Abstract
The random pattern skin flap has been an important procedure in plastic and reconstructive surgery to cover various challenging defects. However, distal flap necrosis is problematic. Crocin is a natural carotenoid compound, which have been reported to possess a wide spectrum of properties and induce pleiotropic anti-inflammatory, anti-oxidative and cytoprotective effects. We explored whether crocin enhanced random skin flap survival. Thirty-six male SD rats were distributed to two groups randomly: the crocin and control groups. Flap survival areas were measured 7 days after surgery. Neutrophil numbers and microvascular density were evaluated via haematoxylin and eosin staining, and blood perfusion via laser Doppler imaging. Vascular endothelial growth factor (VEGF) levels were measured by immunohistochemical staining and Western blotting. We also measured the levels of markers of ischaemia-reperfusion injury [malondialdehyde (MDA) and superoxide dismutase (SOD)]. With regard to flap survival area assay, a significant between-group difference of survival area for the experimental flap was evident. As for flap blood flow test in Area II, the crocin group was statistically better than that of the control group. And in the histological result, the mean vessel density and VEGF level were statistically higher when treated with crocin. Crocin also decreased the MDA but increased the SOD level. Crocin thus improved random skin flap viability, enhancing angiogenesis and inhibiting ischaemia-reperfusion injury.
Keywords: Crocin, random pattern skin flap, laser Doppler imaging, VEGF
Introduction
In plastic and reconstructive surgery, soft tissue defect is a common and challenging problem which mainly caused by traumatic or cancer-associated deformities. Due to its simple surgery procedure, skin random flap is widely used in transplantation to functional and aesthetic reconstruction by plastic surgeons, especially for tissue repair [1]. However, necrosis of the flap distal end (partial or complete) is a common phenomenon after surgery, which leads to severe complications. In the clinical settings, the length-to-width ratio of such flaps cannot be increased beyond 1.5:1 to 2:1 [2]. This problem greatly limits the clinical application of random flap application for soft tissue transplantation [3]. One study [4] suggested that ischaemia-reperfusion injury, a poor blood supply, and haemodynamic injury contribute to flap necrosis. We previously showed [5,6] that angiogenesis was important item of flap survival, especially the deliverance of the key protein called vascular endothelial growth factor (VEGF). Generous evidence indicates that many herbs and herbal extracts from traditional Chinese medicine can effectively treat vascular diseases [7,8]. Crocin has been used in traditional Chinese medicine throughout history, which has been traditionally used for the treatment of many types of disease, including neurodegenerative disorders, coronary artery diseases, respiratory diseases, gastrointestinal diseases, fever, diabetes, and so on [9]. Crocin, a major component of crocus sativus, is associated with some of these therapeutic effects [10]. It has been demonstrated that crocin exhibits multiple activities, including anti-cancer, anti-inflammatory and anti-oxidation effects [11,12]; crocin is also found promote burn wound healing in rats, perhaps by reducing inflammation and promoting neovascularization [13]. We thus hypothesised that crocin could improve the survival of random pattern skin flap via an animal model.
Materials and experimental methods
Drugs and reagents
Crocin was obtained from Sigma-Aldrich (Saint Louis, MO, United States). Molecular formula was shown in Figure 1. Anti-VEGF polyclonal antibodies were purchased from Bioworld (Nanjing, China). The malondialdehyde (MDA) and superoxide dismutase (SOD) content were measured using ELISA kit following manufacturer’s instructions (Nanjing Jiancheng Bioengineering Institute. Nanjing, China).
Figure 1.
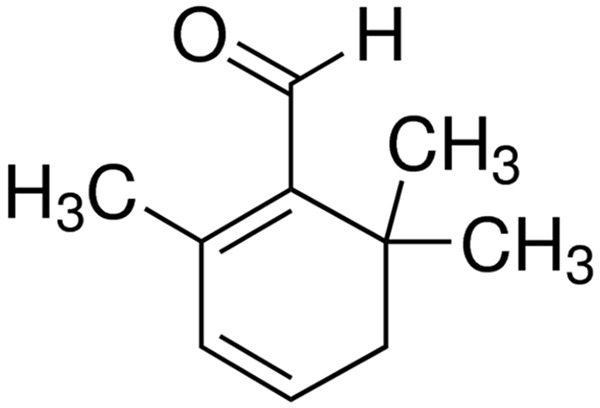
Chemical structure of crocin, a prenylated flavonoid glycoside.
Animals
In total, thirty-six healthy Sprague-Dawley rats (male, body weight 220~250 g, 2-month-old) were acquired from the Animal center of Wenzhou Medical University (Wenzhou, China) (SCXK [Zhe] 2005-0061). The animals were cared as directed by the Guide for the Care and Use of Laboratory Animals of Wenzhou Medical University. Upon arrival, animals were individually caged and kept in rooms where temperature (22±2°C), humidity, and light (12-h/12-h light/dark) were controlled. All experimental animals with free access to standard chow and tap water, were randomly divided into test (crocin) group and control groups by means of random number table, 18 rats in each. Animal center of Wenzhou Medical University Ethical Committee had permitted the study (ID Number: wydw 2019-0145). As for the method of animal killing, we have killed rats using overdoses of narcotics, this procedure was painless and commonly used in animal killing.
Surgical procedure and drug administration
Adapted to one overnight, all experimental animals were shaved and sterilized before surgery. The surgical operation was completed under general anesthesia. The anesthesia of all rats was achieved via an intraperitoneal injection with 40-50 mg.kg-1 sodium pentobarbital. The pinch flexion/withdrawal experiment was used to determine the adequate anaesthesia. A modified McFarlane flap (3×9 cm; Figure 2A) was created on each experimental animal. To ensure accurate positioning, the proximal end of the flap was on the margin of the iliac crest as a maker. To reduce bacterial infections, all instruments and surgical procedures were sterile. Sacral arteries in both sides were sectioned completely to ensure no axial vessel was incorporated into the flap (Figure 2B). Following each model was dissociated from the deep fascia completely, all flaps were repositioned into initially position with 4-0 silk (Figure 2C). To prevent injury from cannibalism activities and beneficial to socialization lives, all rats were then individually caged. To counter self-mutilation, a collar was fitted to every rat [14]. Crocin was dissolve in saline solution and given via intraperitoneal injection at 50 mg kg-1 day-1 for 7 consecutive days; while the control group was treated with saline solution for comparison [15,16].
Figure 2.

A McFarlane flap model was designed (3×9 cm); no axial vessels were incorporated into the flap. A. The modified McFarlane flap model (3×9 cm) was applied to every rat. B. No axial vessels were incorporated into the flap in which the ischemic gradient is proportional to the distance from the base. White arrow refers to the axial vessels. C. Each flap was sutured back to its original position with continuous 4-0 silk sutures.
Flap survival
The random skin flap was separated into three identical areas: the proximal of flap as Area I, the intermediate of flap as Area II, and distal of the flap as Area III. Every area was possessed with equal size (3×3 cm) [17]. After the operation, flap appearance, colour, texture, and hair status were evaluated. All rats were euthanised on the 7 days postoperative. Then all flaps were dissected for further investigation. There was a rough boundary between survival and dead flap. The flaps of each rat were photographed, and the survival areas were measured by superimposition of photographs onto graph paper. On basis of which we measured surviving and total flap areas on transparent cellophane sheets and calculated flap survival percentages [18] as survival area ×100/total area (survival and ischaemic) [19].
Histological staining
After sacrifice, three 1×1 cm central samples were collected from every area, which were fixed in 4% (v/v) paraformaldehyde, embedded in paraffin. Then, samples were made into slices of 4-μm thickness and haematoxylin and eosin stain (H&E) was discovered for next step. Slides were observed under an Olympus BH51 light microscope with 100 magnification (Olympus Corporation, Japan). Then three representative fields in each of these areas were further counted at ×400 magnification. Oedema, granulation thickness, neutrophil numbers, and microvessel numbers/mm2 (an indicator of microvascular density [MVD]) [20] were evaluated. This work was repeatedly finished by two separate physicians.
Laser Doppler imaging
All animals underwent laser Doppler perfusion imaging (MoorLDI-2, Moor Instruments Ltd, Devon, Britain) at room temperature on the last day. The parameters of the instrument were as follows: 633 nm laser wavelength, 55 cm scan and 5 min duration. The results were quantified by the MoorLDI Review V6.1 software. The measurement would repeat three times for every rat and the mean value was calculated after three times repeat [21].
Immunohistochemical staining
All flap zones were subjected to immunohistochemical VEGF staining with the streptavidin-peroxidase way; we blocked all slices with routine goat serum at 25°C for 20 minutes; added 50 µL amounts of primary VEGF antibodies (1:100) followed by hatched at 4°C for 24 hours; rewarmed the slides to 37°C for 50 min; washed them with phosphate buffer saline (PBS); added 50 µL amounts of goat anti-rat antibodies (1:50) followed by incubation for one hour at 37°C; washed the slices with distilled water; added 3,3’ N-diaminobenzidine tetrahydrochloride (for colour development) followed by counterstain for 5 min. Positive VEGF expression in each slice was identified under the inverted BH51 microscope at ×400 magnification in five fields/slice. All images were carefully analyzed through the software Image-Pro Plus ver. 6.0 (Media Cybernetics, MD). After calibration the intensity, we selected the target colors, use the Measurements. Afterwards, the integral absorbance (IA) value was served as a measure of VEGF level.
Western blotting (WB)
After euthanasia, samples (size: 0.5×0.5 cm) from the mid-district of Area II in all rats were kept at -80°C pre-WB; homogenized; and protein concentrations determined using the Bicinchoninic Acid (BCA) assay. Equal amounts of protein (55 μg) were separated by 12% Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to polyvinylidene fluoride (PVDF) membranes (Roche Applied Science, USA). All membranes were incubated with blocking buffer 10% (w/v) non-fat milk for 2 h at room temperature and incubated the primary anti-VEGF (1:1000) and β-actin (1:1000) overnight at 4°C. After being washed three times with TBS containing 0.1% Tween-20 for 5 min, the membranes were incubated at room temperature with HRP-conjugated IgG secondary antibodies (1:5000) for 2 h. Finally, the membranes were detected using an enhanced chemiluminescence (ECL) kit and band intensity quantified with Quantity One (Bio-Rad, Hercules, CA, USA) software. β-Actin was used as an internal control.
Superoxide dismutase (SOD) activity and malondialdehyde (MDA) content
At 7 days postoperatively, flap specimens (0.5×0.5 cm) from each group were acquired from districts between Area II and the other areas, weighed, and muscle tissues prepared as 10% volume fractions after homogenisation in an ice bath. SOD activity was estimated using the enzymatic xanthine oxidase method with the Total Superoxide Dismutase (T-SOD) assay kit (A001-1-2, NJJC). While with the Malondialdehyde (MDA) assay kit (A003-1-2, Nanjing Jiancheng), thiobarbituric acid test was used to determine the malondialdehyde (MDA) content at 95°C [22].
Statistical analysis
All analyses were performed using SPSS software version 17.0 (SPSS Inc., USA). We report means ± standard deviations. Kruskal Wallis and Mann-Whitney U test were applied for statistical analysis to compare the parameters. A p-value <0.05 was considered to be statistically significant. The significance difference between two groups was tested via analysis of t-test.
Results
Crocin improved the flap survival area and ameliorated tissue oedema
All rats were in good condition before execution. All flaps were monitored by the same observer from days 1 to 7 postoperatively, and colour, elasticity, and necrosis recorded. One day after surgery, all flaps were pale and swollen. All flaps were purple and oedematous in Area III, indicating distal ischaemia. On day 3, these regions underwent necrosis, evidenced by the appearance of dark brown nidi. On day 7 postoperatively, all flaps exhibited clearly-dividing line between surviving and ischemia regions. The ischemia areas were scab, harden, inanimate; the surviving areas were ruddy, pastel, active (Figure 3A). A significant between-group difference in terms of survival area was evident: 67.17±9.62 (crocin group) and 49.28±6.2% (control groups) with P<0.01 (Figure 3B). To explore the extent of oedema, all flaps were detached to allow us to view the undersides. As manifested in Figure 3A, the distal regions were swollen and bruised and lacked subcutaneous venous blood flow in the control group.
Figure 3.

Crocin improves flap survival area and ameliorates tissue edema. A. Digital photographs of flap survival in the Control and test groups were taken on day 7. B. Flap survival percentages for each group were then quantified and analyzed. Significance: *P<0.01 versus the Control group. Data represent Mean values ± SEM.
Crocin increased neovascularisation and VEGF expression
To evaluate the effects of crocin on granulation tissue thickness, oedema, and neutrophil infiltration, flap sections were H&E-stained. The distal areas of all flaps were morphologically similar. As shown in Figure 4A, on day 7, all flaps revealed analogous changes, including structural damage, oedema (presence vacuole, hyalinization and spaces separated the connective tissue) and seep inflammatory cells.
Figure 4.

Crocin promoted angiogenesis in the ischemic areas of the flap. A. Histological changes in middle area II of the flaps of the test and control groups. Magnifications: ×200 (hematoxylin and eosin stain). The test group showed more angiogenesis. Black arrows refer to new blood vessels. B. The mean vessel densities (MVDs) in each group were quantified and analyzed. *P<0.01 versus the Control group. Data represent Mean values ± SEM.
Of all images, 90% exhibited muscle fibre degeneration and necrosis. When analyzed the Area II, compared with the control group, the crocin group displayed larger fibroblast multiplication, covered with fresh granulation soft tissue, less oedema, bushier fascia haemorrhage, more extensive inflammatory cells infiltration, and more neovascularization (Figure 4A). What’s more, The MVDs of Area II were, respectively, 22.5±2.64 and 16.11±345/mm2 (P<0.01, Figure 4B); thus, significantly different. On the 7th day after surgery, we used the laser Doppler system to estimate the blood flow situation of all flaps. The flow rates in middle area were 79.11±21.26 (control group) and 146.17±19.86 (test group) perfusion units, respectively; thus, significantly better in the crocin group (Figure 5, P<0.01).
Figure 5.

A. The blood perfusion on day 7 in two groups. B. Mean blood flow of the II zone in the flap in two groups measured by laser Doppler imaging at day 7 after surgery (*P<0.01 vs. the control group).
VEGF is a potent and specific mitogen for vascular endothelia cells. We measured VEGF expression to explore the extent of neovascularisation. As shown in Figure 6A and 6B, in term of VEGF level, there was a statistic difference between the two groups (test group: 112.06±10.24/field; control group: 83.22±12.46/field; P<0.01). Western blotting also demonstrated higher expression of VEGF in the crocin group compared with the control group (P<0.01; Figure 6C, 6D).
Figure 6.

Crocin Increased Vascular Endothelial Growth Factor Expression. A. VEGF protein levels in the test and control groups (immunohistochemical staining; ×400). B. Optical density values of VEGF expressions are quantified and analyzed for each group. C. Western blotting for VEGF protein expressions in area II tissue in the control and test groups. D. Optical density values of VEGF expressions in each group are quantified and analyzed for each group. *P<0.01 versus the control group. Data represent Mean values ± SEM.
Crocin reduced oxidative stress in ischaemic flap areas
Crocin affected SOD and MDA levels, protecting against ischaemia-reperfusion injury. In term of mean SOD activity, the test group showed higher grades than the control group (48.5±4.1 U/mg protein VS. 37.17±4.91 U/mg protein, P<0.01; Figure 7A). Also, the test group (26.89±5.25 nmol/mg protein) exhibited significantly lower mean MDA content compared to the control group (46.39±6.82 nmol/mg protein, P<0.01; Figure 7B).
Figure 7.

A. SOD activities in the test and control groups. SOD, superoxide dismutase. B. MDA levels in the test and control groups. MDA, malondialdehyde. *P<0.01 versus the control group. Data represent Mean values ± SEM.
Discussion
Random skin flap necrosis extends the hospital stay, increases treatment costs, and triggers more surgical interventions. Necrosis, especially in the distal regions of random flaps, compromises flap utility. Many efforts have been made to reduce distal flap necrosis [23,24]. We earlier showed that both traditional Chinese medicine Shuxuetong [5] and Huangqi [6] injections significantly improved flap survival by increasing blood perfusion, and reduced ischaemia-reperfusion injury and necrosis. Here, we found that crocin also reduced ischaemia-reperfusion injury and increased neovascularisation; crocin may thus improve skin flap survival.
Crocin is the principal chemical component extracted from crocus sativus and has been safely used in traditional medicine for a long period of time. In modern studies, its active constituents including protective effects, anti-inflammatory activities and molecular mechanisms of crocin on thimmune system have been demonstrated [25]. It appears that crocin can potentiate the anti-oxidant defense system by modulating MAPK and NF-κB pathways [26]. Yaribey et al. explored crocin potentiated the anti-oxidant defense system by increasing the activity of both SOD and catalase, and improved OS by diminishing MDA production in pancreatic cells of rats [27]. In addition, crocin showed protective effect against ischemia/reperfusion injury and cerebral edema in a rat model of stroke and decreased infarct volume by suppression of the production of free radicals and increased antioxidant enzyme activity [28].
VEGF promotes neovascularisation by promoting vascular permeability, extracellular matrix degeneration, vascular endothelial cell migration, proliferation and angiogenesis. VEGF is expressed in developing blood vessels [29]; VEGF receptors are unique to endothelial cells [30,31]. VEGF stimulates neovascularisation, increases blood flow and pressure, and improves muscle function and tissue viability [32]. We found that crocin increased VEGF expression, as revealed both immunohistochemically and Western blotting. We used H&E staining to measure MVD; flap viability was enhanced. Laser Doppler imaging showed that blood flow in flap of the crocin group was appreciable luxuriant than in controls. Therefore, crocin increased the blood supply to the flap, implying that VEGF played a key role in blood vessel formation. However, further experimental studies are required to elucidate the specific mechanism of crocin promoting the expression of VEGF.
Inflammation and ischaemia-reperfusion injury greatly compromise flap survival [33]. MDA is one of the most important products of ischaemia-reperfusion injury. Its production can also aggravate cell membrane damage. Therefore, MDA content is a commonly used index in senescence physiology and resistance physiology research. MDA can be used to understand the extent of ischaemia-reperfusion injury, and indirectly measure the degree of membrane system damage and tissue stress resistance [34]. SOD is a crucial antioxidant enzyme in organisms and the primary substance to scavenge free radicals in vivo. It has been proved that there are more than 60 diseases caused by oxygen free radicals. It can resist and block the damage caused by oxygen free radicals to cells, and repair damaged cells in time, and restore the damage caused by free radicals [35]. The results showed that the levels of SOD and MDA in the crocin group were statistically different from those in the control group, indicating that crocin can effectively reduce the oxidative stress and alleviate ischaemia-reperfusion injury. Crocin increased endogenous SOD activity and decreased the mean MDA level, indicating that lipid peroxidation was inhibited. By increasing free radical scavenging, crocin protects tissue from ischemia-reperfusion injury.
Crocin may reduce the distal necrosis of the random flap by increasing the number of effective blood vessels and inhibiting the ischemia-reperfusion injury of the flap, which may be helpful for clinical treatment.
Acknowledgements
This study was supported by the Zhejiang Medical and Health Science and Technology Plan Project (No. 2017KY480 and National Natural Science Foundation of China (No. 81701928).
Disclosure of conflict of interest
None.
Abbreviations
- VEGF
vascular endothelial growth factor
- MDA
Malondialdehyde
- SOD
superoxide dismutase
- MVD
microvascular density
- IA
integral absorbance
- EPS
endothelial progenitor cells
- NO
Nitric Oxide
- BCA
bicinchoninic acid
- TBS
Tris-buffered saline
- PVDF
polyvinylidene fluoride
- SDS
sodium dodecyl sulfate
References
- 1.Lucas JB. The physiology and biomechanics of skin flaps. Facial Plast Surg Clin North Am. 2017;25:303–11. doi: 10.1016/j.fsc.2017.03.003. [DOI] [PubMed] [Google Scholar]
- 2.Ali G, Hakan A, Hakan P, Aysun BK, Mehmet C, Nurzen S, Ahmet A. Protective effect of melatonin on random pattern skin flap necrosis in pinealectomized rat. J Pineal Res. 2010;36:58–63. doi: 10.1046/j.1600-079x.2003.00099.x. [DOI] [PubMed] [Google Scholar]
- 3.Changizi AS, Zohrabi M, Hassanpoor A, Hosseini N, Hajihashemi S. Oral administration of the aqueous extract of Rosmarinus officinalis in rats before renal reperfusion injury. Iran J Kidney Dis. 2013;7:367. [PubMed] [Google Scholar]
- 4.Young Seok K, Suk RT, Won-Jai L, Won Min Y, Kwan-Chul T. The effect of botulinum toxin A on skin flap survival in rats. Wound Repair Regen. 2010;17:411–17. doi: 10.1111/j.1524-475X.2009.00477.x. [DOI] [PubMed] [Google Scholar]
- 5.Cai L, Huang W, Lin D. Effects of traditional Chinese medicine shuxuetong injection on random skin flap survival in rats. ScientificWorldJournal. 2014;2014:816545. doi: 10.1155/2014/816545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cai L, Cao B, Lin D. Effects of traditional Chinese medicine huangqi injection (radix astragali) on random skin flap survival in rats. J Reconstr Microsurg. 2015;31:565–570. doi: 10.1055/s-0035-1555142. [DOI] [PubMed] [Google Scholar]
- 7.Shu H, Arita H, Hayashida M, Zhang L, An K, Huang W, Hanaoka K. Anti-hypersensitivity effects of Shu-jing-huo-xue-tang, a Chinese herbal medicine, in CCI-neuropathic rats. J Ethnopharmacol. 2010;131:464–70. doi: 10.1016/j.jep.2010.07.004. [DOI] [PubMed] [Google Scholar]
- 8.Zhai ZJ, Li HW, Liu GW, Qu XH, Tian B, Yan W, Lin Z, Tang TT, Qin A, Dai KR. Andrographolide suppresses RANKL-induced osteoclastogenesis in vitro and prevents inflammatory bone loss in vivo. Br J Pharmacol. 2014;171:663–75. doi: 10.1111/bph.12463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boskabady MH, Farkhondeh T. Antiinflammatory, antioxidant, and immunomodulatory effects of crocus sativus l and its main constituents. Phytother Res. 2016;30:1072–94. doi: 10.1002/ptr.5622. [DOI] [PubMed] [Google Scholar]
- 10.Deng M, Li D, Zhang Y, Zhou G, Liu W, Cao Y, Zhang W. Protective effect of crocin on ultraviolet B-induced dermal fibroblast photoaging. Mol Med Rep. 2018;18:1439–46. doi: 10.3892/mmr.2018.9150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Samarghandian S. Effect of crocin on aged rat kidney through inhibition of oxidative stress and proinflammatory state. Phytother Res. 2016;30:1345–53. doi: 10.1002/ptr.5638. [DOI] [PubMed] [Google Scholar]
- 12.Yao C, Liu BB, Qian XD, Li LQ, Cao HB, Guo QS, Zhou GF. Crocin induces autophagic apoptosis in hepatocellular carcinoma by inhibiting Akt/mTOR activity. Onco Targets Ther. 2018;11:2017–28. doi: 10.2147/OTT.S154586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Alemzadeh E, Oryan A. Effectiveness of a crocus sativus extract on burn wounds in rats. Planta Med. 2018;84:1191–200. doi: 10.1055/a-0631-3620. [DOI] [PubMed] [Google Scholar]
- 14.Cao B, Wang L, Lin D, Cai L, Gao W. Effects of lidocaine on random skin flap survival in rats. Dermatol Surg. 2015;41:53–58. doi: 10.1097/DSS.0000000000000241. [DOI] [PubMed] [Google Scholar]
- 15.Liu W, Sun Y, Cheng Z, Guo Y, Liu P, Wen Y. Crocin exerts anti-inflammatory and anti-arthritic effects on type II collagen-induced arthritis in rats. Pharm Biol. 2018;56:209–16. doi: 10.1080/13880209.2018.1448874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dianat M, Radan M, Badavi M, Mard SA, Bayati V, Ahmadizadeh M. Crocin attenuates cigarette smoke-induced lung injury and cardiac dysfunction by anti-oxidative effects: the role of Nrf2 antioxidant system in preventing oxidative stress. Respir Res. 2018;19:58. doi: 10.1186/s12931-018-0766-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kelly CP, Gupta A, Keskin M, Jackson IT. A new design of a dorsal flap in the rat to study skin necrosis and its prevention. J Plast Reconstr Aesthet Surg. 2010;63:1553–56. doi: 10.1016/j.bjps.2009.08.011. [DOI] [PubMed] [Google Scholar]
- 18.Lin B, Lin Y, Lin D, Cao B. Effects of bezafibrate on the survival of random skin flaps in rats. J Reconstr Microsurg. 2016;32:395–401. doi: 10.1055/s-0036-1571348. [DOI] [PubMed] [Google Scholar]
- 19.Movassaghi K, Lewis M, Shahzad F, May JW Jr. Improved survival rates of random flaps in rabbits with a monoclonal antibody that blocks leukocyte adherence. Plast Reconstr Surg. 1994;93:1035–40. doi: 10.1097/00006534-199404001-00020. [DOI] [PubMed] [Google Scholar]
- 20.Chen Z, Wang T, Cai L, Su C, Zhong B, Lei Y, Xiang AP. Clinicopathological significance of non-small cell lung cancer with high prevalence of Oct-4 tumor cells. J Exp Clin Cancer Res. 2012;31:10. doi: 10.1186/1756-9966-31-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tao XY, Wang L, Gao WY, Ding J, Feng XL, Zhou ZW, Yang LH. The effect of inducible nitric oxide synthase on multiterritory perforator flap survival in rats. J Reconstr Microsurg. 2016;32:643–49. doi: 10.1055/s-0036-1584808. [DOI] [PubMed] [Google Scholar]
- 22.Ozkan F, Senayli Y, Ozyurt H, Erkorkmaz U, Bostan B. Antioxidant effects of propofol on tourniquet-induced ischemia-reperfusion injury: an experimental study. J Surg Res. 2012;176:601–7. doi: 10.1016/j.jss.2011.10.032. [DOI] [PubMed] [Google Scholar]
- 23.Lin R, Chen H, Callow D, Li S, Wang L, Li S, Chen L, Ding J, Gao W, Xu H, Kong J, Zhou K. Multifaceted effects of astragaloside IV on promotion of random pattern skin flap survival in rats. Am J Transl Res. 2017;9:4161–72. [PMC free article] [PubMed] [Google Scholar]
- 24.Serin M, Altinel D, Leblebici C, Biltekin B, Celikten M, Irmak F, Yazar SK. Preoperative subcutaneous sildenafil injection increases random flap survival in rats. Acta Cir Bras. 2018;33:216–22. doi: 10.1590/s0102-865020180030000003. [DOI] [PubMed] [Google Scholar]
- 25.Zeinali M, Zirak MR, Rezaee SA, Karimi G, Hosseinzadeh H. Immunoregulatory and anti-inflammatory properties of (Saffron) and its main active constituents: a review. Iran J Basic Med Sci. 2019;22:334–44. doi: 10.22038/ijbms.2019.34365.8158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.El-Fawal R, El Fayoumi HM, Mahmoud MF. Effects of diosmin and crocin on metabolic syndrome-associated cardio-vascular complications in rats. Naunyn Schmiedebergs Arch Pharmacol. 2019;392 doi: 10.1007/s00210-019-01700-8. undefined. [DOI] [PubMed] [Google Scholar]
- 27.Yaribeygi H, Noroozadeh A, Mohammadi MT, Johnston TP, Sahebkar A. Crocin improves oxidative stress by potentiating intrinsic anti-oxidant defense systems in pancreatic cells during uncontrolled hyperglycemia. J Pharmacopuncture. 2019;22:83–89. doi: 10.3831/KPI.2019.22.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vakili A, Einali MR, Bandegi AR. Protective effect of crocin against cerebral ischemia in a dose-dependent manner in a rat model of ischemic stroke. J Stroke Cerebrovasc Dis. 2014;23:106–13. doi: 10.1016/j.jstrokecerebrovasdis.2012.10.008. [DOI] [PubMed] [Google Scholar]
- 29.Carmeliet P, Ferreira V, Breier G, Pollefeyt S, Kieckens L, Gertsenstein M, Fahrig M, Vandenhoeck A, Harpal K, Eberhardt C, Declercq C, Pawling J, Moons L, Collen D, Risau W, Nagy A. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature. 1996;380:435–39. doi: 10.1038/380435a0. [DOI] [PubMed] [Google Scholar]
- 30.Kenny D, Coughlan MG, Pagel PS, Kampine JP, Warltier DC. Transforming growth factor beta 1 preserves endothelial function after multiple brief coronary artery occlusions and reperfusion. Am Heart J. 1994;127:1456–61. doi: 10.1016/0002-8703(94)90370-0. [DOI] [PubMed] [Google Scholar]
- 31.Keck P, Hauser S, Krivi G, Sanzo K, Warren T, Feder J, Connolly D. Vascular permeability factor, an endothelial cell mitogen related to PDGF. Science. 246:1309–12. doi: 10.1126/science.2479987. [DOI] [PubMed] [Google Scholar]
- 32.Kryger Z, Zhang F, Dogan T, Cheng C, Lineaweaver WC, Buncke HJ. The effects of VEGF on survival of a random flap in the rat: examination of various routes of administration. Br J Plast Surg. 2000;53:234–39. doi: 10.1054/bjps.1999.3315. [DOI] [PubMed] [Google Scholar]
- 33.Cui J, Liu L, Zou J, Qiao W, Liu H, Qi Y, Yan C. Protective effect of endogenous hydrogen sulfide against oxidative stress in gastric ischemia-reperfusion injury. Exp Ther Med. 2013;5:689–94. doi: 10.3892/etm.2012.870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tsikas D. Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: analytical and biological challenges. Anal Biochem. 2017;524:13–30. doi: 10.1016/j.ab.2016.10.021. [DOI] [PubMed] [Google Scholar]
- 35.Jackson MJ. Reactive oxygen species in sarcopenia: should we focus on excess oxidative damage or defective redox signalling? Mol Aspects Med. 2016;50:33–40. doi: 10.1016/j.mam.2016.05.002. [DOI] [PubMed] [Google Scholar]


