Abstract
Background
Head and neck squamous cell carcinoma (HNSC) ranks as the sixth most common malignancy. The identification of highly specific and sensitive prognostic markers and potential drug targets can contribute to enhanced patient prognosis and individualized treatments. Heat shock proteins (HSPs) act as molecular chaperones and play a crucial role in maintaining cell homeostasis. Recently, research has indicated that HSPs also act as “evil chaperones” in cancer development.
Methods
In this study, we assessed the expression of HSPs in HNSC patients using the ONCOMINE, GEPIA, and UALCAN databases. Mutations of HSP genes were also analysed using the cBioPortal database. Additionally, the expression levels of HSPs were verified using the Human Protein Altas (THPA) database.
Results
We found that the expression levels of HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 were significantly higher in tissues from HNSC patients compared with normal tissues. Moreover, HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 expressions were linked to disease progression. Survival analysis with the GEPIA and OncoLnc databases indicated that upregulation of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 was related to poor overall survival (OS).
Conclusion
This study suggests that the HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 genes are potential clinical targets and prognostic biomarkers for patients with HNSC.
Keywords: Biomarkers, Prognosis, Expression, Heat shock proteins, HNSC
Background
Head and neck squamous cell carcinoma (HNSC) commonly occurs in the oral cavity, larynx, and pharynx and ranks as the sixth most common malignancy. In 2018, there was an estimated 43,000 HNSC-associated deaths worldwide and 835,000 new cases [1, 2]. Unfortunately, diagnosis of HNSC is usually made at an advanced disease stage as the clinical symptoms of HNSC are not obvious during the early stage. As a result, the current 5-year survival rate is less than 65% [3]. It is generally understood that the accumulation of genetic mutations in epithelial cells plays a key causal role in the development and progression of HNSC [4]. Hence, the identification of highly specific and sensitive prognostic markers and potential drug targets can contribute to enhanced patient prognosis and individualised treatments.
Heat shock proteins (HSPs) are genetically highly conserved proteins that act as molecular chaperones and play a crucial role in maintaining cell homeostasis [5]. Aside from their cytoprotective effects, recent research has suggested that HSPs also act as “evil chaperones” in the development, progression, metastasis and drug resistance of cancers [6, 7]. Therefore, HSPs have recently been proposed as potential therapeutic targets for tumor therapy [8]. HSPH1 (also named HSP105), a member of the HSP70 superfamily, is a component of the β-catenin degradation complex. Previous studies have demonstrated that overexpression of HSPH1/HSP105 in various cancers is associated with increased levels of nuclear β-catenin protein and upregulation of Wnt target genes [9]. HSPD1 is a molecular chaperone primarily localised in the mitochondrial matrix. It has been described as a potential prognostic and diagnostic biomarker for cancer. Recent studies have demonstrated that HSPD1 not only regulates the stability of survivin protein, but also regulates the mRNA expression of survivin [10]. SERPINH1, also called heat shock protein 47 (HSP47), is a collagen specific molecular chaperone. Several studies have confirmed that SERPINH1 participates in numerous steps of collagen synthesis, blocking the aggregation of procollagen and inducing the hydroxylation of proline and lysine residues. Abnormal expression levels of SERPINH1 are frequently found in a variety of cancers, including cervical, lung and gastric cancers [11–13]. Heat shock protein A4 (HSPA4), a member of the HSP110 family, is widely expressed in a variety of organs and can be induced under different conditions, including carcinogenic stress [14–16]. Recent studies have indicated that knockdown of HSPA4 can significantly reduce the migration, invasion, and transformation activities of tumor cells [17]. Heat shock protein 90α (Hsp90α) is the major cytosolic chaperone in eukaryotes. It is involved in cell protection and intracellular signaling transduction, controls intracellular homeostasis and assemblies of endoplasmic reticulum-secreted peptides, and regulates the translocation of proteins across the membranes of organelles after translation. Upregulated expression of Hsp90α is observed in a variety of cancer tissues, including liver, breast, and pancreatic cancers [18–20]. However, there is limited understanding of the underlying mechanisms and the unique roles of these genes in HNSC.
Although some studies have reported dysregulated expression of HSPs in HNSC and have linked this to patients’ prognosis [21, 22], the overall HSP expression profiles and the prognostic relevance of these expression profiles remain unknown. In the current study, we assessed the expression levels and mutations of HSPs in HNSC patients. The aim was to assess the potential functions, patterns of expression, and prognostic relevance of these genes in HNSC. To achieve this goal, we analysed large datasets available in various public databases.
Methods
Ethics statement
Our study was approved by the Academic Committee of Huazhong University of Science and Technology, and conducted in accordance with the principles expressed in the Declaration of Helsinki. All datasets used in this study were extracted from online public databases or published literature, and thus, written informed consent was not required for the current data analysis.
ONCOMINE analysis
The expression patterns of HSPs in different cancers can be assessed using the online ONCOMINE database (https://www.oncomine.org). ONCOMINE is an integrated data-mining platform that store previously published or open-access cancer microarray data. The results were filtered by selecting HNSC vs. normal tissue.
GEPIA dataset
GEPIA is an online database that incorporates gene expression data from TCGA and GTEx, bringing together 9736 tumor samples and 8587 normal controls. It allows for the assessment of differential expression profiles, patient outcomes, and various other analyses [23]. Through these public bioinformatics platforms, we are able to analyse the expression profiles of HSP genes in HNSC.
UALCAN dataset
The UALCAN database (http://ualcan.path.uab.edu/index.html) is a user-friendly interactive platform for facilitating tumor subgroup gene expression and survival analyses [24]. Using the UALCAN database, we can evaluate the expression levels of HSP genes in HNSC and normal tissues based on tumor stages in the Cancer Genome Atlas (TCGA) HNSC datasets.
The Human Protein Atlas (THPA) database
The Human Protein Atlas (THPA) (https://www.proteinatlas.org) is a public database that can be used to validate the expression of target genes. It contains immunohistochemical expression data for near 20 common kinds of cancers. In this study, we compared the protein expression levels of different HSPs between normal and HNSC tissues using immunohistochemistry images.
Prognostic analysis
Using the GEPIA (http://gepia.cancer-pku.cn) database and OncoLnc (http://www.oncolnc.org) database, we analyzed the relationship between HSP gene expressions and overall survival in HNSC patients using a cox p value threshold of < 0.05.
TCGA data and cBioPortal
The Cancer Genome Atlas (TCGA) database contains sequencing and clinicopathologic data for 30 cancer types [25]. The Head and Neck Squamous Cell Carcinoma (TCGA, Provisional) was selected for further analysis of HSP genes using cBioPortal (https://www.cbioportal.org). Genomic profiles, including mutations and copy-number alterations (CNA) were calculated using the cBioPortal’s online tool.
Protein–protein interaction (PPI) network construction
In order to better understand the molecular mechanisms of HSPs in tumorigenesis, the PPI network for HSP family genes was constructed using STRING database. We choose a minimum interaction score of 0.4 as a cut-off when visualizing this PPI network.
Functional and pathway enrichment analysis
The online STRING (https://string-db.org/) tool provides investigators with systematic and comprehensive functional annotation tools to identify the biological meaning behind an extensive list of genes. In our study, Gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) enrichment analyses were conducted for HSP family genes using STRING. The significance threshold was p < 0.05.
Results
The expression levels of HSPs in HNSC patients
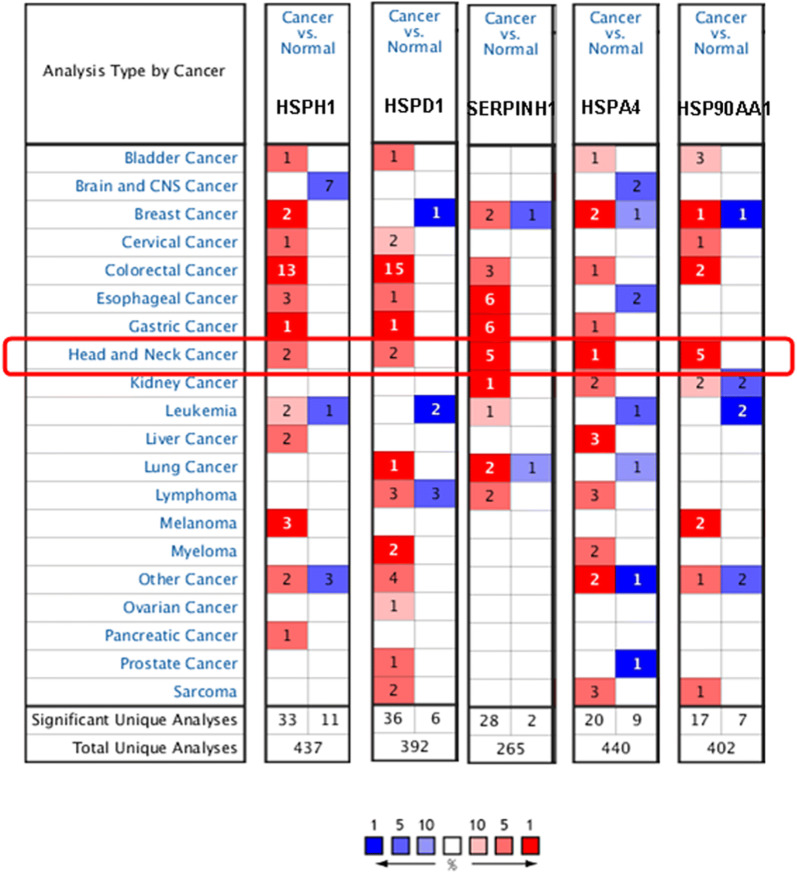
In order to explore the prognostic and potential therapeutic values of different HSP members in HNSC, we analyzed the mRNA and protein expressions of HSPs in HNSC patients using the ONCOMINE database. The expressions of five HSPs members (HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1) in more than 20 types of cancers were detected and compared with expressions in normal tissues using the ONCOMINE database. As were shown in Fig. 1, the expression levels of HSPs were significantly upregulated in HNSC patients. Interestingly, the expression levels of these five HSP genes were significantly upregulated in most tumors. In addition, we searched various HNSC datasets and found that these five genes showed significant upregulation across all datasets (Additional file 1: Fig. S1, Table 1). We further used the UALCAN.database to validate our findings. The results also showed that the expression levels of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 in HNSC were significantly increased (Additional file 2: Fig. S2).
Fig. 1.
The expression levels of HSPs in different types of cancers (ONCOMINE). The expression levels of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in different types of cancers. Red: over-expression; Blue: down-regulated expression
Table 1.
The significant changes of HSP associated genes expression levels between different types of Head and neck cancer and normal tissues (ONCOMINE database)
| Gene | Types of head and neck cancer vs. normal | Fold change | p value | T test | Reference (PMID) |
|---|---|---|---|---|---|
| HSPH1 | Tongue carcinoma vs. normal | 2.902 | 5.56E−7 | 6.528 | Pyeon, 17510386 |
| Tongue squamous cell carcinoma vs. normal | 2.136 | 7.85E−5 | 4.433 | Ye, 18254958 | |
| Head and neck squamous cell carcinoma vs. normal | 1.896 | 1.37E−5 | 5.107 | Ginos, 14729608 | |
| Tall cell variant thyroid gland papillary carcinoma vs. normal | 1.178 | 3.22E−6 | 7.594 | Giordano, 16609007 | |
| Hypopharyngeal squamous cell carcinoma vs. normal | 2.982 | 0.033 | 2.362 | Schlingemann, 16205657 | |
| HSPD1 | Salivary gland adenoid cystic carcinoma vs. normal | 1.500 | 3.23E−4 | 4.102 | FriersonHF, 12368205 |
| Head and neck squamous cell carcinoma vs. normal | 2.282 | 4.67E−7 | 6.773 | Ginos, 14729608 | |
| Floor of the mouth carcinoma vs. normal | 2.488 | 9.57E−7 | 7.144 | Pyeon, 17510386 | |
| Nasopharyngeal carcinoma vs. normal | 1.812 | 3.60E−5 | 5.246 | Sengupta, 16912175 | |
| Tongue squamous cell carcinoma vs. normal | 1.732 | 1.59E−5 | 4.785 | Ye, 18254958 | |
| SERPINH1 | Head and neck squamous cell carcinoma vs. normal | 6.350 | 1.25E−6 | 12.064 | Cromer, 14676830 |
| Tongue squamous cell carcinoma vs. normal | 3.148 | 1.09E−10 | 7.757 | Estilo,19138406 | |
| Salivary gland adenoid cystic carcinoma vs. normal | 308.933 | 3.74E−6 | 7.691 | FriersonHF, 12368205 | |
| Oral cavity squamous cell carcinoma vs. normal | 2.072 | 2.44E−9 | 7.125 | Peng,21853135 | |
| Tongue squamous cell carcinoma vs. normal | 2.022 | 2.05E−9 | 7.405 | Talbot, 15833835 | |
| HSPA4 | Thyroid gland oncocytic follicular carcinoma vs. normal | 1.259 | 1.32E−4 | 6.863 | Giordano, 16609007 |
| Oral cavity carcinoma vs. normal | 2.732 | 4.49E−6 | 6.212 | Pyeon, 17510386 | |
| Tongue squamous cell carcinoma vs. normal | 1.478 | 1.74E−6 | 5.146 | Talbot, 15833835 | |
| Follicular variant thyroid gland papillary carcinoma vs. normal | 1.017 | 4.02E−4 | 3.464 | TCGA, 2013 | |
| Oral cavity squamous cell carcinoma epithelia vs. normal | 1.199 | 0.002 | 3.391 | Toruner, 15381369 | |
| HSP90AA1 | Floor of the mouth carcinoma vs. normal | 3.010 | 1.40E−8 | 8.051 | Pyeon, 17510386 |
| Oral cavity carcinoma vs. normal | 3.878 | 6.57E−9 | 8.959 | Pyeon, 17510386 | |
| Tongue carcinoma vs. normal | 2.549 | 2.98E−7 | 6.220 | Pyeon, 17510386 | |
| Tonsillar carcinoma vs. normal | 2.641 | 1.81E−6 | 6.050 | Pyeon, 17510386 | |
| Hypopharyngeal squamous cell carcinoma vs. normal | 2.931 | 0.011 | 3.044 | Schlingemann, 16205657 |
TCGA the Cancer Genome Atlas
Correlations between the expression levels of HSPs and the clinicopathological parameters of HNSC patients
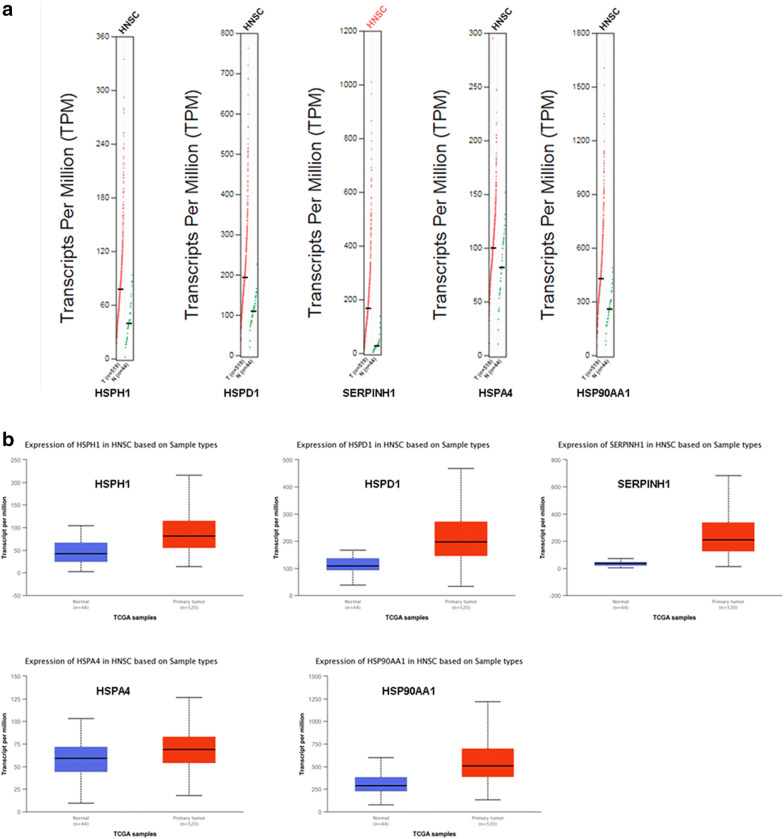
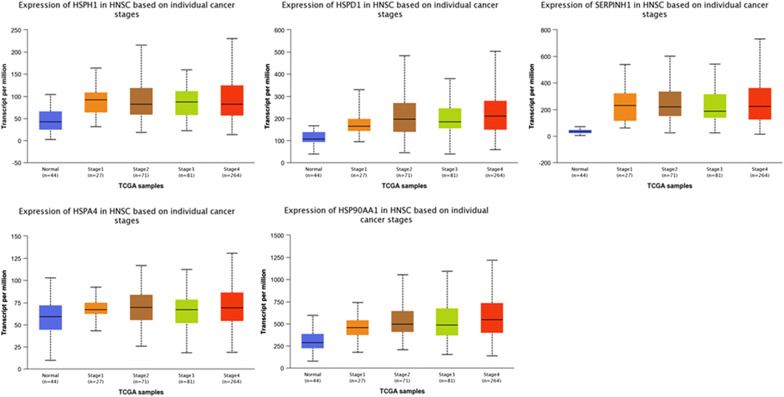
GEPIA was used to compare HSPs between tumor and normal tissues. The findings revealed that the expression of all five genes was higher in HNSC samples than in normal control samples (Fig. 2a, b). We also analyzed HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 expression as a function of the HNSC tumor stage. The results revealed a clear correlation between gene expression and tumor stage, with HNSC patients in more advanced stages tending to exhibit HSP expression levels (Fig. 3). After investigating the expression patterns of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 in HNSC, we examined HSP expression patterns in HNSC using THPA. The results confirmed that the protein levels of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 were elevated in HNSC samples relative to normal control samples (Fig. 4).
Fig. 2.
The expression profiles of HSPs in HNSC patients. The expression profile of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in HNSC patients were analyzed by GEPIA (a) and UALCAN database (b); the p-value was set at 0.05
Fig. 3.
Correlations between expression levels of HSPs and tumor stage in HNSC patients. The relationships between expression levels of HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 and tumor stages in HNSC patients were determined using the UALCAN database. The p-value was set at 0.05. The abscissa indicates the HNSC tumor stage and the ordinate indicates the expression levels of HSPs (gene expression ~ pathological stage)
Fig. 4.
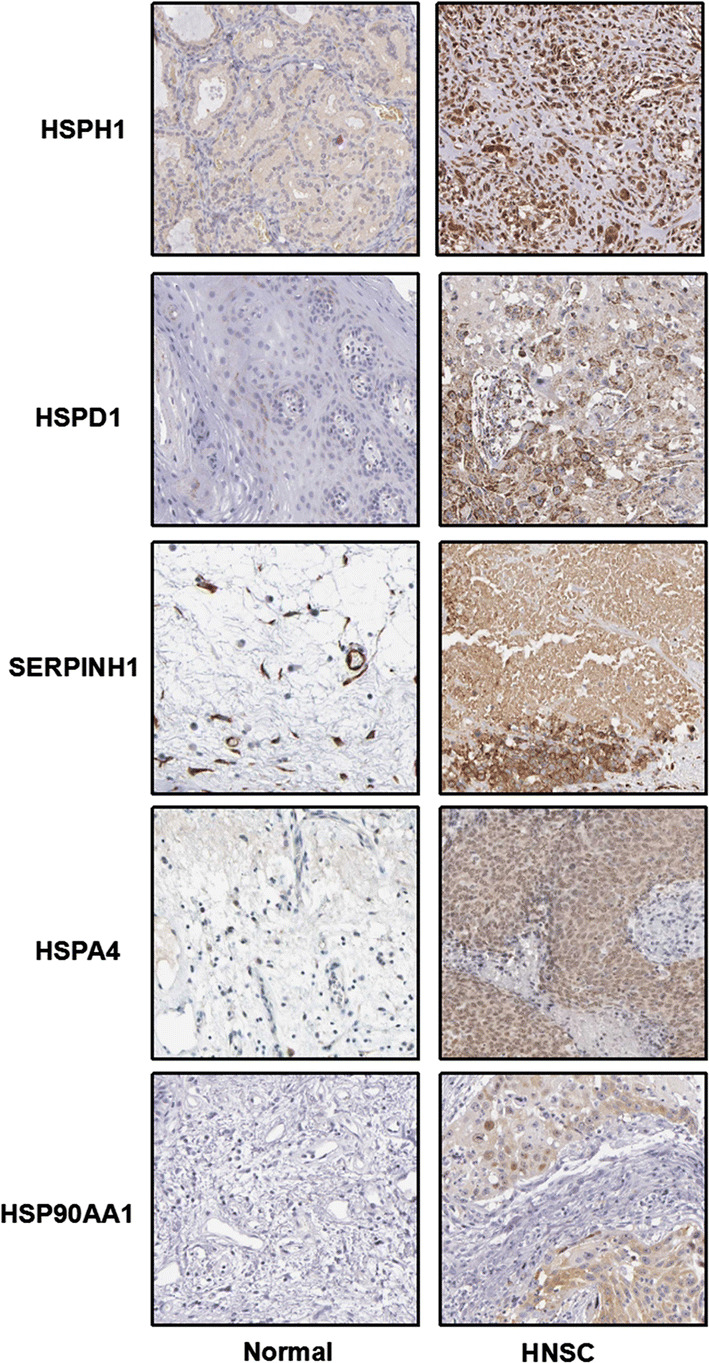
HSP genes were distinctively high expressed in HNSC tissues from Human Protein Atlas. HSP proteins were not expressed in normal head and neck tissues, whereas their high expressions were observed in HNSC tissues
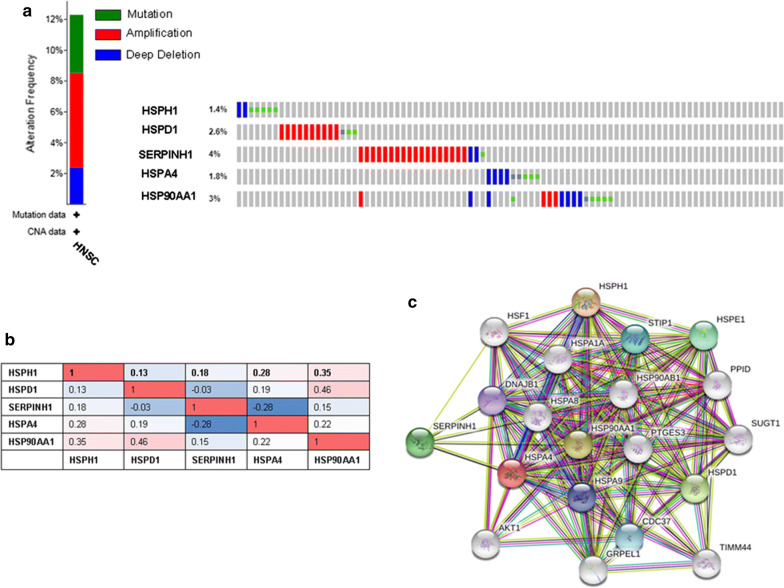
Genetic alterations to HSP-associated genes and neighbor gene networks in HNSC patients
The frequencies of mutations of the HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 genes in HNSC were assessed using cBioPortal. In total, 504 patients from the Head and Neck Squamous Cell Carcinoma (TCGA, Provisional) were analyzed. According to this dataset, the percentages of genetic variations in HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 genes among HNSC patients ranged from 1.4 to 4% for individual genes (HSPH1, 1.4%; HSPD1, 2.6%; SERPINH1, 4%; HSPA4, 1.8%; HSP90AA1, 3%). A total of 62 (12%) samples exhibited gene set/pathway alterations, with mutations in 2.82% of analyzed gene sets (Fig. 5a).The cBioPortal online tool also allows Pearson correlation analysis of HSP expression data (RNA Seq V2 RSEM) in HNSC (TCGA, Provisional). The results revealed a significant negative correlation between HSPD1 and SERPINH1 (Fig. 5b). Next, a PPI enrichment analysis, was then used to explore the relationships among these genes in HNSC. The PPI network was constructed by STRING. The results showed that several HSPs, including DNAJB1, HSPA1A, STIP1, HSPE1, HSPA8, HSPA9, HSF1, and HSP90AB1, were closely associated with HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 (Fig. 5c).
Fig. 5.
Expression of HSP genes and mutation analysis in HNSC. a Summary of alterations in HSPs. b Pearson correlation analysis of HSP family members. c Protein–protein interaction network of HSPs
The prognostic values of HSPs in HNSC
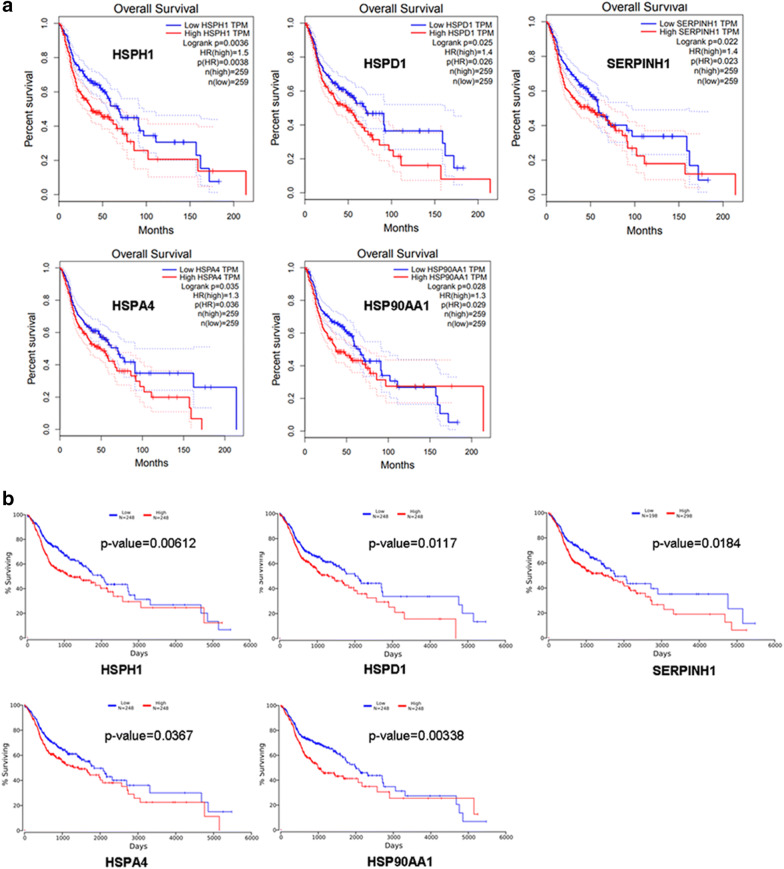
We evaluated the prognostic significance of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 in all HNSC patients using Kaplan–Meier plots and the OncoLnc database. The results revealed that increased expression levels of HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 were strongly associated with poor overall survival (Fig. 6). Thus, the results suggest that highly expressed HSPs (HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1) are prognostic factors for HNSC. Since HSPs are reportedly associated with tumor immunity [26], we used the TIMER (https://cistrome.shinyapps.io/timer/) database to investigate the relationships between the HSP expression levels and the levels of immune infiltration in HNSC. Unfortunately, we did not find any significant correlations between HSP gene expressions and immune infiltration levels (Additional file 3: Fig. S3).
Fig. 6.
The prognostic values of HSP family members in HNSC patients. The overall survival curves comparing HNSC patients with high (red) and low (blue) HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 expression levels were plotted using the GEPIA (a) and OncoLnc databases (b) at the threshold p-value of < 0.05
Functional enrichment analysis of HSPs in HNSC patients
The functions of these five genes were next explored through GO and KEGG analyses. GO analyses allow assessment of the biological process, molecular function, and cellular component annotations of genes of interest. The results revealed that HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1 as well as their neighbor genes, are primarily enriched for regulation of protein ubiquitination, cellular protein metabolic and immune system process, chaperone-mediated autophagy, regulation of apoptotic process, positive regulation of DNA metabolic process, protein targeting to mitochondrion, regulation of cellular response to stress and heat, and protein folding (Additional file 4: Fig S4A). Enriched molecular functions included transcription regulation by Hsp70 protein binding, ATPase regulator activity, enzyme regulator activity, DNA polymerase binding, ubiquitin protein ligase binding, chaperone binding, ATP binding, protein binding, HSP binding, and unfolded protein binding (Additional file 4: Fig S4B). Cellular component annotations for these genes included mitochondrion, cytoplasmic vesicle lumen, intracellular organelle part, protein-containing complex, extracellular exosome, ficolin-1-rich granule lumen, cytosol, intracellular organelle lumen, cytoplasmic part, and chaperone complex (Additional file 4: Fig S4C). The KEGG pathways for these genes are shown in Table 2. Among these pathways, protein processing in the estrogen signaling pathway, endoplasmic reticulum, antigen processing and presentation, Prostate cancer, PI3K-Akt signaling pathway, NOD-like receptor signaling pathway, Epstein-Barr virus infection, MAPK signaling pathway, IL-17 signaling pathway, and Th17 cell differentiation were involved in tumor development and pathogenesis in HNSC (Additional file 5: Fig S5A and B).
Table 2.
KEGG pathway analysis of HSP associated genes in HNSC
| Pathway ID | Pathway name | Gene count | False discovery rate | Genes |
|---|---|---|---|---|
| hsa04141 | Protein processing in endoplasmic reticulum | 6 | 7.82E−07 | DNAJB1, HSP90AA1, HSP90AB1, HSPA1A, HSPA8, HSPH1 |
| hsa04612 | Antigen processing and presentation | 5 | 7.82E−07 | HSP90AA1, HSP90AB1, HSPA1A, HSPA4, HSPA8 |
| hsa04915 | Estrogen signaling pathway | 5 | 7.43E−06 | AKT1, HSP90AA1, HSP90AB1, HSPA1A, HSPA8 |
| hsa05215 | Prostate cancer | 3 | 0.0017 | AKT1, HSP90AA1, HSP90AB1 |
| hsa04151 | PI3K-Akt signaling pathway | 4 | 0.0033 | AKT1, CDC37, HSP90AA1, HSP90AB1 |
| hsa04621 | NOD-like receptor signaling pathway | 3 | 0.0045 | HSP90AA1, HSP90AB1, SUGT1 |
| hsa05169 | Epstein-Barr virus infection | 3 | 0.0061 | AKT1, HSPA1A, HSPA8 |
| hsa04010 | MAPK signaling pathway | 3 | 0.0173 | AKT1, HSPA1A, HSPA8 |
| hsa04657 | IL-17 signaling pathway | 2 | 0.0209 | HSP90AA1, HSP90AB1 |
| hsa04659 | Th17 cell differentiation | 2 | 0.0242 | HSP90AA1, HSP90AB1 |
Discussion
Dysregulation of HSPs is common in cancer development. Research suggests that HSPs are essential for tumor cell proliferation and differentiation. Although rarely used as diagnostic biomarkers for cancer, the expression levels of HSPs may predict the development of various cancers. For example, there is considerable evidence demonstrating that the over-expression of HSP27 may confer poor prognosis in gastric, prostate, and liver cancers [27–29]. Herein, we sought to investigate the expression pattern and prognostic values of different HSP members (HSPH1, HSPD1, SERPINH1, HSPA4 and HSP90AA1) in HNSC. These findings advance our current understanding of HNSC and may offer a means for improving treatment approaches and prognostic accuracy in HNSC patients.
The primary role of HSPs in tumorigenesis involves the stabilisation of functions of mutated or aberrantly-expressed tumor-related genes. Thus, high expression of HSPs is a hallmark of many cancers. In addition, HSPs are released from cancer cells, influencing their properties and functions through receptor-mediated signaling [30]. HSPH1 has been reported to be over-expressed in melanoma and colon cancer patients [31, 32]. The upregulation of HSPH1 is implicated in hyperactivation of the Wnt signalling pathway. The transcription levels of Wnt signalling target genes are significantly downregulated in cell line models with HSPH1 inhibition [9]. In our study, we confirmed that the expression of HSPH1 in HNSC tissue was significantly elevated compared with normal tissue. We also observed a significant correlation between the expression of HSPH1 and tumor stage in HNSC patients. High HSPH1 expression was associated with low overall survival in all HNSC patients.
HSPD1 is a molecular chaperone that is primarily localized in the mitochondrial matrix [33]. Recently, HSPD1 has been found in many extramitochondrial sites, including the extracellular surface, cell surface, intracellular vesicles, nucleus, extracellular fluid, and even the cytoplasm. HSPD1 has been recognized as a potential biomarker for tumor diagnosis and prognosis, especially in colorectal cancer. There is increasing evidence that HSPD1, especially intracellular HSPD1, is involved in the survival and metastasis of various tumors [34–36]. In our study, the ONCOMINE and TCGA datasets revealed that the expression of HSPD1 was higher in HNSC tissue than in normal tissues. High HSPD1 expression was significantly associated with poor overall survival among HNSC patients followed up for more than 6000 days.
SERPINH1 is a collagen-binding protein and has been identified to be a collagen-specific chaperon. It promotes the malignant behavior of cancer cells and predicts the metastatic activity of human cancer cells. SERPINH1 also plays an essential role in regulating the expression of extracellular matrix (ECM) proteins and fibronectin (FN); therefore, dysfunction of SERPINH1 stimulates abnormal expression of ECM proteins, which promotes epithelial-mesenehymaltransition (EMT) [37]. Our data confirmed that dysregulation of SERPINH1 is closely related to the development and progression of HNSC, and further affects the prognosis of HNSC.
HSPA4 is highly expressed in malignant tumor cells and is involved in tumor development and chemotherapy resistance, presumably due to its ability to inhibit tumor cell apoptosis [38]. It has been reported that overexpression of HSPA4 can inhibit cell apoptosis and prevent activation of the caspase signaling pathway, which leads to accumulation of misfolded proteins, ROS, and DNA damage. HSP70 not only affects the apoptotic pathway, but also the autophagic pathway. It prevents the formation of autophagosomes by activating the mTOR pathway. Consistent with these results, Leu et al. found that the inhibition of HSP70 resulted in significant increases in the expression of LC3-II and the number of autophagosomes [39]. In our study, it is clear that HSPA4 acts as an “evil chaperone” in HNSC. Its expression was significantly increased in HNSC patients and was associated with poor prognosis.
HSP90AA1 acts as a highly conserved chaperone protein and participates in tumor cell differentiation, proliferation, and angiogenesis. Recently, it has been regarded as a promising target for specific cancer therapy [40]. Hsp90AA1 is expressed in various cancers, including breast, colon, ovarian, lung and prostate cancers. This may be related to the involvement of HSP90AA1 in the regulation of apoptosis and signaling transduction triggered by growth factors, death receptors, and stress signals [41]. HSP90AA1 inhibits the initiation of apoptosis by blocking the binding of caspase 9 to apoptotic protein 1 activator. Additionally, it promotes the formation of tumor cells by stabilizing mutant p53 complexes, thereby inhibiting the apoptosis of tumor cells [38, 42]. In our systematic analyses of various databases, we found that HSP90AA1 was significantly upregulated in HNSC patients, and its expression level was significantly associated with the tumor stage in HNSC patients. As expected, high HSP90AA1 expression predicted poor overall survival among HNSC patients, implying an oncogenic role of HSP90AA1 in HNSC.
In this study, we also explored the genetic alterations and potential functions of HSP family members. The percentages of genetic mutations in HSPs in HNSC varied from 1.4% to 4% for individual genes. At the same time, we constructed a network for these five genes and their neighbor genes. Functional analysis demonstrated that these five genes were primarily enriched in tumor-related signaling pathways, indicating that HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 play crucial cancer-promoting roles in the development of head and neck cancer.
Conclusion
In summary, our results indicate that HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 are significantly upregulated in HNSC patients and their upregulation is negatively correlated with HNSC tumor stage. Based on the above findings, it is expected that HSPH1, HSPD1, SERPINH1, HSPA4, and HSP90AA1 could act as potential prognostic biomarkers and therapeutic targets for HNSC. Our research contributes to a better understanding of the pathogenesis of HNSC and may assist in the development of more effective targeted drugs for head and neck cancer. However, further mechanistic studies are needed to validate our findings and to promote clinical application of HSPs as prognostic or therapeutic targets in HNSC.
Supplementary information
Additional file 1: Fig. S1. The expression levels of HSPs in HNSC (ONCOIME). The expression levels of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in several head and neck cancer studies. Red: over-expression. The significance threshold was p < 0.05.
Additional file 2: Fig. S2. Expressions of HSPs across TCGA cancers (with tumor and normal samples). The expression levels of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in pan-cancer.
Additional file 3: Fig. S3. The relationships between the HSP expression levels and the levels of immune infiltration in HNSC.
Additional file 4: Fig. S4. Functional Enrichment Analysis of HSPs in patients with HNSC. GO enrichment analysis predicted the function of target genes from three aspects: biological processes (A), cellular components (B), and molecular functions (C).
Additional file 5: Fig. S5. p53 signal pathway and cell cycle pathway regulated by HSPs in HNSC. The MAPK signal pathway (A) and PI3K-Akt signaling pathway (B) regulated by HSPs in HNSC are shown.
Acknowledgements
Not applicable.
Abbreviations
- HNSC
Head and neck squamous cell carcinoma
- HSPs
Heat shock proteins
- THPA
The Human Protein Altas
- OS
Overall survival
- HSP47
Heat shock protein 47
- Hsp90α
Heat shock protein 90α
- ECM
Extracellular matrix
- TCGA
The Cancer Genome Atlas
- CAN
Copy-number alterations
- GO
Gene ontology
- KEGG
Kyoto Encyclopedia of Genes and Genomes
Authors’ contributions
Conceived and designed the study strategy: HJX; Acquisition of data: statistical analysis and interpretation of data GRF, YQT; Drafting or revision of the manuscript: YQT, GRF; Reference collection and data management: NW; Wrote the manuscript: YQT; Prepared the tables and figures: GRF; Study supervision: HJX; All authors read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 81771002 and 81500796) and Research fund of Wuhan Union hospital (No. 02.03.2019-114).
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Guorun Fan and Yaqin Tu contributed equally to this work
Supplementary information
Supplementary information accompanies this paper at 10.1186/s12935-020-01296-7.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65(2):87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A. Cancer treatment and survivorship statistics, 2016. CA Cancer J Clin. 2016;66(4):271–289. doi: 10.3322/caac.21349. [DOI] [PubMed] [Google Scholar]
- 4.Puram SV, Tirosh I, Parikh AS, Patel AP, Yizhak K, Gillespie S, Rodman C, Luo CL, Mroz EA, Emerick KS, et al. Single-cell transcriptomic analysis of primary and metastatic tumor ecosystems in head and neck cancer. Cell. 2017;171(7):1611–1624. doi: 10.1016/j.cell.2017.10.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lindquist S, Craig EA. The heat-shock proteins. Annu Rev Genet. 1988;22:631–677. doi: 10.1146/annurev.ge.22.120188.003215. [DOI] [PubMed] [Google Scholar]
- 6.Cornford PA, Dodson AR, Parsons KF, Desmond AD, Woolfenden A, Fordham M, Neoptolemos JP, Ke Y, Foster CS. Heat shock protein expression independently predicts clinical outcome in prostate cancer. Cancer Res. 2000;60(24):7099–7105. [PubMed] [Google Scholar]
- 7.van de Vijver MJ, He YD, van’t Veer LJ, Dai H, Hart AA, Voskuil DW, Schreiber GJ, Peterse JL, Roberts C, Marton MJ, et al. A gene-expression signature as a predictor of survival in breast cancer. N Engl J Med. 2002;347(25):1999–2009. doi: 10.1056/NEJMoa021967. [DOI] [PubMed] [Google Scholar]
- 8.Bellaye PS, Burgy O, Causse S, Garrido C, Bonniaud P. Heat shock proteins in fibrosis and wound healing: good or evil? Pharmacol Therap. 2014;143(2):119–132. doi: 10.1016/j.pharmthera.2014.02.009. [DOI] [PubMed] [Google Scholar]
- 9.Yu N, Kakunda M, Pham V, Lill JR, Du P, Wongchenko M, Yan Y, Firestein R, Huang X. HSP105 recruits protein phosphatase 2A to dephosphorylate beta-catenin. Mol Cell Biol. 2015;35(8):1390–1400. doi: 10.1128/MCB.01307-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kang BH, Shu CW, Chao JK, Lee CH, Fu TY, Liou HH, Ger LP, Liu PF. HSPD1 repressed E-cadherin expression to promote cell invasion and migration for poor prognosis in oral squamous cell carcinoma. Sci Rep. 2019;9(1):8932. doi: 10.1038/s41598-019-45489-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yamamoto N, Kinoshita T, Nohata N, Yoshino H, Itesako T, Fujimura L, Mitsuhashi A, Usui H, Enokida H, Nakagawa M, et al. Tumor-suppressive microRNA-29a inhibits cancer cell migration and invasion via targeting HSP47 in cervical squamous cell carcinoma. Int J Oncol. 2013;43(6):1855–1863. doi: 10.3892/ijo.2013.2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li T, Gao X, Han L, Yu J, Li H. Identification of hub genes with prognostic values in gastric cancer by bioinformatics analysis. World J Surg Oncol. 2018;16(1):114. doi: 10.1186/s12957-018-1409-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kamikawaji K, Seki N, Watanabe M, Mataki H, Kumamoto T, Takagi K, Mizuno K, Inoue H. Regulation of LOXL2 and SERPINH1 by antitumor microRNA-29a in lung cancer with idiopathic pulmonary fibrosis. J Hum Genet. 2016;61(12):985–993. doi: 10.1038/jhg.2016.99. [DOI] [PubMed] [Google Scholar]
- 14.Li C, Liu D, Yuan Y, Huang S, Shi M, Tao K, Feng W. Overexpression of Apg-2 increases cell proliferation and protects from oxidative damage in BaF3-BCR/ABL cells. Int J Oncol. 2010;36(4):899–904. doi: 10.3892/ijo_00000568. [DOI] [PubMed] [Google Scholar]
- 15.Gotoh K, Nonoguchi K, Higashitsuji H, Kaneko Y, Sakurai T, Sumitomo Y, Itoh K, Subjeck JR, Fujita J. Apg-2 has a chaperone-like activity similar to Hsp110 and is overexpressed in hepatocellular carcinomas. FEBS Lett. 2004;560(1–3):19–24. doi: 10.1016/S0014-5793(04)00034-1. [DOI] [PubMed] [Google Scholar]
- 16.Kaneko Y, Kimura T, Kishishita M, Noda Y, Fujita J. Cloning of apg-2 encoding a novel member of heat shock protein 110 family. Gene. 1997;189(1):19–24. doi: 10.1016/s0378-1119(96)00807-4. [DOI] [PubMed] [Google Scholar]
- 17.Wu CY, Lin CT, Wu MZ, Wu KJ. Induction of HSPA4 and HSPA14 by NBS1 overexpression contributes to NBS1-induced in vitro metastatic and transformation activity. J Biomed Sci. 2011;18:1. doi: 10.1186/1423-0127-18-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lee YC, Chang WW, Chen YY, Tsai YH, Chou YH, Tseng HC, Chen HL, Wu CC, Chang-Chien J, Lee HT, et al. Hsp90alpha mediates BMI1 expression in breast cancer stem/progenitor cells through facilitating nuclear translocation of c-Myc and EZH2. Int J Mol Sci. 2017;18(9):1986. doi: 10.3390/ijms18091986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fu Y, Xu X, Huang D, Cui D, Liu L, Liu J, He Z, Zheng S, Luo Y. Plasma heat shock protein 90alpha as a biomarker for the diagnosis of liver cancer: an official, large-scale, and multicenter clinical trial. EBioMedicine. 2017;24:56–63. doi: 10.1016/j.ebiom.2017.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen CC, Chen LL, Li CP, Hsu YT, Jiang SS, Fan CS, Chua KV, Huang SX, Shyr YM, Chen LT, et al. Myeloid-derived macrophages and secreted HSP90alpha induce pancreatic ductal adenocarcinoma development. Oncoimmunology. 2018;7(5):e1424612. doi: 10.1080/2162402X.2018.1424612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ono K, Eguchi T, Sogawa C, Calderwood SK, Futagawa J, Kasai T, Seno M, Okamoto K, Sasaki A, Kozaki KI. HSP-enriched properties of extracellular vesicles involve survival of metastatic oral cancer cells. J Cell Biochem. 2018;119(9):7350–7362. doi: 10.1002/jcb.27039. [DOI] [PubMed] [Google Scholar]
- 22.Kosec A, Novak R, Konjevoda P, Trkulja V, Bedekovic V, Grgurevic L. Tumor tissue hnRNP M and HSP 90alpha as potential predictors of disease-specific mortality in patients with early-stage cutaneous head and neck melanoma: a proteomics-based study. Oncotarget. 2019;10(62):6713–6722. doi: 10.18632/oncotarget.27333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tang Z, Li C, Kang B, Gao G, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017;45(W1):W98–W102. doi: 10.1093/nar/gkx247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chandrashekar DS, Bashel B, Balasubramanya SAH, Creighton CJ, Ponce-Rodriguez I, Chakravarthi B, Varambally S. UALCAN: a portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017;19(8):649–658. doi: 10.1016/j.neo.2017.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cancer Genome Atlas N Comprehensive molecular portraits of human breast tumors. Nature. 2012;490(7418):61–70. doi: 10.1038/nature11412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shevtsov M, Multhoff G. Heat shock protein-peptide and HSP-based immunotherapies for the treatment of cancer. Front Immunol. 2016;7:171. doi: 10.3389/fimmu.2016.00171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.King KL, Li AF, Chau GY, Chi CW, Wu CW, Huang CL, Lui WY. Prognostic significance of heat shock protein-27 expression in hepatocellular carcinoma and its relation to histologic grading and survival. Cancer. 2000;88(11):2464–2470. doi: 10.1002/1097-0142(20000601)88:11<2464::aid-cncr6>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 28.Kapranos N, Kominea A, Konstantinopoulos PA, Savva S, Artelaris S, Vandoros G, Sotiropoulou-Bonikou G, Papavassiliou AG. Expression of the 27-kDa heat shock protein (HSP27) in gastric carcinomas and adjacent normal, metaplastic, and dysplastic gastric mucosa, and its prognostic significance. J Cancer Res Clin Oncol. 2002;128(8):426–432. doi: 10.1007/s00432-002-0357-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bostwick DG. Immunohistochemical changes in prostate cancer after androgen deprivation therapy. Mol Urol. 2000;4(3):101–106. [PubMed] [Google Scholar]
- 30.Calderwood SK, Gong J. Heat Shock Proteins Promote Cancer: it’s a Protection Racket. Trends Biochem Sci. 2016;41(4):311–323. doi: 10.1016/j.tibs.2016.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Skrzypczak M, Goryca K, Rubel T, Paziewska A, Mikula M, Jarosz D, Pachlewski J, Oledzki J, Ostrowski J. Modeling oncogenic signaling in colon tumors by multidirectional analyses of microarray data directed for maximization of analytical reliability. PloS ONE. 2010;5(10):e13091. doi: 10.1371/journal.pone.0013091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kai M, Nakatsura T, Egami H, Senju S, Nishimura Y, Ogawa M. Heat shock protein 105 is overexpressed in a variety of human tumors. Oncol Rep. 2003;10(6):1777–1782. [PubMed] [Google Scholar]
- 33.Cheng MY, Hartl FU, Martin J, Pollock RA, Kalousek F, Neupert W, Hallberg EM, Hallberg RL, Horwich AL. Mitochondrial heat-shock protein hsp60 is essential for assembly of proteins imported into yeast mitochondria. Nature. 1989;337(6208):620–625. doi: 10.1038/337620a0. [DOI] [PubMed] [Google Scholar]
- 34.Li XS, Xu Q, Fu XY, Luo WS. Heat shock protein 60 overexpression is associated with the progression and prognosis in gastric cancer. PLoS ONE. 2014;9(9):e107507. doi: 10.1371/journal.pone.0107507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Cappello F, David S, Rappa F, Bucchieri F, Marasa L, Bartolotta TE, Farina F, Zummo G. The expression of HSP60 and HSP10 in large bowel carcinomas with lymph node metastase. BMC Cancer. 2005;5:139. doi: 10.1186/1471-2407-5-139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Zhou J, Li XL, Chen ZR, Chng WJ. Tumor-derived exosomes in colorectal cancer progression and their clinical applications. Oncotarget. 2017;8(59):100781–100790. doi: 10.18632/oncotarget.20117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen D, Gassenmaier M, Maruschke M, Riesenberg R, Pohla H, Stief CG, Zimmermann W, Buchner A. Expression and prognostic significance of a comprehensive epithelial-mesenchymal transition gene set in renal cell carcinoma. J Urol. 2014;191(2):479–486. doi: 10.1016/j.juro.2013.08.052. [DOI] [PubMed] [Google Scholar]
- 38.Jego G, Hazoume A, Seigneuric R, Garrido C. Targeting heat shock proteins in cancer. Cancer Lett. 2013;332(2):275–285. doi: 10.1016/j.canlet.2010.10.014. [DOI] [PubMed] [Google Scholar]
- 39.Leu JI, Pimkina J, Frank A, Murphy ME, George DL. A small molecule inhibitor of inducible heat shock protein 70. Mol Cell. 2009;36(1):15–27. doi: 10.1016/j.molcel.2009.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pandey P, Saleh A, Nakazawa A, Kumar S, Srinivasula SM, Kumar V, Weichselbaum R, Nalin C, Alnemri ES, Kufe D, et al. Negative regulation of cytochrome c-mediated oligomerization of Apaf-1 and activation of procaspase-9 by heat shock protein 90. EMBO J. 2000;19(16):4310–4322. doi: 10.1093/emboj/19.16.4310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Li W, Sahu D, Tsen F. Secreted heat shock protein-90 (Hsp90) in wound healing and cancer. Biochim Biophys Acta. 2012;1823(3):730–741. doi: 10.1016/j.bbamcr.2011.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Blagosklonny MV, Toretsky J, Bohen S, Neckers L. Mutant conformation of p53 translated in vitro or in vivo requires functional HSP90. Proc Natl Acad Sci USA. 1996;93(16):8379–8383. doi: 10.1073/pnas.93.16.8379. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Fig. S1. The expression levels of HSPs in HNSC (ONCOIME). The expression levels of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in several head and neck cancer studies. Red: over-expression. The significance threshold was p < 0.05.
Additional file 2: Fig. S2. Expressions of HSPs across TCGA cancers (with tumor and normal samples). The expression levels of HSPH1, HSPD1, Serpinh1, HSPA4 and HSP90AA1 in pan-cancer.
Additional file 3: Fig. S3. The relationships between the HSP expression levels and the levels of immune infiltration in HNSC.
Additional file 4: Fig. S4. Functional Enrichment Analysis of HSPs in patients with HNSC. GO enrichment analysis predicted the function of target genes from three aspects: biological processes (A), cellular components (B), and molecular functions (C).
Additional file 5: Fig. S5. p53 signal pathway and cell cycle pathway regulated by HSPs in HNSC. The MAPK signal pathway (A) and PI3K-Akt signaling pathway (B) regulated by HSPs in HNSC are shown.
Data Availability Statement
All data generated or analyzed during this study are included in this published article.