Abstract
The triple-negative subtype of breast cancer (TNBC) has the bleakest prognosis, owing to its lack of either hormone receptor as well as human epidermal growth factor receptor 2. Henceforth, immunotherapy has emerged as the front-runner for TNBC treatment, which avoids potentially damaging chemotherapeutics. However, despite its documented association with aggressive side effects and developed resistance, immune checkpoint blockade continues to dominate the TNBC immunotherapy scene. These immune checkpoint blockade drawbacks necessitate the exploration of other immunotherapeutic methods that would expand options for TNBC patients. One such method is the exploitation and recruitment of natural killer cells, which by harnessing the innate rather than adaptive immune system could potentially circumvent the downsides of immune checkpoint blockade. In this review, the authors will elucidate the advantageousness of natural killer cell-based immuno-oncology in TNBC as well as demonstrate the need to more extensively research such therapies in the future.
Keywords: Triple negative breast cancer, Natural killer cells, Immune checkpoint blockades, Programmed death-ligand 1, Cytotoxic T-lymphocyte-associated protein 4, Natural killer lectin-like group 2 member D
Core tip: Triple-negative breast cancer patients have the worst prognosis and conventional therapeutic approaches. Triple-negative breast cancer patients are considered the patients of choice for immunotherapy. We are shedding light on immune checkpoint blockades, such as programmed death-ligand 1 inhibitors, and its recently discovered side effects and resistance. In this context, we highlight a potential weapon known as the innate immune system and its native soldiers, which are the natural killer cells.
INTRODUCTION
As the leading type of cancer in women worldwide comprising 24.2% of all female cancer patients as of 2018, breast cancer (BC) is a prime target of oncological research. As of 2018, 626679 deaths due to BC have been documented[1] with the actual mortality rates showing great variance according to stage of detection. This variance is demonstrated by the fact that a 99% survival rate is estimated for patients with the cancer still localized only to the breast, whereas patients in which the cancer has metastasized to more distant parts of the body show a much less encouraging survival rate of just 27%[2].
Diversity of BC
BC is by no means a uniform disease and is in fact represented by a number of different subtypes. Approximately 80% of BCs are estrogen receptor-positive (ER+) with the majority of these (65%) also being progesterone receptor-positive (PR+)[3,4]. This means that in these subsets of BC their proliferation is stimulated by the hormones progesterone and/or estrogen due to their characteristic overexpression of these hormone receptors. As such, the typical method of therapy for ER+/PR+ BC is based on targeting these receptors using blockers such as Tamoxifen™ or by curtailing synthesis of the hormones themselves via aromatase inhibitors such as Anastrazole™ and Letrozole™. About 20% of BCs are also human epidermal growth factor receptor 2 (HER2) positive. The HER2 pathway is a proliferative one, meaning its overexpression results in uncontrolled cell division. The HER2+ BC subtype is characterized as more aggressive in terms of tumor growth and spreading than others. However, despite its poor prognosis in relation to ER+ and PR+ subtypes, there remains a viable treatment strategy for HER2+ BC, which relies on the targeting of HER2 using monoclonal antibodies such as trastuzumab (commercially known as Herceptin™), which abrogates the aforementioned proliferative activity of these cells and consequently attenuates malignancy, both through the direct effects of receptor blockade as well as recruitment of several immune cells through antibody-dependent cellular cytotoxicity (ADCC)[3,5]. However, in roughly 10%-20% of BC cases, tumor cells are classified as negative for both hormone receptors and HER2. This case, known as triple-negative breast cancer (TNBC), is well recognized as the subtype with the poorest prognosis due to the lack of targeted therapeutic options[6,7]. TNBC survival rates are comparatively lower than non-TNBC ones as demonstrated by a study published in 2018 by Gonçalves Jr et al[8] that showed 5-year survival rates of 80.8% and 62.1% for non-TNBC and TNBC patients, respectively.
TNBC patients: Worst prognosis and poorest survival rates
As mentioned, TNBC provides the bleakest outlook of all BC subtypes. Dent et al[9] painted a picture of this in 2007 in an 8-year follow-up study of 1601 BC patients. Whilst a vast minority were TNBC patients (180; 11.2%), a significantly worse prognosis was demonstrated by their higher mortality rate (42.2% in TNBC vs 28% in other BC subtypes), disease recurrence (33.2% vs 20.4%), with all TNBC-related deaths occurring within 10 years of initial diagnosis as opposed to regular BC mortalities stretching up to 18 years post diagnosis[9]. A further study was conducted 1 year later on the same cohort investigating the metastatic effects of TNBC. Results were yet again discouraging: TNBC patients had a 23% risk factor of developing visceral metastasis within 10 years as opposed to just 9% of other BC patients[10]. To this effect, the relative lack of therapeutic options for TNBC is an undoubtedly grave issue.
Chemotherapeutic insufficiency in TNBC
Despite its ominous implications, TNBC responds quite well to traditional chemotherapy. Response rate to neoadjuvant therapy has actually been found to be significantly higher in TNBC patients in comparison with other subtypes, with one comprehensive study by Liedtke et al[11] on 255 TNBC patients (out of a 1118-BC patient cohort) clocking this difference at 22% vs 11%. The real issue of TNBC is the poor survival rate of those who do not respond to such chemotherapies adequately, mainly due to the lack of secondary therapeutic options that would otherwise be available to PR+, ER+ or HER2+ patients. In an attempt to alleviate this dilemma, researchers identified the defective DNA repair pathways characteristic of TNBC as a potential target. The enzyme poly (ADP-ribose) polymerase, normally known to contribute to base-excision DNA repair, has been shown to be dysfunctional in TNBC and contributes to the genetic instability of the disease[12]. As such, the poly (ADP-ribose) polymerase inhibitor iniparib has been tested in a combinatorial capacity with the chemotherapeutics gemcitabine and carboplatin. Whilst phase II trials were promising[13], phase III trials showed no considerable difference between combined therapy and sole chemotherapy[14]. This yet again underlines the immense struggle to find targeted therapies in TNBC.
WHY IMMUNOTHERAPY IN TNBC?
However, TNBC is associated with a high degree of chromosome instability and mutation, such as that of the tumor suppressor gene TP53[15,16]. Owing to this, mutant proteins produced by TNBC are hypothesized to be recognized by the immune system as unfamiliar antigens (i.e. neoantigens). This is one possible explanation to the characteristic and notable elevation of tumor-infiltrating lymphocyte (TIL) levels within the tumor microenvironments of TNBC patients, whereby lymphocytes are aggressively recruited to the site of malignancy. It is this observation of elevated TILs that prompted the reasoning that immunotherapy (i.e. stimulation of TILs or induced overexpression of neoantigens) would be an effective strategy to combat TNBC. This is further reinforced by the finding that higher TIL levels in TNBC are correlated to better prognoses amongst patients following the administration of immune-stimulating chemotherapeutic agents such as anthracyclines. The heterogeneity of TNBC cells has in recent years been thoroughly examined to identify any potential candidates for targeted therapy[17]. This has proven difficult, despite the identification of TNBC-specific antigens such as MAGE-A and NY-ESO-1. Usage of such antigens is merely confined to cancer “vaccines,” which enhance tumor immunogenicity by improving the action of immunotherapeutic agents such as immune checkpoint blockers (ICB)[18].
ICB in TNBC
The era of immuno-oncology has borne with it fresh hope for TNBC patients. As previously mentioned, the abundance of TILs in the TNBC microenvironment is indicative of immunotherapy being a promising treatment approach due to the disease’s evident immunogenicity. Naturally, evasion measures are undertaken by the disease that allows it to prosper even under the immune-heavy climate in which it grows. Upon analysis of expression patterns, the immune checkpoint ligand programmed-death ligand 1 was found to be significantly overexpressed amongst TNBC cell surfaces as one such evasion measure[19]. As such, the programmed death-1/programmed-death ligand 1 axis has risen as the chief target of TNBC immunotherapies with a number of programmed-death ligand 1-directed monoclonal antibodies are currently in clinical trial phases[20]. As is apparent from Table 1, the immunotherapeutic strategies of TNBC treatment are overwhelmingly biased towards ICB, with full approval still far off for most. Despite the promise of ICB in TNBC (well indicated by the progress into later trials for most monoclonal antibodies), some TNBC patients could yet find themselves short of options.
Table 1.
List of immunotherapeutic agents in various clinical trial phases and earliest expected completion dates of most significant trials[20]
| Drug | Function | Trial phase | Est. date of completion |
| Pembrolizumab | PD-1 Inhibitor | III | December 2019 |
| Nivolumab | PD-1 Inhibitor | II | August 2022 |
| Atezolizumab | PDL-1 Inhibitor | III | April 2020 |
| Avelumab | PDL-1 Inhibitor | III | June 2023 |
| Durvalumab | PD-1 Inhibitor | I/II | October 2019 |
| Tremelimumab | CTLA-4 Inhibitor | II | October 20181 |
Phase II complete, phase III not yet begun. PD: Programmed death; PDL: Programmed death ligand; CTLA-4: Cytotoxic T-lymphocyte-associated protein 4.
Limitations of ICB
Adverse inflammatory reactions: From a purely therapeutic standpoint, the effects of ICB treatment are extremely promising. Its positive response rates through the elevation of cytotoxic T-lymphocyte levels and efficacy as well as increased generation of T-helper cells are widely recognized. However, ICB by no means represents an anticancer “magic bullet” and poses issues of its own. Apart from the astronomical prices of ICB agents, a worrying problem commonly rears its head during ICB administration in the form of autoimmune events. Seeing as immune checkpoints normally serve as natural brakes to prevent prolonged or excessively severe immune responses, their blockade has predictably resulted in adverse inflammatory reactions in several cases. These responses vary greatly in terms of localization as well as severity; hypophysitis, gastroenteritis, enterocolitis, hepatitis and many other immune-mediated side effects have been observed in clinical studies, with their intensities ranging from grade 2 (moderate) to grade 5 (life-threatening or fatal)[21].
Primary and/or secondary resistance: In addition to autoimmune tendencies, another issue with ICB therapy is the emergence of resistance against it amongst treated individuals. According to clinical trial data, in addition to responders and innate nonresponders, a third category of patients who initially respond to ICB therapy but subsequently acquire resistance to it has been defined. ICB is based mainly on the regeneration of exhausted T-cells and thus largely relies on the three elements of T-cell expansion, T-cell potency, and T-cell memory formation. All three of these mechanisms have been found to be impaired in cases of ICB resistance[22].
While unnaturally mutated neoantigens serve as the basis for immune recognition of malignant cells, the selection pressure applied by the immune system results in a remainder of cancer cells that have never innately expressed such antigens or have adapted to effectively “conceal” themselves from TILs either by shedding their neoantigens or the ability to present them through mutational loss[23]. The downregulation of major histocompatibility complex (MHC) class I elements such as the β2-microglobulin domain has been documented amongst patients who acquired ICB therapy resistance. The lack of a proper antigen presentation apparatus in malignant cells in such cases greatly decreases the potential for antigen recognition and subsequent priming and clonal expansion of the T-cell pool, rendering ICB administration redundant whereby there is a numeric shortage of T-cells to be targeted[24].
The efficacy of cytotoxic T-cells has been found to be lacking within resistant individuals. Even after antigen recognition and proper expansion/activation of the T-cell population, the tumor microenvironment is far from an ideal place for T-cells to exert their effector functions. A cocktail of immunosuppressive cytokines, alternate immune checkpoints, inhibitory receptors and immunosuppressive leukocytes are all characteristic of the tumor microenvironment and contribute to the attenuation of T-cell effector functions even after ICB therapy. Most notably, a trend of loss-of-function mutations to the genes encoding Janus kinases 1 and 2 has been highlighted amongst the tumors of resistant patients. These two proteins are components of the Janus kinase/STAT pathway, which in an immune context is crucial to the production of numerous stimulatory cytokines necessary for proper T-cell efficacy[24].
The formation of memory antitumor CD8+ cells is one of the hallmarks of ICB therapy despite the unelucidated mechanism. Also characteristic of resistant patients is the impairment of this memory formation and consequent short-lived nature of reactivated cytotoxic T-lymphocytes[25]. This could possibly be attributed to epigenetic factors that influence exhausted cells away from memory cell formation and thereby result in long-term resistance to ICB[26].
As has been mentioned, ICB represents a new source of hope for problematic cancers such as TNBC. However, the combined prominence of autoimmunity and/or resistance in clinical studies of various malignancies indicates that TNBC will not behave any differently and will indeed display these same tendencies; once again leaving TNBC patients bereft of options. Henceforth, it is necessary to look into tertiary methods of TNBC treatment in tandem with ICB research in order to provide alternative pathways for cases in which both chemotherapy and ICB should fail.
HUMAN IMMUNE SYSTEM
Innate and adaptive immune arms
The three major types of lymphocytes that comprise the mammalian immune system are T-cells, B-cells and NK cells; each playing its own indisputable role in the eternal fight between host and foreign bodies or malignancies. Whilst T-cells and B-cells are members of the adaptive immune system that is triggered by the detection of foreign antigens in the blood, NK cells belong to the innate immune system[27]. This in turn implies a nonspecific mode of action whereby NK cells are not armed against particular antigens but are instead primed to fight any unnaturally altered cell. This is mainly achieved through immunoglobulin-like MHC class I-specific receptors on NK cell surfaces that phosphorylate internal inhibitory immunoreceptor tyrosine-based inhibitory motifs upon recognition of normally expressed MHC class I on functional host cells. When MHC class I is lacking or defective, as is common in virally infected and malignant cells, positive transcriptional signals are fed back to the NK cell that triggers their active cytolytic state. Other modes of activation, such as the recognition of stress-induced NK cell ligands or ADCC, also exist. It is because of this search and destroy mode of action that NK cells are often referred to as the “first line of defense” with regards to mammalian immunity[28].
Intricate cross talk between the innate and the adaptive arms of the immune system
The innate and adaptive immune systems are by no means secluded from one another; a constant flux of crosstalk between the two arms is characteristic of any properly functioning immune system as shown in Figure 1. An example of this is the “beacon” function of NK cells upon their encounter with malignant cells and consequent secretion of interferon-gamma. Interferon-gamma is noted for its activation of antigen-presenting cells such as macrophages and dendritic cells, which subsequently induce the effector functions of T-helper cells initiating a comprehensive cascade of adaptive immune responses[29]. ADCC, where NK cells are recruited to constant regions of antibodies (derived from adaptive B-cells) coating malignant cells, is a demonstration of how the adaptive arm can potentiate the innate arm (and not only vice versa) in what has grown to be an attractive immunotherapeutic strategy[30]. However, despite this evident harmonious relationship between the two arms, it has been demonstrated that, to a degree, the innate immune system can work alone in an antitumor capacity. O’Sullivan et al[31] illustrated this in a 2012 study involving groups of mice that were either: (1) Wild type; (2) Lacking an adaptive immune system; or (3) Deficient of both the adaptive and innate immune systems. Upon tumor induction, wild type mice naturally displayed the lowest rate of tumor growth. However, of the two remaining groups, mice in group 2 showed significantly more impaired tumor growth than group 3 mice, indicating a notably prominent role for innate level effector functions in antitumor immunity[31].
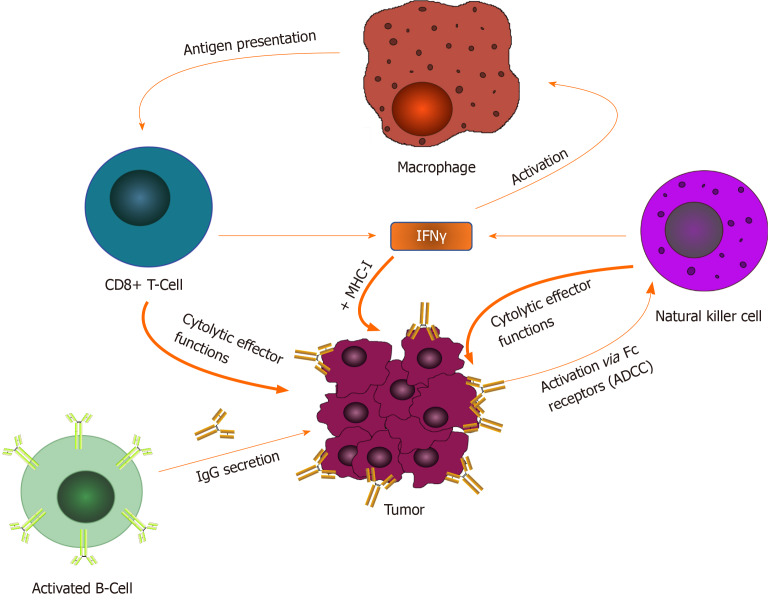
Figure 1.
Adaptive/innate intricate relationship. A demonstration of the interplay between various adaptive (IgG, cytotoxic T-lymphocytes, etc.) and innate (Natural killer cells, macrophages, etc.) factors in the mounting of a full antitumor immune response via cytokines such as interleukin-2 and interferon-gamma. IFNγ: Interferon-gamma: MHC-1: Major histocompatibility complex-1; ADCC: Antibody-dependent cellular cytotoxicity.
EXPLOITATION OF THE INNATE IMMUNE SYSTEM: A GAP IN THE IMMUNO-ONCOLOGICAL LANDSCAPE
Why innate-mediated immunotherapy?
As discussed previously, the modern immuno-oncology scene is dominated by ICB, whereby our understanding of immune checkpoints has led to the commercial release of various cytotoxic T-lymphocyte-associated protein 4 and programmed death-1 blockers such as ipilimumab, pembrolizumab and nivolumab[32,33]. Despite the evident focus of research on ICB and the resounding progress made over the past decade, only a small fraction (approximately 20%) of patients enjoy long-term benefits from these therapies[34]. This could, in part or in full, be attributed to the previously discussed resistance mechanisms of malignancies against ICB. Most of these mechanisms, such as the shedding of the antigen-presenting apparatus on malignant cell surfaces, the upregulation of alternative immune checkpoints or the reduction of memory cell formation, are evolutionary measures directed against T-cells in the tumor microenvironment, i.e. the adaptive immune system[23,33,35].
The incidence of resistance amongst cancer patients triggers the need to investigate on a molecular level. Whilst potential biomarkers for ICB efficacy have been investigated in a predictive sense[36], little progress has been made in elucidating the molecular bases by which acquired resistance in individual patients comes about[23]. Henceforth, it is imperative that these intricacies be clarified with a view of adopting different immunotherapeutic strategies.
The attractiveness of innate-mediated immunotherapy comes from the fact that, whilst not fully understood, ICB resistance mechanisms may be adaptive-specific and leave the door open for an innate approach. An example of this is the well-characterized shedding of human leukocyte antigen (HLA) molecules in cancer[37]. This disarming of tumor cells’ antigen-presenting capability results in their significantly decreased immunogenicity with regards to the adaptive immune system. However, given NK cells’ inherent tendency to fight off cells with improper MHC function, a gateway is opened for alternative treatment for patients whose cancer has evaded ICB through this adaptive-specific mutation. It is in scenarios such as this that the innate immune system should be explored as a viable treatment option.
With regards to autoimmunity, NK cell stimulation poses a much lower risk than their T-cell counterparts. NK cells are conventionally short-lived in their active form, indicating that therapies that rely on their stimulation and recruitment are unlikely to result in any prolonged inflammatory reaction leading to autoreactivity. Interestingly, an inverse relation was in fact proposed. This was brought about by the observation of NK cell deficiency or dysfunction in patients of autoimmune disorders[38]. One hypothesis for this is an ongoing “battle” between NK cells and autoreactive T-cells, which would indeed result in NK cell clearance during T-cell overstimulation. As such, therapies that involve NK cell stimulation could and have indeed in some studies shown positive effects on autoimmunity by the induced clearance of autoreactive T-cells[39].
NK CELLS: NATIVE SENTINELS OF THE INNATE IMMUNE ARM
Human NK cells are the natural guards of the innate immune system. They originate from hematopoietic stem cells and undergo maturation in the bone marrow[40]. NK cells are categorized as the third largest lymphocyte population[41]. NK cells represent 2%-18% in human peripheral blood[41]; they are also found in peripheral tissues like the liver, peritoneal cavity and placenta[42,43]. NK cells are phenotypically defined by the expression of CD56 and/or CD16 and the absence of the T-cell receptor CD3[44]. “Natural cytotoxicity” describes an effectively contributing phenomenon of NK cells as a first line of defense against viral infections and more in general against pathogens without prior sensitization as previously mentioned[28,45-47]. NK cells are also involved in immune surveillance against tumors and prevent dissemination of metastatic tumors[48,49]. These effector functions are mediated through cellular cytotoxicity (mainly through perforins and granzymes), in addition to secretion of several noncellular mediators such as chemokines and cytokines[49]. NK cells cross-talking among immune cells also play a regulatory control in mediating the anti-tumor adaptive immunity of T- and B-cells that in contrast require initial priming for the expression of their activity[50-52].
Subsets of NK cells
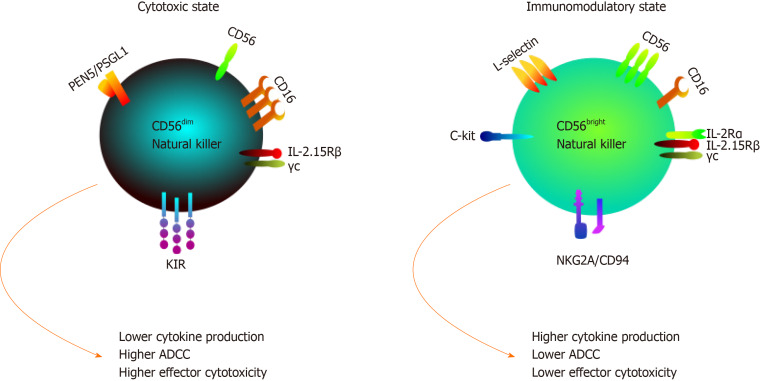
Human NK cells are classified according to the relative expression of the surface markers CD16 and CD56 into two major subsets/classes: CD56 dim and CD56 bright. These subsets differ in their function, phenotype and tissue localization[53]. The low density CD56 (CD56dim) subset comprises the majority (almost 90%) of peripheral blood NK cells and are characterized by their high expression of CD16, killer Ig-like receptors (KIRs) and perforins, making them more potent cytotoxic lymphocytes as illustrated in Figure 2. However, CD56bright NK cells are rarely present in peripheral blood but are predominant in lymph nodes, inflamed tissues and decidua[54,55]. The latter subset has a lower cytotoxicity as it expresses low levels of perforin and KIRs[56]. CD56bright NK cells are known as immunoregulatory NK cells as its functions are mainly mediated through the cytokine production such as interferon-gamma, tumor necrosis factor-α and transforming growth factor-β as shown in Figure 2[57,58].
Figure 2.
Natural killer cell subsets differ both functionally and phenotypically. Functionally, CD56dim natural killer (NK) cells are cytotoxic cells that produce low levels of cytokines in response to monokine stimulation. Yet, they are potent mediators of cytotoxic effector functions due to high levels of CD16 surface expression. Meanwhile CD56bright NK cells are known as immunoregulatory cells that produce high levels of cytokines such as interferon-gamma, interleukin-10 and transforming growth factor-β upon activation. It has low expression of CD16, thus performing reduced cytotoxic functions. Morphologically, CD56dim and CD56bright exhibit differential receptor profiles; for instance CD56dim NK cells exhibit much higher levels of killer Ig-like receptors, whereas resting CD56bright NK cells have high expression of CD94/NKG2A. ADCC: Antibody-dependent cellular cytotoxicity; IL: Interleukin; KIR: Killer Ig-like receptor.
REPERTOIRE OF RECEPTORS ORCHESTRATING NK CELL SIGNALS
NK cells have a unique feature of discriminating infected or malignant cells from normal “self” cells via a complex balance between activating and inhibitory receptor-ligand interactions[59]. In addition, the resting inactivated NK cell surface constitutively expresses a wide range of receptors, which upon their activation by different ligands initiate several downstream signaling pathways and result in boosting NK cytotoxicity and cytokine production[60].
Inhibitory receptors
Upon recognition of their respective ligands, inhibitory receptors on NK cells phosphorylate associated immunoreceptor tyrosine-based inhibitory motifs, which in turn recruit the phosphatases SHP and SHIP. They dephosphorylate transcriptional signaling molecules Lck, Syk, ZAP70, Vav1 and Fyn, which results in prevention of active-form transcription patterns. This pathway, along with the simultaneous dephosphorylation of activating immunoreceptor tyrosine-based activator motif domains, ensures that NK cells are not converted into an active state upon healthy host cell encounter and thus avoids events of autoimmunity. The aforementioned MHC class I-specific receptors are commonly referred to as KIRs and are almost all inhibitory in nature. KIRs are essential for tolerance towards host cells expressing classical MHC molecules (namely HLA-A, HLA-B and HLA-C). As for nonclassical MHCs, a heterodimer of CD94 and natural killer group 2 member A is responsible for detecting molecules such as HLA-E, which despite not being an archetypal MHC can be expressed amongst healthy host cells[61].
Activating receptors
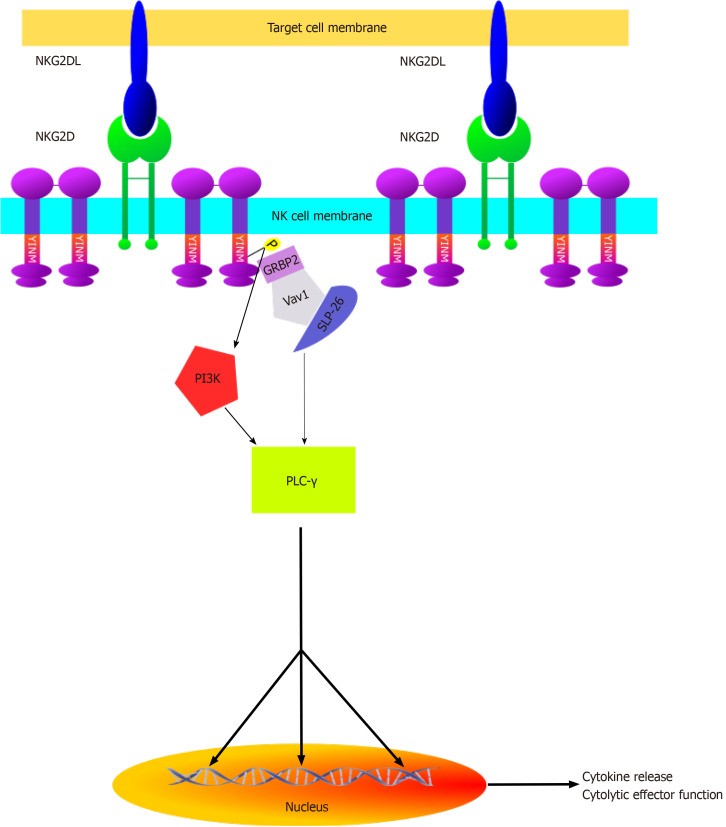
Natural killer lectin-like group 2, member D (NKG2D) is a hexameric transmembrane protein that acts as the principal activating receptor on NK cell surfaces. Ligands of NKG2D, termed NKG2DLs, act as triggers to the cytolytic mode of NK cells. As can be seen in Figure 3, NKG2D is associated with two DAP10 homodimers that each harbor two YINM motifs. Upon association with NKG2DLs, these motifs are phosphorylated and proceed to recruit the Grb2/Vav1/SLP-76 signaling complex and/or the p85 subunit of PI3-kinase. Taken together with the recruitment of the Syk/Zap70 complex by the alternate activating receptor Ly49D, these comprise the initial steps of three different cascades that have the downstream additive effect of amplifying both cytokine and cytotoxic molecule production within NK cells. Effectively “armed” for attack, NK cells proceed to kill their target either directly through apoptotic mechanisms such as the tumor necrosis factor, Fas/FasL, TRAIL and perforin/granzyme pathways or indirectly through stimulation of both innate and adaptive immune response via overproduction of cytokines such as IL-7, IL-12, and IL-15, which serve to upregulate NKG2D expression both on fellow NK cells and adaptive CD8+ cells[61].
Figure 3.
Natural killer lectin-like group 2, member D mode of action. Illustration of activation cascades initiated by the active conformations of natural killer lectin-like group 2, member D receptors in natural killer cells. NKG2D: Natural killer lectin-like group 2, member D; NKG2DL: Ligands of natural killer lectin-like group 2, member D.
NK cell ligands
NKG2DLs are mainly divided into two classes: MHC class I-related proteins (comprising MIC-A and MIC-B) and UL16 binding proteins (comprising ULBPs 1-6). Given the prominence of NKG2D amongst NK cells, the most integral aspect for the effective facilitation of tumor immunosurveillance is the differential expression pattern of NKG2DLs. To this effect, NKG2DLs are commonly overexpressed amongst malignancies. Indeed, studies have shown evidence for the increased expression of MIC-A and ULBPs 1-5 in all of colorectal cancer, ovarian cancer and BC along with further evidence of MIC-B expression in BC. As prognostic factors, NKG2DLs tend to be inconsistent. An example of this being ULBP 5 overexpression indicating a positive prognosis in colorectal cancer whereas ULBPs 2 and 4 correlate with a negative prognosis in ovarian cancer. The prognostic effects of various NKG2DLs on BC were elucidated in a comprehensive 2012 study by de Kruijf et al[62] of Leiden University. The results of the study yielded the finding that high MIC-A/B and ULBP 2 expression correlated with significantly more favorable prognoses as opposed to low expression with regards to tumor size, tumor grade and relapse rates.
NK CELLS IN CANCER
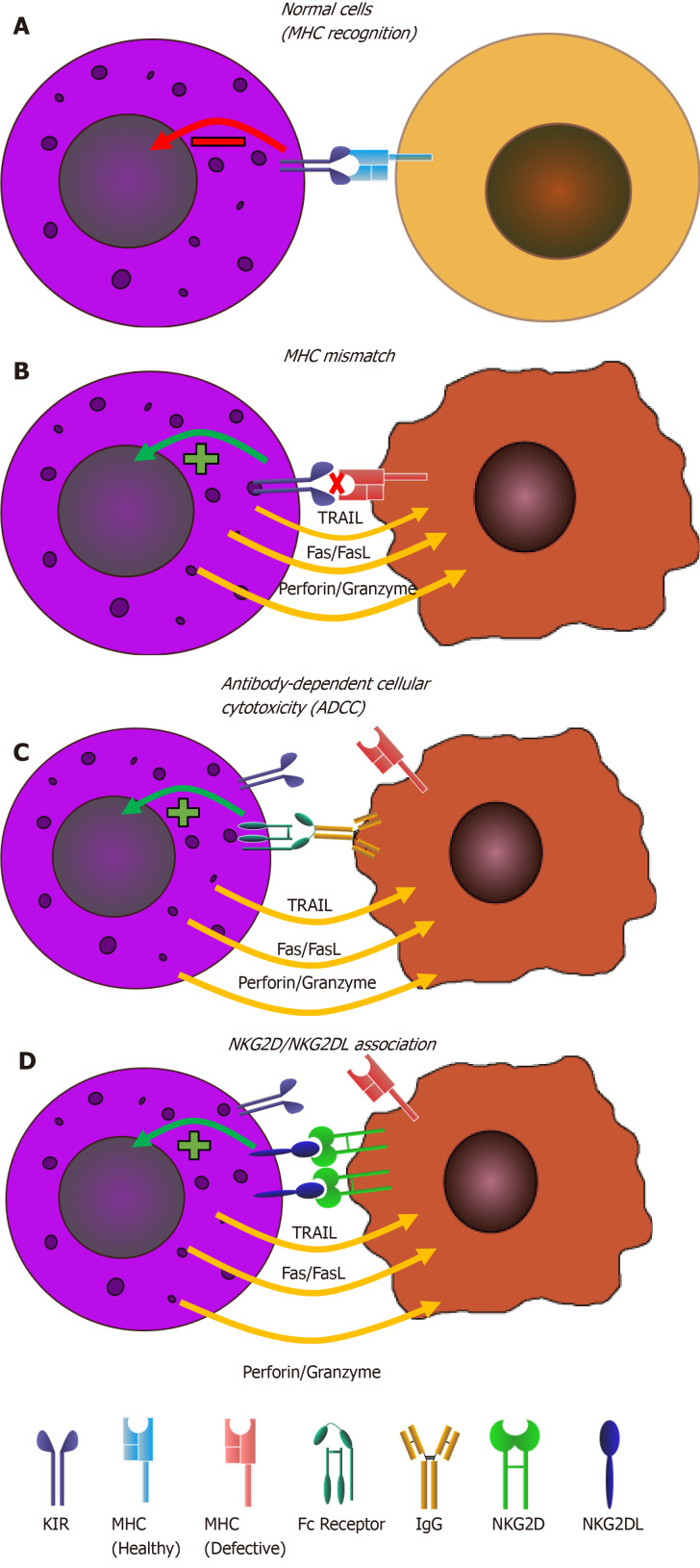
As shown in Figure 4, NK cells are equipped with these thorough mechanisms of host and nonhost recognition and therefore are important mediators of tumor immunosurveillance and eradication. This point is underlined by the higher susceptibility to cancer yielded by mice that are NK cell-deficient in comparison with wild type mice[28] as well as the higher susceptibility of mice lacking both adaptive and innate immune components in comparison with those lacking just the adaptive arm[31]. Consistent with these findings, the increased risk of cancer within humans with lower NK cell counts has been demonstrated through an 11-year follow-up study in 2000[63]. This is further corroborated by the observation of NK cell cytolytic impairment in various forms of cancer, such as non-small cell lung carcinoma and BC.
Figure 4.
Different modes of natural killer cell activation. Natural killer cell differential modes of activation when encountered with A: Healthy cells; B: Diseased cells with natural killer Ig-like receptors mismatch; C: Antibody-coated diseased cells; D: Diseased cells expressing natural killer lectin-like group 2, member D. NKG2D: Natural killer lectin-like group 2, member D; NKG2DL: Ligands of natural killer lectin-like group 2, member D; MHC: Major histocompatibility complex; KIR: Killer Ig-like receptor.
NK CELLS IN IMMUNO-ONCOLOGY
Given their favorable characteristics in terms of resistivity and autoimmunity in comparison with adaptive methods, NK cells are a prime candidate for novel immunotherapeutic research, and their exploitation has long been appealing. Attempts to achieve this include the upregulation of stimulatory receptors, downregulation of inhibitory ones, ADCC mediation, manipulation of cytolytic pathways such as Fas/FasL and TRAIL and NK cell activation through external agents such as vaccines and chemotherapeutic drugs[38]. In adoptive NK cell treatment involving external cell transfusions, it has indeed been demonstrated that increased NK cell count correlates to a favorable outcome in diseases such as non-Hodgkin lymphoma[64]. With NK cells having been proven integral to anticancer immunity, recent significant attention has turned to manipulating the tumor microenvironment in an NK cell-stimulatory capacity in ways such as induced expression of activating NKG2DLs on cancer cell surfaces.
Despite such promise, NK cell-based therapies have hardly made any clinical progress as opposed to more popular immunotherapeutic strategies such as ICB. Given ICB’s limitations, it is imperative that more attention be turned to this scope of research in the near future as a means to provide patients of aggressive and difficult-to-treat cancers more gravely needed leeway in terms of therapy options, one of the biggest examples being TNBC patients.
Footnotes
Conflict-of-interest statement: The authors declare no conflicts of interest.
Manuscript source: Invited manuscript
Peer-review started: March 12, 2020
First decision: April 22, 2020
Article in press: May 29, 2020
Specialty type: Oncology
Country/Territory of origin: Egypt
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Papazafiropoulou A S-Editor: Wang JL L-Editor: Filipodia E-Editor: Liu MY
Contributor Information
Mustafa Abdel-Latif, Biotechnology Program, Faculty of Pharmacy and Biotechnology, German University in Cairo, Cairo 11835, Egypt.
Rana Ahmed Youness, Pharmaceutical Biology Department, Faculty of Pharmacy and Biotechnology, German University in Cairo, Cairo 11835, Egypt. rana.youness21@gmail.com.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Noone AM, Howlader N, Krapcho M, Miller D, Brest A, Yu M, Ruhl J, Tatalovich Z, Mariotto A, Lewis DR, Chen HS, Feuer EJ, Cronin KA, editors. SEER Cancer Statistics Review, 1975-2015. Bethesda: National Cancer Institute, 2018. [Google Scholar]
- 3.Youness RA, Gad MZ. Long non-coding RNAs: Functional regulatory players in breast cancer. Noncoding RNA Res. 2019;4:36–44. doi: 10.1016/j.ncrna.2019.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Youness RA, Hafez HM, Khallaf E, Assal RA, Abdel Motaal A, Gad MZ. The long noncoding RNA sONE represses triple-negative breast cancer aggressiveness through inducing the expression of miR-34a, miR-15a, miR-16, and let-7a. J Cell Physiol. 2019;234:20286–20297. doi: 10.1002/jcp.28629. [DOI] [PubMed] [Google Scholar]
- 5.Junttila TT, Parsons K, Olsson C, Lu Y, Xin Y, Theriault J, Crocker L, Pabonan O, Baginski T, Meng G, Totpal K, Kelley RF, Sliwkowski MX. Superior in vivo efficacy of afucosylated trastuzumab in the treatment of HER2-amplified breast cancer. Cancer Res. 2010;70:4481–4489. doi: 10.1158/0008-5472.CAN-09-3704. [DOI] [PubMed] [Google Scholar]
- 6.Youness RA, Assal RA, Abdel Motaal A, Gad MZ. A novel role of sONE/NOS3/NO signaling cascade in mediating hydrogen sulphide bilateral effects on triple negative breast cancer progression. Nitric Oxide. 2018;80:12–23. doi: 10.1016/j.niox.2018.07.004. [DOI] [PubMed] [Google Scholar]
- 7.Awad AR, Youness RA, Ibrahim M, Motaal AA, El-Askary HI, Assal RA, Gad MZ. An acetylated derivative of vitexin halts MDA-MB-231 cellular progression and improves its immunogenic profile through tuning miR- 20a-MICA/B axis. Nat Prod Res. 2019:1–5. doi: 10.1080/14786419.2019.1686372. [DOI] [PubMed] [Google Scholar]
- 8.Gonçalves H Jr, Guerra MR, Duarte Cintra JR, Fayer VA, Brum IV, Bustamante Teixeira MT. Survival Study of Triple-Negative and Non-Triple-Negative Breast Cancer in a Brazilian Cohort. Clin Med Insights Oncol. 2018;12:1179554918790563. doi: 10.1177/1179554918790563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dent R, Trudeau M, Pritchard KI, Hanna WM, Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P, Narod SA. Triple-negative breast cancer: clinical features and patterns of recurrence. Clin Cancer Res. 2007;13:4429–4434. doi: 10.1158/1078-0432.CCR-06-3045. [DOI] [PubMed] [Google Scholar]
- 10.Dent R, Hanna WM, Trudeau M, Rawlinson E, Sun P, Narod SA. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res Treat. 2009;115:423–428. doi: 10.1007/s10549-008-0086-2. [DOI] [PubMed] [Google Scholar]
- 11.Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B, Green M, Cristofanilli M, Hortobagyi GN, Pusztai L. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26:1275–1281. doi: 10.1200/JCO.2007.14.4147. [DOI] [PubMed] [Google Scholar]
- 12.Alli E, Sharma VB, Sunderesakumar P, Ford JM. Defective repair of oxidative dna damage in triple-negative breast cancer confers sensitivity to inhibition of poly(ADP-ribose) polymerase. Cancer Res. 2009;69:3589–3596. doi: 10.1158/0008-5472.CAN-08-4016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.O'Shaughnessy J, Osborne C, Pippen JE, Yoffe M, Patt D, Rocha C, Koo IC, Sherman BM, Bradley C. Iniparib plus chemotherapy in metastatic triple-negative breast cancer. N Engl J Med. 2011;364:205–214. doi: 10.1056/NEJMoa1011418. [DOI] [PubMed] [Google Scholar]
- 14.O'Shaughnessy J, Schwartzberg L, Danso MA, Miller KD, Rugo HS, Neubauer M, Robert N, Hellerstedt B, Saleh M, Richards P, Specht JM, Yardley DA, Carlson RW, Finn RS, Charpentier E, Garcia-Ribas I, Winer EP. Phase III study of iniparib plus gemcitabine and carboplatin versus gemcitabine and carboplatin in patients with metastatic triple-negative breast cancer. J Clin Oncol. 2014;32:3840–3847. doi: 10.1200/JCO.2014.55.2984. [DOI] [PubMed] [Google Scholar]
- 15.Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature. 2012;490:61–70. doi: 10.1038/nature11412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.ElKhouly AM, Youness RA, Gad MZ. MicroRNA-486-5p and microRNA-486-3p: Multifaceted pleiotropic mediators in oncological and non-oncological conditions. Noncoding RNA Res. 2020;5:11–21. doi: 10.1016/j.ncrna.2020.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu T, Di G. Role of tumor microenvironment in triple-negative breast cancer and its prognostic significance. Chin J Cancer Res. 2017;29:237–252. doi: 10.21147/j.issn.1000-9604.2017.03.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yuan J, Adamow M, Ginsberg BA, Rasalan TS, Ritter E, Gallardo HF, Xu Y, Pogoriler E, Terzulli SL, Kuk D, Panageas KS, Ritter G, Sznol M, Halaban R, Jungbluth AA, Allison JP, Old LJ, Wolchok JD, Gnjatic S. Integrated NY-ESO-1 antibody and CD8+ T-cell responses correlate with clinical benefit in advanced melanoma patients treated with ipilimumab. Proc Natl Acad Sci USA. 2011;108:16723–16728. doi: 10.1073/pnas.1110814108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mittendorf EA, Philips AV, Meric-Bernstam F, Qiao N, Wu Y, Harrington S, Su X, Wang Y, Gonzalez-Angulo AM, Akcakanat A, Chawla A, Curran M, Hwu P, Sharma P, Litton JK, Molldrem JJ, Alatrash G. PD-L1 expression in triple-negative breast cancer. Cancer Immunol Res. 2014;2:361–370. doi: 10.1158/2326-6066.CIR-13-0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Katz H, Alsharedi M. Immunotherapy in triple-negative breast cancer. Med Oncol. 2017;35:13. doi: 10.1007/s12032-017-1071-6. [DOI] [PubMed] [Google Scholar]
- 21.Fellner C. Ipilimumab (yervoy) prolongs survival in advanced melanoma: serious side effects and a hefty price tag may limit its use. P T. 2012;37:503–530. [PMC free article] [PubMed] [Google Scholar]
- 22.Jenkins RW, Barbie DA, Flaherty KT. Mechanisms of resistance to immune checkpoint inhibitors. Br J Cancer. 2018;118:9–16. doi: 10.1038/bjc.2017.434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Anagnostou V, Smith KN, Forde PM, Niknafs N, Bhattacharya R, White J, Zhang T, Adleff V, Phallen J, Wali N, Hruban C, Guthrie VB, Rodgers K, Naidoo J, Kang H, Sharfman W, Georgiades C, Verde F, Illei P, Li QK, Gabrielson E, Brock MV, Zahnow CA, Baylin SB, Scharpf RB, Brahmer JR, Karchin R, Pardoll DM, Velculescu VE. Evolution of Neoantigen Landscape during Immune Checkpoint Blockade in Non-Small Cell Lung Cancer. Cancer Discov. 2017;7:264–276. doi: 10.1158/2159-8290.CD-16-0828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zaretsky JM, Garcia-Diaz A, Shin DS, Escuin-Ordinas H, Hugo W, Hu-Lieskovan S, Torrejon DY, Abril-Rodriguez G, Sandoval S, Barthly L, Saco J, Homet Moreno B, Mezzadra R, Chmielowski B, Ruchalski K, Shintaku IP, Sanchez PJ, Puig-Saus C, Cherry G, Seja E, Kong X, Pang J, Berent-Maoz B, Comin-Anduix B, Graeber TG, Tumeh PC, Schumacher TN, Lo RS, Ribas A. Mutations Associated with Acquired Resistance to PD-1 Blockade in Melanoma. N Engl J Med. 2016;375:819–829. doi: 10.1056/NEJMoa1604958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ribas A, Shin DS, Zaretsky J, Frederiksen J, Cornish A, Avramis E, Seja E, Kivork C, Siebert J, Kaplan-Lefko P, Wang X, Chmielowski B, Glaspy JA, Tumeh PC, Chodon T, Pe'er D, Comin-Anduix B. PD-1 Blockade Expands Intratumoral Memory T Cells. Cancer Immunol Res. 2016;4:194–203. doi: 10.1158/2326-6066.CIR-15-0210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sen DR, Kaminski J, Barnitz RA, Kurachi M, Gerdemann U, Yates KB, Tsao HW, Godec J, LaFleur MW, Brown FD, Tonnerre P, Chung RT, Tully DC, Allen TM, Frahm N, Lauer GM, Wherry EJ, Yosef N, Haining WN. The epigenetic landscape of T cell exhaustion. Science. 2016;354:1165–1169. doi: 10.1126/science.aae0491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Youness RA, Rahmoon MA, Assal RA, Gomaa AI, Hamza MT, Waked I, El Tayebi HM, Abdelaziz AI. Contradicting interplay between insulin-like growth factor-1 and miR-486-5p in primary NK cells and hepatoma cell lines with a contemporary inhibitory impact on HCC tumor progression. Growth Factors. 2016;34:128–140. doi: 10.1080/08977194.2016.1200571. [DOI] [PubMed] [Google Scholar]
- 28.Vivier E, Tomasello E, Baratin M, Walzer T, Ugolini S. Functions of natural killer cells. Nat Immunol. 2008;9:503–510. doi: 10.1038/ni1582. [DOI] [PubMed] [Google Scholar]
- 29.Guermonprez P, Valladeau J, Zitvogel L, Théry C, Amigorena S. Antigen presentation and T cell stimulation by dendritic cells. Annu Rev Immunol. 2002;20:621–667. doi: 10.1146/annurev.immunol.20.100301.064828. [DOI] [PubMed] [Google Scholar]
- 30.Zhu EF, Gai SA, Opel CF, Kwan BH, Surana R, Mihm MC, Kauke MJ, Moynihan KD, Angelini A, Williams RT, Stephan MT, Kim JS, Yaffe MB, Irvine DJ, Weiner LM, Dranoff G, Wittrup KD. Synergistic innate and adaptive immune response to combination immunotherapy with anti-tumor antigen antibodies and extended serum half-life IL-2. Cancer Cell. 2015;27:489–501. doi: 10.1016/j.ccell.2015.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.O'Sullivan T, Saddawi-Konefka R, Vermi W, Koebel CM, Arthur C, White JM, Uppaluri R, Andrews DM, Ngiow SF, Teng MW, Smyth MJ, Schreiber RD, Bui JD. Cancer immunoediting by the innate immune system in the absence of adaptive immunity. J Exp Med. 2012;209:1869–1882. doi: 10.1084/jem.20112738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Allard B, Aspeslagh S, Garaud S, Dupont FA, Solinas C, Kok M, Routy B, Sotiriou C, Stagg J, Buisseret L. Immuno-oncology-101: overview of major concepts and translational perspectives. Semin Cancer Biol. 2018;52:1–11. doi: 10.1016/j.semcancer.2018.02.005. [DOI] [PubMed] [Google Scholar]
- 33.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer. 2012;12:252–264. doi: 10.1038/nrc3239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Morfouace M, Hewitt SM, Salgado R, Hartmann K, Litiere S, Tejpar S, Golfinopoulos V, Lively T, Thurin M, Conley B, Lacombe D. A transatlantic perspective on the integration of immuno-oncology prognostic and predictive biomarkers in innovative clinical trial design. Semin Cancer Biol. 2018;52:158–165. doi: 10.1016/j.semcancer.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 35.O'Donnell JS, Long GV, Scolyer RA, Teng MW, Smyth MJ. Resistance to PD1/PDL1 checkpoint inhibition. Cancer Treat Rev. 2017;52:71–81. doi: 10.1016/j.ctrv.2016.11.007. [DOI] [PubMed] [Google Scholar]
- 36.Topalian SL, Taube JM, Anders RA, Pardoll DM. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer. 2016;16:275–287. doi: 10.1038/nrc.2016.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shukla SA, Rooney MS, Rajasagi M, Tiao G, Dixon PM, Lawrence MS, Stevens J, Lane WJ, Dellagatta JL, Steelman S, Sougnez C, Cibulskis K, Kiezun A, Hacohen N, Brusic V, Wu CJ, Getz G. Comprehensive analysis of cancer-associated somatic mutations in class I HLA genes. Nat Biotechnol. 2015;33:1152–1158. doi: 10.1038/nbt.3344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Terme M, Ullrich E, Delahaye NF, Chaput N, Zitvogel L. Natural killer cell-directed therapies: moving from unexpected results to successful strategies. Nat Immunol. 2008;9:486–494. doi: 10.1038/ni1580. [DOI] [PubMed] [Google Scholar]
- 39.Bielekova B, Catalfamo M, Reichert-Scrivner S, Packer A, Cerna M, Waldmann TA, McFarland H, Henkart PA, Martin R. Regulatory CD56(bright) natural killer cells mediate immunomodulatory effects of IL-2Ralpha-targeted therapy (daclizumab) in multiple sclerosis. Proc Natl Acad Sci USA. 2006;103:5941–5946. doi: 10.1073/pnas.0601335103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Freud AG, Becknell B, Roychowdhury S, Mao HC, Ferketich AK, Nuovo GJ, Hughes TL, Marburger TB, Sung J, Baiocchi RA, Guimond M, Caligiuri MA. A human CD34(+) subset resides in lymph nodes and differentiates into CD56bright natural killer cells. Immunity. 2005;22:295–304. doi: 10.1016/j.immuni.2005.01.013. [DOI] [PubMed] [Google Scholar]
- 41.Zimmer J. Natural Killer Cells at the Forefront of Modern Immunology. Springer, 2009. [Google Scholar]
- 42.Grégoire C, Chasson L, Luci C, Tomasello E, Geissmann F, Vivier E, Walzer T. The trafficking of natural killer cells. Immunol Rev. 2007;220:169–182. doi: 10.1111/j.1600-065X.2007.00563.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Smyth MJ, Hayakawa Y, Takeda K, Yagita H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat Rev Cancer. 2002;2:850–861. doi: 10.1038/nrc928. [DOI] [PubMed] [Google Scholar]
- 44.Farag SS, Fehniger TA, Ruggeri L, Velardi A, Caligiuri MA. Natural killer cell receptors: new biology and insights into the graft-versus-leukemia effect. Blood. 2002;100:1935–1947. doi: 10.1182/blood-2002-02-0350. [DOI] [PubMed] [Google Scholar]
- 45.Jost S, Altfeld M. Control of human viral infections by natural killer cells. Annu Rev Immunol. 2013;31:163–194. doi: 10.1146/annurev-immunol-032712-100001. [DOI] [PubMed] [Google Scholar]
- 46.Watzl C, Urlaub D. Molecular mechanisms of natural killer cell regulation. Front Biosci (Landmark Ed) 2012;17:1418–1432. doi: 10.2741/3995. [DOI] [PubMed] [Google Scholar]
- 47.Herberman RB, Nunn ME, Holden HT, Lavrin DH. Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic and allogeneic tumors. II. Characterization of effector cells. Int J Cancer. 1975;16:230–239. doi: 10.1002/ijc.2910160205. [DOI] [PubMed] [Google Scholar]
- 48.Herberman RB, Nunn ME, Lavrin DH. Natural cytotoxic reactivity of mouse lymphoid cells against syngeneic acid allogeneic tumors. I. Distribution of reactivity and specificity. Int J Cancer. 1975;16:216–229. doi: 10.1002/ijc.2910160204. [DOI] [PubMed] [Google Scholar]
- 49.Peter HH, Kalden JR, Seeland P, Diehl V, Eckert G. Humoral and cellular immune reactions 'in vitro' against allogeneic and autologous human melanoma cells. Clin Exp Immunol. 1975;20:193–207. [PMC free article] [PubMed] [Google Scholar]
- 50.Kiessling R, Klein E, Wigzell H. "Natural" killer cells in the mouse. I. Cytotoxic cells with specificity for mouse Moloney leukemia cells. Specificity and distribution according to genotype. Eur J Immunol. 1975;5:112–117. doi: 10.1002/eji.1830050208. [DOI] [PubMed] [Google Scholar]
- 51.Kiessling R, Klein E, Pross H, Wigzell H. "Natural" killer cells in the mouse. II. Cytotoxic cells with specificity for mouse Moloney leukemia cells. Characteristics of the killer cell. Eur J Immunol. 1975;5:117–121. doi: 10.1002/eji.1830050209. [DOI] [PubMed] [Google Scholar]
- 52.Agaugué S, Marcenaro E, Ferranti B, Moretta L, Moretta A. Human natural killer cells exposed to IL-2, IL-12, IL-18, or IL-4 differently modulate priming of naive T cells by monocyte-derived dendritic cells. Blood. 2008;112:1776–1783. doi: 10.1182/blood-2008-02-135871. [DOI] [PubMed] [Google Scholar]
- 53.Trinchieri G. Biology of natural killer cells. Adv Immunol. 1989;47:187–376. doi: 10.1016/S0065-2776(08)60664-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Paust S, Senman B, von Andrian UH. Adaptive immune responses mediated by natural killer cells. Immunol Rev. 2010;235:286–296. doi: 10.1111/j.0105-2896.2010.00906.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Di Santo JP. Natural killer cell developmental pathways: a question of balance. Annu Rev Immunol. 2006;24:257–286. doi: 10.1146/annurev.immunol.24.021605.090700. [DOI] [PubMed] [Google Scholar]
- 56.Orange JS, Ballas ZK. Natural killer cells in human health and disease. Clin Immunol. 2006;118:1–10. doi: 10.1016/j.clim.2005.10.011. [DOI] [PubMed] [Google Scholar]
- 57.Lanier LL, Le AM, Civin CI, Loken MR, Phillips JH. The relationship of CD16 (Leu-11) and Leu-19 (NKH-1) antigen expression on human peripheral blood NK cells and cytotoxic T lymphocytes. J Immunol. 1986;136:4480–4486. [PubMed] [Google Scholar]
- 58.Fehniger TA, Cooper MA, Nuovo GJ, Cella M, Facchetti F, Colonna M, Caligiuri MA. CD56bright natural killer cells are present in human lymph nodes and are activated by T cell-derived IL-2: a potential new link between adaptive and innate immunity. Blood. 2003;101:3052–3057. doi: 10.1182/blood-2002-09-2876. [DOI] [PubMed] [Google Scholar]
- 59.Fontenot JD, Rasmussen JP, Gavin MA, Rudensky AY. A function for interleukin 2 in Foxp3-expressing regulatory T cells. Nat Immunol. 2005;6:1142–1151. doi: 10.1038/ni1263. [DOI] [PubMed] [Google Scholar]
- 60.Ferlazzo G, Thomas D, Lin SL, Goodman K, Morandi B, Muller WA, Moretta A, Münz C. The abundant NK cells in human secondary lymphoid tissues require activation to express killer cell Ig-like receptors and become cytolytic. J Immunol. 2004;172:1455–1462. doi: 10.4049/jimmunol.172.3.1455. [DOI] [PubMed] [Google Scholar]
- 61.Bryceson YT, Fauriat C, Nunes JM, Wood SM, Björkström NK, Long EO, Ljunggren HG. Functional analysis of human NK cells by flow cytometry. Methods Mol Biol. 2010;612:335–352. doi: 10.1007/978-1-60761-362-6_23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Cooper MA, Fehniger TA, Turner SC, Chen KS, Ghaheri BA, Ghayur T, Carson WE, Caligiuri MA. Human natural killer cells: a unique innate immunoregulatory role for the CD56(bright) subset. Blood. 2001;97:3146–3151. doi: 10.1182/blood.v97.10.3146. [DOI] [PubMed] [Google Scholar]
- 63.Lanier LL. NK cell recognition. Annu Rev Immunol. 2005;23:225–274. doi: 10.1146/annurev.immunol.23.021704.115526. [DOI] [PubMed] [Google Scholar]
- 64.Kovanen PE, Leonard WJ. Cytokines and immunodeficiency diseases: critical roles of the gamma(c)-dependent cytokines interleukins 2, 4, 7, 9, 15, and 21, and their signaling pathways. Immunol Rev. 2004;202:67–83. doi: 10.1111/j.0105-2896.2004.00203.x. [DOI] [PubMed] [Google Scholar]
- 65.Paul S, Lal G. The Molecular Mechanism of Natural Killer Cells Function and Its Importance in Cancer Immunotherapy. Front Immunol. 2017;8:1124. doi: 10.3389/fimmu.2017.01124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.de Kruijf EM, Sajet A, van Nes JG, Putter H, Smit VT, Eagle RA, Jafferji I, Trowsdale J, Liefers GJ, van de Velde CJ, Kuppen PJ. NKG2D ligand tumor expression and association with clinical outcome in early breast cancer patients: an observational study. BMC Cancer. 2012;12:24. doi: 10.1186/1471-2407-12-24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Imai K, Matsuyama S, Miyake S, Suga K, Nakachi K. Natural cytotoxic activity of peripheral-blood lymphocytes and cancer incidence: an 11-year follow-up study of a general population. Lancet. 2000;356:1795–1799. doi: 10.1016/S0140-6736(00)03231-1. [DOI] [PubMed] [Google Scholar]
- 68.Porrata LF, Inwards DJ, Ansell SM, Micallef IN, Johnston PB, Gastineau DA, Litzow MR, Winters JL, Markovic SN. Early lymphocyte recovery predicts superior survival after autologous stem cell transplantation in non-Hodgkin lymphoma: a prospective study. Biol Blood Marrow Transplant. 2008;14:807–816. doi: 10.1016/j.bbmt.2008.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]