al. Epidemiology and patterns of care of patients admitted to Italian intensive cardiac care units: the BLITZ‐3 registry. J Cardiovasc Med (Hagerstown). 2010;11(6):450‐461. [DOI] [PubMed] [Google Scholar]
- 4. Holland EM, Moss TJ. Acute noncardiovascular illness in the cardiac intensive care unit. J Am Coll Cardiol. 2017;69(16):1999‐2007. [DOI] [PubMed] [Google Scholar]
- 5. Goldfarb M, van Diepen S, Liszkowski M, Jentzer JC, Pedraza I, Cercek B. Noncardiovascular disease and critical care delivery in a contemporary cardiac and medical intensive care unit. J Intensive Care Med. 2017;34(7):537‐543. [DOI] [PubMed] [Google Scholar]
- 6. Keegan MT, Gajic O, Afessa B. Severity of illness scoring systems in the intensive care unit. Crit Care Med. 2011;39(1):163‐169. [DOI] [PubMed] [Google Scholar]
- 7. Vincent JL, Moreno R. Clinical review: scoring systems in the critically ill. Crit Care. 2010;14(2):207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Vincent JL, Moreno R, Takala J, et al. The SOFA (sepsis‐related organ failure assessment) score to describe organ dysfunction/failure. On behalf of the working group on sepsis‐related problems of the European Society of intensive care medicine. Intensive Care Med. 1996;22(7):707‐710. [DOI] [PubMed] [Google Scholar]
- 9. Minne L, Abu‐Hanna A, de Jonge E. Evaluation of SOFA‐based models for predicting mortality in the ICU: a systematic review. Crit Care. 2008;12(6):R161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Moreno R, Vincent JL, Matos R, et al. The use of maximum SOFA score to quantify organ dysfunction/failure in intensive care. Results of a prospective, multicentre study. Working group on sepsis related problems of the ESICM. Intensive Care Med. 1999;25(7):686‐696. [DOI] [PubMed] [Google Scholar]
- 11. Harrison AM, Yadav H, Pickering BW, Cartin‐Ceba R, Herasevich V. Validation of computerized automatic calculation of the sequential organ failure assessment score. Crit Care Res Pract. 2013;2013:975672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Aakre C, Franco PM, Ferreyra M, Kitson J, Li M, Herasevich V. Prospective validation of a near real‐time EHR‐integrated automated SOFA score calculator. Int J Med Inform. 2017;103:1‐6. [DOI] [PubMed] [Google Scholar]
- 13. Jentzer JC, Bennett C, Wiley BM, et al. Predictive value of the sequential organ failure assessment score for mortality in a contemporary cardiac intensive care unit population. J Am Heart Assoc. 2018;7(6):e008169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Shankar‐Hari M, Harrison DA, Rowan KM. Differences in impact of definitional elements on mortality precludes international comparisons of sepsis epidemiology‐a cohort study illustrating the need for standardized reporting. Crit Care Med. 2016;44(12):2223‐2230. [DOI] [PubMed] [Google Scholar]
- 15. Argyriou G, Vrettou CS, Filippatos G, Sainis G, Nanas S, Routsi C. Comparative evaluation of acute physiology and chronic health evaluation II and sequential organ failure assessment scoring systems in patients admitted to the cardiac intensive care unit. J Crit Care. 2015;30(4):752‐757. [DOI] [PubMed] [Google Scholar]
- 16. Jentzer JC, Bennett C, Wiley BM, Murphree DH, Keegan MT, Barsness GW. Predictive value of individual sequential organ failure assessment sub‐scores for mortality in the cardiac intensive care unit. PLoS One. 2019;14(5):e0216177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bennett CE, Wright RS, Jentzer J, et al. Severity of illness assessment with application of the APACHE IV predicted mortality and outcome trends analysis in an academic cardiac intensive care unit. J Crit Care. 2019;50:242‐246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Herasevich V, Pickering BW, Dong Y, Peters SG, Gajic O. Informatics infrastructure for syndrome surveillance, decision support, reporting, and modeling of critical illness. Mayo Clin Proc. 2010;85(3):247‐254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Knaus WA, Wagner DP, Draper EA, et al. The APACHE III prognostic system. Risk prediction of hospital mortality for critically ill hospitalized adults. Chest. 1991;100(6):1619‐1636. [DOI] [PubMed] [Google Scholar]
- 20. Chandra S, Kashyap R, Trillo‐Alvarez CA, et al. Mapping physicians' admission diagnoses to structured concepts towards fully automatic calculation of acute physiology and chronic health evaluation score. BMJ Open. 2011;1(2):e000216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Johnson AE, Kramer AA, Clifford GD. A new severity of illness scale using a subset of acute physiology and chronic health evaluation data elements shows comparable predictive accuracy. Crit Care Med. 2013;41(7):1711‐1718. [DOI] [PubMed] [Google Scholar]
- 22. Zimmerman JE, Kramer AA, McNair DS, Malila FM. Acute physiology and chronic health evaluation (APACHE) IV: hospital mortality assessment for today's critically ill patients. Crit Care Med. 2006;34(5):1297‐1310. [DOI] [PubMed] [Google Scholar]
- 23. Singh B, Singh A, Ahmed A, et al. Derivation and validation of automated electronic search strategies to extract Charlson comorbidities from electronic medical records. Mayo Clin Proc. 2012;87(9):817‐824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Rocca WA, Yawn BP, St Sauver JL, Grossardt BR, Melton LJ III. History of the Rochester epidemiology project: half a century of medical records linkage in a US population. Mayo Clin Proc. 2012;87(12):1202‐1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Vallabhajosyula S, Dunlay SM, Prasad A, et al. Acute noncardiac organ failure in acute myocardial infarction with cardiogenic shock. J Am Coll Cardiol. 2019;73(14):1781‐1791. [DOI] [PubMed] [Google Scholar]
- 26. Knox DB, Lanspa MJ, Pratt CM, Kuttler KG, Jones JP, Brown SM. Glasgow coma scale score dominates the association between admission sequential organ failure assessment score and 30‐day mortality in a mixed intensive care unit population. J Crit Care. 2014;29(5):780‐785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Toma T, Abu‐Hanna A, Bosman RJ. Discovery and integration of univariate patterns from daily individual organ‐failure scores for intensive care mortality prediction. Artif Intell Med. 2008;43(1):47‐60. [DOI] [PubMed] [Google Scholar]
- 28. Gupta T, Puskarich MA, DeVos E, et al. Sequential organ failure assessment component score prediction of in‐hospital mortality from sepsis. J Intensive Care Med. 2018; [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1 Supporting Information


