Figure 1.
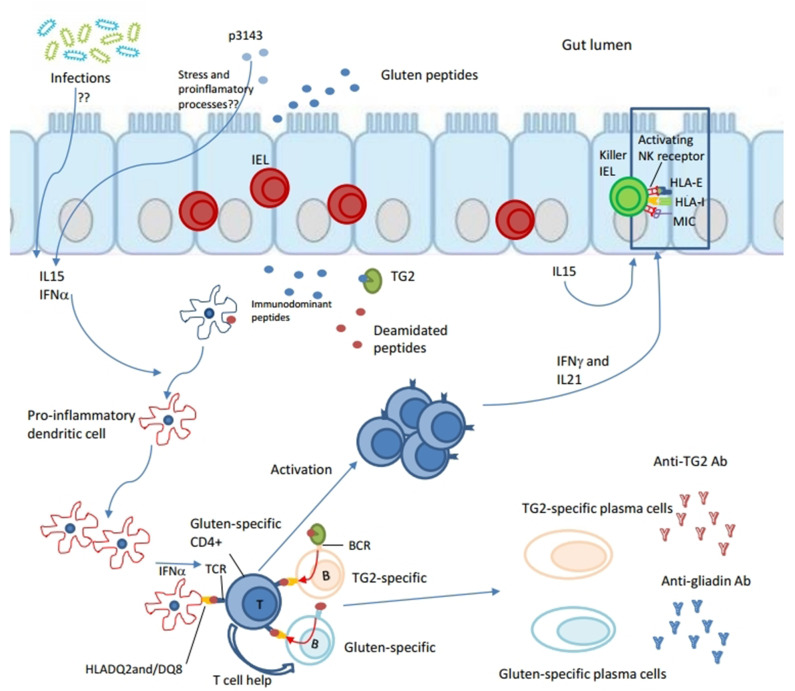
Both innate and adaptive immune responses are induced in the pathogenesis of celiac disease. Gliadin peptides resulting from the partial degradation of gluten in the intestinal lumen cross the epithelial barrier through the transepithelial way or passively by paracellular flux gaining access to lamina propria. Some gliadin peptides, such as p31–43, are thought to induce epithelial stress and inflammation. Other, the immunodominant ones such as the 33-mer, are deamidated by tissue transglutaminase 2 (TG2), resulting in higher affinity for HLA-DQ2 or DQ8 molecules. Deamidated gliadin-peptides are taken up by antigen presenting cells (APCs), such as pro-inflammatory dendritic cells, which promote activation of gluten-specific CD4+ T cell responses in lamina propria. Because of an already inflamed environment (by virus, gliadin? eliciting IL15 and type 1 interferons response), gluten-specific CD4+ T cells express a Th1 phenotype dominated by interferon (IFN)-γ and IL-21. Furthermore, gluten-specific T cells are thought to give help to both TG2-specific and gluten-specific B cells to differentiate into secreting anti-TG2 and anti-gliadin antibodies plasma cells, respectively. In particular, B cell recognizes its antigen (TG2-gliadin complex or gliadin peptides) by BCR, internalizes it, processes and presents gliadin in the context of HLA-DQ2 or DQ8 molecules to gliadin-specific CD4+ T cell. Because of their interaction both T cell and B cell would be activated producing pro-inflammatory cytokines, the first one, and differentiating in secreting plasma cell, the second. Other cytokines, such as IL15 and INF α are thought to be produced by stressed intestinal epithelial cells and/or dendritic cells. They are involved into innate immune responses. IL15 seems to have a central role in the recruitment of intraepithelial lymphocytes (IELs) and in their licensing to become pathogenic killer cells. In fact, IL15 seems involved in the expression of activating natural killer (NK) receptors CD94 and NKG2D, as well as in epithelial expression of stress molecules such as HLA-E and MIC. Finally, a complex remodeling of small intestinal mucosa takes place downstream of T-cell activation that leads to the classical “flat mucosa” of celiac disease.