Abstract
The term autologous fecal microbiota transplantation (a-FMT) refers herein to the use of one's feces during a healthy state for later use to restore gut microbial communities after perturbations. Generally, heterologous fecal microbiota transplantation (h-FMT), where feces from a ``healthy” donor is transplanted into a person with illness, has been used to treat infectious diseases such as recurrent Clostridioides difficile infection (CDI), with cure rates of up to 90%. In humans, due to limited response to medicines, h-FMT has become a hallmark intervention to treat CDI. Extrapolating the benefits from CDI, h-FMT has been attempted in various diseases, including inflammatory bowel disease (IBD), but clinical response has been variable and less effective (ranging between 24% and 50%). Differences in h-FMT clinical response could be because CDI is caused by a Clostridial infection, whereas IBD is a complex, microbiome-driven immunological inflammatory disorder that presents predominantly within the gut wall of genetically-susceptible hosts. FMT response variability could also be due to differences in microbiome composition between donors, recipients, and within individuals, which vary with diet, and environments, across regions. While donor selection has emerged as a key factor in FMT success, the use of heterologous donor stool still places the recipient at risk of exposure to infectious/pathogenic microorganisms. As an implementable solution, herein we review the available literature on a-FMT, and list some considerations on the benefits of a-FMT for IBD.
Abbreviation: a-FMT, autologous fecal microbiota transplantation; CD, Crohn's disease; CDI, Clostridium difficile infection; CI, Confidence interval; FMT, Fecal microbiota transplantation; hGM, human gut microbiota; h-FMT, heterologous fecal microbiota transplantation; IBD, inflammatory bowel disease; IBS, irritable bowel syndrome; RCT, randomized controlled trial; UC, ulcerative colitis
Introduction
The human gut microbiota (hGM) is comprised of bacteria, viruses, fungi, protozoa and archaea, which collectively, may have functionally distinct proinflammatory or anti-inflammatory roles, as described in animal models.1 Alterations in the compositional balance of microbial communities, also termed “dysbiosis,” has been hypothesized to play a central role in human diseases. However, the causal connection between the gut microbiome and the pathogenesis of human diseases remains uncertain, as pathogenicity studies with animal models have been deemed implausibly biased, “exaggerating causality.”2
Irrespective of the complexity of understanding of the role of the gut microbiome in complex multimodal diseases,3 fecal microbiota transplantation (FMT), where feces from a `healthy” donor are transplanted into a patient (``heterologous-FMT”; h-FMT), has been increasingly investigated as a treatment option to help restore the functional balance between pro- and anti-inflammatory microbial communities. In clinical practice, h-FMT has proven highly successful for the treatment of recurrent Clostridioides difficile infection (CDI), with cure rates of up to 90%.4 , 5 By contrast, the clinical response rate from h-FMT in patients with inflammatory bowel disease (IBD) has been less impressive (clinical remission ranging from 24% to 50%).6 Moreover, some studies have reported a worsening of IBD activity in patients following transplantation of ``healthy” donor feces.7
Studies indicate that FMT response in IBD depends on the composition of the donor microbiome,8 , 9 leading to the perhaps overly simplistic notion of ``super-donors,” which by definition, are people who have gut microbiota that induce a beneficial response in numerous individuals when heterologously transplanted into patients.10 While identification and characterization of ``super-donors, could help researchers to refine h-FMT formulations and improve clinical efficacy, the use of heterologous stool does not prevent the infectious/safety concerns of h-FMT. Thus, there is a need for clinical rationalization and solutions to the causes of poor FMT response in IBD, while addressing safety concerns.
Current h-FMT donor screening practices focus on safety, by excluding known pathogens, however, the long-term safety of the procedure remains unclear. Microbiological safety in IBD is relevant considering the frequent use of corticosteroid, immunomodulators, and anti-TNF-alpha antibody therapy in these patients.11 , 12 Especially, after the recent FDA report of mortality and morbidity in immunocompromised patients due to an FMT-acquired enteropathogenic Escherichia coli infection, as well as other reports of Shigatoxin-producing Escherichia coli, following the investigational use of FMT.13 More recent studies documenting the presence of the respiratory SARS-CoV-2 (COVID-19) virus in the stool of infected individuals,14, 15, 16, 17, 18 also further illustrate the need for proper screening and selection of donor stool for FMT. Since the presence of pathogens in donors could alter/influence the response in patients,19 , 20 improved strategies to avoid the risk of unwanted infections are needed.
FMT has been proposed to revert dysbiosis in the gut microbial community of the patient, however, it is unclear whether dysbiosis is a cause or a consequence of IBD. Recently, studies of the long-term dynamics of the IBD microbiome indicate that the gut microbiome transitions over time between dysbiotic and healthy states.21 In this context, our studies with germ-free animals transplanted with hGM experimentally demonstrated that the effect of FMT response in mice varies across donors, and it is person-specific, and not IBD-specific.22 , 23 Since FMT response variability is presumed due to factors intrinsic to the person as a donor (genetics, diet, other environmental exposures),24 there is a need to control for such factors, and most importantly to further study the therapeutic potential for autologous FMT (a-FMT). The implementation of a-FMT, where the patients’ own stool is banked during a time of ``health” (eg, remission, when a patient is not experiencing symptoms), as an alternative solution to circumvent the risks associated with heterologous donor stool, could be a valuable clinical strategy to improve FMT response rates, while fully abolishing infectious/safety concerns. Herein, we review the benefits and considerations for a-FMT in IBD. Where possible, we report confidence intervals (CIs) to facilitate interpretation of data.
Overall clinical effect of FMTs
For the treatment of recurrent and refractory CDI, h-FMT has proved to be a highly clinically effective and cost-effective therapy, with a meta-analysis of studies reporting a 92% (95% CI, 89%–94%) clinical resolution rate compared to the use of one of the first choice antibiotics against CDI, vancomycin (relative risk:0.23, 95% CI = 0.07–0.80).4 Differences in CDI cure rates have, however, been observed between the lower (enema-colonic) vs upper (oral-gastric) gastrointestinal tract delivery (95% [95% CI = 92%–97%] vs 88% [95% CI = 82%–94%], respectively).4 Even for the treatment of CDI in patients with underlying IBD, arguably comparable FMT success rates have been reported, with a recent meta-analysis showing an initial cure rate of 81% (95% CI = 76%–85%), and overall cure rate of up to 89% (95% CI = 83%–93%).25
The treatment of IBD with h-FMT to induce clinical remission has, however, been less effective,26, 27, 28 with clinical remission rates ranging from 24% to 50%.6 In a recent meta-analysis,26 encompassing 53 studies, only 36% (201/555, 95% CI = 32.2%–40.4%) of UC and 50.5% (42/83, 95% CI = 39.4%–61.8%) of CD patients achieved clinical remission (primary outcome, as described by the respective study authors). Heterogeneity was moderate in the meta-analysis of cohort studies and that of the four included randomized controlled trials (RCTs) in UC. Of concern, some studies have reported worsening of IBD activity following h-FMT, with a recent meta-analysis revealing a rate of worsening, defined to include escalation of medical therapy or surgical intervention after FMT, of 14% (95% CI = 10.0%–21.0%).7 Such discrepancies in h-FMT success could be due to temporal and spatial variability of the hGM for both donors and recipient populations.29
The effectiveness of FMT has been mostly examined using microbiome sequencing technology, primarily 16S rRNA gene sequencing (used to study bacteria), with less studies focusing on the role of nonbacterial microbiome members (viruses, fungi). Positive clinical outcomes in IBD have been associated with donor microbiome composition, specifically higher bacterial richness,8 , 26 , 30, 31, 32, 33, 34 as well as increased relative abundance in butyrate-producing Clostridium clusters IV and XIVa (eg, genera from the Ruminococcaceae and Lachnospiraceae family).8 , 9 , 30 , 35, 36, 37, 38 However, analysis of microbiome data from three completed IBD h-FMT trials39, 40, 41 showed there was no association between the presence and abundance of butyrate-producers in the feces of donors and the clinical response after h-FMT, suggesting that butyrate alone is not sufficient as a predictor for FMT efficacy.42 In addition to the microbiota, some studies have suggested that the donor virome (``phagebiota”) and fungal composition43 , 44 play a critical role in determining h-FMT success,44, 45, 46, 47 although this appears to be disease specific, with studies demonstrating that phages are more relevant to FMT efficacy in CDI (eg, Caudovirales),43 , 48 compared to the presence of eukaryotic viruses in donor stool for UC.49 In addition to the relevance of the donor microbiome, studies showed that the gut microbiome composition in the recipient also plays a role in their clinical response to h-FMT.50 , 51 Thus, antibiotic pretreatment of recipient patients has been effectively used to eliminate the potential competitive advantage of existing microorganisms facilitating the colonization by microbes present in the transplanted feces.52, 53, 54
To improve FMT response, there is a need to control for person/microbiome variation. In FMT studies for CDI, interindividual heterogeneity in donor stools do not seem to affect clinical response (cure) rates, and thus, does not play a role in donor selection.55 , 56 However, in the case of more complex diseases, such as IBD, which involves a multi-faceted interaction between host and microbiome, donor stool composition is emerging as one of the most important factors in determining poor/variable patient response.8, 9, 10 , 22 , 42 Supporting this, results of several h-FMT trials in IBD patients (active UC) have demonstrated significant treatment benefit from some donor stool, but not others.8 , 9 Highlighting donor-dependence in h-FMT success, a double-blind RCT conducted by Moayyedi et al8 in patients with active UC (where stools from 5 different donors vs placebo was used), showed that 78% of patients (7 out of 9) achieved remission (Mayo score ≤2 with an endoscopic Mayo score of 0 at week 7) from a single donor, while only 10% of patients achieved remission with the other donors, with 1 donor eliciting no success at all. These findings highlight that not all ``healthy” people are ``good” donors, or that their microbiomes can be considered comparably ``anti-inflammatory,” and further emphasize the variable nature of the human gut microbiome. Furthermore, findings emphasize the importance of proper selection of donors to improve the likelihood of FMT clinical response, if heterologous stool is to be preserved as the central strategy for microbiome therapeutics. Notably, autologous reconstitution of the gut microbiota has proven effective to restore depleted commensals following antibiotic treatment57 , 58 and is often used as the placebo FMT treatment in RCTs on the basis that it is most physiologically relevant.59
Autologous-FMT as placebo for heterologous-FMT research vs. enhanced clinical benefits
Autologous FMTs have been used for clinical and research purposes. To date, the primary clinical benefit of using autologous samples is that it circumvents current safety concerns regarding infectious pathologies, an important concern, especially in high-risk populations such as the elderly, those with co-morbidities and immunocompromised patients. For instance, in hematopoietic stem cell transplantation patients, a high-risk population due to immunodeficiency, autologous reconstitution of the gut microbiota after antibiotic therapy, using feces banked prior to initiation of antibiotic therapy, has proven highly effective.60 , 61 Thus far, postantibiotic microbial reconstitution of the gut microbiome via a-FMT has been shown more effective than probiotics.57 Here, a-FMT served as a safe and rapid approach to restoring depleted ``commensal” taxa and other community-level metabolic deficiencies that are associated with pathogen/indigenous microbial recolonization during the vulnerable period after antibiotic consumption.57 , 58
Current guidelines for RCT trials for FMT recommend the use of autologous transplants (``a-FMT”) as a placebo ``control” intervention treatment because it is assumed that autologous stool represents the most inert and physiologically similar research option compared to donor feces.59 However, it is important to highlight that often RCTs collect samples from the recipient at the time of investigational FMT treatment, indicating that if patients are sick/ill at the time of FMT, their own sample would be unhealthy (presumably dysbiotic) and unable to have a beneficial effect. Therefore, most RCTs would be biased against autologous transplants as a therapeutic option, favoring h-FMT. In this context, our studies in germ-free animal model of IBD indicate that the feces of IBD patients in complete remission may have proinflammatory or anti-inflammatory potential, depending on the donor, and therefore autologous transplantation require further re-consideration for clinical purposes, well beyond the current use for research control purposes.
Several studies have used autologous transplants, collecting feces from the patient at time of treatment (when patient is presumed sick), as the comparative placebo control treatment to donor feces. Their findings highlight the therapeutic potential for a-FMT via two key concepts. First, the microbiome of a sick person can deviate from an ``inflammatory” (dysbiotic) state to an ``anti-inflammatory” (healthy) state. Second, the microbiome of a healthy individual can deviate from an ``anti-inflammatory” (healthy) state to that of ``inflammatory” state of dysbiosis, during which, FMT would result in a suboptimal clinical response.
In RCTs for irritable bowel syndrome (IBS) where h-FMT has been compared to autologous transplants as the control intervention, trials have provided dichotomous FMT clinical response data regarding the proportion of h-FMT treated patients who achieved a successful outcome, compared to the FMT response reported in those treated with autologous stool.62 Table 1 summarizes data from available RCT, and illustrates that in most cases, although there is overlap of the predicted effects for h-FMT vs a-FMT (notice 95% CI lower and upper limits), often, autologous had a lower response rate, indicating that patients often recover on their own, without the need of FMT. However, the increased proportion of responsive patients to treatment indicate that FMT holds benefits beyond the basic baseline ability of patients to recover on their own. In this regard, the improvement of FMT by using a well-designed autologous fecal transplantation program, where banking of one's feces occurs during a healthy state for later use to restore gut microbial communities after perturbation, would improve the rate of responses in patients.
Table 1.
Summary of outcomes from RCTs using feces for autologous-FMT collected at time of investigation for use as placebo vs heterologous-FMT
| Study design |
Achieved clinical benefit/remission |
|||||
|---|---|---|---|---|---|---|
| Author | Case/control | Disease | Donor screening | Primary outcome and follow up | Heterologous-FMT (95%CI) | Autologous-FMT (95%CI) |
| Johnsen91 IBS |
55/28 | IBS-D 53%, IBS-M 47% |
pooled; 2 healthy volunteers |
decrease in IBS-severity scoring system >75 points at 12 wks. R: colonoscopy, once |
3 mo: 65% (51.4%–77.8%)* 12 mo: 56% (42.3%–69.7%)* |
3 mo: 43% (24.5%–62.8%)* 12 mo: 35.7% (18.6%–55.9%)* |
| Holvoet92 IBS |
42/22 | 100% IBS‐D or IBS‐M | unpooled; 2 healthy donors |
self-reported questionnaire on symptoms at 12 wks. R: nasojejunal, once. |
50% (34.2%–65.8%)* | 29% (13.9–54.9)* |
| Holster93 IBS |
8/8 | IBS‐C 25.0%, IBS‐D, 56.2% IBS‐M 18.8% |
unpooled; healthy donor | decrease in gastrointestinal symptom rating scale‐IBS of ≥30% at 2 and 8 wks. R: colonoscopy, once |
50% (15.7–84.3). Note: 66% (2/3) responded to ``donor 1” while 40% (2/5) responded to ``donor 2”. |
12.5 (0.3%–52.7%) |
| Kelly63 CDI |
22/24 | ≥3 CDI recurrences; course of vancomycin received |
unpooled; healthy volunteers | resolution of diarrhea w/out need for further anti-CDAD therapy at 8 wks. R: colonoscopy |
90.9% (70.8%–98.9%) | 62.5% (40.6%–81.2%) Resolution after autologous FMT differed by site. |
| Rossen65 UC |
23/25 | Mild to moderate UC | unpooled; healthy subjects (partner, friend, volunteer) | clinical remission and endoscopic response at 12 wks. R: nasoduodenal, wk 0 and 3. |
At 6-wk: 26.1% (10.2%–48.4%)* 12-wk: 30.4% (13.2%–52.9%)* |
6-wk: 32% (14.9%–53.5%)* 12-wk: 32.0% (14.9%–53.5%)* |
| Costello66 UC |
38/35 | Mild to moderate UC | pooled; healthy volunteers. |
steroid-free remission and 8 wks and 12 mo. R: colonoscopy followed by 2 enemas over 7 days. |
32% (17.5–48.7)* | 9% (1.8–23.1)* |
R, route of administration and frequency of transplantation; mo, month; wk, week.
95%CI computed based on reported N/N using normal approximation to the binomial calculation.
Exemplifying the variability of healthy human microbiomes, a meta-analysis of 5 RCTs for IBS showed no significant difference in IBS symptom improvement between h-FMT compared to placebo treatment encompassing autologous transplants or ‘inert’ capsules (RR = 0.98, 95% CI = 0.58–1.66); however, subgroup analysis showed that ``inert” placebo capsules were superior to h-FMT (2 pooled trials; RR = 1.96; 95% CI = 1.19–3.20), but, when administered via colonoscopy, h-FMT was superior to autologous stool (2 pooled trials; RR = 0.63; 95% CI = 0.43–0.93).62 By comparison, a RCT of FMT for CDI demonstrated that, although the overall FMT cure rate for a-FMT patients was significantly lower (62.5%, 15/24 [95% CI = 41.6%–79.6%]) to that of h-FMT patients (90.9%, 20/22 [95% CI = 69.2%–97.8%]), cure rates differed by medical center, with one site achieving a cure rate of 90% (95% CI = 51.8%–98.7%) via autologous transplants compared to 91.7% (95% CI = 57.2%–98.9%) via donor stool (``h-FMT”).63 Of note, comparison of patient cohorts by site showed that the site with the lower autologous stool cure rate had a significantly higher proportion of patients reporting prior probiotic treatment, although how this may have affected the observed differences in the pre- and post-FMT microbiome profiles between treatment groups by site was unclear.
Of the limited number of RCTs conducted for FMT in IBD (UC) to date,8 , 9 , 64, 65, 66 only two have used a-FMT as the placebo comparator.65 , 66 Using h-FMT vs a-FMT for treatment of mild-to-moderate UC, Roosen et al,65 suggested that autologous stool could be as effective as healthy donor feces in inducing clinical remission (a-FMT 32%, 8/25 [95% CI = 14.9%–53.5%] vs h-FMT 26.1%, 6/23 [95% CI = 10.2%–48.4%]). Microbiome analysis showed no significant difference in baseline gut microbiota composition between recipient patient groups, whereas a significantly difference in donor and UC patient stool composition was identified. However, at 12 weeks of treatment, microbiome diversity was significantly increased in all FMT ``responders” (UC patients who achieved clinical and endoscopic remission) irrespective of whether donor or autologous stool was used. Analytically, a-FMT responders shifted away from nonresponders, but in a different direction to that of h-FMT responders, suggesting differences in the post-FMT effect on microbiota composition and clinical response when donor and autologous stool is used. By comparison, the recent RCT of Costello et al66 in patients with mild-to-moderate UC found FMT achieved remission in 32% (12/38, 95% CI = 17.5%–48.7%) of h-FMT compared to 9% (3/35, 95% CI = 1.8%–23.1%) in a-FMT patients. From a methodological perspective, it is important to make a distinction of how a-FMT samples intended for placebo purposes have been handled differently to that of h-FMT stool samples. In their study,66 donor stools intended for the h-FMT groups were prepared anaerobically (pooled from 3 to 4 donors at 16 different collection time points; from the 16 batches produced, each provided treatment for 1–7 participants), whereas, the autologous stool intended to be used as placebo a-FMT were processed aerobically (from a single time point), highlighting the potential for study and selection/test bias against autologous stool for FMT. Such fundamental methodological difference introduces a confounding factor (reducing the anaerobic diversity in feces for the a-FMT group) favoring the presence of anaerobic bacteria/microorganisms in stools intended for use in the h-FMT group of patients.
Conceptually, the clinical benefit observed in a-FMT placebo groups, where feces were collected from the patient at time of treatment (when patient is sick), suggest that a patients’ own feces could hold therapeutic potential for administration during a disease flare in some patients if a fecal sample were to be collected/stored during a time of ``health” (eg, remission, when a patient is not experiencing symptoms).
Considerations on the selection of stools for a-FMT
To date, there are no established guidelines or clear microbial/clinical parameters for the identification of appropriate donors and standardization of donor stool selection for h-FMT, or alternatively, for the ‘optimal’ timing for the collection and functional selection of feces intended for a-FMT. Since the IBD microbiome has been shown to transition from ``inflammatory” (dysbiosis) to ``anti-inflammatory” (healthy microbiome),21 , 29 the observed benefit in the discussed RCTs support the need for proper selection of feces for transplantation.
While some studies have used single donor h-FMT, the variability in patient response based on donor stool has elicited others to consider the use of multiple, pooled donor stool inoculum. Pooling of donor stool has been proposed as a method to improve remission rates by limiting the chance for a patient to receive an ``ineffective”/``inflammatory” stool sample.67 However, clinical benefit in RCTs for IBD has been reported when multidonor-pooled9 , 66 and single-donor-unpooled h-FMT protocols were used,8 irrespective of the intensity/duration of FMT administration, stool preparation methods (anaerobic vs anaerobic, fresh vs frozen), or if donor stool came from healthy partners, relatives, or healthy volunteers. Specifically, in a double blind placebo controlled RCT9 where an intensive dosing approach was combined with a multidonor pooling for patients with mild-to-moderate UC, post hoc analysis revealed a higher remission rate in patients treated with pooled h-FMT batches that contained stool from 1 specific donor compared to recipients whose h-FMT batches did not contain stool from that donor (37%, 14/38 [95% CI = 21.8–54.0] vs 18%, 7/40 [95% CI = 0.73–32.8], respectively). While findings are not surprising considering that multidonor pooling using a set of randomly selected donors will not ensure the inclusion of an ``effective” (anti-inflammatory) donor, or ensure the ability for one (or more) included ``effective” donors to ``outweigh” the effect of other ``ineffective” donors in a pooled sample, findings do emphasize that pooling of donor stool does not predict a reproducible h-FMT response. Moreover, it is less likely that the ``effective” donor microbiome will stay intact due to increased microbial interactions/incompatibilities which occur between transplanted microbiomes.32 , 34 The problem of pooling samples is that it increases the risk of having a pool of potential pathogens that cannot be traced back to the donor, and therefore does not improve the safety of FMT compared to h-FMT from a single donor or the use of a-FMT (one/self-donor).
Diet compatibility has been proposed as a method to improve donor-recipient matching and durability of FMT response,68 assuming individuals with the same diet would have compatible microbiota, and future trials should collect dietary data for better understanding of the role of donor diet on FMT durability post-FMT. In this regard, metabolic profiling could be used to guide the selection of fecal samples for FMT. Taking into consideration the temporal, variable behavior of the human gut microbiome29 and the variability of FMT response, future stool selection strategies could consider more refined profiling methods such as metagenomics to determine community-level functionality of the banked stool and those currently deficient in the recipient, in combination with direct metabolite measurement (eg, SCFA, secondary bile acids69) which could serve as a superior proxy of community function. However, in the case of fecal samples for h-FMT, such methods would not account for host-dependent differences in absorption rates of target molecules (eg, butyrate), or other donor-dependent factors such as dietary habits, which are more likely to differ from those of the recipient.42 Given that diet can change microbiome profiles in as little as 24 hours,70 FMT protocols, whether using donor stool or the patients’ own feces, may benefit from using dietary strategies both prior to stool banking and following stool administration, to support the transplanted microbiome, potentially extending the sustainability of the post-FMT microbiota.71
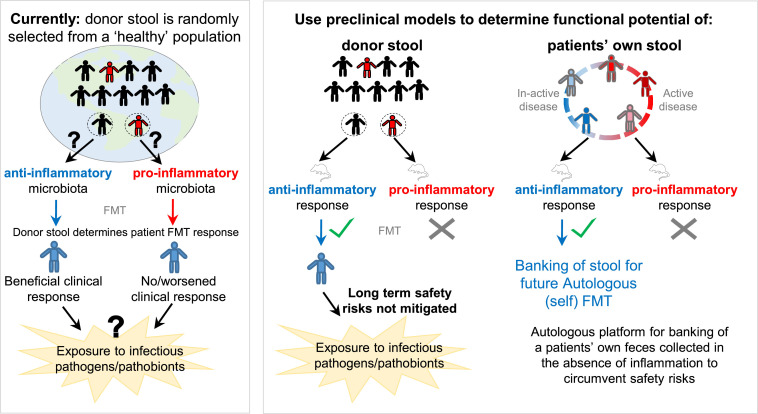
Functional assays, such as fecal calprotectin and myeloperoxidase activity, are used in IBD (humans, animals) to assess underlying host intestinal inflammation.72, 73, 74 Previous preclinical studies have demonstrated that alterations in the gut microbiota, favoring proinflammatory communities, can sensitize mice to the development of several inflammatory diseases.75 , 76 hGM-transplanted mouse models of chemically-induced colitis have also been widely used to elucidate the functional role of hGM FMT in IBD.1 Thus, interested in using a functionality-based method approach for selection of donor stool, we conducted inflammation-based tests on germ-free animals. Our preclinical studies using germ-free mice transplanted with hGM support the notion that the IBD microbiome can periodically transition away from an ``inflammatory,” dysbiotic state to that resembling an ``anti-inflammatory” healthy donor. Using feces from IBD (in clinical remission) and healthy control donors transplanted into a germ-free mouse model of spontaneous cobblestone/ileal Crohn's disease (SAMP1/YitFc), we showed (with optimal, >90% engraftment of human taxa) that the hGM-FMT effect on mouse IBD-phenotype was independent of the disease state of the donor.22 That is, feces from some IBD and some healthy donors did not affect the severity of intestinal inflammation in mice, while the other donors worsened inflammation. Comparably, Baxter et al23 demonstrated, using a hGM-FMT mouse model of chemically induced colorectal cancer (CRC), that feces from some CRC patients can have an anti-inflammatory tumorigenesis effect in mice, whereas the opposite effect was seen with some healthy donors. Results highlight that the distinct, person-specific structure of the microbiome, and not the disease status of the individual (CRC patient vs healthy control) is associated with the functional effect of hGM FMT. Furthermore, results collectively emphasize that selection of fecal samples, regardless of whether for a-FMT or h-FMT, need to be tested using a functional assay, for instance preclinical hGM mouse models. Fig 1 provides a schematic overview of the current vs the approach herein proposed for FMT stool selection, whereby preclinical mouse models serve as a functional tool to determine the ``inflammatory” potential of human feces for heterologous and autologous FMT.
Fig 1.
Schematic overview of the current vs. the proposed approach for selection of FMT stool. Figure illustrates that the current approach to donor stool selection for FMT perpetuates the unpredictability of patient clinical response in IBD, as well as the risk of exposure to infectious pathogens/pathobionts. By comparison, preclinical mouse models could help researchers determine the functional potential of a fecal sample prior to transplantation. By doing so, the likelihood of a beneficial clinical response to donor feces would improve, and, if used to test a patients’ own feces (for use as autologous FMT), potentially abolish current safety concerns of the procedure.
Collectively, preliminary preclinical and human evidence supports two concepts. The first is the use of hGM mouse models as a screening tool for hGM ``inflammatory potential,” to help guide selection of ``anti-inflammatory” donor stool for use in FMT clinical studies, as a complimentary approach to current donor screening procedures. The second is the use of such preclinical hGM models for identification of ``anti-inflammatory” patient feces for a-FMT. That is, the use of hGM FMT rodent models to determine whether a fecal sample, isolated in the absence of inflammation, for instance, from an IBD patient during times of remission, holds ``anti-inflammatory” potential as a therapy for autologous (self) transplantation during times of a disease flare.22 Similarly, such models could help guide personalized therapies, tailored to the individual patient, that will redirect the gut microbiome toward an ``anti-inflammatory” healthy state. Despite the benefits of using mice as functional avenues to guide FMT therapy, their implementation in clinical settings is expected to be challenging and/or possibly unrealistic at large scale.
Autologous transplants to reduce safety/infectious concerns for h-FMT
Despite generally low rates of reported adverse events, the most limiting caveat about FMT using whole donor stool is the concern of long-term safety of the procedure.77 By contrast, autologous transplantation circumvents the potential for transmission of both communicable and noncommunicable diseases, as well the transfer of yet to be discovered microbial drivers of disease present in the gut microbiota. Autologous FMT also reduces the potential for donor-recipient microbial-interactions/incompatibilities which have been shown to influence strain engraftment outcomes.32 , 34
Current FMT donor selection procedures rely on available technologies for infectious (eg, cytomegalovirus78) and pathogenicity screening,79 including the exclusion of donors testing positive for known antibiotic-resistant bacteria,68 among several other factors.13, 14, 15, 16 , 59 However, in an attempt to minimize the risk of infectious pathogen transmission, and the notion that a similarity in microbial composition might be more tolerated by the mucosal immune system of the patient,28 most studies have selected h-FMT donors based on their genetic and environmental proximity (healthy adults or children family members) to the recipient.80
One highly relevant, example of exposure to unwanted life-threatening infectious pathogens present in donor stool is the recently issued FDA safety alert based on the documented presence of the respiratory causing virus SARS-CoV-2 (COVID-19) in the stool of infected individuals,14, 15, 16 and in turn, the possible risk of transmission via FMT.17 To avoid such risk, the FDA recommends using stool donated before December 1, 2019. For stool donated after this date, the FDA recommends additional precautions should be taken, including, (1) the inclusion of donor screening questions related to COVID-19 infection, (2) the testing of both donors and their stool for COVID-19 (as feasible), (3) development of criteria for exclusion of donors and donor stool based on screening and testing, and (4) to include information pertaining to the potential risk of transmission of the virus in the informed consent.18
Several preclinical studies in mice transplanted with hGM have demonstrated the transfer of host phenotype through microbiota transplantation.51 , 81, 82, 83, 84 In this context, the potential for transmission of non-infectious diseases associated with microbially-derived metabolites present in donor stools has been hypothesized.85, 86, 87 In particular, the association between dietary animal protein and trimethylamine N-oxide (TMAO) production, a bacterially-derived metabolite associated with cardiovascular risk modulation.88 While machine learning has been proposed as a method to identify complex community signatures in healthy donor stool that could reproducibly distinguish/predict ``effective” donors and a phenotype/clinical response of interest,32 , 42 , 89 , 90 such computational approach does not address current safety concerns of h-FMT.
Limitations of therapeutic benefit of FMT in IBD
The challenging component of a program of a-FMT is the implementation of a functional testing platform to enable the accurate identification of ``anti-inflammatory” fecal samples from an IBD patient because the nature of their disease makes them prone to be often inflamed, even in remission, as we showed.22 Having a well-documented bank of autologous samples could be implementable among patients soon after diagnosis as a scalable measure to use therapeutic a-FMT, however, this may not be feasible for all facilities given the considerable resources required to maintain stool banking programs.
Although a-FMT holds therapeutic potential, it is possible that with the multi-factorial nature of IBD, a patients’ own stool would not be able to effectively treat 100% of cases. IBD is a multimodal disease3 (not infectious, and more predictable and responsive to FMT as with CDI) for which the complex nature of disease severity, including the person-specific temporal and phenotypic variability in gut microbiota, are analytically challenging to study and interpret. We have extensively discussed the multimodality of disease subtypes underlying IBD, from analytical perspective, elsewhere.3
Conclusion
Autologous-FMT is preferable to h-FMT to prevent infectious complications, however, there is a need to identify functionally optimal stool samples based on inflammation relevant to IBD. The implementation of a-FMT patient stool banks using functional assays, for instance hGM mouse models, to identify/confirm the anti-inflammatory potential of a fecal sample for use during a disease flare could be more clinically beneficial than screening banked fecal samples from multiple randomly selected healthy donors when needed.
ACKNOWLEDGMENTS
Conflicts of Interest: Authors declare that there are no conflicts of interest or competing financial/non-financial interests to disclose. We confirm that this work is original and has not been published as peer-reviewed material elsewhere, nor is it currently under consideration for publication elsewhere.
Funding: No financial support to disclose.
The manuscript was written by the authors without external editorial support. All authors have read the journal's authorship statement. The manuscript was reviewed and approved by all named authors.
All authors have read the journal's policy on disclosure of potential conflicts of interest, and declare no potential conflicts of interest for this work.
References
- 1.Quraishi M.N., Shaheen W., Oo Y.H., Iqbal T.H. Immunological mechanisms underpinning faecal microbiota transplantation for the treatment of inflammatory bowel disease. Clin Exp Immunol. 2020;199:24–38. doi: 10.1111/cei.13397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Walter J., Armet A.M., Finlay B.B., Shanahan F. Establishing or exaggerating causality for the gut microbiome: lessons from human microbiota-associated rodents. Cell. 2020;180:221–232. doi: 10.1016/j.cell.2019.12.025. [DOI] [PubMed] [Google Scholar]
- 3.Basson A., Cominelli F., Rodriguez-Palacios A. Patterns of ‘analytical irreproducibility’ in multimodal diseases. bioRxiv. 2020 10.1101/2020.03.22.002469. [Google Scholar]
- 4.Quraishi M.N., Widlak M., Bhala N. Systematic review with meta-analysis: the efficacy of faecal microbiota transplantation for the treatment of recurrent and refractory Clostridium difficile infection. Aliment Pharmacol Ther. 2017;46:479–493. doi: 10.1111/apt.14201. [DOI] [PubMed] [Google Scholar]
- 5.Moayyedi P., Yuan Y., Baharith H., Ford A.C. Faecal microbiota transplantation for Clostridium difficile-associated diarrhoea: a systematic review of randomised controlled trials. Med J Aust. 2017;207:166–172. doi: 10.5694/mja17.00295. [DOI] [PubMed] [Google Scholar]
- 6.Colman R.J., Rubin D.T. Fecal microbiota transplantation as therapy for inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis. 2014;8:1569–1581. doi: 10.1016/j.crohns.2014.08.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Qazi T., Amaratunga T., Barnes E.L., Fischer M., Kassam Z., Allegretti J.R. The risk of inflammatory bowel disease flares after fecal microbiota transplantation: Systematic review and meta-analysis. Gut Microbes. 2017;8:574–588. doi: 10.1080/19490976.2017.1353848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Moayyedi P., Surette M.G., Kim P.T. Fecal microbiota transplantation induces remission in patients with active ulcerative colitis in a randomized controlled trial. Gastroenterology. 2015;149:102–109. doi: 10.1053/j.gastro.2015.04.001. e6. [DOI] [PubMed] [Google Scholar]
- 9.Paramsothy S., Kamm M.A., Kaakoush N.O. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: a randomised placebo-controlled trial. Lancet. 2017;389:1218–1228. doi: 10.1016/S0140-6736(17)30182-4. [DOI] [PubMed] [Google Scholar]
- 10.Wilson B.C., Vatanen T., Cutfield W.S., O'Sullivan J.M. The super-donor phenomenon in fecal microbiota transplantation. Front Cell Infect Microbiol. 2019;9 doi: 10.3389/fcimb.2019.00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lukin D.J., Lawlor G., Hudesman D.P. Escalation of immunosuppressive therapy for inflammatory bowel disease is not associated with adverse outcomes after infection with Clostridium difficile. Inflamm Bowel Dis. 2019;25:775–781. doi: 10.1093/ibd/izy308. [DOI] [PubMed] [Google Scholar]
- 12.Zabana Y., Rodriguez L., Lobaton T. Relevant infections in inflammatory bowel disease, and their relationship with immunosuppressive therapy and their effects on disease mortality. J Crohns Colitis. 2019;13:828–837. doi: 10.1093/ecco-jcc/jjz013. [DOI] [PubMed] [Google Scholar]
- 13.McSeveney M. FDA In Brief: FDA warns about potential risk of serious infections caused by multi-drug resistant organisms related to the investigational use of Fecal Microbiota for Transplantation. July 29th. https://www.fda.gov/news-events/fda-brief/fda-brief-fda-warns-about-potential-risk-serious-infections-caused-multi-drug-resistant-organisms
- 14.Tang A., Tong Z.D., Wang H.L. Detection of novel coronavirus by RT-PCR in stool specimen from asymptomatic child, China. Emerg Infect Dis. 2020;26 doi: 10.3201/eid2606.200301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Xiao W.J., Wang M.L., Wei W. Detection of SARS-CoV and RNA on aerosol samples from SARS-patients admitted to hospital. Zhonghua Liu Xing Bing Xue Za Zhi. 2004;25:882–885. [PubMed] [Google Scholar]
- 16.Wang W., Xu Y., Gao R. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020 doi: 10.1001/jama.2020.3786. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gu J., Han B., Wang J. COVID-19: Gastrointestinal manifestations and potential fecal-oral transmission. Gastroenterology. 2020;158:1518–1519. doi: 10.1053/j.gastro.2020.02.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.FDA. Safety alert regarding use of fecal microbiota for transplantation and additional safety protections pertaining to SARS-CoV-2 and COVID-19. March 31, 2020. https://www.fda.gov/vaccines-blood-biologics/safety-availability-biologics/safety-alert-regarding-use-fecal-microbiota-transplantation-and-additional-safety-protections
- 19.Wida EC. Chipotle employees say managers still 'cut corners,' putting food safety at risk. April 7, 2020. https://www.today.com/food/chipotle-employees-say-managers-still-cut-corners-putting-food-safety-t173461
- 20.Wu M.Y., Hsu M.Y., Chen S.J., Hwang D.K., Yen T.H., Cheng C.M. Point-of-care detection devices for food safety monitoring: proactive disease prevention. Trends Biotechnol. 2017;35:288–300. doi: 10.1016/j.tibtech.2016.12.005. [DOI] [PubMed] [Google Scholar]
- 21.Halfvarson J., Brislawn C.J., Lamendella R. Dynamics of the human gut microbiome in inflammatory bowel disease. Nat Microbiol. 2017;2:17004. doi: 10.1038/nmicrobiol.2017.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Basson A., Gomez-Nguyen A., Menghini P. Human gut microbiome transplantation in ileitis prone mice: a tool for the functional characterization of the microbiota in inflammatory bowel disease patients. Inflamm Bowel Dis. 2019;26:347–359. doi: 10.1093/ibd/izz242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Baxter N.T., Zackular J.P., Chen G.Y., Schloss P.D. Structure of the gut microbiome following colonization with human feces determines colonic tumor burden. Microbiome. 2014;2:20. doi: 10.1186/2049-2618-2-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Basson A., Trotter A., Rodriguez-Palacios A., Cominelli F. Mucosal interactions between genetics, diet, and microbiome in inflammatory bowel disease. Front Immunol. 2016;7 doi: 10.3389/fimmu.2016.00290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Chen T., Zhou Q., Zhang D. Effect of Faecal microbiota transplantation for treatment of clostridium difficile infection in patients with inflammatory bowel disease: a systematic review and meta-analysis of cohort studies. J Crohns Colitis. 2018;12:710–717. doi: 10.1093/ecco-jcc/jjy031. [DOI] [PubMed] [Google Scholar]
- 26.Paramsothy S., Paramsothy R., Rubin D.T. Faecal microbiota transplantation for inflammatory bowel disease: a systematic review and meta-analysis. J Crohns Colitis. 2017;11:1180–1199. doi: 10.1093/ecco-jcc/jjx063. [DOI] [PubMed] [Google Scholar]
- 27.Vaughn B.P., Vatanen T., Allegretti J.R. Increased intestinal microbial diversity following fecal microbiota transplant for active crohn's disease. Inflamm Bowel Dis. 2016;22:2182–2190. doi: 10.1097/MIB.0000000000000893. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cui B., Feng Q., Wang H. Fecal microbiota transplantation through mid-gut for refractory Crohn's disease: safety, feasibility, and efficacy trial results. J Gastroenterol Hepatol. 2015;30:51–58. doi: 10.1111/jgh.12727. [DOI] [PubMed] [Google Scholar]
- 29.Parfrey L.W., Knight R. Spatial and temporal variability of the human microbiota. Clin Microbiol Infect. 2012;18(Suppl 4):8–11. doi: 10.1111/j.1469-0691.2012.03861.x. [DOI] [PubMed] [Google Scholar]
- 30.Vermeire S., Joossens M., Verbeke K. Donor species richness determines faecal microbiota transplantation success in inflammatory bowel disease. J Crohns Colitis. 2016;10:387–394. doi: 10.1093/ecco-jcc/jjv203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Staley C., Kaiser T., Vaughn B.P. Predicting recurrence of Clostridium difficile infection following encapsulated fecal microbiota transplantation. Microbiome. 2018;6:166. doi: 10.1186/s40168-018-0549-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Smillie C.S., Sauk J., Gevers D. Strain tracking reveals the determinants of bacterial engraftment in the human gut following fecal microbiota transplantation. Cell Host Microbe. 2018;23:229–240. doi: 10.1016/j.chom.2018.01.003. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Seekatz A.M., Aas J., Gessert C.E. Recovery of the gut microbiome following fecal microbiota transplantation. Mbio. 2014;5:e00893. doi: 10.1128/mBio.00893-14. -14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Li S.S., Zhu A., Benes V. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science. 2016;352:586–589. doi: 10.1126/science.aad8852. [DOI] [PubMed] [Google Scholar]
- 35.Rossen N.G., Fuentes S., van der Spek M.J. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology. 2015;149:110–118. doi: 10.1053/j.gastro.2015.03.045. e4. [DOI] [PubMed] [Google Scholar]
- 36.Fuentes S., Rossen N.G., van der Spek M.J. Microbial shifts and signatures of long-term remission in ulcerative colitis after faecal microbiota transplantation. ISME J. 2017;11:1877–1889. doi: 10.1038/ismej.2017.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Paramsothy S., Nielsen S., Kamm M.A. Specific bacteria and metabolites associated with response to fecal microbiota transplantation in patients with ulcerative colitis. Gastroenterology. 2019;156:1440–1454. doi: 10.1053/j.gastro.2018.12.001. e2. [DOI] [PubMed] [Google Scholar]
- 38.Kellingray L., Gall G.L., Defernez M., Beales I.L.P., Franslem-Elumogo N., Narbad A. Microbial taxonomic and metabolic alterations during faecal microbiota transplantation to treat Clostridium difficile infection. J Infect. 2018;77:107–118. doi: 10.1016/j.jinf.2018.04.012. [DOI] [PubMed] [Google Scholar]
- 39.Kump P., Wurm P., Grochenig H.P. The taxonomic composition of the donor intestinal microbiota is a major factor influencing the efficacy of faecal microbiota transplantation in therapy refractory ulcerative colitis. Aliment Pharmacol Ther. 2018;47:67–77. doi: 10.1111/apt.14387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Goyal A., Yeh A., Bush B.R. Safety, clinical response, and microbiome findings following fecal microbiota transplant in children with inflammatory bowel disease. Inflamm Bowel Dis. 2018;24:410–421. doi: 10.1093/ibd/izx035. [DOI] [PubMed] [Google Scholar]
- 41.Jacob V., Crawford C., Cohen-Mekelburg S. Single delivery of high-diversity fecal microbiota preparation by colonoscopy is safe and effective in increasing microbial diversity in active ulcerative colitis. Inflamm Bowel Dis. 2017;23:903–911. doi: 10.1097/MIB.0000000000001132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Duvallet C., Zellmer C., Panchal P., Budree S., Osman M., Alm E.J. Framework for rational donor selection in fecal microbiota transplant clinical trials. PLoS One. 2019;14 doi: 10.1371/journal.pone.0222881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ott S.J., Waetzig G.H., Rehman A. Efficacy of sterile fecal filtrate transfer for treating patients with Clostridium difficile infection. Gastroenterology. 2017;152:799–811. doi: 10.1053/j.gastro.2016.11.010. e7. [DOI] [PubMed] [Google Scholar]
- 44.Zuo T., Wong S.H., Lam K. Bacteriophage transfer during faecal microbiota transplantation in Clostridium difficile infection is associated with treatment outcome. Gut. 2018;67:634–643. doi: 10.1136/gutjnl-2017-313952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.DuPont H.L., Jiang Z.D., DuPont A.W., Utay N.S. Abnormal intestinal microbiome in medical disorders and potential reversibility by fecal microbiota transplantation. Dig Dis Sci. 2020;65:741–756. doi: 10.1007/s10620-020-06102-y. [DOI] [PubMed] [Google Scholar]
- 46.Park H., Millan B.T., Hotte N., Kao D.H., Madsen K. A11 effect of fecal microbial transplant on microbial and phage composition in patients with Clostridium difficile infection. J Can Assoc. 2018;1:20–21. [Google Scholar]
- 47.Sartor R.B., Wu G.D. Roles for intestinal bacteria, viruses, and fungi in pathogenesis of inflammatory bowel diseases and therapeutic approaches. Gastroenterology. 2017;152:327–339. doi: 10.1053/j.gastro.2016.10.012. e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Courtiss E.H., Goldwyn R.M. The effects of nasal surgery on airflow. Plast Reconstr Surg. 1983;72:9–21. doi: 10.1097/00006534-198307000-00003. [DOI] [PubMed] [Google Scholar]
- 49.Conceição-Neto N., Duboutte W., Dierckx T. DOP080 Low viral richness at baseline in ulcerative ulcerative colitis associated with faecal microbiota transplantation success. J Crohn's Colitis. 2017;11:S73–SS4. [Google Scholar]
- 50.Kootte R.S., Levin E., Salojarvi J. Improvement of insulin sensitivity after lean donor feces in metabolic syndrome is driven by baseline intestinal microbiota composition. Cell Metab. 2017;26:611–619. doi: 10.1016/j.cmet.2017.09.008. e6. [DOI] [PubMed] [Google Scholar]
- 51.Vrieze A., Van Nood E., Holleman F. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143:913–916. doi: 10.1053/j.gastro.2012.06.031. e7. [DOI] [PubMed] [Google Scholar]
- 52.Keshteli A.H., Millan B., Madsen K.L. Pretreatment with antibiotics may enhance the efficacy of fecal microbiota transplantation in ulcerative colitis: a meta-analysis. Mucosal Immunol. 2017;10:565–566. doi: 10.1038/mi.2016.123. [DOI] [PubMed] [Google Scholar]
- 53.Ji S.K., Yan H., Jiang T. Preparing the gut with antibiotics enhances gut microbiota reprogramming efficiency by promoting xenomicrobiota colonization. Front Microbiol. 2017;8:1208. doi: 10.3389/fmicb.2017.01208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Millan B., Park H., Hotte N. Canadian journal of gastroenterology and hepatology conference. 2016. Antibiotics and bowel preparation enhance the ability of fecal microbial transplantation to reshape the gut microbiota in IL-10-/-mice. [Google Scholar]
- 55.Kassam Z., Lee C.H., Yuan Y., Hunt R.H. Fecal microbiota transplantation for Clostridium difficile infection: systematic review and meta-analysis. Am J Gastroenterol. 2013;108:500–508. doi: 10.1038/ajg.2013.59. [DOI] [PubMed] [Google Scholar]
- 56.Gough E., Shaikh H., Manges A.R. Systematic review of intestinal microbiota transplantation (fecal bacteriotherapy) for recurrent Clostridium difficile infection. Clin Infect Dis. 2011;53:994–1002. doi: 10.1093/cid/cir632. [DOI] [PubMed] [Google Scholar]
- 57.Suez J., Zmora N., Zilberman-Schapira G. Post-antibiotic gut mucosal microbiome reconstitution is impaired by probiotics and improved by autologous FMT. Cell. 2018;174:1406–1423. doi: 10.1016/j.cell.2018.08.047. e16. [DOI] [PubMed] [Google Scholar]
- 58.Bulow C., Langdon A., Hink T. Impact of amoxicillin-clavulanate followed by autologous fecal microbiota transplantation on fecal microbiome structure and metabolic potential. mSphere. 2018;3 doi: 10.1128/mSphereDirect.00588-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kellermayer R. Fecal microbiota transplantation: great potential with many challenges. Transl Gastroenterol Hepatol. 2019;4:40. doi: 10.21037/tgh.2019.05.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Taur Y., Coyte K., Schluter J. Reconstitution of the gut microbiota of antibiotic-treated patients by autologous fecal microbiota transplant. Sci Transl Med. 2018;10 doi: 10.1126/scitranslmed.aap9489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van Nood E., Vrieze A., Nieuwdorp M. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407–415. doi: 10.1056/NEJMoa1205037. [DOI] [PubMed] [Google Scholar]
- 62.Ianiro G., Eusebi L.H., Black C.J., Gasbarrini A., Cammarota G., Ford A.C. Systematic review with meta-analysis: efficacy of faecal microbiota transplantation for the treatment of irritable bowel syndrome. Aliment Pharmacol Ther. 2019;50:240–248. doi: 10.1111/apt.15330. [DOI] [PubMed] [Google Scholar]
- 63.Kelly C.R., Khoruts A., Staley C. Effect of fecal microbiota transplantation on recurrence in multiply recurrent Clostridium difficile infection: a randomized trial. Ann Intern Med. 2016;165:609–616. doi: 10.7326/M16-0271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sood A., Mahajan R., Singh A. Role of faecal microbiota transplantation for maintenance of remission in patients with ulcerative colitis: a pilot study. J Crohns Colitis. 2019;13:1311–1317. doi: 10.1093/ecco-jcc/jjz060. [DOI] [PubMed] [Google Scholar]
- 65.Rossen N., S. F., van der Spek M.J., Tijssen J.G. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology. 2015;149:110–8e4. doi: 10.1053/j.gastro.2015.03.045. [DOI] [PubMed] [Google Scholar]
- 66.Costello S.P., Hughes P.A., Waters O. Effect of fecal microbiota transplantation on 8-week remission in patients with ulcerative colitis: a randomized clinical trial. JAMA. 2019;321:156–164. doi: 10.1001/jama.2018.20046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Kazerouni A., Wein L.M. Exploring the efficacy of pooled stools in fecal microbiota transplantation for microbiota-associated chronic diseases. PLoS One. 2017;12 doi: 10.1371/journal.pone.0163956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Woodworth M.H., Carpentieri C., Sitchenko K.L., Kraft C.S. Challenges in fecal donor selection and screening for fecal microbiota transplantation: a review. Gut Microbes. 2017;8:225–237. doi: 10.1080/19490976.2017.1286006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Barnes D., Ng K., Smits S., Sonnenburg J., Kassam Z., Park K.T. Competitively selected donor fecal microbiota transplantation: butyrate concentration and diversity as measures of donor quality. J Pediatr Gastroenterol Nutr. 2018;67:185–187. doi: 10.1097/MPG.0000000000001940. [DOI] [PubMed] [Google Scholar]
- 70.David L.A., Maurice C.F., Carmody R.N. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505:559–563. doi: 10.1038/nature12820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Thompson S., Guetterman H., Taylor A. dietary predictors of fecal microbiota transplantation success. J Acad Nutr Dietet. 2016;116:A76. [Google Scholar]
- 72.von Roon A.C., Karamountzos L., Purkayastha S. Diagnostic precision of fecal calprotectin for inflammatory bowel disease and colorectal malignancy. Am J Gastroenterol. 2007;102:803–813. doi: 10.1111/j.1572-0241.2007.01126.x. [DOI] [PubMed] [Google Scholar]
- 73.Rodriguez-Palacios A., Aladyshkina N., Cominelli F. Stereomicroscopy and 3D-target myeloperoxidase intestinal phenotyping following a fecal flora homogenization protocol. Protocol Exch. 2015 doi: 10.1038/protex.2015.065. [DOI] [Google Scholar]
- 74.Konikoff M.R., Denson L.A. Role of fecal calprotectin as a biomarker of intestinal inflammation in inflammatory bowel disease. Inflamm Bowel Dis. 2006;12:524–534. doi: 10.1097/00054725-200606000-00013. [DOI] [PubMed] [Google Scholar]
- 75.Machiels K., Joossens M., Sabino J. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut. 2014;63:1275–1283. doi: 10.1136/gutjnl-2013-304833. [DOI] [PubMed] [Google Scholar]
- 76.Moeinian M., Abdolghaffari A.H., Nikfar S., Momtaz S., Abdollahi M. Effects of alpha lipoic acid and its derivative “andrographolid-lipoic acid-1” on ulcerative colitis: a systematic review with meta-analysis of animal studies. J Cell Biochem. 2019;120:4766–4782. doi: 10.1002/jcb.27807. [DOI] [PubMed] [Google Scholar]
- 77.Lee C.H., Chai J., Hammond K. Long-term durability andsafety of fecal microbiota transplantation for recurrent or refractory Clostridioides difficile infection with or without antibiotic exposure. Eur J Clin Microbiol Infect Dis. 2019;38:1731–1735. doi: 10.1007/s10096-019-03602-2. [DOI] [PubMed] [Google Scholar]
- 78.Hohmann E.L. Case 25-2014: a man with ulcerative colitis and bloody diarrhea. N Engl J Med. 2014;371:1848–1849. doi: 10.1056/NEJMc1410949. [DOI] [PubMed] [Google Scholar]
- 79.D'Odorico I., Di Bella S., Monticelli J., Giacobbe D.R., Boldock E., Luzzati R. Role of fecal microbiota transplantation in inflammatory bowel disease. J Dig Dis. 2018;19:322–334. doi: 10.1111/1751-2980.12603. [DOI] [PubMed] [Google Scholar]
- 80.Fang H., Fu L., Wang J. Protocol for fecal microbiota transplantation in inflammatory bowel disease: a systematic review and meta-analysis. Biomed Res Int. 2018;2018 doi: 10.1155/2018/8941340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Schulz M.D., Atay C., Heringer J. High-fat-diet-mediated dysbiosis promotes intestinal carcinogenesis independently of obesity. Nature. 2014;514:508–512. doi: 10.1038/nature13398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Collins S.M., Kassam Z., Bercik P. The adoptive transfer of behavioral phenotype via the intestinal microbiota: experimental evidence and clinical implications. Curr Opin Microbiol. 2013;16:240–245. doi: 10.1016/j.mib.2013.06.004. [DOI] [PubMed] [Google Scholar]
- 83.Turnbaugh P.J., Ridaura V.K., Faith J.J., Rey F.E., Knight R., Gordon J.I. The effect of diet on the human gut microbiome: a metagenomic analysis in humanized gnotobiotic mice. Sci Transl Med. 2009;1 doi: 10.1126/scitranslmed.3000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Fei N., Zhao L. An opportunistic pathogen isolated from the gut of an obese human causes obesity in germfree mice. ISME J. 2013;7:880–884. doi: 10.1038/ismej.2012.153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sonnenburg J.L., Backhed F. Diet-microbiota interactions as moderators of human metabolism. Nature. 2016;535:56–64. doi: 10.1038/nature18846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.De Filippo C., Cavalieri D., Di Paola M. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc Natl Acad Sci U S A. 2010;107:14691–14696. doi: 10.1073/pnas.1005963107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Barnes D., Park K.T. Donor considerations in fecal microbiota transplantation. Curr Gastroenterol Rep. 2017;19:10. doi: 10.1007/s11894-017-0548-y. [DOI] [PubMed] [Google Scholar]
- 88.Koeth R.A., Wang Z., Levison B.S. Intestinal microbiota metabolism of L-carnitine, a nutrient in red meat, promotes atherosclerosis. Nat Med. 2013;19:576–585. doi: 10.1038/nm.3145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Stein R.R., Tanoue T., Szabady R.L. Computer-guided design of optimal microbial consortia for immune system modulation. Elife. 2018;7 doi: 10.7554/eLife.30916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Zou M., Jie Z., Cui B. Fecal microbiota transplantation results in bacterial strain displacement in patients with inflammatory bowel diseases. FEBS Open Bio. 2020;10:41–55. doi: 10.1002/2211-5463.12744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Johnsen P.H., Hilpusch F., Cavanagh J.P. Faecal microbiota transplantation versus placebo for moderate-to-severe irritable bowel syndrome: a double-blind, randomised, placebo-controlled, parallel-group, single-centre trial. Lancet Gastroenterol Hepatol. 2018;3:17–24. doi: 10.1016/S2468-1253(17)30338-2. [DOI] [PubMed] [Google Scholar]
- 92.Holvoet T., Joossens M., Jerina B. Fecal microbiota transplantation in irritable bowel syndrome with predominant abdominal bloating: results from a double blind, placebo‐controlled clinical trial. Gastroenterology. 2018;154:S–130. doi: 10.1053/j.gastro.2020.07.013. [DOI] [PubMed] [Google Scholar]
- 93.Holster S., Lindqvist C.M., Repsilber D. The effect of allogenic versus autologous fecal microbiota transfer on symptoms, visceral perception and fecal and mucosal microbiota in irritable bowel syndrome: a randomized controlled study. Clin Transl Gastroenterol. 2019;10:e00034. doi: 10.14309/ctg.0000000000000034. [DOI] [PMC free article] [PubMed] [Google Scholar]