Abstract
Background
The level of lipoprotein-associated phospholipase A2 (LP-PLA2) in serum is independently correlated to coronary artery diseases (CAD). The aim of the study was to determine whether LP-PLA2 activity is positively associated with the seriousness of CAD.
Methods
Amount to 1056 patients suspected of having CAD underwent coronary angiography (CAG) to determine the seriousness of CAD. According to the amount of diseased coronary branches, the 1056 patients were split into three groups: single-vessel stenosis group, multiple-vessels stenosis group (> or = 2 diseased coronary branches),and control group (no diseased coronary branches). According to CAG results, electrocardiography, cardiac biomarker, and clinical presentation, all patients were split into four groups: acute myocardial infarction (AMI), unstable angina (UA), stable angina (SA), and control groups (excluding CAD). The activity of LP-PLA2 was compared statistically among the subgroups. Receiver operating characteristic analysis was applied to investigate the role of LP-PLA2 in evaluating the presence and seriousness of CAD.
Results
The level of LP-PLA2 increased in line with the number of diseased coronary branches. The levels of LP-PLA2 in the AMI and UA groups were observably higher when compared with the control and SA groups. LP-PLA2 had 75.6% sensitivity and 67.3% specificity for recognizing CAD, and 53.0% sensitivity and 80.3% specificity for recognizing severe coronary artery lesions.
Conclusion
The activity of LP-PLA2 is positively correlated to the seriousness of CAD.
Keywords: Lipoprotein-associated phospholipase A2, Cardiovascular disease, Coronary artery diseases, Coronary angiography
Background
Coronary artery diseases (CAD) remain the main cause of mortality and functional impairment all over the world, in spite of huge progress on medical therapeutics in recent decades. It is widely acknowledged that inflammatory reactions contribute considerably to the production of atherosclerosis, from original atherosclerotic plaque to subsequent destabilization and eventual rupture [1]. Several previous researches have suggested that numerous inflammatory biomarkers are relevant to cardiovascular diseases (CVD); one of the most typical of these is C-reactive protein (CRP) [2, 3].
LP-PLA2 is an enzyme produced mainly by neutrophils and macrophages from atherosclerotic plaques, which are then transferred to the bloodstream via high-density lipoprotein-cholesterol (HDL-C) as well as low-density lipoprotein-cholesterol (LDL-C) [4, 5]. Plenty of evidences have indicated that, as an important biomarker of inflammation, Lp-PLA2 is an independent risk factor for CVD [6]. Several researches have demonstrated that higher plasma levels of LP-PLA2 activity further increase the risk of cardiovascular events, for instance ischemic stroke and myocardial infarction [7]. However, the correlation between the level of LP-PLA2 and CVD has not necessarily been confirmed in other research [7–9]. Besides, two recent clinical studies suggested that darapladib, as a selective LP-PLA2 antagonist, may not improve clinical outcomes in patients with CAD [10, 11]. The results of previous research appear to be contradictory.
In view of these conflicting findings, we carried out a targeted clinical research to observe whether increased activity of LP-PLA2 is positively correlate to the seriousness of CAD. The findings of our study would provide meaningful information to confirm whether the risk degree of CAD could be predicted independently by LP-PLA2 level.
Methods
Patients and protocols
All the patients were consecutively recruited from December 2016 to June 2019 in the Department of Cardiology, First Affiliated Hospital of Nanjing Medical University. All the patients gave informed consent, and the Ethical Committee of Nanjing Medical University approved the research. All enrolled subjects underwent coronary angiography (CAG) to establish the severity of CAD. All patients underwent CAG in accordance with the requirements of coronary angiography guidelines [12]. According to the number of diseased coronary branches (≥50% stenosis), 1056 patients were split into three groups: single-vessel stenosis (SVS) group, multiple-vessel stenosis (MVS) group (≥2 diseased coronary branches), and control group (< 50% stenosis). According to the CAG results, cardiac biomarker, electrocardiography, as well as clinical presentation, subjects were split into four groups: AMI, UA, SA, and control groups (excluding CAD).
The demographic characteristics of all patients, including age, gender, smoking status, and clinical characteristics (such as diabetes mellitus and hypertension), were obtained via questionnaire [13]. Blood samples were collected from these subjects for various blood index analyses. Fasting blood glucose (FBG), lipoprotein(a) (Lp(a)), total cholesterol (TC), high-density lipoprotein-cholesterol (HDL-C), low-density lipoprotein-cholesterol (LDL-C) and triglyceride (TG) were tested using fasting venous blood on day 1 of hospital admission. The measurement of blood lipid is carried out by the automatic biochemical analyzer (Tokyo Medical 1024i, Japan). Determination of the plasma level of LP-PLA2 was done using an enzyme-linked immune-absorbent assay (ELISA) kit (commercially bought from Nanjing Norman Biological Technology Co. Ltd., China). The process of the testing the level of LP-PLA2 was conducted according to the manufacturer’s instructions to maintain quality control. Meanwhile, three independent processes were conducted in duplicate, and the mean value was calculated. Receiver operating characteristic (ROC) curves were drawn, and areas under the curve (AUC) were compared to assess the cut-off values of LP-PLA2 to judge the presence and severity of CAD. The severity of CAD is mainly judged by comparing single-vessel lesions with multiple-vessel lesions.
Analysis of data and statistics
Continuous variables that satisfied the normal distribution were shown as means ± standard deviation (SD). All measured data were analyzed to compare differences between groups, using one-way ANOVA and Student’s t-test. Data described as proportions were assessed with a Chi-square test. For non-normally distributed variables,we use the median and inter-quartile range to represent. Evaluation of the relationship between the seriousness of CAD and the plasma level of LP-PLA2 was performed using univariate and multivariate regression analyses. P values were two-sided, and P values≤0.05 were considered as statistically significant. All analyses were conducted using SPSS software package version 21.0 (Chicago, IL, USA).
Results
In all, 1056 studied subjects underwent CAG. The results showed that the coronary artery of 294 patients were without significant stenosis (< 50%), who were regarded as the control group. Meanwhile, 762 patients were diagnosed as having coronary atherosclerotic heart disease (at least one coronary artery stenosis ≥50%); this group contained 366 patients with SVS and 396 with MVS. According to the severity of clinical presentation, all patients diagnosed as having CAD were divided into SA (n = 192), UA (n = 330), and AMI (n = 240) groups.
Assessment of the relationship between the level of LP-PLA2 and degree of coronary artery stenosis
As shown in Table 1, participants diagnosed as being in the MVS group had several risk factors compared to the control group. Generally, participants with MVS were older. Their levels of serum Lp(a) and FBG were visibly higher, while the level of HDL-C was dramatically lower in patients with MVS in comparison to the controls. The basic data of the SVS group showed no significant difference compared with the controls and MVS group. This shows that those in the SVS group expressed an obviously high level of LP-PLA2 in comparison to the control group, while the MVS group also showed a apparent increase in expression of LP-PLA2. After adjusting for gender, age, smoking, hypertension, diabetes mellitus, FBG, Lp(a), TC, TG, LDL-C, and HDL-C, the level of LP-PLA2 expression was still independently associated with the degree of coronary artery stenosis, with an odd ratio (OR) of 1.011 (MVS versus control group, 95% confidence interval (CI) 1.006–1.017, P < 0.05).
Table 1.
The relationship between the level of LP-PLA2 and degree of coronary artery stenosis
| Group | Control | Single | Multiple | P |
|---|---|---|---|---|
| N | 294 | 366 | 396 | |
| Age (years) | 60.63 ± 11.96 | 63.16 ± 12.88 | 65.23 ± 10.93* | 0.113 |
| Male(%) | 204 (69.4) | 306 (83.6) | 282 (71.2) | 0.237 |
| Smoking(%) | 84 (28.6) | 150 (41.0) | 162 (40.9) | 0.315 |
| Hypertension(%) | 168 (57.1) | 216 (59.0) | 276 (69.7) | 0.305 |
| Diabetes (%) | 54 (18.4) | 96 (26.2) | 120 (30.3) | 0.345 |
| FBG (mmol/L) a | 5.35 (4.39–8.56) | 6.04 (5.32–10.25) | 6.27 (4.28–10.45)* | 0.204 |
| Lp(a)(mmol/L) a | 230.39 (112.06–428.32) | 262.12 (121.46–675.28) | 346.55 (176.55–728.36)* | 0.059 |
| TC (mmol/L) | 4.40 ± 1.13 | 4.20 ± 0.20 | 4.52 ± 1.28 | 0.291 |
| TG (mmol/L) | 1.52 ± 0.97 | 1.51 ± 0.87 | 1.78 ± 0.87 | 0.213 |
| LDL-C (mmol/L) | 2.82 ± 0.93 | 2.59 ± 0.84 | 2.88 ± 1.02 | 0.192 |
| HDL-C (mmol/L) | 1.17 ± 0.28 | 1.09 ± 0.23 | 1.04 ± 0.26 * | 0.027 |
| Lp-PLA2(ng/ml) a | 172.83 (92.35–296.42) | 230.95 (115.69–406.35)* | 289.24 (132.11–430.21)*# | < 0.001 |
aindicated median and inter-quartile range, FBG Fasting blood glucose, Lp(a) Lipoprotein(a), TC Total cholesterol, TG Triglyceride, LDL-C Low density lipoprotein-cholesterol, HDL-C High density lipoprotein-cholesterol, Lp-PLA2 Lipoprotein associated-phospholipase A2. *P < 0.05 versus control group, #P < 0.05 versus single group
Assessment of the relationship between the level of LP-PLA2 and seriousness of clinical presentation
All studied subjects were split into four groups according to the seriousness of clinical presentation, as demonstrated in Table 2. The HDL-C level of the UA and AMI groups showed a apparent increase in comparison to the control group. In comparison to the SA and control groups, LP-PLA2 level was dramatically higher in the UA and AMI groups. No obvious difference were observed between the UA and AMI groups, while no obvious difference between the SA and control groups were observed. After adjusting for gender, age, smoking, hypertension, diabetes mellitus, FBG, Lp(a), TC, TG, LDL-C, and HDL-C, the level of LP-PLA2 expression was still independently associated with the seriousness of clinical presentation, with an OR of 1.014 (AMI versus control group, 95% CI 1.008–1.021, P < 0.05).
Table 2.
The relationship between the level of LP-PLA2 and seriousness of clinical Presentation
| Group | Control | SA | UA | AMI | P |
|---|---|---|---|---|---|
| N | 294 | 192 | 330 | 240 | |
| Age (years) | 60.63 ± 11.96 | 66.00 ± 13.66 | 64.80 ± 10.99 | 62.05 ± 11.58 | 0.130 |
| Male(%) | 204 (69.4) | 150 (78.1) | 252 (76.4) | 186 (77.5) | 0.762 |
| Smoking(%) | 84 (28.6) | 66 (34.3) | 138 (41.8) | 108 (45) | 0.362 |
| Hypertension(%) | 168 (57.1) | 120 (62.5) | 252 (76.4) | 120 (50) | 0.051 |
| Diabetes (%) | 54 (18.4) | 60 (31.2) | 84 (25.5) | 72 (30) | 0.514 |
| FBG (mmol/L)a | 5.35 (4.39–8.56) | 6.13 (4.69–10.36) | 6.14 (4.72–10.38) | 6.23 (4.56–10.42) | 0.071 |
| Lp(a) (mmol/L)a | 230.39 (112.06–428.32) | 282.16 (115.22–652.36) | 308.20 (122.08–721.12) | 322.03 (130.06–732.16) | 0.390 |
| TC (mmol/L) | 4.40 ± 1.13 | 4.25 ± 1.13 | 4.31 ± 1.19 | 4.53 ± 1.15 | 0.724 |
| TG (mmol/L) | 1.52 ± 0.97 | 1.65 ± 0.96 | 1.64 ± 0.96 | 1.65 ± 0.69 | 0.782 |
| LDL-C (mmol/L) | 2.82 ± 0.93 | 2.65 ± 0.89 | 2.65 ± 1.02 | 2.94 ± 0.88 | 0.432 |
| HDL-C (mmol/L) | 1.17 ± 0.28 | 1.11 ± 0.22 | 1.05 ± 0.27* | 1.04 ± 0.24* | 0.056 |
| Lp-PLA2(ng/ml) a | 172.83 (92.35–296.42) | 193.84 (102.35–372.35) | 263.95 (172.32–396.45)*& | 311.45 (185.24–412.35)*& | < 0.001 |
aindicated median and inter-quartile range, *P < 0.05 versus control group, &P < 0.05 versus SA group
Assessment of the presence and severity of CAD by cut-off values of LP-PLA2
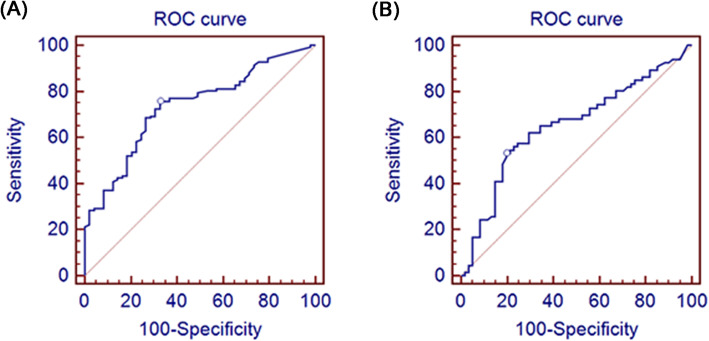
The ROC curve analysis demonstrated the AUC of LP-PLA2 in judging the presence and severity of CAD (Fig. 1). The AUC were 0.734 (Fig. 1a) and 0.652 (Fig. 1b), respectively. LP-PLA2 had 75.6% sensitivity and 67.3% specificity for recognizing CAD, and 53.0% sensitivity and 80.3% specificity for recognizing severe CAD.
Fig. 1.
ROC analysis results for LP-PLA2 in assessing the presence and severity of CAD. a ROC analysis results for LP-PLA2 in assessing the presence of CAD; b ROC analysis results for LP-PLA2 in assessing the severity of CAD
Discussion
The previous study has confirmed that the development of CAD is closely related to the inflammatory response, while the dynamic process of CAD is resulted from the interaction between the inflammatory response and vascular endothelial dysfunction. LP-PLA2 is an enzyme mainly separated from atherosclerotic plaques, which was produced inflammatory cells and circulates in the bloodstream. Due to its important effects on atherosclerosis [14, 15], LP-PLA2 was considered a promising biomarker for the evaluation of CVD risk. Evidence from basic and clinical studies also indicated that decreased LP-PLA2 level played important role in slowing atherosclerosis and cutting down the occurrence of CAD [16, 17].
Some studies have shown that the increase of LP-PLA2 level is closely correlated to the incidence of acute coronary syndrome. At the same time, LP-PLA2 can also predict the recurrence of angina, heart failure, myocardial infarction and other cardiovascular events [18]. And there is evidence that LP-PLA2 has a significant clinical predictive value for the occurrence and development of CAD [19]. Therefore, it is reasonable to speculate that elevated plasma level of LP-PLA2 might contribute to accelerating the progression of coronary artery stenosis as well as atherosclerotic plaque rupture. Results of our cross-sectional research also support the hypothesis.
Previous studies from Shahar Lavi and institution have hinted that the production of LP-PLA2 is correlated to endothelial dysfunction as well as early coronary atherosclerosis [20]. Different from previous studies, our study is the first to explore the correlation between phospholipase A2 and the amount of coronary artery lesions. The results demonstrate that LP-PLA2 level in the SVS and MVS groups was obviously increased compared to the control group. Therefore, we speculated that the more serious the endothelial dysfunction and coronary atherosclerosis, the higher the plasma LP-PLA2 level would be. Although it is regrettable that we did not study the endothelial function of each group, several clinical and basic experiments have confirmed the positive correlation between LP-PLA2 and endothelial dysfunction. Research from Iwase M showed that, in addition to endothelial-associated mechanisms, oxidized fatty acid and hemolytic lecithin play vital roles in the development of atherosclerotic plaques as well [21]. In general, we believe that LP-PLA2 could accelerate atherosclerotic plaque development.
LP-PLA2 can provide important value for the prevention as well as treatment of cardiovascular and cerebrovascular diseases. Data from Berger JS show that an LP-PLA2 inhibitor can significantly ameliorate the occurrence of cardiovascular events compared with a placebo [22]. Daida H also believed that decreasing the level of plasma LP-PLA2 could not only delay the process of atherosclerosis but also decrease the occurrence of cardiovascular events [17]. However, data from the latest clinical trials suggested that using the LP-PLA2 inhibitor darapladib treatment may not decrease the occurrence of CAD [11, 23], which seems to contradict previous research. Although in our research, the plasma level of LP-PLA2 in the AMI and UA groups was remarkably higher than that in the control group after adjustment of several basic data, and the LP-PLA2 level gradually rose with the number of coronary artery lesions, we think this is due to the continuous release of LP-PLA2 from unstable and ruptured plaques. All this indicates that detecting LP-PLA2 activity is useful not only for judging the severity of CAD but also for estimating the severity of coronary atherosclerosis, which is consistent with data from Blankenberg [24]. For the first time, we used ROC curve to assess the diagnostic value of LP-PLA2 for CAD. ROC curve analysis reveals that LP-PLA2 can be used not only to evaluate the occurrence of CAD but also to judge its severity. We speculate that the risk of cardiovascular events will increase with the rising level of LP-PLA2 in plasma.
There are some limitations in this study. First, the time of research was relatively short and the number of cases relatively small, so it still needs a major case supplement. Besides, we did not exclude patients who need to take hypolipidemic drugs and aspirin before drawing blood, which may have had an impact on the results. However, Anping Cai thought that statins do not affect the correlation between LP-PLA2 level and the seriousness of CAD [13]. Finally, there may be cases of that certain patients with non-significant CAD (small lesions) develop unstable angina due to plaque instability, this may have some influence on the research results, however, we believe that the impact is limited.
Conclusions
In conclusion, as a relatively new inflammatory marker, the effect of LP-PLA2 in atherosclerosis is still controversial, but more and more evidence indicates that it plays a an important role in the occurrence and development of coronary atherosclerosis. The current study also shows that LP-PLA2 activity is positively correlated to the seriousness of CAD, and this can provide a strong basis for predicting the occurrence and prognosis of CAD, at the same time, it may provide a new way to treat CAD in the future.
Acknowledgements
To our Nurses’ and PCI surgeons’ team, and technicians in executing this study.
Declarations
Not applicable.
Abbreviations
- CAD
Coronary artery diseases
- CAG
Coronary angiography
- SA
Stable angina
- UA
Unstable angina
- AMI
Acute myocardial infarction
- CVD
Cardiovascular diseases
- CRP
C-reactive protein
- LP-PLA2
Lipoprotein-associated phospholipase A2
- SVS
Single-vessel stenosis
- MVS
Multiple-vessel stenosis
- FBG
Fasting blood glucose
- Lp(a)
Lipoprotein(a)
- TC
Total cholesterol
- TG
Triglyceride
- LDL-C
Low-density lipoprotein-cholesterol
- HDL-C
High-density lipoprotein-cholesterol
- ELISA
Enzyme-linked immune-absorbent assay
- ROC
Receiver operating characteristic
- AUC
Areas under the curve
- OR
Odd ratio
Authors’ contributions
HZ and DZ designed the study. HZ, YG and DZ drafted the manuscript; HZ, YG, and DW collected the clinical information; HZ and DW are responsible for statistics and data analysis of data. The final manuscript was approved by all authors .
Funding
The research was funded by the grant from National Natural Science Foundation of China (Grant Number. 81000058). The fund mainly provides research funding support.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
All the research was approved by the institutional review board of Ethical Committee of Nanjing Medical University. And all the enrolled subjects provided written informed consent. The administrative permissions were acquired by our team to access the data we used in our research.
Consent for publication
Signed consents were obtained from the enrolled patients to use their clinical information and sequencing data for publications.
Competing interests
All authors declare no conflicts of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Hao Zhang and Yang Gao contributed equally to this work.
References
- 1.Garcia de Tena J. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;353(4):429–430. doi: 10.1056/NEJM200507283530425. [DOI] [PubMed] [Google Scholar]
- 2.Ridker PM, Hennekens CH, Buring JE, Rifai N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N Engl J Med. 2000;342(12):836–843. doi: 10.1056/NEJM200003233421202. [DOI] [PubMed] [Google Scholar]
- 3.Moutzouri E, Tsimihodimos V, Tselepis AD. Inflammatory biomarkers and cardiovascular risk assessment. Current knowledge and future perspectives. Curr Pharm Des. 2013;19(21):3827–3840. doi: 10.2174/13816128113199990307. [DOI] [PubMed] [Google Scholar]
- 4.Macphee CH, Nelson J, Zalewski A. Role of lipoprotein-associated phospholipase A2 in atherosclerosis and its potential as a therapeutic target. Curr Opin Pharmacol. 2006;6(2):154–161. doi: 10.1016/j.coph.2005.11.008. [DOI] [PubMed] [Google Scholar]
- 5.Zalewski A, Macphee C. Role of lipoprotein-associated phospholipase A2 in atherosclerosis: biology, epidemiology, and possible therapeutic target. Arterioscler Thromb Vasc Biol. 2005;25(5):923–931. doi: 10.1161/01.ATV.0000160551.21962.a7. [DOI] [PubMed] [Google Scholar]
- 6.Tousoulis D, Papageorgiou N, Androulakis E, Stefanadis C. Lp-PLA2--a novel marker of atherosclerosis: to treat or not to treat? Int J Cardiol. 2013;165(2):213–216. doi: 10.1016/j.ijcard.2012.09.210. [DOI] [PubMed] [Google Scholar]
- 7.Ballantyne CM, Hoogeveen RC, Bang H, Coresh J, Folsom AR, Heiss G, et al. Lipoprotein-associated phospholipase A2, high-sensitivity C-reactive protein, and risk for incident coronary heart disease in middle-aged men and women in the atherosclerosis risk in communities (ARIC) study. Circulation. 2004;109(7):837–842. doi: 10.1161/01.CIR.0000116763.91992.F1. [DOI] [PubMed] [Google Scholar]
- 8.Lind L, Simon T, Johansson L, Kotti S, Hansen T, Machecourt J, Ninio E, et al. Circulating levels of secretory- and lipoprotein-associated phospholipase A2 activities: relation to atherosclerotic plaques and future all-cause mortality. Eur Heart J. 2012;33(23):2946–2954. doi: 10.1093/eurheartj/ehs132. [DOI] [PubMed] [Google Scholar]
- 9.Katan M, Moon YP, Paik MC, Wolfert RL, Sacco RL, Elkind MS. Lipoprotein-associated phospholipase A2 is associated with atherosclerotic stroke risk: the northern Manhattan study. PLoS One. 2014;9(1):e83393. doi: 10.1371/journal.pone.0083393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Investigators S, White HD, Held C, Stewart R, Tarka E, Brown R, et al. Darapladib for preventing ischemic events in stable coronary heart disease. N Engl J Med. 2014;370(18):1702–1711. doi: 10.1056/NEJMoa1315878. [DOI] [PubMed] [Google Scholar]
- 11.O'Donoghue ML, Braunwald E, White HD, Lukas MA, Tarka E, Steg PG, et al. Effect of darapladib on major coronary events after an acute coronary syndrome: the SOLID-TIMI 52 randomized clinical trial. Jama. 2014;312(10):1006–1015. doi: 10.1001/jama.2014.11061. [DOI] [PubMed] [Google Scholar]
- 12.Silber S. ESC guidelines 2019 on chronic coronary syndrome (CCS, previously "stable coronary artery disease") : What is new? What is particularly important? Herz. 2019;44(8):676–683. doi: 10.1007/s00059-019-04862-6. [DOI] [PubMed] [Google Scholar]
- 13.Cai A, Li G, Chen J, Li X, Li L, Zhou Y. Increased serum level of Lp-PLA2 is independently associated with the severity of coronary artery diseases: a cross-sectional study of Chinese population. BMC Cardiovasc Disord. 2015;15(1):14. doi: 10.1186/s12872-015-0001-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wilensky RL, Macphee CH. Lipoprotein-associated phospholipase a (2) and atherosclerosis. Curr Opin Lipidol. 2009;20(5):415–420. doi: 10.1097/MOL.0b013e3283307c16. [DOI] [PubMed] [Google Scholar]
- 15.Cai A, Zheng D, Qiu R, Mai W, Zhou Y. Lipoprotein-associated phospholipase A2 (Lp-PLA (2)): a novel and promising biomarker for cardiovascular risks assessment. Dis Markers. 2013;34(5):323–331. doi: 10.1155/2013/432136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mohler ER, 3rd, Ballantyne CM, Davidson MH, Hanefeld M, Ruilope LM, et al. The effect of darapladib on plasma lipoprotein-associated phospholipase A2 activity and cardiovascular biomarkers in patients with stable coronary heart disease or coronary heart disease risk equivalent: the results of a multicenter, randomized, double-blind, placebo-controlled study. J Am Coll Cardiol. 2008;51(17):1632–1641. doi: 10.1016/j.jacc.2007.11.079. [DOI] [PubMed] [Google Scholar]
- 17.Daida H, Iwase T, Yagi S, Ando H, Nakajima H. Effect of darapladib on plasma lipoprotein-associated phospholipase A2 activity in Japanese dyslipidemic patients, with exploratory analysis of a PLA2G7 gene polymorphism of Val279Phe. Circ J. 2013;77(6):1518–1525. doi: 10.1253/circj.CJ-12-0813. [DOI] [PubMed] [Google Scholar]
- 18.Lp PLASC, Thompson A, Gao P, Orfei L, Watson S, Di Angelantonio E, et al. Lipoprotein-associated phospholipase a (2) and risk of coronary disease, stroke, and mortality: collaborative analysis of 32 prospective studies. Lancet. 2010;375(9725):1536–1544. doi: 10.1016/S0140-6736(10)60319-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.May HT, Horne BD, Anderson JL, Wolfert RL, Muhlestein JB, Renlund DG, Clarke JL, Kolek MJ, Bair TL, Pearson RR, et al. Lipoprotein-associated phospholipase A2 independently predicts the angiographic diagnosis of coronary artery disease and coronary death. Am Heart J. 2006;152(5):997–1003. doi: 10.1016/j.ahj.2006.01.011. [DOI] [PubMed] [Google Scholar]
- 20.Lavi S, Mcconnell JP, Rihal CS, Prasad A, Mathew V, Lerman LO, et al. Local production of lipoprotein-associated phospholipase A2 and Lysophosphatidylcholine in the coronary circulation association with early coronary atherosclerosis and endothelial dysfunction in humans. Circulation. 2007;115(21):2715–2721. doi: 10.1161/CIRCULATIONAHA.106.671420. [DOI] [PubMed] [Google Scholar]
- 21.Iwase M, Sonoki K, Sasaki N, Ohdo S, Higuchi S, Hattori H, et al. Lysophosphatidylcholine contents in plasma LDL in patients with type 2 diabetes mellitus: relation with lipoprotein-associated phospholipase A2 and effects of simvastatin treatment. Atherosclerosis. 2008;196(2):931–936. doi: 10.1016/j.atherosclerosis.2007.02.012. [DOI] [PubMed] [Google Scholar]
- 22.Berger JS, Ballantyne CM, Davidson MH, Johnson JL, Tarka EA, Lawrence D, et al. Peripheral artery disease, biomarkers, and darapladib. Am Heart J. 2011;161(5):972–978. doi: 10.1016/j.ahj.2011.01.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Buszman P. Darapladib for preventing ischemic events in stable coronary heart disease — NEJM. New Eng J Med. 2014;370(18):1702–1711. doi: 10.1056/NEJMoa1315878. [DOI] [PubMed] [Google Scholar]
- 24.Blankenberg S, Stengel D, Rupprecht HJ, Bickel C, Meyer J, Cambien F, et al. Plasma PAF-acetylhydrolase in patients with coronary artery disease results of a cross-sectional analysis. J Lipid Res. 2003;44(7):1381–1386. doi: 10.1194/jlr.M300086-JLR200. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.