Abstract
The field of nanomedicine is constantly expanding. Since the first work dated in 1999, almost 28 thousand articles have been published, and more and more are published every year: just think that only in the last five years 20,855 have come out (source PUBMED) including original research and reviews. The goal of this review is to present the current knowledge about nanomedicine in Alzheimer’s disease, a widespread neurodegenerative disorder in the over 60 population that deeply affects memory and cognition. Thus, after a brief introduction on the pathology and on the state-of-the-art research for NPs passing the BBB, special attention is placed to new targets that can enter the interest of nanoparticle designers and to new promising therapies. The authors performed a literature review limited to the last three years (2017–2020) of available studies with the intention to present only novel formulations or approaches where at least in vitro studies have been performed. This choice was made because, while limiting the sector to nanotechnology applied to Alzheimer, an organic census of all the relevant news is difficult to obtain.
Keywords: Alzheimer’s disease, therapy, nanomedicine, nanoparticles, nutraceutical, intranasal route, green synthesis, stem cells
Introduction
Dementia, to date, affects ~47 million of people worldwide, and, since the average age of the patients is over 60, this number is expected to reach ~131 million by 2050, mostly because of the rapid expansion of lifespan.1 Alzheimer’s disease (AD) is one of the most common progressive neurodegenerative disorders that leads to dementia among the elderly. The first clinical manifestations of the disease are usually short-term memory loss, cognitive impairment, psychiatric symptoms, behaviors disturbances and then dementia, physical impairment and premature death.2 Typical AD pathological hallmarks are extracellular deposition of amyloid plaques, containing aggregates of Amyloid-beta (Aβ) peptides, and intracellular accumulation of neurofibrillary tangles (NFTs). Aβ fibrils are formed during the processing of transmembrane amyloid precursor protein (APP), a protein that plays an important role in neurons but whose functions have yet to be completely defined. NFTs are, instead, composed by hyperphosphorylated tau protein, a microtubule-binding protein acting to maintain the proper cytoskeleton and cytoskeletal network.3 Moreover, the reduced synthesis of acetylcholine, the neurotransmitter related to learning, memory, performance, behavior, and emotional responses in AD patients' brains causes a deterioration of basal forebrain cholinergic neurons (BFCNs), cerebral cortex atrophy, cortical neurons reduction,4 strong oxidative stress, and inflammation signs.5
The onset of the disease depends on several elements including inheritance, sex, aging, environmental, social factors as well as various pathologies (ie, atherosclerosis, hypertension, hypercholesterolemia, type II diabetes, stroke, transient ischemic attack and brain trauma).6 Also epigenetic mechanisms, as DNA methylation or histones acetylation/deacetylation, are reported to affect memory processes, thus they may contribute to the complexity of AD.3 Despite the majority (90–95% of total cases) of AD patients are late and sporadic (sAD), some early familiar forms (5–10% of incidence; fAD) have been identified, mainly due to variants in APP, PS1 and PS2 genes (encoding presenilins, proteins that are part of the multicomplex that cleaves APP). All these variants, overall, contribute to an increased Aβ accumulation. Apolipoprotein E (APOE) is, instead, the main genetic modulator in sporadic patients. APOE proteins are involved in the amyloid deposition and some isoforms might act as either risk (APOE4) or protective (APOE2) factors towards AD.7
AD Pathophysiological Main Hypothesis
Since the first disease-causing hit has not been clearly identified yet, the discussion about the etiology of AD is still open. Are the lesions and their constituent proteins the true initiators of the disease due to their toxicity, or rather are they indicative of an active host response or an environmental adaptation? Among the many hypothesis, the “amyloid cascade” is the most common and it involves APP neuronal cleavage pathways. APP may alternatively be cleaved by α- and consecutively γ-secretases, leading to nonamyloidogenic processing. In healthy people, Aβ production rate (ie, 7.6%) is normally lower than its rate of clearance (ie, 8.3%) per hour.8 In AD, APP is processed by β- followed by γ-secretases, resulting in amyloidogenic pathway and causing the formation of Aβ protein fragments of 40 (Aβ40) or of 42 (Aβ42) amino acid length. As this latter can aggregate in insoluble amyloid fibrils, when the ratio between Aβ42/Aβ40 is imbalanced towards Aβ42, oligomers formation prevails, extracellular amyloid plaques are deposited and, in turn, synaptic structure and plasticity alterations sustained a cognitive decline.2 Moreover, AD patients show a reduction in Aβ degradation rate and clearance, both contributing to a net Aβ accumulation.9 The Aβ polymerization has a variety of cytotoxic effects both in vitro and in vivo: it affects the mitochondrial redox balance, therefore causing oxidative stress and calcium overload, it disrupts the autophagic pathway and the acetylcholine and glutamate neurotransmission. Growing evidence suggests that Aβ toxicity also involves downregulation of specific miRNAs targeting β-secretase 1, thus contributing to its own overproduction. It activates the hyperphosphorylation of tau protein through the impairment of the insulin-signalling pathway, that, in turn, affects microtubule stabilization and leads to NFTs formation, severely compromising neuronal communication. Aβ and hyperphosphorylated tau aggregates contribute together to the recruitment and to the activation of microglia, causing a neuroinflammation picture that spreads neuronal and vascular dysfunctions through proinflammatory cytokines secretion.3,6
Clinical trials, despite enhanced removal of amyloid plaques, failed to prevent cognitive deterioration and even accelerated neuronal loss. Hence, the correlation of the plaques presence with the severity of the disease is weak and, although an important role in the disease etiology is still recognized for Aβ aggregates, some theories are now scaling it back. Indeed, Aβ accumulation seems to be necessary but not sufficient to cause AD and it happens several years before the first clinical manifestations of the disease.7
The “two-hit vascular hypothesis” suggests that AD is the result of a two-steps process in which a first damage within brain circulation leads to a reduced cerebral blood flow and to BBB dysfunction. Then, these alterations contribute together to the second “hit”, consisting in the accumulation of neurotoxic substances and of Aβ aggregates due to both a decrease in its clearance and a rise in its production. This hypothesis supports the idea that AD and vascular dementia are not two separate pathologies, rather a continuum disease manifesting with several degree of heterogeneity and with a still not well-established temporal link.9
Another reported theory is the “AD cholinergic hypothesis”, that focuses on the alterations in choline uptake, acetylcholine synthesis and acetylcholine esterase activity; all of them resulting in a progressive and irreversible impairment of BFCNs involved in the control of memory and learning.2 Moreover, an upstream neurotrophic deficit has been proposed as a linking mechanism between Aβ dysfunction, tau accumulation and cholinergic impairment.2
In the “mitochondrial cascade hypothesis”, the abnormal neuronal functions are thought to be a consequence of subsequent mutations on mitochondrial DNA, resulting in a lower ATP production and a higher oxidative stress. Furthermore, these events should also be responsible for proteasome dysfunctions, and for an increase in NFTs and Aβ aggregates.10
Besides these principal hypothesis, other mechanisms have been suggested in order to explain the pathophysiology of AD,7 however, at now, none of them is exhaustive probably due to the fact that various simultaneous processes contribute to the high complexity of the disease.
Currently FDA-Approved Drugs for AD Therapy
Nowadays, only five drugs are approved by the FDA for AD treatment: tacrine, donepezil, rivastigmine, galantamine, and memantine. The first four are acetylcholine esterase inhibitors (AchEIs), while the latter is an N-methyl-D-aspartate receptor antagonist.11 However, the current status about AD therapeutic options mirrors the incomplete understanding of the pathological mechanisms of disease, thus these drugs only address symptoms. Despite they could not block the progression of the disease, they temporarily increase cognitive functions through a partial amelioration of cholinergic and glutamatergic neurotransmission.10 However, AchEIs manifest some limitations as they are unstable in the circulation, show unpredictable uptake and bioavailability and may cause gastrointestinal complications.12
Of course, there is a great effort to identify new therapeutic strategies in order to overcome the limitations of those that are now available. Despite the difficulties related to the incomplete knowledge of the mechanisms of disease, the anatomical structure of the brain-blood barrier (BBB) represents a further intrinsic obstacle to obtain an efficient drug delivery.
Blood-Brain Barrier
BBB structure is not the focus of this review, hence some essential key points about this topic are given and the reader is suggested to eventually look through other papers that better present its structure, physiological, immunological and pathological characteristics and BBB overcoming strategies.9,12,13 In summary, BBB is composed by brain microvascular endothelial cells, pericytes, astrocytes, tight junctions, neurons, and basal membrane that create a continuous barrier whose functions are the protection of the brain and the strict control of the passage of solutes.14 Molecules that can overcome the BBB through passive diffusion should be liposoluble and generally not charged at physiological pH, should have a <500 Da molecular weight and a partition coefficient between 0.5 and 6.0.4 Selective carriers, instead, move nutrients and signal molecules from the blood to the brain and vice versa.9 BBB disruption is a typical sign of various pathological conditions. Despite some conflicting data, it is generally believed that AD patients show an increase in BBB permeability in the presence of a comorbidity with vascular dementia, that may be a consequence of alterations on adherens junctions.14 Moreover, BBB permeability is also altered by age-dependent changes affecting both its structure and its functions.14
Considering that the therapeutics delivery to the brain is limited by the need and the difficulties to overcome the BBB and given the modest efficacy of the now available AD drugs, nanotechnologies may provide a promising strategy in the field of neurodegenerative diseases due to nanoparticles low toxicity, high biocompatibility, good stability in blood, and capability to deliver functional cargo.15
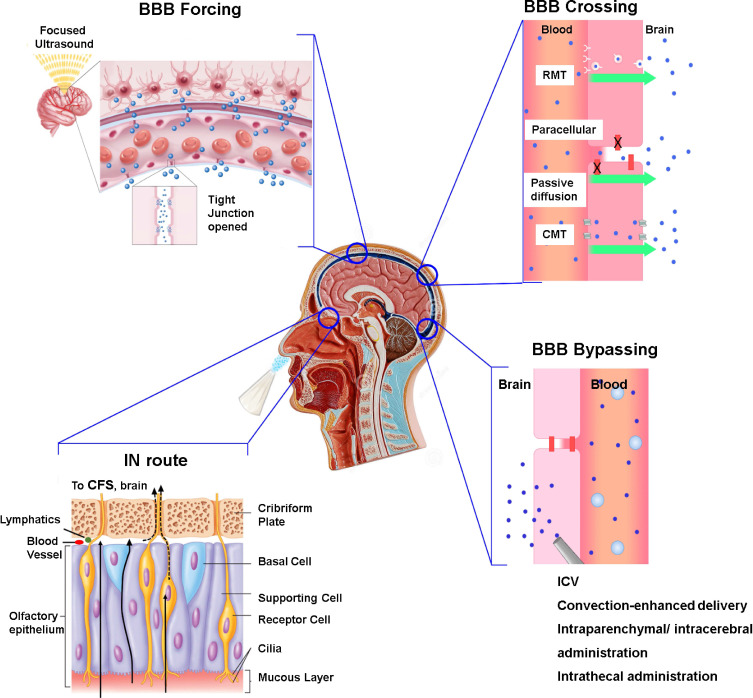
In general, there are two main pathways that could be exploited to overcome the BBB and thus to facilitate the drug delivery to the central nervous system (CNS), the first one is “crossing” it while the second one is “bypassing” it (Figure 1). Nanoparticles (NPs) may represent a useful tool to favor brain delivery; indeed, their external surface can be properly tuned through the functionalization with a specific ligand, designed to improve BBB passage. Moreover, the functionalizations should be multiple in order, not only to ensure BBB passage, but also to target molecular mechanisms of disease (Table 1).
Figure 1.
Summary of the strategies to overcome or bypass the BBB.
Notes: Adapted from Stanfield CL, Germann WJ. Fisiologia Terza Edizione. [Physiology Third Edition] with permission from EdiSES srl16 and adapted with permission from Focused Ultrasound Foundation.17
Abbreviations: CMT, carrier-mediated transport; RMT, receptor-mediated transcytosis; ICV, intracerebroventricular; IN route, Intranasal route; CSF, Cerebrospinal fluid.
Table 1.
Characteristics of the Most Promising Brain-Targeting Functionalized NPs Listed in the Text
| Specific Target | NPs | Characteristics | REF | ||||
|---|---|---|---|---|---|---|---|
| Mean Size (nm) | Polydispersity Index | Zeta Potential (mV) | Encapsulation Efficiency (%) | ||||
| Aβ plaques, tau fibrils | mf-LIPs | 155 ±30 | 0.213±0.055 | −3.31±1.51 | NA | 125 | |
| PEG-BTH-nanoliposomes | 102–109 | 170±0.005 | −3.13 ± 0.235 −2.27 ± 0.165) |
NA | 126 | ||
| NPs functionalized Aβ1-42 monoclonal antibody |
125 | 0.1–0.2 | −20 to −30 | NA | 46 | ||
| PEGylated nanosweepers composed of cationic CS | 200 | NA | NA | NA | 47 | ||
| TA–AuNP | 21.6 ± 2.4 | 0.235 | −30 and −40 | NA | 48 | ||
| Sulfur NPs | Met@TS | 120.21±4.36 | NA | −34.2±0.45 | NA | 49 | |
| RVG@Met@TS | 123.12±4.27 | NA | −35.2±0.36 | NA | |||
| Met@VS | 109.15±3.56 | NA | −33.7±0.42 | NA | |||
| RVG@Met@VS | 110.25±3.29 | NA | −36.8±0.29 | NA | |||
| Met@SS | 52.33±1.27 | NA | −36.2±0.39 | NA | |||
| RVG@Met@SS | 56.57±2.23 | NA | −37.6±0.21 | NA | |||
| OPCDs | 7.3 | NA | 17,4 | NA | 50 | ||
| IDA-NPs | 88.0 ± 5.9 | NA | −41.3 ± 7.7 | NA | 51 | ||
| D-Pen@Se NPs | 113.5 | 0.31 | − 8.36 | NA | 52 | ||
| PEG/CS-PZn NPs | NA | NA | 15 | 12.5 ± 1.2 | 53 | ||
| CMCDs | 1–5 | NA | NA | NA | 54 | ||
| MoO3−x nanodots | 31 ±0.7 | NA | −13.4 ± 1.6 | 58.5 Mo6+; 41.5 Mo5+ |
54 | ||
| β-NaYF4:Yb/Er | 38.3 ± 3.7 | NA | NA | 55 | |||
| KLVFF-PLGA NPs | 216 ± 34 | 0,25 ± 0.03 | −34 ± 3 | NA | 56 | ||
| D/L-FexCuySe NPs | 50 | NA | NA | NA | 57 | ||
| DBP-PLGA | 226.6 ± 44.4 | 0.039 ± 0.013 | −0.144 | 89.3 ± 3.3 | 59 | ||
| Nicotinamide-loaded SLNs | S80-SLN (Polysorbate80) | 112 ± 1.6 | NA | NA | 1 | 60 | |
| PS-SLN (Phosphatidylserine) | 124 ± 0.8 | NA | −46.1 ± 0.6 | 5 | |||
| PA-SLN (Phosphatidic acid) | 137 ± 1.05 | NA | −50.6 ± 0.8 | 10 | |||
| PC-CdS NPs | 50–60 | NA | NA | NA | 61 | ||
| HMPWCs | 10–30 | NA | −30 | NA | 62 | ||
| CS@Se | 89.1 ± 4.5 | 0.27 ± 0.05 | −41.7 ± 3.5 | NA | 63, 64 | ||
| Reducing intracellular calcium | Amlodipine functionalized nanodiamond particles | 31.1 ± 8.2 | NA | NA | NA | 69 | |
| Lomerizine modified nanospheres (LNP+PEI) | 210 | NA | NA | 7.1 ± 1.3 | 71 | ||
| Felodipine loaded NPs | 651 ±2.10 | 0.780 ± 0.074 | 14.1 ± 0.67 | 68.78 ± 0.680 | 72, 73 | ||
| Nimodipine encapsulated in Chitosan NPs | 5.2 ± 1.1 | NA | NA | NA | 74 | ||
| PPARs agonists | Polymeric PLGA-PEG NPs carrying PGZ | 155.0 ± 1.8 | 0.1 | −13.0 ± 0.5 | 92.5 | 76 | |
| Neuronal regeneration | Iron oxide NPs NGF-MNPs | 45 ± 17 | NA | NA | NA | 78 | |
| SPIO–AuNPs | 20.8 ± 2.7 | NA | −25.1 | NA | 79, 80 | ||
| 6MP-AuNPs-RDP | 24.6 | NA | −25.8 | NA | 81 | ||
Abbreviations: mf-LIPs, multifunctional liposomes with TREG-mAPOE and Mab; TA–AuNP, tannic acid–stabilized gold nanoparticle; OPCDs, polymerized o-phenylenediamine-derived carbon dots; IDA-NPs, iminodiacetic acid-conjugated NPs; D-Pen@Se NPs, D-penicillamine-capped selenium NPs; PEG/CS-PZn NPs, PEG-modified-CS-NPs loaded with a Zn(II); CMCDs, cow milk-derived carbon dots; β-NaYF4:Yb/Er, upconverted NPs co-doped it with rare earth elements, ytterbium and erbium; KLVFF-PLGA NPs, PLGA NPs functionalized with KLVFF; D/L-FexCuySe NPs, Chiral Iron Copper Selenide NPs; DBP-PLGA, PLGA NPs functionalized with a vitamin D-binding protein; PC-CdS NPs, protein-capped metal NPs; HMPWCs, manganese Prussian white NPs; CS@Se, chondroitin sulphate nano-selenium; 6MP-AuNPs-RDP, AuNPs loaded 6-mercaptopurine functionalized peptide RDP; NA, not available.
Main Families of NPs
For a matter of clarity, before exploring how BBB passage could benefit from nanotechnologies, the principal families of NPs that are reported in the manuscript are here introduced. The following is just a brief description, including very few and basic details, as each type of NPs can be seen as a “building block” in which theoretically infinite combinations of surface modifications could be properly designed.
Polymer-Based Nanoparticles
The most attractive features of polymeric NPs are biodegradability, biocompatibility, long shelf life, and stability during storage; moreover they ensure a controlled and sustained release of the load. Their dimensions are in the range of 10–1000 nm. Natural polymers are usually polysaccharides (chitosan CS is the most used), while synthetic ones are poly (lactic acid) (PLA), poly (glycolic acid) (PGA), and poly (D,L-Lactic-co-Glycolic acid) (PLGA). Depending on the method of preparation, the drug can be either seat within the NP, that functions as a reservoir (nanocapsules), or embedded in the matrix (nanospheres).18
Lipid-Based NPs
The use of liposomes (size range 100–200 nm), the hallmark of lipid-based NPs, benefits of a high drug-to-lipid ratio, an excellent retention of the encapsulated drug, and a long circulation lifetime (>6 h). Their lipidic bilayer is primarily composed of amphipathic phospholipids (phosphatidylcholines) enclosing an interior aqueous space. Phosphatidylethanolamine (PE) is included in the formulation when the goal is the fusion of the liposome with cell membranes because its spontaneous negative curvature favor this event.
Solid Lipid Nanoparticles (SLNs, size range 50–1000 nm) are based on lipid components other than phospholipids (triglycerides, glyceride mixtures), and were born as an alternative approach to liposomes. In fact, they have a hydrophobic core that provides a suitable environment for a better entrapment and efficient load of hydrophobic drugs, moreover, a finely controlled drug release, and an improved stability. Their synthesis is cheap and can be easily scaled up.19
Inorganic NPs
Inorganic NPs in the biomedical field are non-toxic, hydrophilic, biocompatible, and highly stable under physiological conditions. Their physical properties are tailored by controlling size, shape, surface, and domain interactions. They can exhibit intrinsic surface plasmon resonance and electrical properties of metal NPs (gold NPs, silver NPs, size range 2–100 nm), magnetic properties (iron oxide NPs), or optical properties (rare-earth-doped NPs). Thanks to their large surface-to-volume ratio, a suitable decoration of their surface might be exploited to positively regulate in vivo behavior and targeting. Inorganic ceramic NPs, such as calcium phosphates (eg, hydroxyapatite), silica, and titania, offer higher thermal and chemical stability than polymeric ones. Thus, encapsulation of drugs within ceramic particles would give a better protection of labile agents against denaturation. This large and heterogeneous family of NPs has been studied in preclinical development as potential diagnostic and therapeutic systems both separately or in a combined approach, referred as “theragnostics”. In this regard, iron oxide NPs, and in particular super paramagnetic iron oxide NPs (SPIONs, size 60–150 nm), represent one of the most promising theranostic nanocarriers: the inorganic core of the magnetic NP acts as a contrast agent detectable through magnetic resonance imaging (MRI) and surface modifications allow a targeted drug delivery (hyperthermia).20
Carbon Nanotubes
Carbon nanotubes (CNTs) are nanostructures with a length in the range of micrometers. They consist of one or several concentric graphite-like layers and are classified according to the number of carbon layers as single-walled carbon nanotubes (SWCNTs, single graphene layer with diameter varying between 0.4 and 2 nm) or multi-walled carbon nanotubes (MWCNTs, comprised of two or several cylinders with a diameter up to 100 nm).
For their exceptional properties, such as ultra-light weight, special electronic structures, and high chemical and thermal stability, they have been involved in several applications, from clinical diagnostics to therapy. In fact, MWCNTs, thanks to their large surface area and hydrophobic nature prolonging their halflives, have been used for targeting and controlling the delivery of drugs by several stimuli (magnetic, electric, or temperature changes).21
Microbubbles
Microbubbles were initially used as diagnostic agents but are now emerging as a promising alternative therapeutic tool because ultrasonication provides a non-invasive technique with unique ability to penetrate biological tissues. Bio-medically microbubbles have erythrocytes-like dimensions and are comprised of three phases: the innermost part is filled with gases, the middle layer or “shell” can contain proteins, surfactant lipids, or biodegradable polymers, and the outermost layer is covered up with the liquid phase. Microbubbles carry drugs or genes to any specific tissues; when ultrasounds are administered, they resonate to expand, shrink and finally burst, causing site-specific delivery of bioactive materials through the opening of the BBB tight junctions.22
Janus Particles
Janus particles are named after the two-faced Roman God Janus. They represent a unique class of particles with two distinct anisotropic sides in terms of composition and surface features, which allow customization of physicochemical properties down to the nanoscale. Indeed, in one single particle, complementary physical, chemical and magnetic properties are combined to ensure diverse and synergic functionalization. Their dimensions fall into the submicrometer scale, from a few hundred nanometers to a few micrometers with scarce examples smaller than 100 nm, while shapes might be spherical, ellipsoidal, cylindrical, or disk-like. The two distinguished compartments on the two sides, usually made of materials with incompatible surfaces or chemistries such as hydrophilic versus hydrophobic (polymer–polymer, polymer–metal, metal–metal, lipid–lipid, and lipid–polymer) confer to the Janus particles asymmetric structure and properties that make possible the incorporation of multiple functionalities in a spatially controlled manner. Biomedical applications range from dual-release drug delivery (one compartment host a hydrophilic drug whilst the second a hydrophobic one), to synergistic actions of combined therapy (ie, chemo-photothermal), theranostics, multimodal imaging and biosensing.23
Strategies to Overcome the BBB
Nanotechnologies to Cross the BBB
“BBB crossing” refers to the many ways by which molecules can endogenously pass through the BBB, ie, paracellular transport, passive transcellular diffusion, carrier-mediated transport (CMT), receptor-mediated transcytosis (RMT), adsorptive-mediated transcytosis (AMT), and cell-mediated transport (Figure 1).9
Actually, the most common strategies used to ameliorate CNS delivery of nanodevices are CMT and RMT.24 Since the number of specific carriers and receptors expressed on the plasma membrane is limited, CMT and RMT are both saturating mechanisms. In physiological conditions, they move different molecules across the BBB: carriers mediate the passage of glucose and amino acids, while receptors control the trafficking of proteins, hormones, growth factors, enzymes, and plasma proteins.13 Several NPs have been functionalized in order to target the receptors of the most common mediators such as transferrin (TfR), insulin and insulin-like growth factors, low-density lipoprotein, leptin, heparin-binding EGF and tumor necrosis factors, and glucose transporter.13,24 Among the others, lipidic NPs are definitively one of the main nanostructures whose therapeutic efficacy was improved by an increase in BBB passage due to functionalization with specific ligands able to bind carriers or receptors.25–27 It is worth to say that, in general, BBB crossing through CMT and RMT are nowadays well-known strategies and they have been extensively reviewed.25,28,29 As far as concerned CMT, a descriptive case was reported about liposomes modified with glucose transporter-1 targeting ligands on their surface used as nonviral gene delivery vectors with a plasmid encoding BDNF encapsulated inside. Internalization and a high yield of transfection were demonstrated in several neuronal cell lines, plus the ability to cross the BBB more efficiently than control liposomes. Brain tissue targeting was evaluated in vivo in C57BL/6 mice: intravenously injected modified liposomal formulation was found in the amount of 4.6% in the post-euthanized brain tissues.30 A factual example of RMT is insulin-coated gold NPs (INS-GNPs; 20 nm diameter) whose BBB passage is mediated by the insulin receptors. About 5% of the intravenously injected GNPs were found in the brain 2 h post-administration. Brain ex vivo CT scan demonstrated that the highest amount of INS-GNPs per mg tissue resided within the hippocampus, the cortex, and the cerebellum.31
Unlike CMT and RMT, AMT is a non-selective pathway, indeed it depends on the electrostatic interactions between positively charged molecules and the negatively charged plasma membrane. AMT is another common strategy exploited to improve brain delivery, usually involving cationic proteins or cell penetrating peptides (CPPs).13,24 In particular, CPPs are short amphipatic or cationic peptides (10/27 amino acids) that can enter the tissues in a non-invasive manner.13,32 Although they could not provide brain selectivity, CPPs can be used to functionalize NPs, preventing brain efflux mechanisms and rapid degradation.33 Several CPPs have been extensively studied, among all the TAT and the Angiopep-2 peptides, but novel carriers are now emerging.32 Bi-functional fusion peptides, that consist of the fusion of two brain-targeting peptides aiming at increasing CNS targeting efficiency, were also designed. TPL-polymeric NPs were obtained by the combination of TGN, a BBB-penetrating peptide, and Tet1, a neuronal receptor-binding peptide (GT1b). TPL bipeptide improved the passage of NPs respect to mono-ligands both in an in vitro BBB model and in an in vivo AD mouse model. Furthermore, the functionalization of the TPL-NPs with the neuroprotective peptide NAP ameliorated cognitive impairment in the AD mouse model in terms of spatial learning and memory dysfunction.34
Nanotechnologies to Bypass the BBB
“BBB bypassing” collects all those routes of administration that avoid a direct passage of the BBB; indeed, drugs reach CNS without physically interacting with the BBB. In particular, the therapeutic strategies aimed to bypass the BBB could be injected either through a surgical exposure to the brain via a pump or a catheter (intracerebroventricular (ICV), convection-enhanced delivery and intraparenchymal/intracerebral administration) or through a lumbar puncture (intrathecal administration) (Figure 1).9 However, although they can guarantee a quite large amount of drug delivered to the CNS, these techniques are quite invasive and carry a not negligible risk of neurotoxicity and/or CNS infections.35 For all these reasons, routes of administration that bypass BBB through sensory organs, as the intranasal (IN) one,9 are preferred (Figure 1).
The IN route is composed of three different tracks: the olfactory, the trigeminal and the systemic one, all of which are well described in recent reviews.36–38 Among its pros, IN is a non-invasive procedure, has a rapid kinetics of delivery (minutes), an ease of administration (by the patients themselves or by the caregivers, yielding an increased compliance), and a reduction in the risks of systemic side effects, as the systemic circulation is only partially involved. The main control is the low nasal drug absorption, particularly for hydrophilic drugs, peptides, and proteins. In this sense, nanotechnology has made a strong contribution since 2007,39 and, thereafter, polymeric or lipid-based NPs have been developed with the aim of ameliorating the nose-to-brain delivery of a variety of drugs.40 Both these classes of NPs present advantages and disadvantages38 and, although an updated revision of the recent literature has been done by Akel and colleagues.,37 still more comparative studies are needed.
Just to complete the scenario, two interesting formulations are here included. Hierarchical porous carbon encapsulated into PLLA and PLGA NPs revealed their efficiency in adsorbing drugs and in successfully delivering the cargo to the hippocampus after a single IN dose.41 In the field of lipid-based NPs, a thermogelling system containing liposomes loaded with an active pharmaceutical ingredient behaved as a solution at room temperature, facilitating the administration, then became a gel in the nasal cavity at 34°C. This property might represent an advantage; indeed, considering the IN route, attention should be given to the mucociliary clearance of the vestibular region, as it severely limits the time available for drug absorption and effectively rules out sustained nasal drug administration. In this case, liposomes had increased interactions with the mucin, that, in turn, increased nasal residence time, thus ameliorating the drug bioavailability in the olfactory mucosa, while maintaining a good drug release from liposome in nasal stimulated fluid.42
In order to raise the drug residence time in the nasal cavity, biological adhesives and mucoadhesive polymers, such as chitosan (CS), have been studied. Moreover, to increase drug absorption, especially through nasal epithelium, CPPs have been used as drugs or carriers surface modifiers.38,43 Both these goals have to be reached avoiding causing irritations or damages to the nasal mucosa.
Overall, all the studies suggest that the application of the nanotechnologies to the IN route represents a synergistic add-on. Considering that a specific region of the brain expresses specific receptors, the functionalization and tuning technology of the NPs may be extremely useful. Still, up to now, no IN nanosystem has reached the clinical development phase and more studies, especially on the pharmacokinetics of drugs delivered through the IN route, are required.
Nanotechnologies Exploiting New Tools to Force BBB Opening
Some technologies are aimed to force the BBB opening and, therefore, improve CNS drug delivery (Figure 1). This approach was recently used with microbubbles loaded with quercetin-modified sulfur NPs (Qc@SNPs-MB). In this study, the “sonoporation”, ie, the application of non-invasive ultrasounds, resulted in a transient opening of the BBB in an AD mouse model. Moreover, sonoporation induced microbubbles destabilization and release of Qc@SNPs, that accumulated in the brain parenchyma. Following the treatment, a general cognitive amelioration and a reduction in Aβ accumulation and in neuronal loss were observed in AD mice.44 Near infrared light (NIR) irradiation has also been used in several studies to improve NPs brain delivery as NIR application leads to a temporal disruption of the BBB, which enhances its opening due to a local heating effect.24 There are a few NPs having photothermal effects, as 2D black phosphorus (BP) nanosheets, flower-shaped hollow nano-ruthenium (Ru NPs), and Janus nanomotor (JNM). BP nanosheets, a promising neuroprotective agent due to their ability to capture the neurotoxic Cu2+, showed increased BBB permeability both in vitro and in vivo after NIR irradiation.45 The same was true also for NGF-PCM@Ru NPs that are nanocomposites loaded with nerve growth factor (NGF) and sealed with phase change material (PCM). When the temperature became higher than 40°C, PCM melted and induced the release of NGF, which inhibited tau hyperphosphorylation and aggregation, restored oxidative stress and nerve damage and improved learning and memory deficiency in an AD mouse model. Thus, in this case, NIR application not only improved BBB passage but also ensured a controlled drug release.46 JNM conjugated with an Aβ aggregation inhibitor (JNM-I), whose motion is constitutively propelled by NIR, showed an enhanced ability to cross the BBB after the laser application. Although it has not yet been tested in vivo, this method might be promising since NIR application should, on one side, improve CNS targeting, on the other side, increase JNM-I motion, promoting the contact between the Aβ inhibitor and the Aβ fibrils.47 Although these strategies seem to be quite hopeful, it is not clear whether BBB opening may be translational, moreover whether repetitive and prolonged BBB openings may induce neurotoxicity.35
Specific Targets After BBB Overcoming
Aβ Plaques, Tau Fibrils Reducing and Anti-Inflammatory Agents
Regardless of how the nanoparticles managed to overcome the barrier, one of the goals that is principally pursued is the reduction of Aβ plaques. Anti-Aβ1-42-functionalized polymer NPs removed Aβ peptides from the brain with a concomitant correction of memory deficits in AD-like transgenic mice.48 Self-destructive PEGylated nanosweepers composed of cationic CS captured Aβ and promoted their degradation through cell-mediated autophagy.49 Metal–phenolic networks-coated AuNPs inhibited Aβ fibrillation,50 sulfur NPs,51 and polymerized o-phenylenediamine-derived carbon dots52 decreased Cu2+-mediated Aβ aggregation, while iminodiacetic acid-conjugated,53 d-penicillamine-capped selenium,54 and PEG-modified-CS-NPs loaded with a Zn(II)-binding peptide55 decreased Zn2+-mediated Aβ aggregation. MoO3−x nanodots acted on the same target through the mimic of catalase and SOD activities,56 and upconversion NPs disrupted amyloid plaques after NIR irradiation.57 PLGA NPs functionalized with KLVFF, a pentapeptide specifically designed to target a central region of Aβ, inhibited Aβ elongation and fibrils formation.58 Chiral Iron Copper Selenide NPs (D-FexCuySe NPs) under NIR radiation reduced Aβ aggregation and favor fibrils disaggregation in a mouse dopaminergic cell line. Furthermore, these data were confirmed in vivo; indeed, D-FexCuySe NPs removed amyloid plaques with a concomitant significant recovery of the cognitive abilities of AD mice.59 The application of some NPs led to the observation of other potential benefits, probably related to the anti-Aβ activities. Indeed, the chronic treatment of AD mice with redox NPs reduced Aβ plaques, increased antioxidant enzymatic activity, and ameliorated cognitive deficits.60 PLGA NPs functionalized with a vitamin D-binding protein showed not only in vitro inhibition of Aβ aggregation, but also reduction of neuroinflammation, neuronal loss, and cognitive dysfunction in AD mice.61
However, the disruption of Aβ aggregates is sometimes mediated by ROS generation,57,59 therefore, for these nanodevices, the eventual worsening of AD neuronal damages and neuroinflammation due to an increased oxidative stress seems mandatory to be tested.
As far as concerned tau protein, the inhibition of its fibrillization is the main target of nicotinamide-loaded solid lipid nanoparticles (SLNs),62 and protein-capped metal NPs,63 while a reduction of tau hyperphosphorylation, oxidative stress damage and neuroinflammation were observed testing in vitro and in vivo self-assembly hollow manganese Prussian white nanocapsules, that are nanozymes, ie, nanomaterials with enzyme-like characteristics,64 and CS sulphate nano-selenium.65,66 All these NPs succeeded in their goal, however their actions in the control of other clinical hallmarks could not be excluded and should be further evaluated.
Reducing Intracellular Calcium
The increase in calcium accumulation in neurons correlates with aging to the point that it can trigger mechanisms of degenerative processes supporting the “calcium hypothesis” for AD excitotoxic neurodegeneration. Intracellular calcium accumulation can also be a downstream contingency of Aβ accumulation. Thus, excitotoxicity may be attenuated by reducing calcium load from the extracellular environment by blocking calcium influx in both synaptic terminals as well as postsynaptic sites. These conditions may be mediated by L-type and N-type calcium channels and NMDA-receptors. Moreover, relaxation of the smooth muscle in cerebral vasculature, obtained through calcium blockade, may improve cerebrovascular perfusion, usually reduced in AD, and counteract hypertension, detrimental on AD development.67,68
Among the calcium channel blockers (CCBs) studies, nitrendipine seems to be the most promising: it reduced the incidence of dementia by 55% in the SYST-EUR trial, penetrated the BBB and blocked the uncontrolled influx of calcium into the neurons; finally, it decreased the accumulation of Aβ in the brain of a murine AD model, a property shared with another CCB, nilvadipine.69,70 The larger part of CCBs, though, are hydrophobic molecules unable to pass the BBB. Below are some of them that have already been successfully loaded in NPs as anti-hypertensive therapy. Amlodipine into nanodiamond particles is ready to be tested in vitro,71 liposomes enhanced lacidipine bioavailability,72 lomerizine in nanoparticle-based carriers suppressed Ca2+ influx in PC12 cells in response to glutamate.73 In vitro testinnig revealed that PLGA NPs showed longer duration of felodipine release with reduced burst release compared with pure drug paralleled by a complete biocompatibility in in vivo toxicity study in albino mice.74,75 Nimodipine, another CCB known for its selectivity for cerebral vascular district, in spite of its lipophilicity, when encapsulated in CS NPs benefits of a better delivery.76
These are just examples of how, besides working on new CCBs molecules, a novel therapeutic strategy may involve the encapsulation of already known CCBs in appropriately functionalized NPs to improve the bioavailability and to limit the pleiotropic effects that especially dihydropyridines manifest.
PPARs Agonists
Interestingly, all the pathways related to the initiation of AD are directly or indirectly regulated by peroxisome proliferator-activated receptors (PPARs), a family of type II nuclear receptors ubiquitously expressed, with tissue-, cell type-, and developmental stage-specific patterns. Their transcriptional activity controls a unique subset of genes, responsible for lipid and energy metabolism. In addition, a nongenomic action of PPARs interferes with several cellular pathways.77 Some studies have been focused on the relation between AD and γ isotype and have demonstrated that the administration of PPARγ agonists, pioglitazone (PGZ) among all, could be a promising strategy. Polymeric PLGA-PEG NPs carrying PGZ showed encouraging results both in vitro and in vivo; indeed, no cytotoxicity and no alterations of the permeability of a BBB model were observed. Furthermore, their chronical administration in a murine AD mouse model induced a reduction of Aβ deposition in the cortical cortex and ameliorated memory deficits.78
PPARα is also now emerging in AD pathogenesis. Its expression is significantly decreased in AD brain and its neuronal form was demonstrated to be involved in the regulation of Aβ production and clearance, in cellular metabolism, in oxidative stress, and in neuroinflammation. Therefore, a few clinical trials with PPARα agonists, including natural and synthetic compounds, have been recently reported.
Overall, the efficacy of PPARs agonists in the treatment of neurodegenerative diseases is controversial. Some data suggest beneficial effects, other highlight ineffectiveness or side effects, mainly related to the cardiovascular system, the metabolic rate, and the body weight. Hence, other studies are needed in the view of a pharmacological personalized medicine.77
Neuronal Regeneration
To counteract the AD progressive neuronal degeneration, one potential therapeutic approach might consist of the administration of molecular factors inducing neuronal regeneration.79 There are many neurotrophic factors that could positively promote neuronal proliferation and neurite growth and whose properties have been tested in nanoformulations. Nerve growth factor (NGF) is probably the most studied and it was tested in vitro in several nanoformulations. Iron oxide NPs NGF-MNPs, whose selective deliver was controlled by magnetic fields,80 and superparamagnetic iron oxide (SPIO)–AuNPs, activated by a low-intensity light-emitting diode source,81,82 promoted neurite outgrowth and neuronal differentiation and enhanced the complexity of the neuronal branching network in PC12 cells. Moreover, the addition of the flavonoid quercetin in the functionalization of NGF-SPIO-NPs turned out to accelerate this last process.83 AuNPs loaded with the anti-inflammatory drug 6-mercaptopurine and functionalized with neuron-targeting peptide RDP to increase the cellular uptake were tested in an SH-SY5Y in vitro model and showed increased cell proliferation and neurite growth.79
Innovative Rising Therapies
As we already described, FDA-approved AD drugs have several limits; indeed, they can only partially and temporarily restore the cognitive decline of the patients, furthermore some side effects could arise. Consequently, a great interest turns around the development of novel therapeutic approaches, starting from the in vitro and the in vivo preclinical evaluation of new molecules. In the next section, new pharmacological agents that are emerging in the field of nanomedicine will be described.
Nutraceutical Loaded NPs and Green NPs
The term “nutraceutical” derives from the combination of “nutrition” and “pharmaceutical”,84 thus it is used to identify natural compounds present in the food that improve mental and physical health. In the last decade, an exponential increased interest in nutraceuticals has been observed because of the several advantages that could be linked to their application due to their beneficial antioxidant and anti-inflammatory properties and to their possibility to regulate intra- and extracellular signalling pathways. The rational in the use of nutraceuticals in AD therapy is coherently related to the high level of inflammation and oxidative stress observed as clinical hallmarks.
Nanoformulations are particularly interesting in this field because they can improve some chemical and biological limits of nutraceuticals (poor solubility, stability, and bioavailability), and pharmacokinetics feature in order to favor brain targeting. Today (April, 14th 2020) in PUBMED database, a search through the keywords “nutraceutical” and “Alzheimer” retrieves more than 200 papers in a limited selection to the last five years, but only 1 (a review) if the keyword “nanomedicine” or “nanoparticle” was added.85 This indicates that a comprehensive revision of the literature in this field is lacking and that dedicated reviews may be needed. Here, only the more recent advances in this field are indicated (Table 2).
Table 2.
Mean Characteristics and Principal in vitro and in vivo Results of Nutraceuticals Loaded Nanoformulations
| Nutraceutical | NP | Characteristics | RESULTS | REF | ||||
|---|---|---|---|---|---|---|---|---|
| Mean Size (nm) | Polydispersity Index | Zeta Potential (mV) | Encapsulation Efficiency (%) | In Vitro | In Vivo | |||
| Resveratrol | SLNs | 142 ± 10 | 0.12 ± 0.04 | −0.08 ± 0.05 | 75–100% | In BBB models (human endothelial cells/pericytes and hCMEC/D3 cells): improved BBB passage | 74 | |
| Nano capsules |
249 ± 5 | 0.12 ± 0.05 | −14 ± 1.8 | 99.89 ± 1.3 | In Aβ-injected AD rat model: impaired learning and memory abilities, increased astrocyte and microglia recruitment | |||
| Quercetin | PLGA | 100–150 | NA | NA | NA | In SH-SY5Y cells: reduced Zn2+-Aβ42-induced cytotoxicity | In APP/PS1 mouse model: ameliorated spatial learning and memory impairments without signs of adverse effects | 78 |
| SLNs | 234±18 | 0.06±0.01 | −32±8 | 77±11 | In hCMEC/D3 cells (BBB model): high BBB permeability | 75 | ||
| SNPs-MB | NA | NA | NA | NA |
In a bEnd.3 cells/SH-SY5Y BBB model: improved NPs entry In SH-SHY5Y cells: reduced ER stress levels and ER stress-mediated hallmarks (inflammatory response, calcium homeostasis imbalance, and oxidative stress) |
In APP/PS1 mouse model: reduced Aβ deposition and neuronal loss, improved learning and memory behavior | 34 | |
| Anthocyanins | PLGA | 165 | 0.4 | −12 | 60 | In SH-SY5Y cells: increased cell viability against Aβ42, abrogated ROS generation, attenuated AD and neuroapoptotic markers | 77 | |
| AuNPs | 135 ± 5 | 0.5 | −11.5 ± 2 | 34 | In BV2 cells: prevented tau hyperphosphorylation, reduced protein expression levels of neuroinflammatory and neuroapoptotic markers |
In Aβ-injected AD mouse model: prevented tau hyperphosphorylation, reduced microglia and astrocytes activation, reduced neuroinflammatory and neuroapoptotic markers, attenuated neurodegeneration In Aβ-injected AD mouse model: prevented tau hyperphosphorylation, reduced protein expression levels of apoptosis and neurodegeneration markers, mitigated synaptic dysfunctions and ameliorated memory impairments |
81 79 |
|
| Rutin | Lipid polymer hybrid NPs | 232.4 ± 4.01 | 0.339 ± 0.010 | −1.76 ± 0.33 | 68.06 ± 1.50 | In erythrocytes separated from rat blood: hemolysis test confirmed biocompatibility | In white male albino rats: biodistribution study confirmed brain accumulation | 78 |
| HST | Nanocrystals | NA | NA | NA | NA | In unilateral icv-STZ rat AD model: improved memory functions, reduced levels of hippocampal oxidative stress | 82 | |
| Curcumin | Nanogels | 201 ± 19 | NA | − 25.2 ± 2.0 | NA | In SH-SY5Y cells: protected cells from Aβ-induced cytotoxicity | 74 | |
| Polymeric NPs | 197 ± 2.3 | 0.18 | −22.44 ± 2.3 | 77.99 ± 0.91 |
In SH-SY5Y cells: protected cells from Aβ-induced cytotoxicity In SK-N-SH cells: protected cells from oxidative stress damages |
|||
| Liposomes | 168.9±1.3 | 0.164 | 20.3±1.4 | NA | In APP/PS1 mouse model: acted on Aβ aggregates | |||
| Berberine | Multi-walled carbon nanotubes | 125–295 | 0,3–0,25 | NA | NA | In SH-SY5Y cells: efficient cellular uptake of the NPs | In Aβ-injected AD rat model: recovered memory performance, reduced Aβ aggregates and oxidative stress damages | 93 |
| α-bisabolol | SLNs | 139.5 | 0.244 | −31.7 | 98.44 | In Neuro-2A cells: reduced Aβ-induced free radicals levels and oxidative stress damages, showed neuroprotection from Aβ-induced apoptosis | 94 | |
| Apocynin | Poly anhydride NPs | 385±1,17 | 0.029 | −12.5 ± 4.7 | 42.7 ± 1.8 |
In N27 cells: no signs of cytotoxicity, NPs internalization In primary cortical neurons: no signs of cytotoxicity, NPs internalization, protection against oxidative stress damages In LUHMES cells: protection against oxidative stress damages |
95 | |
| Ginsenoside Rg3 | PLGA | 115 | 0.081 | NA | 65–70 |
In C6 cells: cellular uptake In THP-1 cells: reduced Aβ-induced amyloid plaques formation, oxidative stress damages and pro-inflammatory cytokines levels, reduced expression of gene encoding the β-amyloid A4 precursor In BMVECs/C6 cells BBB model: BBB crossing |
96 | |
| Phytol | PLGA | 177.4 ± 5.9 | 0.2 ± 0.06 | −32.8 ± 2.2 | 92.14 ± 0.29 | In Neuro-2A cells: protected cells from Aβ-induced cytotoxicity | 97 | |
| EGCG | PLGA | 124.8 ± 5.2 | 0.054 ± 0.013 | −15.1 ± 1.7 | 97.1 ± 2.4 |
In primary brain microvascular endothelial cells (BBB model): alterations of the BBB integrity through tight junctions disruption |
In APP/PS1 mouse model: increased synapses, reduced amyloid plaques and neuroinflammation, ameliorated spatial learning and memory abilities |
98 |
| RIN | PLGA | 145.2 | 0133 | NA | 60 |
In erythrocytes separated from rabbit blood: hemolysis test confirmed biocompatibility In bEnd.3 cells (BBB model): high intracellular uptake and passage In PC12 cells: reduced apoptosis due to Aβ-induced cytotoxicity |
In C57BL/6 mice: biodistribution study confirmed brain accumulation | 99 |
Abbreviations: SLNs, solid lipid nanoparticles; PLGA, poly(lactide-co-glycolide); AuPdNPs, gold/palladium nanoparticles; SNPs-MB, sulfur nanoparticles in microbubbles; HST, hesperetin; Icv, intracerebroventricular; STZ, streptozotocin; EGCG, epigallocatechin-3-gallate; RIN, rhynchophylline .
Flavonoids are compounds extracted from plants and classified in several classes upon their chemical structure. All of them, though, share the same health-promoting effects. One of the most popular flavonoids is resveratrol, extracted from grape skin and seed. Its role in AD therapy is related to its capability of attenuating neuronal injuries and lessening the progression of neurodegeneration. In fact, it regulates signalling pathways, autophagy, it is a free-radicals scavenger, an AchEI and its neuroprotective role in the brain is also linked to its potential to interact with neuronal and glial pathways. In order to counteract its poor solubility and its rapid metabolism, it has been encapsulated or conjugated in SLNs and in nanocapsules. Data showed how resveratrol could reach the brain and inhibit the formation and aggregation of Aβ peptides.8,85 Another very interesting flavonoid is quercetin. Its applications are best studied in targeting cancer. Recently, few studies have demonstrated that, when delivered through functionalized PLGA NPs or SLNs, quercetin played an important role as antioxidative agent in disassembling Aβ fibrils in SH-SY5Y cells and abrogated memory impairments and ameliorated cognition in AD mice.86,87 Quercetin had also been loaded in microbubbles to improve the BBB crossing, as we already described above.44 Several other NPs loaded with flavonoids were designed and tested, some of them in vitro, other in vivo, confirming their antioxidant, anti-inflammatory and neuroprotective properties. Anthocyanins encapsulated by PEG-PLGA NPs,88 and rutin-loaded lipid polymer hybrid NPs89 belong to the first group (in vitro tested flavonoids), while anthocyanin-loaded PEG-AuNPs90,91 and hesperetin nanocrystals92 belong to the second one as they were tested in a mouse and in a rat AD model, respectively.
Curcumin is a chemical product of turmeric, belonging to ginger family, well known for its antioxidant and anti-inflammatory properties. Therapeutic applications are limited by its hydrophobic nature. Kanubaddi and colleagues reported improvements in its solubility, stability, and ability to bind and dissolve Aβ aggregates when conjugated with AuNPs, nanogels, polymeric NPs, and nanoliposomes.85 More recently, natural micellar-forming polymers, such as casein, have attracted interest in the development of micelle delivery system thanks to their safety, biodegradability, and ability to not affect the immune system. Many casein-based pharmaceutical products are in clinical evaluation. The novelty in this recent approach is to transform micellar curcumin into a solid dispersion powder, in order to enhance curcumin solubility and bioavailability upon oral administration. The study revealed that a micellar curcumin-spray dried powder (MC-SDP) with self-assembled casein and the addition of sucrose as a protectant showed a higher percentage of dissolved curcumin compared to control MC-SPD without casein and sucrose. This suggests a possible oral delivery of ease-handling MC-SDP.93 A novel curcumin nanodelivery system with high encapsulation efficiency, loading capacity and better stability, are zein-hyaluronic acid (HA) NPs (ZH-NPs).94
Other nutraceuticals carried by NPs were tested and gave successful preliminary results, among them berberine decorating multi-walled carbon nanotubes,95 α-bisabolol SLNs,96 apocynin polyanhydride NPs,97 ginsenoside Rg3-,98 phytol-,99 epigallocatechin-3-gallate-,100 and rhynchophylline-loaded101 PLGA NPs.
Recently, it has been shown that the combined use of various nutraceuticals could improve their preventive or therapeutic effect. Curcumin and the flavonoid quercetagetin can be co-delivered through layer-by-layer composite ZH-NPs.102 Another cocktail that should be improved by the help of NPs and seems worth to be tested in nanoformulations is ferulic acid with curcumin and phosphatidylserine. These three compounds together showed a synergistic therapeutic effect ameliorating cognitive dysfunction in AD mice.103
In the last five years, nanotechnologies have been approaching to the “green synthesis” of NPs, in line with the principles of green, cost-effective, and eco-friendly chemistry. This green synthesis approach replaces the use of toxic chemicals, as reducing and stabilizing agents, with phytochemicals during NPs synthesis.104 Phytochemicals coating onto NPs surface guarantee their biocompatibility and bacteriostatic properties. Thanks to their antioxidant and anti-inflammatory properties, green synthetized nanoparticles may play an important role in many therapeutic applications. Very recent studies demonstrated that green zinc oxide NPs maintained their high antioxidant properties,104 procyanidins fractions from Leucosidea sericea were employed in the synthesis of stable and active AuNPs,105 quercetin was used in the synthesis of gold and silver NPs,106 and, finally, silver NPs synthesized by macerating in the powdered fresh aerial parts of two plants, Lampranthus coccineus and Malephora lutea, demonstrated, in vitro, an anti-AchE and an antioxidant activities comparable to rivastigmine, suggesting their potential application in AD.107
Nucleic Acid Applications in AD
Preliminary results suggest that gene therapy combined with NPs to improve brain delivery might have a great potential in AD treatment.108 Various nucleic acid molecules are in development. siRNA against BACE1, an enzyme involved in the amyloidogenic processing, is one of the most studied; beside the lipid based, the most promising nanoformulation is nano-carriers based on PEGylated poly(2-(N,N-dimethylamino) ethyl methacrylate) functionalized with CPPs to improve BBB crossing and neuron-specific-targeting.109,110 These NPs reduced in vivo Aβ plaques, tau hyperphosphorylation and promoted hippocampal neurogenesis and cognitive performance.110 Tests in vitro and in vivo demonstrated, instead, that liposomes, double functionalized with TfR ligands and a CPP to favor BBB passing improved the therapeutic efficiency of the neuroprotective ApoE2 plasmids.108
A very promising and extremely specific approach is represented by antisense oligonucleotides (ASOs), which are small RNA sequences inducing protein repression. An overexpression of the histone deacetylase 2 (HDAC2) protein was observed in AD patients, therefore HDAC2-directed ASOs were designed.111 However, ASOs seem unable to cross the BBB, thus their conjugation with NPs might be examined.
Finally, an innovative way to employ nucleic acids in AD therapy was described by Shao et al,112 tetrahedral DNA nanostructures (TDNs) composed by 3D framework nucleic acid molecules could reduce apoptotic pathways in PC12 cells. When administered in an AD rat model, they also reduced Aβ40 deposition and apoptosis in the hippocampus, improved learning, memory and caused a partial restoration of the morphological anomalies.
Stem Cell Therapies and Personalized Medicine in AD
In the view of a personalized medicine for AD patients, an innovative therapeutic approach is definitively represented by “stem cell therapy”. Stem cells have self-renewal ability and high differentiation potential towards mature cells in the correct microenvironment. Thus, they represent a valuable research tool for basic, pre-clinical, and clinical studies. Four types of stem cells have been classified and here below a summary of the recent findings for each of them is provided.
Embryonic stem cells (ESCs) are self-renewing, totipotent stem cells. When transplanted in an AD model, they showed therapeutic efficacy with an improvement in memory impairment.113 Several studies, though, pointed out their association with high risks of transplantation rejection and immune responses and to the possibility of tumorigenesis and teratoma formation.114 Moreover, they bring along ethical issues, as they are extracted from the inner cell mass of blastocysts. For these reasons, other approaches are preferred.
Neural stem cells (NSCs) are relatively more accessible than ESCs as can be isolated from primary tissues, from post-mortem adult brains, and NSC can be derived from induced pluripotent stem cells (iPSCs). They improved the delivery of neurotrophic factors, learning, memory, and behaviors in vivo AD mouse model where, due to the need of been transplanted into the hippocampus, they can generate non-neuronal glial cells. This limitation could be overcome whether growth factors are co-transplanted.113,114
Mesenchymal stem cells (MSCs) are a type of multipotent somatic stem cells, easily accessible and highly used in stem cell studies. Their main advantage is providing a reduced risk of an immunological attack because they can be derived from the recipients themselves. Their enormous potential is related to their paracrine effects by secretion of neurotrophic factors, chemokines, anti-inflammatory cytokines, immunoregulatory function, stimulating in situ neurogenesis, synaptogenesis, angiogenesis, neuroprotection effects, cell replacement, and decreasing oxidative stress and apoptosis. In vitro, human MSCs are able to dramatically increase hippocampal neurogenesis and to trigger the differentiation of NPCs into mature neurons.113 In vivo MSCs transplantation has shown to decrease the formation of Aβ deposits and plaques and tau-related cell death. Like NSCs, MSCs secretions, due to their paracrine effects, stimulate neurogenesis, synaptogenesis, and neuronal differentiation and have neuroprotective functions.114 This strong evidence about their therapeutic efficacy may be a consequence of MSCs-immunoregulation because of the modulation of microglia/astrocytes activity state and of the mediation of neuroinflammation via several transcription factors signaling pathways.115 In terms of their use for human therapy, MSCs intravenously infusion seems not to be the best route of administration because, despite the non-invasivity, very few stem cells reach the brain, indicating the need for a more effective approach. The magnetic targeting cell delivery may represent an alternative: cells should be labeled with magnetic agents such as superparamagnetic iron oxide nanoparticles (SPIONs) and the application of an external magnet should deliver them in the region of interest. In a recent paper, cells derived from Wharton’s jelly (WJ) of human umbilical cord, a well-known source of MSCs, have been labelled through a biomimicry method in order to avoid the use of potentially cytotoxic transfection agents.116 The biomimicry method considers the SPIONs labeling of WJ-derived MSCs after a co-incubation with non-adherent cells derived from the same umbilical cord. This passage yields to an acceptable amount of intracellular iron content, comparable to the one obtained using a more typical transfection agent. It is supposed that the microenvironment created by the biomimicry method resembles the WJ-MSCs niche and acts as a natural transfection agent. In this microenvironment, MSCs increase their phagocytosis by signaling interactions and retaining their survival and reproduction capabilities. After intravenously infusion, SPIONs-labeled WJ-MSCs were found in the hippocampus areas. BBB passage and MSCs homing are somehow expected not only because of a partial leakage of the BBB but also because of the signaling connections between the lesion area and the injected cells through the secretion of paracrine modulators and the expression of chemokine receptors and adhesion molecules. Moreover, the magnetic targeted cell delivery (MTCD) technique was more effective in the retention of SPIONs-labeled WJ-MSCs toward the hippocampal region of AD rats. Other analyses showed improvements in AD rats memory and learning modulating the cholinergic system by increasing the expression level of AChE and ChAT proteins and decreasing neuronal loss of the hippocampal area. Moreover, there were no significant differences between direct ICV-cell-injection and IV-cell-injection followed by magnetic targeting. Thus, MTCD technique confirms to be an effective and less invasive tool in AD regenerative medicine.
iPSCs, as the name suggest, are cells that maintain their capability of generate endoderm, mesoderm, and ectoderm germ layers. They share the most identical properties compared to ESCs, without ethical issues, as they can be generated from several somatic sources like fibroblasts, peripheral blood mononuclear cells and, more recently, from urine-derived epithelial cells with less invasive procedures.117 Different strategies for reprogramming have been developed that allow to originate any kind of cells with an unlimited supply.118 On the one hand, the establishment of a neuron derived human-iPSCs model is useful for in vitro testing of small molecular chemical compounds that could be preclinically screened through low-and high-throughput approaches; on the other, it has facilitated the generation of human models of neurodegenerative disorders that can overcome the species gap involved in the use of animal to mimic human diseases. Indeed, in the absence of biologically-relevant human disease models, animal ones represent the only available approach to recapitulate a physiological and anatomical condition in vivo. However, the coming of neurons derived from human-iPSCs will allow to explore the pathophysiology of neurodegenerative diseases at cellular level and to have a platform for the development of disease-modifying agents for AD in a future towards personalized therapy for the individual patient. As a proof of concept, iPSC-derived cholinergic neuronal precursors transplanted in the hippocampus of transgenic AD mice successfully differentiated into mature cholinergic neurons with a concomitant reversal of spatial memory impairment.114 As far as concern humans, patient-specific neural cells have been derived more frequently from fAD and more rarely on sAD cases.118 The first is expected to recapitulate disease-specific phenotypes and to elucidate the molecular mechanisms that drive neurodegenerative diseases. In fact, neurons differentiated from patient-specific iPSCs harboring genetic variants in PS1 and PS2 showed impaired neurite outgrowth and increased Aβ42 secretion, responding to β- and γ-secretase inhibitors that reduced the Aβ42 consequential tau phosphorylation too.119,120 Also, the few studies conducted on iPSCs from sAD patients presented increased Aβ levels with an altered Aβ42/40 ratio and an increased APP expression,121 but the results were somehow inhomogeneous, reflecting the complexity of the pathogenesis of sAD.120
Despite iPSCs are the newest form of stem cell therapies, neurons derived from iPSCs are not devoid of limitations. Indeed, their high proliferative capacity may cause the risk of tumor formation. To overcome this problem, a recent work has demonstrated that adult cells could be reprogrammed into mature neurons without taking them to their precursor stem cell phenotypic form.114
iPSCs-derived neurons differentiation and maintenance require long-term culture, proving costly and time-consuming. Moreover, differentiation efficiency can vary among clones and their heterogeneity complicate data reproducibility and analyses based on high-throughput screening. Therefore, more work is required to reach a good level of homogeneity and differentiation efficiency.
As the 2D in vitro culture may not be sufficient to reproduce the complexity of the nervous system, a novel approach shows the possibilities to model a neurodegenerative disease transfecting episomal vectors on isogenic iPSCs differentiated in cerebral organoid, called human mini-brains, three-dimensional (3D) structures highly reminiscent of certain human brain regions.122 Thanks to a miniaturized spinning bioreactor, other groups generated forebrain-specific organoids from human iPSCs that recapitulate key features of human cortical development.120 These systems can be used to evaluate the development of a pathological state and eventually to test therapeutics. Because of its embryonic/fetal nature, some criticisms have been raised about the unsuitableness of this model to recapitulate events occurring in the late phases of the disease. However specific AD markers, such as an imbalance of Aβ secretion, tau hyperphosphorylation and protein aggregation leading to the formation of amyloid fibrils, have been recently described in organoids.122,123 Moreover, to circumvent the issues surrounding this comparison, researchers have used additional gene transfer strategies to express proteins of interest in a stable manner through lentiviral infection, or, more recently with episomal vector. The episomal vector is extrachromosomal, it allows the expression of transgene without the usual risk of random insertion encountered with lentiviral approaches.124
Theragnostics in AD
Theragnostics is a quite new strategy that combines diagnosis and therapeutic approaches. The winning card of this match is the potential to have a unique device that on one side recognizes and identifies a marker of the disease and on the other side reaches it and releases a specific drug. In AD, markers will be the Aβ plaques and the tau tangles; the theragnostic agent should allow both the molecular imaging recognition of the AD hallmarks (diagnosis) and the deliver of disaggregating or aggregation-inhibiting drugs (therapy).
Useful dyes with great potential in theragnostic applications, namely radiolabelled or fluorescent probes, polymeric NPs and SPIONs, developed for the imaging of amyloid aggregated, have been previously reviewed.4,125
Whether appropriately labelled, ASOs may be promising in theragnostics as they can regulate the expression of key proteins by selectively targeting their mRNAs. Studies demonstrated that radiolabelled ASOs directed against APP reduced its expression, improved cognitive functions and reversed learning and memory deficits in mice. Similarly, aptamers, short single-stranded RNA or DNA oligonucleotides with a unique three-dimensional structure, can bind target proteins with high affinity, as in the case of Aβ1-40 fibrils (thus exert their role as biosensors for molecular imaging) and efficiently inhibit their aggregation (therapeutic application).126
More recently, multifunctionalized liposomes with both amyloid and BBB affinity were described as potentially theragnostics agents. They displayed three ligands on their surface: mAPOE, Mab to target the LDL and the Tf receptors of the BBB (for a targeted delivery) and TREG, a curcumin-lipid ligand with affinity towards amyloid species (as therapy). In vitro characterization confirmed their interaction with Aβ peptides and their uptake by a cellular BBB model. Preliminary in vivo studies demonstrated their brain localization.127 The same authors described also pegylated benzothiazoles nanoliposomes with high affinity towards Aβ species. Despite they showed only in vitro studies, also in this case the results seem promising for a theragnostic approach.128
Overall, besides the first exciting theragnostics examples were mostly applied in oncology, emerging possibilities are continuously increasing and seem to be likely suitable also for neurodegenerative disorders.
Conclusions
In this review, a state-of-the-art of the latest developed NPs and of the newest promising strategies for AD therapy are presented. However, only preliminary in vitro results are now available for the majority of the most recent applications. Although much has been achieved so far, more can be further done only just taking advantage of the current knowledge acquired in specific sub-fields and integrating these different perspectives. A promising approach may come from NPs with a multi-drugs payload; indeed, a multiple payload combined within a single delivery platform could either avoid the possibility to escape the treatment by attacking with more than one drug or allow to reach different tasks in one shot. Other improvements are also expected whether the final shape of the NPs is reconsidered, since this physical parameter influences the ability to overcome biological barriers and might be exploited to increase the efficiency of the functionalization. The fact that a specific NP shape is better achieved by means of a top-down fabrication method is known since 2010,129 The importance of the shape of the ligand was recently demonstrated as discoidal HDL passed the BBB, enhanced the Aβ efflux and strongly reduced the Aβ fibrils concentration and extension in vitro to a much higher extent compared to other shapes and other ligands.130 Another poorly investigated aspect is the effect of the NPs per sé on the functional activity of the tissue or cells they encounter while moving towards their target. Only few data are, indeed, available about mechanical tension exerted on the membranes when NPs enter or pass through the cells and also about whether and how electrical activity of the targeted neurons is affected by the NPs.131
Finally, neurotoxicity that could arise after NPs administration is an aspect to consider in their design. The nanomaterial itself could be cytotoxic, protein corona formation could promote the BBB passage of neurotoxic (but also neuroprotective) factors, and some routes of administration could be more invasive or even induce a BBB breakdown, favoring brain entry of toxic substances or pathogens.35 Complement system (CS) activation should also be better evaluated, since hypersensitivity reactions and all the CS pathways (classical, alternative and lectine) were observed in some cases after NPs administration.4 Moreover, NPs might have direct effects on cellular metabolism, as an increase of ROS production, inflammation and oxidative stress,35 and/or a modification of gene expression,26 potentially promoting autophagy or apoptotic/necrotic pathways.4
In conclusion, as previously suggested,132 AD is a disorder with multifactorial causes, multifaceted nature of symptoms and numerous consequences on patient’s cognitive impairment. To answer to the complexity surrounding this heterogeneous disease, an approach that combines the perspectives from different fields seems to be the best now available option.
Acknowledgments
The authors acknowledge Samuele Barbuti for his help in preparing the figure included.
Author Contributions
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; gave final approval of the version to be published; and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Yiannopoulou KG, Papageorgiou SG. Current and future treatments in alzheimer disease: an update. J Cent Nerv Syst Dis. 2020;12:1179573520907397. doi: 10.1177/1179573520907397 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Faustino C, Rijo P, Reis CP. Nanotechnological strategies for nerve growth factor delivery: therapeutic implications in Alzheimer’s disease. Pharmacol Res. 2017;120:68–87. doi: 10.1016/j.phrs.2017.03.020 [DOI] [PubMed] [Google Scholar]
- 3.Tiwari S, Atluri V, Kaushik A, Yndart A, Nair M. Alzheimer’s disease: pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine. 2019;14:5541–5554. doi: 10.2147/IJN.S200490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ahmad J, Akhter S, Rizwanullah M, et al. Nanotechnology based theranostic approaches in Alzheimer’s Disease management: current status and future perspective. Curr Alzheimer Res. 2017;14(11):1164–1181. doi: 10.2174/1567205014666170508121031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Martín-Rapun R, De Matteis L, Ambrosone A, Garcia-Embid S, Gutierrez L, de la Fuente JM. Targeted nanoparticles for the Treatment of Alzheimer’s Disease. Curr Pharm Des. 2017;23(13):1927–1952. doi: 10.2174/1381612822666161226151011 [DOI] [PubMed] [Google Scholar]
- 6.Moreno S, Cerù MP. In search for novel strategies towards neuroprotection and neuroregeneration: is PPARα a promising therapeutic target? Neural Regen Res. 2015;10(9):1409–1412. doi: 10.4103/1673-5374.165313 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Karran E, De Strooper B. The amyloid cascade hypothesis: are we poised for success or failure? J Neurochem. 2016;139(Suppl 2):237–252. doi: 10.1111/jnc.13632 [DOI] [PubMed] [Google Scholar]
- 8.Uddin MS, Kabir MT, Niaz K, et al. Molecular Insight into the therapeutic promise of flavonoids against Alzheimer’s Disease. Molecules. 2020;25:6. doi: 10.3390/molecules25061267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Furtado D, Björnmalm M, Ayton S, Bush AI, Kempe K, Caruso F. Overcoming the blood-brain barrier: the role of nanomaterials in treating Neurological Diseases. Adv Mater. 2018;30(46):e1801362. [DOI] [PubMed] [Google Scholar]
- 10.Harilal S, Jose J, Parambi DGT, et al. Advancements in nanotherapeutics for Alzheimer’s disease: current perspectives. J Pharm Pharmacol. 2019;71(9):1370–1383. doi: 10.1111/jphp.13132 [DOI] [PubMed] [Google Scholar]
- 11.Kevadiya BD, Ottemann BM, Thomas MB, et al. Neurotheranostics as personalized medicines. Adv Drug Deliv Rev. 2019;148:252–289. doi: 10.1016/j.addr.2018.10.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ross C, Taylor M, Fullwood N, Allsop D. Liposome delivery systems for the treatment of Alzheimer’s disease. Int J Nanomedicine. 2018;13:8507–8522. doi: 10.2147/IJN.S183117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong KH, Riaz MK, Xie Y, et al. Review of current strategies for delivering Alzheimer’s Disease drugs across the blood-brain barrier. Int J Mol Sci. 2019;20:2. doi: 10.3390/ijms20020381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dong X. Current Strategies for Brain Drug Delivery. Theranostics. 2018;8(6):1481–1493. doi: 10.7150/thno.21254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tosi G, Pederzoli F, Belletti D, et al. Nanomedicine in Alzheimer’s disease: amyloid beta targeting strategy. Prog Brain Res. 2019;245:57–88. [DOI] [PubMed] [Google Scholar]
- 16.Stanfield CL, Germann WJ. Nervoso Il Sistema; il Sistema nervoso Centrale [The Nervous System; the Central Nervous System]. In: Molinari CG, Marini HR, editors. Edizione Fisiologia Terza. [Physiology Third Edition]. Naples, Italy: EdiSES Srl; 2009:216–251. Italian.
- 17.Freeman T. Focused ultrasound opens the blood-brain barrier. Physics World Available from: https://physicsworld.com/a/focused-ultrasound-opens-the-blood-brain-barrier-2/. Accessed August 4, 2020.
- 18.Vasile C. Chapter 1 Polymeric Nanomaterials: Recent Developments, Properties and Medical Applications in Polymeric Nanomaterials in Nanotherapeutics. Elsevier; 2019. [Google Scholar]
- 19.Puri A, Loomis K, Smith B, et al. Lipid-based nanoparticles as pharmaceutical drug carriers: from concepts to clinic. Crit Rev Ther Drug Carrier Syst. 2009;26(6):523–580. doi: 10.1615/CritRevTherDrugCarrierSyst.v26.i6.10 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pandey P, Dahiya M. A brief review on inorganic nanoparticles. J Critical Rev. 2016;3:18–26. [Google Scholar]
- 21.Anzar N, Hasan R, Tyagi M, Yadav N, Narang J. Carbon Nanotube - a Review on Synthesis, Properties and Plethora of Applications in the Field of Biomedical Science. 1 Sensors International; 2020. [Google Scholar]
- 22.Bhattacharya S, Prajapati B, Paul A. A conceptual review on micro bubbles. Biomed J Sci & Tech Res. 2017. [Google Scholar]
- 23.Le TC, Zhai J, Chiu WH, Tran PA, Tran N. Janus particles: recent advances in the biomedical applications. Int J Nanomedicine. 2019;14:6749–6777. doi: 10.2147/IJN.S169030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Praça C, Rai A, Santos T, et al. A nanoformulation for the preferential accumulation in adult neurogenic niches. J Control Release. 2018;284:57–72. doi: 10.1016/j.jconrel.2018.06.013 [DOI] [PubMed] [Google Scholar]
- 25.Agrawal M, Ajazuddin TDK, et al. Recent advancements in liposomes targeting strategies to cross blood-brain barrier (BBB) for the treatment of Alzheimer’s disease. J Control Release. 2017;260:61–77. doi: 10.1016/j.jconrel.2017.05.019 [DOI] [PubMed] [Google Scholar]
- 26.Niu X, Chen J, Gao J. Nanocarriers as a powerful vehicle to overcome blood-brain barrier in treating neurodegenerative diseases: focus on recent advances. Asian J Pharm Sci. 2019;14(5):480–496. doi: 10.1016/j.ajps.2018.09.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Teixeira MI, Lopes CM, Amaral MH, Costa PC. Current insights on lipid nanocarrier-assisted drug delivery in the treatment of neurodegenerative diseases. Eur J Pharm Biopharm. 2020;149:192–217. doi: 10.1016/j.ejpb.2020.01.005 [DOI] [PubMed] [Google Scholar]
- 28.Gao H. Progress and perspectives on targeting nanoparticles for brain drug delivery. Acta Pharm Sin B. 2016;6(4):268–286. doi: 10.1016/j.apsb.2016.05.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pardridge WM. Re-engineering therapeutic antibodies for Alzheimer’s disease as blood-brain barrier penetrating bi-specific antibodies. Expert Opin Biol Ther. 2016;16(12):1455–1468. doi: 10.1080/14712598.2016.1230195 [DOI] [PubMed] [Google Scholar]
- 30.Arora S, Sharma D, Singh J. GLUT-1: an effective target to deliver brain-derived neurotrophic factor gene across the blood brain barrier. ACS Chem Neurosci. 2020;11:1620–1633. doi: 10.1021/acschemneuro.0c00076 [DOI] [PubMed] [Google Scholar]
- 31.Betzer O, Shilo M, Motiei M, Popovtzer R. Insulin-Coated Gold Nanoparticles as an Effective Approach for Bypassing the Blood-Brain Barrier.Proc. SPIE 10891, Nanoscale Imaging, Sensing, and Actuation for Biomedical Applications XVI, 108911H. 2019. doi: 10.1117/12.2510353 [DOI] [Google Scholar]
- 32.Majerova P, Hanes J, Olesova D, Sinsky J, Pilipcinec E, Kovac A. Novel blood-brain barrier shuttle peptides discovered through the phage display method. Molecules. 2020;25:4. doi: 10.3390/molecules25040874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McCully M, Sanchez-Navarro M, Teixido M, Giralt E. Peptide mediated brain delivery of nano- and submicroparticles: a synergistic approach. Curr Pharm Des. 2018;24(13):1366–1376. doi: 10.2174/1381612824666171201115126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guo Q, Xu S, Yang P, et al. A dual-ligand fusion peptide improves the brain-neuron targeting of nanocarriers in Alzheimer’s disease mice. J Control Release. 2020;320:347–362. doi: 10.1016/j.jconrel.2020.01.039 [DOI] [PubMed] [Google Scholar]
- 35.Formicola B, Cox A, Dal Magro R, Masserini M, Re F. Nanomedicine for the treatment of Alzheimer’s Disease. J Biomed Nanotechnol. 2019;15(10):1997–2024. doi: 10.1166/jbn.2019.2837 [DOI] [PubMed] [Google Scholar]
- 36.Crowe TP, Greenlee MHW, Kanthasamy AG, Hsu WH. Mechanism of intranasal drug delivery directly to the brain. Life Sci. 2018;195:44–52. doi: 10.1016/j.lfs.2017.12.025 [DOI] [PubMed] [Google Scholar]
- 37.Akel H, Ismail R, Csóka I. Progress and perspectives of brain-targeting lipid-based nanosystems via the nasal route in Alzheimer’s disease. Eur J Pharm Biopharm. 2020;148:38–53. doi: 10.1016/j.ejpb.2019.12.014 [DOI] [PubMed] [Google Scholar]
- 38.Samaridou E, Alonso MJ. Nose-to-brain peptide delivery - The potential of nanotechnology. Bioorg Med Chem. 2018;26(10):2888–2905. doi: 10.1016/j.bmc.2017.11.001 [DOI] [PubMed] [Google Scholar]
- 39.Gao X, Wu B, Zhang Q, et al. Brain delivery of vasoactive intestinal peptide enhanced with the nanoparticles conjugated with wheat germ agglutinin following intranasal administration. J Control Release. 2007;121(3):156–167. doi: 10.1016/j.jconrel.2007.05.026 [DOI] [PubMed] [Google Scholar]
- 40.Agrawal M, Saraf S, Antimisiaris SG, Chougule MB, Shoyele SA, Alexander A. Nose-to-brain drug delivery: an update on clinical challenges and progress towards approval of anti-Alzheimer drugs. J Control Release. 2018;281:139–177. doi: 10.1016/j.jconrel.2018.05.011 [DOI] [PubMed] [Google Scholar]
- 41.Nanaki SG, Spyrou K, Bekiari C, et al. Hierarchical porous carbon-PLLA and PLGA hybrid nanoparticles for intranasal delivery of galantamine for Alzheimer’s Disease therapy. Pharmaceutics. 2020;12:3. doi: 10.3390/pharmaceutics12030227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Adnet T, Groo AC, Picard C, et al. Pharmacotechnical development of a nasal drug delivery composite nanosystem intended for Alzheimer’s Disease treatment. Pharmaceutics. 2020;12:3. doi: 10.3390/pharmaceutics12030251 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kamei N, Takeda-Morishita M. Brain delivery of insulin boosted by intranasal coadministration with cell-penetrating peptides. J Control Release. 2015;197:105–110. doi: 10.1016/j.jconrel.2014.11.004 [DOI] [PubMed] [Google Scholar]
- 44.Liu Y, Gong Y, Xie W, et al. Microbubbles in combination with focused ultrasound for the delivery of quercetin-modified sulfur nanoparticles through the blood brain barrier into the brain parenchyma and relief of endoplasmic reticulum stress to treat Alzheimer’s disease. Nanoscale. 2020;12(11):6498–6511. doi: 10.1039/C9NR09713A [DOI] [PubMed] [Google Scholar]
- 45.Chen W, Ouyang J, Yi X, et al. Black phosphorus nanosheets as a neuroprotective nanomedicine for neurodegenerative disorder therapy. Adv Mater. 2018;30:3. [DOI] [PubMed] [Google Scholar]
- 46.Zhou H, Gong Y, Liu Y, et al. Intelligently thermoresponsive flower-like hollow nano-ruthenium system for sustained release of nerve growth factor to inhibit hyperphosphorylation of tau and neuronal damage for the treatment of Alzheimer’s disease. Biomaterials. 2020;237:119822. doi: 10.1016/j.biomaterials.2020.119822 [DOI] [PubMed] [Google Scholar]
- 47.Liu W, Wang W, Dong X, Sun Y. Near-infrared light-powered janus nanomotor significantly facilitates inhibition of amyloid-β fibrillogenesis. ACS Appl Mater Interfaces. 2020;12(11):12618–12628. doi: 10.1021/acsami.0c02342 [DOI] [PubMed] [Google Scholar]
- 48.Carradori D, Balducci C, Re F, et al. Antibody-functionalized polymer nanoparticle leading to memory recovery in Alzheimer’s disease-like transgenic mouse model. Nanomedicine. 2018;14(2):609–618. doi: 10.1016/j.nano.2017.12.006 [DOI] [PubMed] [Google Scholar]
- 49.Luo Q, Lin YX, Yang PP, et al. A self-destructive nanosweeper that captures and clears amyloid β-peptides. Nat Commun. 2018;9(1):1802. doi: 10.1038/s41467-018-04255-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhang W, Christofferson AJ, Besford QA, et al. Metal-dependent inhibition of amyloid fibril formation: synergistic effects of cobalt-tannic acid networks. Nanoscale. 2019;11(4):1921–1928. doi: 10.1039/C8NR09221D [DOI] [PubMed] [Google Scholar]
- 51.Sun J, Xie W, Zhu X, Xu M, Liu J. Sulfur nanoparticles with novel morphologies coupled with brain-targeting peptides RVG as a new type of inhibitor against metal-induced aβ aggregation. ACS Chem Neurosci. 2018;9(4):749–761. doi: 10.1021/acschemneuro.7b00312 [DOI] [PubMed] [Google Scholar]
- 52.Chung YJ, Lee BI, Park CB. Multifunctional carbon dots as a therapeutic nanoagent for modulating Cu(ii)-mediated β-amyloid aggregation. Nanoscale. 2019;11(13):6297–6306. doi: 10.1039/C9NR00473D [DOI] [PubMed] [Google Scholar]
- 53.Liu H, Dong X, Liu F, Zheng J, Sun Y. Iminodiacetic acid-conjugated nanoparticles as a bifunctional modulator against Zn. J Colloid Interface Sci. 2017;505:973–982. doi: 10.1016/j.jcis.2017.06.093 [DOI] [PubMed] [Google Scholar]
- 54.Sun D, Zhang W, Yu Q, et al. Chiral penicillamine-modified selenium nanoparticles enantioselectively inhibit metal-induced amyloid β aggregation for treating Alzheimer’s disease. J Colloid Interface Sci. 2017;505:1001–1010. doi: 10.1016/j.jcis.2017.06.083 [DOI] [PubMed] [Google Scholar]
- 55.Zhang X, Zhong M, Zhao P, et al. Screening a specific Zn(ii)-binding peptide for improving the cognitive decline of Alzheimer’s disease in APP/PS1 transgenic mice by inhibiting Zn. Biomater Sci. 2019;7(12):5197–5210. doi: 10.1039/C9BM00676A [DOI] [PubMed] [Google Scholar]
- 56.Han Q, Wang X, Liu X, et al. MoO3-X nanodots with dual enzyme mimic activities as multifunctional modulators for amyloid assembly and neurotoxicity. J Colloid Interface Sci. 2019;539:575–584. doi: 10.1016/j.jcis.2018.12.093 [DOI] [PubMed] [Google Scholar]
- 57.Zhang Z, Wang J, Song Y, Wang Z, Dong M, Liu L. Disassembly of Alzheimer’s amyloid fibrils by functional upconversion nanoparticles under near-infrared light irradiation. Colloids Surf B Biointerfaces. 2019;181:341–348. doi: 10.1016/j.colsurfb.2019.05.053 [DOI] [PubMed] [Google Scholar]
- 58.Pederzoli F, Ruozi B, Duskey J, et al. Nanomedicine against Aβ aggregation by β-sheet breaker peptide delivery: in vitro evidence. Pharmaceutics. 2019;11:11. doi: 10.3390/pharmaceutics11110572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang H, Hao C, Qu A, et al. Light-induced chiral iron copper selenide nanoparticles prevent β-Amyloidopathy in vivo. Angew Chem Int Ed Engl. 2020. [DOI] [PubMed] [Google Scholar]
- 60.Boonruamkaew P, Chonpathompikunlert P, Vong LB, et al. Chronic treatment with a smart antioxidative nanoparticle for inhibition of amyloid plaque propagation in Tg2576 mouse model of Alzheimer’s disease. Sci Rep. 2017;7(1):3785. doi: 10.1038/s41598-017-03411-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Jeon SG, Cha MY, Kim JI, et al. Vitamin D-binding protein-loaded PLGA nanoparticles suppress Alzheimer’s disease-related pathology in 5XFAD mice. Nanomedicine. 2019;17:297–307. doi: 10.1016/j.nano.2019.02.004 [DOI] [PubMed] [Google Scholar]
- 62.Vakilinezhad MA, Amini A, Akbari Javar H, Baha’addini Beigi Zarandi BF, Montaseri H, Dinarvand R. Nicotinamide loaded functionalized solid lipid nanoparticles improves cognition in Alzheimer’s disease animal model by reducing Tau hyperphosphorylation. Daru. 2018;26(2):165–177. doi: 10.1007/s40199-018-0221-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sonawane SK, Ahmad A, Chinnathambi S. Protein-capped metal nanoparticles inhibit tau aggregation in Alzheimer’s Disease. ACS Omega. 2019;4(7):12833–12840. doi: 10.1021/acsomega.9b01411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cai X, Zhang K, Xie X, et al. Self-assembly hollow manganese Prussian white nanocapsules attenuate Tau-related neuropathology and cognitive decline. Biomaterials. 2020;231:119678. doi: 10.1016/j.biomaterials.2019.119678 [DOI] [PubMed] [Google Scholar]
- 65.Ji D, Wu X, Li D, et al. Protective effects of chondroitin sulphate nano-selenium on a mouse model of Alzheimer’s disease. Int J Biol Macromol. 2020;154:233–245. doi: 10.1016/j.ijbiomac.2020.03.079 [DOI] [PubMed] [Google Scholar]
- 66.Gao F, Zhao J, Liu P, et al. Preparation and in vitro evaluation of multi-target-directed selenium-chondroitin sulfate nanoparticles in protecting against the Alzheimer’s disease. Int J Biol Macromol. 2020;142:265–276. doi: 10.1016/j.ijbiomac.2019.09.098 [DOI] [PubMed] [Google Scholar]
- 67.Sierra C. Hypertension and the Risk of Dementia. Front Cardiovasc Med. 2020;7:5. doi: 10.3389/fcvm.2020.00005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wahidi N, Lerner AJ. Blood pressure control and protection of the aging Brain. Neurotherapeutics. 2019;16(3):569–579. doi: 10.1007/s13311-019-00747-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Novotny M, Klimova B, Valis M. Nitrendipine and dementia: forgotten positive facts? Front Aging Neurosci. 2018;10:418. doi: 10.3389/fnagi.2018.00418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Nimmrich V, Eckert A. Calcium channel blockers and dementia. Br J Pharmacol. 2013;169(6):1203–1210. doi: 10.1111/bph.12240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Alawdi SH, Eidi H, Safar MM, Abdel-Wahhab MA. Loading amlodipine on diamond nanoparticles: a novel drug delivery system. Nanotechnol Sci Appl. 2019;12:47–53. doi: 10.2147/NSA.S232517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Li T, Liang W, Xiao X, Qian Y. Nanotechnology, an alternative with promising prospects and advantages for the treatment of cardiovascular diseases. Int J Nanomedicine. 2018;13:7349–7362. doi: 10.2147/IJN.S179678 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Evans CW, Viola HM, Ho D, et al. Nanoparticle-mediated internalisation and release of a calcium channel blocker. RSC Adv. 2012;2(23):8587–8590. doi: 10.1039/c2ra21058d [DOI] [Google Scholar]
- 74.Jana U, Mohanty AK, Pal SL, Manna PK, Mohanta GP. Felodipine loaded PLGA nanoparticles: preparation, physicochemical characterization and in vivo toxicity study. Nano Convergence. 2014;1(1):31. doi: 10.1186/s40580-014-0031-5 [DOI] [Google Scholar]
- 75.Jana U, Mohanty AK, Manna PK, Mohanta GP. Preparation and characterization of nebivolol nanoparticles using Eudragit® RS100. Colloids Surf B Biointerfaces. 2014;113:269–275. doi: 10.1016/j.colsurfb.2013.09.001 [DOI] [PubMed] [Google Scholar]
- 76.Tóth OM, Menyhárt Á, Varga V, et al. Chitosan nanoparticles release nimodipine in response to tissue acidosis to attenuate spreading depolarization evoked during forebrain ischemia. Neuropharmacology. 2020;162:107850. doi: 10.1016/j.neuropharm.2019.107850 [DOI] [PubMed] [Google Scholar]
- 77.Wójtowicz S, Strosznajder AK, Jeżyna M, Strosznajder JB. The novel role of PPAR alpha in the brain: promising target in therapy of Alzheimer’s Disease and other neurodegenerative disorders. Neurochem Res. 2020;45:972–988. doi: 10.1007/s11064-020-02993-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Silva-Abreu M, Calpena AC, Andrés-Benito P, et al. PPARγ agonist-loaded PLGA-PEG nanocarriers as a potential treatment for Alzheimer’s disease: in vitro and in vivo studies. Int J Nanomedicine. 2018;13:5577–5590. doi: 10.2147/IJN.S171490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Xiao Y, Zhang E, Fu A. Promotion of SH-SY5Y cell growth by gold nanoparticles modified with 6-mercaptopurine and a neuron-penetrating peptide. Nanoscale Res Lett. 2017;12(1):641. doi: 10.1186/s11671-017-2417-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Marcus M, Smith A, Maswadeh A, et al. Magnetic targeting of growth factors using iron oxide nanoparticles. Nanomaterials. 2018;8:9. doi: 10.3390/nano8090707 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Yuan M, Wang Y, Qin YX. Engineered nanomedicine for neuroregeneration: light emitting diode-mediated superparamagnetic iron oxide-gold core-shell nanoparticles functionalized by nerve growth factor. Nanomedicine. 2019;21:102052. doi: 10.1016/j.nano.2019.102052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yuan M, Wang Y, Qin YX. Promoting neuroregeneration by applying dynamic magnetic fields to a novel nanomedicine: superparamagnetic iron oxide (SPIO)-gold nanoparticles bounded with nerve growth factor (NGF). Nanomedicine. 2018;14(4):1337–1347. doi: 10.1016/j.nano.2018.03.004 [DOI] [PubMed] [Google Scholar]
- 83.Katebi S, Esmaeili A, Ghaedi K, Zarrabi A. Superparamagnetic iron oxide nanoparticles combined with NGF and quercetin promote neuronal branching morphogenesis of PC12 cells. Int J Nanomedicine. 2019;14:2157–2169. doi: 10.2147/IJN.S191878 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kalra EK. Nutraceutical–definition and introduction. AAPS PharmSci. 2003;5(3):E25. doi: 10.1208/ps050325 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kanubaddi KR, Yang SH, Wu LW, Lee CH, Weng CF. Nanoparticle-conjugated nutraceuticals exert prospectively palliative of amyloid aggregation. Int J Nanomedicine. 2018;13:8473–8485. doi: 10.2147/IJN.S179484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Pinheiro RGR, Granja A, Loureiro JA, et al. Quercetin lipid nanoparticles functionalized with transferrin for Alzheimer’s disease. Eur J Pharm Sci. 2020;148:105314. doi: 10.1016/j.ejps.2020.105314 [DOI] [PubMed] [Google Scholar]
- 87.Sun D, Li N, Zhang W, et al. Design of PLGA-functionalized quercetin nanoparticles for potential use in Alzheimer’s disease. Colloids Surf B Biointerfaces. 2016;148:116–129. doi: 10.1016/j.colsurfb.2016.08.052 [DOI] [PubMed] [Google Scholar]
- 88.Amin FU, Shah SA, Badshah H, Khan M, Kim MO. Anthocyanins encapsulated by PLGA@PEG nanoparticles potentially improved its free radical scavenging capabilities via p38/JNK pathway against Aβ. J Nanobiotechnol. 2017;15(1):12. doi: 10.1186/s12951-016-0227-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Ishak RAH, Mostafa NM, Kamel AO. Stealth lipid polymer hybrid nanoparticles loaded with rutin for effective brain delivery comparative study with the gold standard (Tween 80): optimization, characterization and biodistribution. Drug Deliv. 2017;24(1):1874–1890. doi: 10.1080/10717544.2017.1410263 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ali T, Kim MJ, Rehman SU, Ahmad A, Kim MO. Anthocyanin-loaded PEG-gold nanoparticles enhanced the neuroprotection of anthocyanins in an Aβ. Mol Neurobiol. 2017;54(8):6490–6506. doi: 10.1007/s12035-016-0136-4 [DOI] [PubMed] [Google Scholar]
- 91.Kim MJ, Rehman SU, Amin FU, Kim MO. Enhanced neuroprotection of anthocyanin-loaded PEG-gold nanoparticles against Aβ. Nanomedicine. 2017;13(8):2533–2544. doi: 10.1016/j.nano.2017.06.022 [DOI] [PubMed] [Google Scholar]
- 92.Kheradmand E, Hajizadeh Moghaddam A, Zare M. Neuroprotective effect of hesperetin and nano-hesperetin on recognition memory impairment and the elevated oxygen stress in rat model of Alzheimer’s disease. Biomed Pharmacother. 2018;97:1096–1101. doi: 10.1016/j.biopha.2017.11.047 [DOI] [PubMed] [Google Scholar]
- 93.Wijiani N, Isadiartuti D, Rijal MAS, Yusuf H. Characterization and dissolution study of micellar curcumin-spray dried powder for oral delivery. Int J Nanomedicine. 2020;15:1787–1796. doi: 10.2147/IJN.S245050 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Chen S, Han Y, Sun C, et al. Effect of molecular weight of hyaluronan on zein-based nanoparticles: fabrication, structural characterization and delivery of curcumin. Carbohydr Polym. 2018;201:599–607. doi: 10.1016/j.carbpol.2018.08.116 [DOI] [PubMed] [Google Scholar]
- 95.Lohan S, Raza K, Mehta SK, Bhatti GK, Saini S, Singh B. Anti-Alzheimer’s potential of berberine using surface decorated multi-walled carbon nanotubes: A preclinical evidence. Int J Pharm. 2017;530(12):263–278. doi: 10.1016/j.ijpharm.2017.07.080 [DOI] [PubMed] [Google Scholar]
- 96.Sathya S, Shanmuganathan B, Devi KP. Deciphering the anti-apoptotic potential of α-bisabolol loaded solid lipid nanoparticles against Aβ induced neurotoxicity in Neuro-2a cells. Colloids Surf B Biointerfaces. 2020;190:110948. doi: 10.1016/j.colsurfb.2020.110948 [DOI] [PubMed] [Google Scholar]
- 97.Brenza TM, Ghaisas S, Ramirez JEV, et al. Neuronal protection against oxidative insult by polyanhydride nanoparticle-based mitochondria-targeted antioxidant therapy. Nanomedicine. 2017;13(3):809–820. doi: 10.1016/j.nano.2016.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Aalinkeel R, Kutscher HL, Singh A, et al. Neuroprotective effects of a biodegradable poly(lactic-co-glycolic acid)-ginsenoside Rg3 nanoformulation: a potential nanotherapy for Alzheimer’s disease? J Drug Target. 2018;26(2):182–193. doi: 10.1080/1061186X.2017.1354002 [DOI] [PubMed] [Google Scholar]
- 99.Sathya S, Shanmuganathan B, Saranya S, Vaidevi S, Ruckmani K, Pandima Devi K. Phytol-loaded PLGA nanoparticle as a modulator of Alzheimer’s toxic Aβ peptide aggregation and fibrillation associated with impaired neuronal cell function.. Artif Cells Nanomed Biotechnol. 2018;46(8):1719–1730. doi: 10.1080/21691401.2017.1391822 [DOI] [PubMed] [Google Scholar]
- 100.Cano A, Ettcheto M, Chang JH, et al. Dual-drug loaded nanoparticles of Epigallocatechin-3-gallate (EGCG)/Ascorbic acid enhance therapeutic efficacy of EGCG in a APPswe/PS1dE9 Alzheimer’s disease mice model. J Control Release. 2019;301:62–75. doi: 10.1016/j.jconrel.2019.03.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Xu R, Wang J, Xu J, et al. Rhynchophylline Loaded-mPEG-PLGA nanoparticles coated with tween-80 for preliminary study in Alzheimer’s Disease. Int J Nanomedicine. 2020;15:1149–1160. doi: 10.2147/IJN.S236922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chen S, Han Y, Huang J, et al. Fabrication and characterization of layer-by-layer composite nanoparticles based on zein and hyaluronic acid for codelivery of Curcumin and Quercetagetin. ACS Appl Mater Interfaces. 2019;11(18):16922–16933. doi: 10.1021/acsami.9b02529 [DOI] [PubMed] [Google Scholar]
- 103.Okuda M, Fujita Y, Sugimoto H. The additive effects of low dose intake of ferulic acid, phosphatidylserine and curcumin, not alone, improve cognitive function in APPswe/PS1dE9 transgenic mice. Biol Pharm Bull. 2019;42(10):1694–1706. doi: 10.1248/bpb.b19-00332 [DOI] [PubMed] [Google Scholar]
- 104.Agarwal H, Shanmugam V. A review on anti-inflammatory activity of green synthesized zinc oxide nanoparticle: mechanism-based approach. Bioorg Chem. 2020;94:103423. doi: 10.1016/j.bioorg.2019.103423 [DOI] [PubMed] [Google Scholar]
- 105.Badeggi UM, Ismail E, Adeloye AO, et al. Green synthesis of gold nanoparticles capped with procyanidins from. Biomolecules. 2020;10:3. doi: 10.3390/biom10030452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lee YJ, Park Y. Green synthetic nanoarchitectonics of gold and silver nanoparticles prepared using quercetin and their cytotoxicity and catalytic applications. J Nanosci Nanotechnol. 2020;20(5):2781–2790. doi: 10.1166/jnn.2020.17453 [DOI] [PubMed] [Google Scholar]
- 107.Youssif KA, Haggag EG, Elshamy AM, et al. Anti-Alzheimer potential, metabolomic profiling and molecular docking of green synthesized silver nanoparticles of Lampranthus coccineus and Malephora lutea aqueous extracts. PLoS One. 2019;14(11):e0223781. doi: 10.1371/journal.pone.0223781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Dos Santos Rodrigues B, Kanekiyo T, Singh J. ApoE-2 brain-targeted gene therapy through transferrin and penetratin tagged liposomal nanoparticles. Pharm Res. 2019;36(11):161. doi: 10.1007/s11095-019-2691-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Rassu G, Soddu E, Posadino AM, et al. Nose-to-brain delivery of BACE1 siRNA loaded in solid lipid nanoparticles for Alzheimer’s therapy. Colloids Surf B Biointerfaces. 2017;152:296–301. doi: 10.1016/j.colsurfb.2017.01.031 [DOI] [PubMed] [Google Scholar]
- 110.Wang P, Zheng X, Guo Q, et al. Systemic delivery of BACE1 siRNA through neuron-targeted nanocomplexes for treatment of Alzheimer’s disease. J Control Release. 2018;279:220–233. doi: 10.1016/j.jconrel.2018.04.034 [DOI] [PubMed] [Google Scholar]
- 111.Poplawski SG, Garbett KA, McMahan RL, et al. An antisense oligonucleotide leads to suppressed transcription of hdac2 and long-term memory enhancement. Mol Ther Nucleic Acids. 2020;19:1399–1412. doi: 10.1016/j.omtn.2020.01.027 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Shao X, Cui W, Xie X, Ma W, Zhan Y, Lin Y. Treatment of Alzheimer’s disease with framework nucleic acids. Cell Prolif. 2020;e12787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zhang FQ, Jiang JL, Zhang JT, Niu H, Fu XQ, Zeng LL. Current status and future prospects of stem cell therapy in Alzheimer’s disease. Neural Regen Res. 2020;15(2):242–250. doi: 10.4103/1673-5374.265544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Reddy AP, Ravichandran J, Carkaci-Salli N. Neural regeneration therapies for Alzheimer’s and Parkinson’s disease-related disorders. Biochim Biophys Acta Mol Basis Dis. 2020;1866(4):165506. doi: 10.1016/j.bbadis.2019.06.020 [DOI] [PubMed] [Google Scholar]
- 115.Zhang L, Dong ZF, Zhang JY. Immunomodulatory role of mesenchymal stem cells in Alzheimer’s disease. Life Sci. 2020;246:117405. doi: 10.1016/j.lfs.2020.117405 [DOI] [PubMed] [Google Scholar]
- 116.Hour FQ, Moghadam AJ, Shakeri-Zadeh A, Bakhtiyari M, Shabani R, Mehdizadeh M. Magnetic targeted delivery of the SPIONs-labeled mesenchymal stem cells derived from human Wharton’s jelly in Alzheimer’s rat models. J Control Release. 2020;321:430–441. doi: 10.1016/j.jconrel.2020.02.035 [DOI] [PubMed] [Google Scholar]
- 117.Sato M, Takizawa H, Nakamura A, Turner BJ, Shabanpoor F, Aoki Y. Application of urine-derived stem cells to cellular modeling in neuromuscular and neurodegenerative diseases. Front Mol Neurosci. 2019;12:297. doi: 10.3389/fnmol.2019.00297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Mungenast AE, Siegert S, Tsai LH. Modeling Alzheimer’s disease with human induced pluripotent stem (iPS) cells. Mol Cell Neurosci. 2016;73:13–31. doi: 10.1016/j.mcn.2015.11.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Chang KH, Lee-Chen GJ, Huang CC, et al. Modeling Alzheimer’s Disease by induced pluripotent stem cells carrying APP D678H mutation. Mol Neurobiol. 2019;56(6):3972–3983. doi: 10.1007/s12035-018-1336-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Zhang X, Hu D, Shang Y, Qi X. Using induced pluripotent stem cell neuronal models to study neurodegenerative diseases. Biochim Biophys Acta Mol Basis Dis. 2020;1866(4):165431. doi: 10.1016/j.bbadis.2019.03.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Hossini AM, Megges M, Prigione A, et al. Induced pluripotent stem cell-derived neuronal cells from a sporadic Alzheimer’s disease donor as a model for investigating AD-associated gene regulatory networks. BMC Genomics. 2015;16:84. doi: 10.1186/s12864-015-1262-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Pavoni S, Jarray R, Nassor F, et al. Small-molecule induction of Aβ-42 peptide production in human cerebral organoids to model Alzheimer’s disease associated phenotypes. PLoS One. 2018;13(12):e0209150. doi: 10.1371/journal.pone.0209150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Raja WK, Mungenast AE, Lin YT, et al. Self-Organizing 3D human neural tissue derived from induced pluripotent stem cells recapitulate Alzheimer’s Disease phenotypes. PLoS One. 2016;11(9):e0161969. doi: 10.1371/journal.pone.0161969 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Nassor F, Jarray R, Biard DSF, et al. Long term gene expression in human induced pluripotent stem cells and cerebral organoids to model a Neurodegenerative Disease. Front Cell Neurosci. 2020;14:14. doi: 10.3389/fncel.2020.00014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Aulić S, Bolognesi ML, Legname G. Small-molecule theranostic probes: a promising future in neurodegenerative diseases. Int J Cell Biol. 2013;2013:150952. doi: 10.1155/2013/150952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Chakravarthy M, Chen S, Dodd PR, Veedu RN. Nucleic acid-based theranostics for tackling Alzheimer’s Disease. Theranostics. 2017;7(16):3933–3947. doi: 10.7150/thno.21529 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Papadia K, Markoutsa E, Mourtas S, et al. Multifunctional LUV liposomes decorated for BBB and amyloid targeting. A. In vitro proof-of-concept. Eur J Pharm Sci. 2017;101:140–148. doi: 10.1016/j.ejps.2017.02.019 [DOI] [PubMed] [Google Scholar]
- 128.Mourtas S, Christodoulou P, Klepetsanis P, Gatos D, Barlos K, Antimisiaris SG. Preparation of Benzothiazolyl-Decorated Nanoliposomes. Molecules. 2019;24:8. doi: 10.3390/molecules24081540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Caldorera-Moore M, Guimard N, Shi L, Roy K. Designer nanoparticles: incorporating size, shape and triggered release into nanoscale drug carriers. Expert Opin Drug Deliv. 2010;7(4):479–495. doi: 10.1517/17425240903579971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Dal Magro R, Simonelli S, Cox A, et al. The extent of human apolipoprotein a-i lipidation strongly affects the β-amyloid efflux across the blood-brain barrier. Front Neurosci. 2019;13:419. doi: 10.3389/fnins.2019.00419 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Binda A, Panariti A, Barbuti A, et al. Modulation of the intrinsic neuronal excitability by multifunctional liposomes tailored for the treatment of Alzheimer’s disease. Int J Nanomedicine. 2018;13:4059–4071. doi: 10.2147/IJN.S161563 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Rodriguez Then FS, Jackson J, Ware C, Churchyard R, Hanseeuw B. Interdisciplinary and transdisciplinary perspectives: on the road to a holistic approach to dementia prevention and care. J Alzheimers Dis Rep. 2020;4(1):39–48. doi: 10.3233/ADR-180070 [DOI] [PMC free article] [PubMed] [Google Scholar]