Abstract
Hepatocellular carcinoma (HCC) remains a challenge in the medical field due to its high malignancy and mortality rates particularly for HCC, which has developed multidrug resistance. Therefore, the identification of efficient chemotherapeutic drugs for multidrug resistant HCC has become an urgent issue. Natural products have always been of significance in drug discovery. In the present study, a cell-based method was used to screen a natural compound library, which consisted of 78 compounds, and the doxorubicin-resistant cancer cell line, HepG2/ADM, as screening tools. The findings of the present study led to the shortlisting of one of the compounds, digitoxin, which displayed an inhibitory effect on HepG2/ADM cells, with 50% inhibitory concentration values of 132.65±3.83, 52.29±6.26, and 9.13±3.67 nM for 24, 48, and 72 h, respectively. Immunofluorescence, western blotting and cell cycle analyses revealed that digitoxin induced G2/M cell cycle arrest via the serine/threonine-protein kinase ATR (ATR)-serine/threonine-protein kinase Chk2 (CHK2)-M-phase inducer phosphatase 3 (CDC25C) signaling pathway in HepG2/ADM cells, which may have resulted from a DNA double-stranded break. Digitoxin also induced mitochondrial apoptosis, which was characterized by changes in the interaction between Bcl-2 and Bax, the release of cytochrome c, as well as the activation of the caspase-3 and −9. To the best of our knowledge, the present study is the first report that digitoxin displays an anti-HCC effect on HepG2/ADM cells through G2/M cell cycle arrest, which was mediated by the ATR-CHK2-CDC25C signaling pathway and mitochondrial apoptosis. Therefore, digitoxin could be a promising chemotherapeutic agent for the treatment of patients with HCC.
Keywords: digitoxin, liver cancer, multidrug resistance, G2/M phase arrest, apoptosis
Introduction
Liver cancer, which is one of the most common tumors worldwide, has become difficult to treat due to its high malignancy and mortality rates (1). The mortality rate of liver cancer has increased from 7.5 to 11.2 in men and from 2.8 to 3.8 in women (per 100,000 persons) between 2000 and 2015, worldwide (2). A high amount of research has been performed to identify effective reagents for the treatment liver cancer (3). So far, chemotherapy remains the most widely used treatment for liver cancer; however, most conventional chemotherapeutic drugs, including cisplatin, 5-fluorouracil, and doxorubicin (Dox) exhibit poor efficiency in the treatment of hepatocellular carcinoma (HCC), with a <10% inhibition growth rate (4). A previous study has also confirmed the ineffectiveness of conventional chemotherapy whether they were administered intravenously or intra-arterially (5). Sorafenib® is the only tyrosine kinase inhibitor for HCC treatment approved by the Food and Drug Administration (FDA) of the United States of America; however, its limited efficacy and adverse side effects, including hand/food/skin reactions, asthenia, diarrhea, and arterial hypertension, highlights whether it is suitable for use in a clinical setting (5,6).
Multidrug resistance (MDR) also contributes to the intractability of HCC. MDR is defined as a process in which cancer cells gain resistance to multiple chemotherapeutic drugs with different structures and mechanisms of action (7). MDR has been reported to be responsible for >90% of cases in which chemotherapy had failed and the tumor had recurred (8). Therefore, identification of efficient drugs to combat MDR has become an important issue in the medical field. Natural products have great potential for drug discovery and constitute a large number of chemotherapy agents in cancer treatment. For example, the discovery of vinblastine and vincristine have been developed for use as anticancer agents from natural sources (9). An increasing number of compounds derived from natural resources have been approved as anticancer drugs by the FDA, such as camptothecin, paclitaxel, anthracyclines and taxanes (10–12). Among these, drugs which induce cancer cell cycle arrest or apoptosis are a big part. It is widely accepted that cell cycle arrest may result from DNA damage (13). ATM, a serine/threonine protein kinase, activates checkpoint signaling upon DNA double-stranded breaks (DSBs), thereby acting as a DNA damage sensor and playing a significant role in cell cycle arrest (14).
Digitoxin is a natural cardiac glycoside derived from Digitalis (15). As a potent inhibitor of Na+/K+-ATPase, digitoxin has been clinically used for congestive heart failure for more than 40 years (16). Previously, a number of studies have focused on the anticancer potential of digitoxin and verified notable antitumor activities of digitoxin in lung cancer (17), pancreatic cancer (18), glioma (19), liver cancer (20), prostate cancer (21) and melanoma (22). Mechanistic studies have revealed that the growth inhibitory effect of digitoxin was associated with the induction of apoptosis (23), inhibition of epithelial-mesenchymal transition (21) and suppression of cancer cell stemness (24); however, the underlying mechanism of action of digitoxin against multidrug-resistant HCC cells has not been fully elucidated.
In the present study, a library of 78 natural compounds, including digitoxin was screened in the Dox-resistant cancer cell line, HepG2/ADM. Further investigations demonstrated that digitoxin displayed an inhibitory effect on multidrug-resistant HepG2/ADM cells through G2/M cell cycle arrest via the serine/threonine-protein kinase ATR (ATR)-serine/threonine-protein kinase Chk2 (CHK2)-M-phase inducer phosphatase 3 (CDC25C) signaling pathway and mitochondrial apoptosis. The findings of the present study suggested that digitoxin may be developed into a chemotherapeutic agent for patients with HCC.
Materials and methods
Reagents and antibodies
A library of 78 natural compounds was obtained from Target Molecule Corp. Digitoxin (≥98% pure) was purchased from Baoji Herbest Bio-Tech Co., Ltd. MTT was supplied by Sigma-Aldrich (Merck KGaA). An Annexin-V-FITC/propidium iodide (PI) staining assay kit was obtained from Beyotime Institute of Biotechnology. The bicinchoninic protein assay kit (BCA) was purchased from Thermo Fisher Scientific Inc., while PI and 4′,6-dimidyl-2-phenylindole (DAPI) were purchased from Roche Diagnostics (Shanghai) Co. Ltd. Primary antibodies against cyclin-dependent kinase 1 (CDK1, #9116), cyclin B1 (#4138), phosphorylated (p)-CDK1 (Thr14) (#2543), p-histone H2AX (γH2AX, #9718), ATR (#2790), p-ATR (Ser428) (#2853), CHK2 (#6334), p-Chk2 (Thr68) (#2197), CDC25C (#4688), p-CDC25C (Thr48) (#12028), Bax (#5023), Bcl-2 (#15071), cytochrome c (#11940), caspase-9 (#9508) and-3 (#9662), cleaved-caspase-3 (#9579) and −9 (#20750), cleaved poly (ADP-ribose) polymerase (PARP) (#5625), β-actin (#4970) and the horse-radish peroxidase (HRP)-conjugated secondary antibodies (Anti-mouse IgG, #7076; Anti-rabbit IgG, #7074), Alexa Fluor 647-conjugated anti-rabbit IgG (H+L) (#4414) were obtained from Cell Signaling Technology Inc., (dilution of primary antibodies, 1:1,000; dilution of secondary antibodies, 1:2,000).
Cell line and cell culture
The Dox-resistant human HCC cell line, HepG2/ADM was provided by Professor Kwok-Pui Fung (The Chinese University of Hong Kong, Hong Kong, China). HepG2/ADM cells were cultured in RPMI 1640 medium supplemented with Dox (1.2 µM, Sigma-Aldrich), 1% penicillin-streptomycin (PS), and 10% fetal bovine serum (FBS) to maintain the multidrug-resistant characteristics of the HepG2/ADM cell line. RPMI 1640 medium, PS, and FBS were supplied by Thermo Fisher Scientific Inc.. Cells were incubated at 37°C in a humidified incubator with 5% CO2.
Compound library screening
The cytotoxicity screening of the 78 natural compounds in the library against HepG2/ADM cells was performed via the MTT assay. Cells (5,000/well) were seeded into 96-well plates and cultured overnight at 37°C. After treatment with 78 natural compounds (0.1 µM) for 72 h at 37°C, respectively, cells were incubated with 20 µl MTT (5 mg/ml) at 37°C for 3 h. The formazan crystals were dissolved in 100 µl dimethlysulfoxide (DMSO) and the absorbance of each well was recorded at 595 nm wavelengths using a microplate reader (Beckman Coulter Inc.).
Cell viability assay
Viability of HepG2/ADM cells was determined using a MTT assay. Cells (5,000/well) were seeded in 96-well plates and cultured overnight. Following treatment with digitoxin at concentrations ranging from 3.906–1,000.000 nM for 24, 48 and 72 h, respectively, cells were exposed to 20 µl MTT (5 mg/ml) and incubated at 37°C for 3 h. The formazan crystals were dissolved with 100 µl DMSO and the absorbance was measured at 595 nm using a microplate reader (Beckman Coulter Inc.). As previously described (25), cells treated with medium containing 0.2% DMSO for 24, 48 or 72 h were considered as 100% viable, respectively.
Cell cycle analysis
HepG2/ADM cells (3×105/well) were seeded in 6-well plates and cultured overnight, then treated with digitoxin at 4, 20 and 100 nM for 24 h or 20 nM digitoxin for 12, 24 and 36 h, respectively. Following fixation and permeabilization with pre-cooled 75% ethanol at 4°C overnight, cells were stained with 0.2 mg/ml PI and 0.1 mg/ml RNase in the dark at room temperature for 15 min. The PI fluorescence of the cells was analyzed using an EPICS-X flow cytometry (Beckman Coulter, Inc.) Then, the phase distribution of cell cycle was analyzed using ModFit LT v3.1 software (Verity Software House, Inc.).
Western blot analysis
Following treatment with digitoxin for 24 h, HepG2/ADM cells were collected using trypsinization. Total cellular protein was extracted using the radioimmunoprecipitation assay (RIPA) lysis buffer (containing 1 mM PMSF, 1X phosphatase inhibitor and 1X protease inhibitor, Beyotime Institute of Biotechnology). Then, a BCA assay kit was used to quantify the protein concentration. Proteins (30 µg/lane) were separated using 12% SDS-PAGE gels and then transferred onto PVDF membranes. The membranes were blocked with 5% skimmed milk at room temperature for 1 h and probed with the primary antibodies at a dilution of 1:1,000 overnight at 4°C. After incubation with secondary antibody at a dilution of 1:2,000 for 1 h at room temperature, the protein bands were visualized using an ECL detection kit (Millipore, Merck KGaA) and quantified using the ImageJ software v1.8.0 (National Institutes of Health). β-actin was used as the loading control.
Immunofluorescence
HepG2/ADM cells were treated with digitoxin for 24 h. Then, cells were fixed with 4% paraformaldehyde (PFA) for 15 min at room temperature. After blocking with 5% bovine serum albumin (BSA; Sigma-Aldrich; Merck KGaA) containing 0.4% Triton X-100 (Sigma-Aldrich; Merck KGaA) for 1 h at room temperature, the cells were incubated with the γH2AX primary antibody (1:1,000) overnight at 4°C and Alexa Fluor 647-conjugated secondary antibody (1:2,000) for 1 h at room temperature. Fluorescence was observed using a confocal microscope with a 40× magnification (Axio Vert.A1; Zeiss GmbH).
Annexin-V-FITC/PI staining assay
Following treatment with digitoxin for 24 and 48 h, respectively, HepG2/ADM cells were collected and stained with Annexin-V-FITC/PI for 15 min in darkness at room temperature. The cell apoptotic rates were analyzed using an Epics XL flow cytometer (Beckman Coulter Inc.). The following wavelengths were used 488 (excitation) and 525 (emission) nm for Annexin V-FITC; and 488 (excitation) and 620 nm (emission) for PI. The data was quantified using the FlowJo v7.6 software (FlowJo LLC).
Statistical analysis
All experiments were performed at least three times. Results are presented as the mean ± SEM. GraphPad Prism v7.0 (GraphPad Software Inc.) was used for statistical analysis. One-way analysis of variance (ANOVA) followed by a Tukey's post hoc test was used for multiple comparison. P<0.05 was considered to indicate a statistically significant difference.
Results
Digitoxin shows cytotoxicity towards HepG2/ADM cells
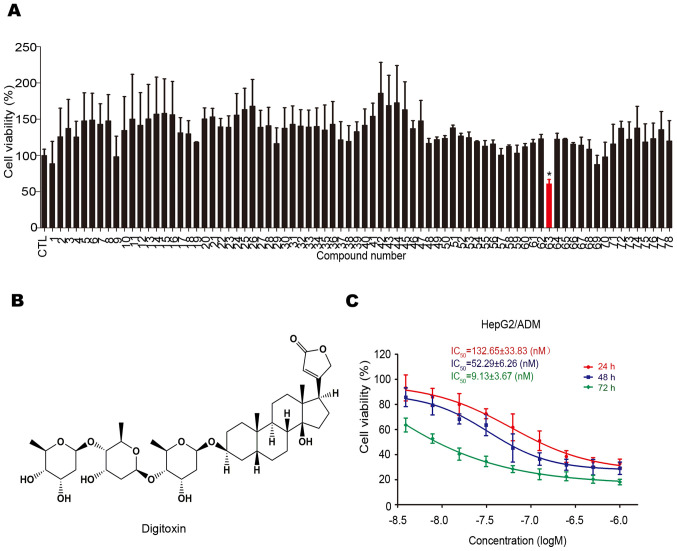
The cytotoxicity of 78 natural compounds on HepG2/ADM cells were determined using the MTT assay. As shown in Fig. 1A, digitoxin (No. 63; chemical structure shown in Fig. 1B) was found to have a greater cytotoxic effect on HepG2/ADM cells. Subsequently, the anti-HCC effect of digitoxin on HepG2/ADM cells was assessed using the MTT assay. As shown in Fig. 1C, digitoxin decreased the viability of HepG2/ADM cells in a dose-dependent manner, with IC50 values of 132.65±33.83, 52.29±6.26 and 9.13±3.67 nM following treatment for 24, 48 and 72 h, respectively. The concentrations used in the cell viability assay were based on the results of the pre-experiment.
Figure 1.
Digitoxin shows cytotoxicity towards HepG2/ADM cells. (A) HepG2/ADM cells were treated with 78 compounds at 0.1 µM for 24 h and the viability was measured using the MTT assay. Digitoxin (No. 63) was identified as the most cytotoxic compound towards HepG2/ADM cells among the different compounds, indicated by the red color. *P<0.05 vs. the control group. (B) Chemical structure of digitoxin. (C) HepG2/ADM cells were treated with digitoxin at different concentrations for 24, 48 or 72 h and the viability was measured using the MTT assay. Digitoxin exerted an anti-HCC effect on HepG2/ADM cells in a dose- and time-dependent manner.
Digitoxin blocks the HepG2/ADM cell cycle at G2/M phase
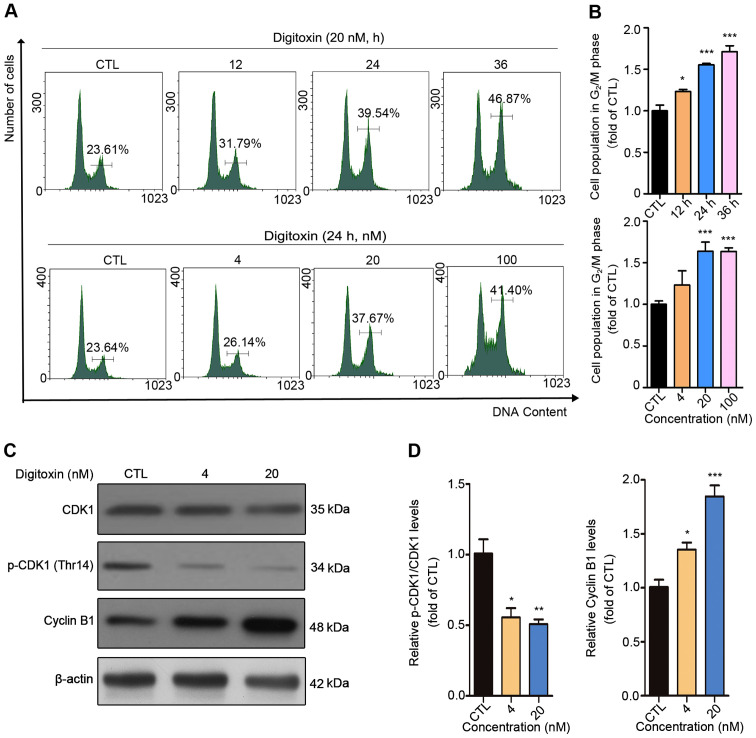
To investigate whether the inhibitory effect of digitoxin on HepG2/ADM cells was associated with cell cycle arrest, DNA content analysis was performed using flow cytometry. The cell population at G2/M phase increased from 23.61 to 46.87% with 0, 12, 24, and 36 h (0 h as the CTL group) following 20 nM digitoxin treatment, while 0, 4, 20, and 100 nM digitoxin treatment (0 nM as the CTL group) for 24 h also resulted in an increase in the number of cells at G2/M phase from 23.64 to 41.40%, indicating that digitoxin induced G2/M cell cycle arrest in HepG2/ADM cells (Fig. 2A and B). CDK1 and cyclin B1, two key regulators of G2/M transition, have been found to be involved in modulating the cell cycle by forming the CDK1/Cyclin B1 complex (26). Digitoxin treatment caused significant downregulation of p-CDK1 (Thr14) and accumulation of cyclin B1 compared with the CTL group, which further confirms that digitoxin blocked the HepG2/ADM cell cycle at G2/M phase (Fig. 2C and D).
Figure 2.
Digitoxin blocks HepG2/ADM cells in the G2/M phase of the cell cycle. (A) HepG2/ADM cells were treated with different concentrations of digitoxin (0, 4, 20, 100 nM) for 24 h or 20 nM of digitoxin for 0, 12, 24 and 36 h, then the cell cycle distributions were detected using flow cytometry. The cell population in the G2/M phase was increased following digitoxin treatment. (B) The cell population in the G2/M phase was quantified using Prism. Each column represents the mean ± SEM (n=3). *P<0.05, ***P<0.001 vs. the control group. (C) HepG2/ADM cells were treated with or without digitoxin (4 and 20 nM) for 24 h, and the protein expression levels of CDK1, p-CDK1 (Thr14) and cyclin B1 were measured using western blot analysis. β-actin served as the loading control. Digitoxin-induced G2/M phase arrest was associated with CDK1 and cyclin B1. (D) Quantitative analysis of the relative protein expression. Data are presented as the mean ± SEM. *P<0.05, **P<0.01, ***P<0.001 vs. the control group. CTL, control; CDK1, cyclin-dependent kinase 1; p, phosphorylated.
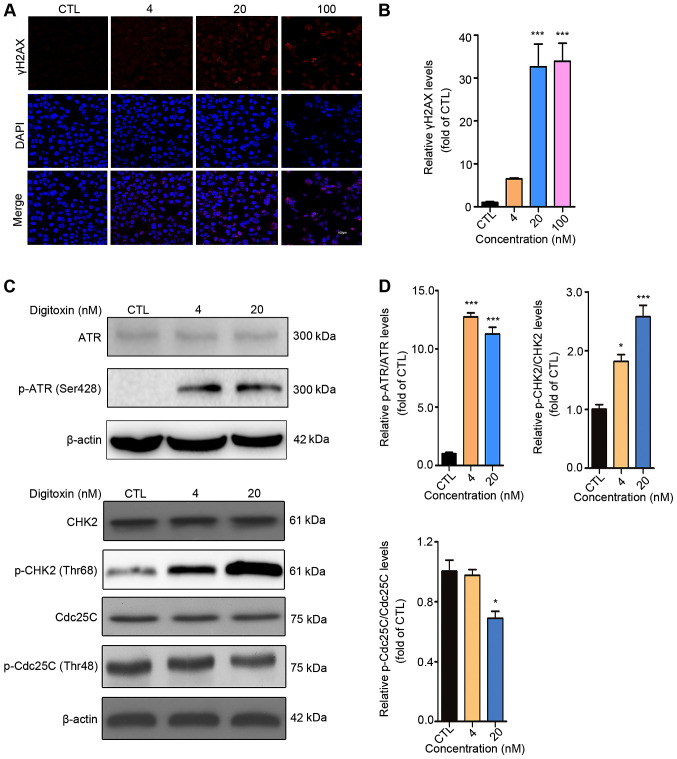
It is well-known that cell cycle arrest may result from DNA damage, in which the serine-protein kinase (ATM)/ATR-CHK1/CHK2-CDC25C signaling pathway plays significant role (27). To determine whether DNA lesions were responsible for the G2/M cell cycle arrest induced by digitoxin, an immunofluorescent staining assay was performed to detect the expression level of γH2AX, a marker of DNA double-stranded break (DSB) (28). The numbers of punctuate γH2AX foci significantly increased in a dose-dependent manner following digitoxin treatment compared with the CTL group. The highest accumulation of γH2AX foci was detected following treatment with 100 nM digitoxin (Fig. 3A and B). ATM and ATR are pivotal sensors of the DNA damage response pathway (29) and regulate the cell cycle partly through activating cell cycle checkpoint kinases CHK1 and CHK2 (30). Active CHK1 and CHK2 decrease the activity of CDC25C, thus inhibiting the dephosphorylation of CDK1 to maintain the inactive status of the CDK1-cyclin B1 complex (31,32). Digitoxin significantly increased the protein expression levels of p-CHK2 (Thr68) in a dose-dependent manner as well as p-ATR (Ser428) compared with the CTL group. In addition, digitoxin inhibited the phosphorylation of CDC25C (Fig. 3C and D). Taken together, these results indicated that digitoxin induced G2/M phase arrest via the ATR-Chk2-Cdc25C signaling pathway following DNA damage.
Figure 3.
Digitoxin induces G2/M phase arrest via the ATR-CHK2-CDC25C signaling pathway. (A) Digitoxin increased the expression level of γH2AX. HepG2/ADM cells were treated with or without digitoxin (4, 20 and 100 nM) for 24 h, then, the expression level of γH2AX was measured using an immunofluorescence assay. Images were obtained at a magnification of ×200. (B) γH2AX fluorescence intensity was subsequently quantified. Data are shown as the mean ± SEM (n=3). ***P<0.001 vs. the control group. (C) The effect of digitoxin on the ATR-CHK2-CDC25C signaling pathway. HepG2/ADM cells were treated with or without digitoxin (4 and 20 nM) for 24 h, then western blot analysis was used to detect the protein expression levels of ATR, p-ATR (Ser428), CHK2, p-CHK2 (Thr68), CDC25C, p-CDC25C (Thr48). β-actin was used as the loading control. (D) The relative protein expression was quantified. Data are presented as the mean ± SEM (n=3). *P<0.05, ***P<0.001 vs. the control group. γH2AX, p-histone H2AX; ATR, serine/threonine-protein kinase ATR; CHK2, serine/threonine-protein kinase chk2; CDC25C, M-phase inducer phosphatase 3; p, phosphorylated; CTL, control.
Digitoxin induces HepG2/ADM cell apoptosis through the mitochondrial apoptotic pathway
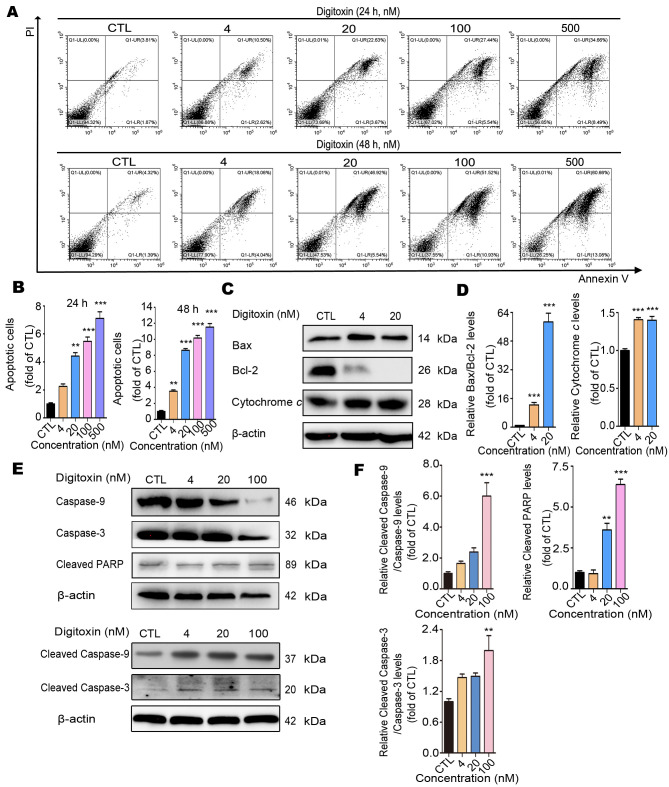
To investigate the underlying mechanism of digitoxin-induced HepG2/ADM cell death, the Annexin V-FITC/PI staining flow cytometry assay was performed to determine whether digitoxin induced apoptosis in HepG2/ADM cells. Different concentrations (CTL, 4, 20, 100 and 500 nM) of digitoxin treatment resulted in a significant augmentation of apoptotic cells (Fig. 4A and B). The apoptotic ratio of HepG2/ADM cells increased by approximately 7-fold from 5.68 to 43.15% for 24 h and 13-fold from 5.71 to 73.74% for 48 h. In addition, the ratio of cleaved caspase-3/caspase-3 and cleaved caspase-9/caspase-9 increased following different concentrations of digitoxin in a dose-dependent manner, and the level of cleaved PARP also increased, confirming that the apoptosis-related genes in the caspase family were activated (Fig. 4E and F). Mitochondrial apoptosis is regarded as the most common mode of cell apoptosis, which is characterized by the release of cytochrome c and changes in the interaction between Bcl-2 (apoptosis inhibitor) and Bax (apoptosis promotor) (33,34). As expected, digitoxin slightly increased the protein expression level of Bax and notably decreased the expression level of Bcl-2. As a consequence, the value of Bax/Bcl-2 increased approximately 58-fold by 20 nM digitoxin treatment compared with the CTL group. Since the level of Bcl-2 after 20 nM digitoxin treatment is very low, higher concentrations of digitoxin were not used in this assay. In addition, digitoxin treatment increased the protein expression level of cytochrome c (Fig. 4C and D). Taken together, these results indicated that digitoxin induced HepG2/ADM cell apoptosis through the mitochondrial apoptotic pathway.
Figure 4.
Digitoxin induces HepG2/ADM cell apoptosis via the mitochondrial apoptotic pathway. (A) Following treatment with different concentrations (0, 4, 20, 100 and 500 nM) of digitoxin for 24 or 48 h, the apoptotic ratio of HepG2/ADM cells was determined using the Annexin V-FITC/PI staining assay. Digitoxin induced HepG2/ADM cell apoptosis. (B) The data of apoptotic cells was quantified and illustrated as the mean ± SEM (n=3). **P<0.01, ***P<0.001 vs. the control group. (C) The effect of digitoxin on the protein expression levels of mitochondrial apoptosis-related proteins. Following treatment with or without digitoxin (4 and 20 nM) for 24 h, the expression levels of Bax, Bcl-2 and cytochrome c were determined using western blot analysis. β-Actin was used as the loading control. (D) The relative protein expression levels were quantified, and the data are presented as the mean ± SEM (n=3). ***P<0.001 vs. the control group. (E) The effect of digitoxin on the expression levels of the apoptosis-related proteins. Following treatment with or without digitoxin (4, 20 and 100 nM) for 24 h, the expression levels of caspase-3 and −9, cleaved-caspase-3, and −9, and cleaved PARP were detected using western blot analysis. β-actin was used as the loading control. (F) The relative protein expression levels were quantified, and the data are presented as the mean ± SEM (n=3). **P<0.01, ***P<0.001 vs. the control group. PARP, cleaved poly (ADP-ribose) polymerase; PI, propidium iodide; CTL, control group.
Discussion
Resistance to chemotherapy is the primary problem for effective treatment in patients with liver cancer (35). A high amount of research has been performed to overcome the problem of multidrug resistance (36–38); however, no important breakthroughs have been achieved (39). Thus, there remains an urgent requirement to identify novel anticancer agents that are effective against chemotherapy-resistant tumors. In the present study, a Dox-resistant HCC cell line, HepG2/ADM was used to screen a library of 78 natural compounds, in which digitoxin was selected due to its effective anti-HCC action on HepG2/ADM cells. In addition, the molecular and/or cellular mechanisms of the apoptotic effect of digitoxin on HepG2/ADM cells were also investigated. The findings of the present study demonstrated that digitoxin induced G2/M cell cycle arrest via the ATR-CHK2-CDC25C signaling pathway, which may result from DNA DSB. Furthermore, digitoxin was found to induce mitochondrial apoptosis in HepG2/ADM cells. The notable findings of the present study indicate that digitoxin could be a potential novel anti-HCC drug, particularly in chemotherapy-resistant HCC.
Digitoxin, a cardenolide, is an inhibitor of Na+/K+- ATPase (16). A number of studies have revealed the anticancer activities of digitoxin against various human cancer cell lines, including hematological, solid, drug-sensitive and/or drug-resistant cancer cells in vitro and in vivo (21,40–42). In liver cancer, a previous study found that the combination of sorafenib and digitoxin significantly inhibited primary HepG2 cell growth, which was potentially through suppression of ERK and hypoxia signaling (43). To the best of our knowledge, no study has addressed the effect of digitoxin on Dox-resistant HepG2/ADM cells. The present study performed purely in vitro work. In vivo studies are required to investigate digitoxin efficacy in detail, and whether ERK and hypoxia signaling pathways play a role in digitoxin-induced HepG2/ADM cell death also requires further investigation.
Numerous studies performed by different laboratories have demonstrated that digitoxin induces G2/M cell cycle arrest in several human cancer cell lines, including the KG1a acute myelogenous leukemia cell line and the K562 chronic myelogenous leukemia cell line (41), 786-O and A498 renal cell carcinoma cell lines (44), and the non-small cell lung cancer cell lines, NCI-H460 (45) and H1975 (17). Previous mechanistic studies have revealed that the downregulation of the cyclin B1/CDK1 complex, and the protein expression levels of CHK1/2 and p53, with the decrease in the protein levels of E3 ubiquitin-protein ligase CCNB1IP1, cyclin-A1, p21, p27, c-Myc and p-5′AMP-activated protein kinase catalytic subunit α-2 have been associated with digitoxin-induced G2/M cell cycle arrest (17,45). The findings of the present study demonstrated cell cycle arrest at the G2/M phase in HepG2/ADM cells following digitoxin treatment; however, reduced level of p-CDK1 (Thr14) and accumulation of cyclin B1 caused by digitoxin were detected in the present study (Fig. 2C and D), which may promote cell cycle progression according to the majority of studies (27,46). In eukaryotic cells, the expression level of cyclin B1 is very low in the G1 phase, and significantly increases during the S phase and peaks at the late G2 phase and early mitosis. When cells enter late mitosis, the expression level of cyclin B1 was found to be significantly decreased (47–51). Therefore, in the present study the increase of cyclin B1 further confirms that a higher proportion of HepG2/ADM cells were in the G2/M phase following digitoxin treatment. With respect to decreasing level of p-CDK1 (Thr14), some signaling pathways, which are activated to regulate cell cycle events such as P21 and P53 (52,53) may be responsible for this phenomenon. Similar results have been reported in other studies. Lee et al (54) demonstrated that p-CDK1 dephosphorylation at Tyr15 and the upregulation of cyclin B1 expression level were detected in 2-methoxyestradiol-induced G2/M arrest in Jurkat T cells. Mak et al (55) reported that small-molecule inhibitors of CHK1 (AZD7762) or WEE1 (MK-1775) induced mitotic arrest, as characterized by the dephosphorylation of p-CDK1 (Tyr15) in HeLa cells. Rong et al (56) also found that p-CDK1 was dephosphorylated at Thr161 following gambogic acid-induced DNA damage and G2/M arrest in HepG2 and A549 cells. However, the reasons for these different effects of digitoxin are complex and require further investigation.
A number of small molecules can arrest the cell cycle at G1/S or S phase to prevent incorrect DNA replication or at G2/M phase to prevent entry into mitosis with damaged DNA (57). The present study found that digitoxin impeded cell cycle progression at the G2/M phase, suggesting that digitoxin may not block DNA replication but induce DNA damage instead. In addition, the activation of the DNA damage response ATR-CHK2-CDC25C pathway and the increase of γH2AX (Ser139) were also found, which confirmed that the molecular mechanism of modulating the cell cycle by digitoxin was induction of DNA damage.
Previous studies have demonstrated that Na+/K+-ATPase was considered as a potential target for cardenolides to combat some cancers (58,59). The protein expression levels of Na+/K+-ATPase in tumor tissues, such as HCC, renal carcinoma cells, non-small cell lung carcinoma, colon carcinoma, prostate carcinoma, and glioma was higher compared with that in normal tissues (60,61). In addition, the colocalization of Na+/K+-ATPase and caveolin on the plasma membrane induced by the knockdown of apolipoprotein E increased the sensitivity of Hep3B cells towards cardenolides, confirming the role of Na+/K+-ATPase in the cytotoxicity of cardenolides (20). These findings demonstrate that the anticancer effects of cardenolides were associated with Na+/K+-ATPase. However, to the best of our knowledge there is a shortage of literature demonstrating that digitoxin-induced cell cycle arrest was attributed to the inhibition of Na+/K+-ATPase. Therefore, the exact molecular mechanism of how digitoxin induces DNA DSB, as well as whether Na+/K+-ATPase inhibition was responsible for apoptotic effect of digitoxin in HepG2/ADM cells requires further investigation. Furthermore, in vivo effects and cardiotoxicity of digitoxin also require further investigation.
In conclusion, our study demonstrated that digitoxin displays an anti-HCC effect on HepG2/ADM cells through ATR-CHK2-CDC25C-mediated G2/M cell cycle arrest and Bax/Bcl-2-mediated mitochondrial apoptosis, making digitoxin a promising chemotherapeutic agent for the treatment of patients with HCC.
Acknowledgements
The authors would like to thank Professor Dong-Mei Zhang (College of Pharmacy, Jinan University) and Dr Jun-Shan Liu (Traditional Chinese Medicine, Southern Medical University) for their guidance in the design of the present study.
Funding
This study was supported by the National Natural Science Foundation of China (grant nos. 81803790 and 81703975), National Natural Science Foundation of Guangdong (grant no. 2020A1515011090), Project of Administration of Traditional Chinese Medicine of Guangdong Province of China (grant no. 20181069) and the Fundamental Research Funds for the Central Universities (grant no. 21618336).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Authors' contributions
LJD and FFX designed the study and revised the manuscript for important intellectual content. YHL and HG performed the experiments and drafted the manuscript. YQH, JYZ, HZ, MSW, XJL and QYM made contributions to analysis and interpretation of data. LC and AYS performed the flow cytometry experiments and analyze the data. JW, YXL, EXZ and YYC assisted with the revision of the manuscript and performed experiments to update the data. All authors have read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Altekruse SF, Henley SJ, Cucinelli JE, McGlynn KA. Changing hepatocellular carcinoma incidence and liver cancer mortality rates in the United States. Am J Gastroenterol. 2014;109:542–553. doi: 10.1038/ajg.2014.11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ma J, Siegel RL, Islami F, Jemal A. Temporal trends in liver cancer mortality by educational attainment in the United States, 2000–2015. Cancer. 2019;125:2089–2098. doi: 10.1002/cncr.32023. [DOI] [PubMed] [Google Scholar]
- 3.Anwanwan D, Singh SK, Singh S, Saikam V, Singh R. Challenges in liver cancer and possible treatment approaches. Biochim Biophys Acta Rev Cancer. 2020;1873:188314. doi: 10.1016/j.bbcan.2019.188314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Yeo W, Mok TS, Zee B, Leung TW, Lai PB, Lau WY, Koh J, Mo FK, Yu SC, Chan AT, et al. A randomized phase III study of doxorubicin versus cisplatin/interferon alpha-2b/doxorubicin/fluorouracil (PIAF) combination chemotherapy for unresectable hepatocellular carcinoma. J Natl Cancer Inst. 2005;97:1532–1538. doi: 10.1093/jnci/dji315. [DOI] [PubMed] [Google Scholar]
- 5.Bruix J, Reig M, Sherman M. Evidence-based diagnosis, staging, and treatment of patients with hepatocellular carcinoma. Gastroenterology. 2016;150:835–853. doi: 10.1053/j.gastro.2015.12.041. [DOI] [PubMed] [Google Scholar]
- 6.Zhu AX, Kudo M, Assenat E, Cattan S, Kang YK, Lim HY, Poon RT, Blanc JF, Vogel A, Chen CL, et al. Effect of everolimus on survival in advanced hepatocellular carcinoma after failure of sorafenib: The EVOLVE-1 randomized clinical trial. JAMA. 2014;312:57–67. doi: 10.1001/jama.2014.7189. [DOI] [PubMed] [Google Scholar]
- 7.Holohan C, Van Schaeybroeck S, Longley DB, Johnston PG. Cancer drug resistance: An evolving paradigm. Nat Rev Cancer. 2013;13:714–726. doi: 10.1038/nrc3599. [DOI] [PubMed] [Google Scholar]
- 8.Mansoori B, Mohammadi A, Davudian S, Shirjang S, Baradaran B. The different mechanisms of cancer drug resistance: A brief review. Adv Pharm Bull. 2017;7:339–348. doi: 10.15171/apb.2017.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rajabalian S. Methanolic extract of Teucrium polium L. potentiates the cytotoxic and apoptotic effects of anticancer drugs of vincristine, vinblastine and doxorubicin against a panel of cancerous cell lines. Exp Oncol. 2008;30:133–138. [PubMed] [Google Scholar]
- 10.Khanna C, Rosenberg M, Vail DM. A review of paclitaxel and novel formulations including those suitable for use in dogs. J Vet Intern Med. 2015;29:1006–1012. doi: 10.1111/jvim.12596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mukherjee AK, Basu S, Sarkar N, Ghosh AC. Advances in cancer therapy with plant based natural products. Curr Med Chem. 2001;8:1467–1486. doi: 10.2174/0929867013372094. [DOI] [PubMed] [Google Scholar]
- 12.Gokduman K. Strategies targeting DNA Topoisomerase I in cancer chemotherapy: Camptothecins, nanocarriers for camptothecins, organic non-camptothecin compounds and metal complexes. Curr Drug Targets. 2016;17:1928–1939. doi: 10.2174/1389450117666160502151707. [DOI] [PubMed] [Google Scholar]
- 13.Fu XY, Zhang S, Wang K, Yang MF, Fan CD, Sun BL. Caudatin inhibits human glioma cells growth through triggering DNA damage-mediated cell cycle arrest. Cell Mol Neurobiol. 2015;35:953–959. doi: 10.1007/s10571-015-0190-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Li Y, Xiong H, Yang DQ. Functional switching of ATM: Sensor of DNA damage in proliferating cells and mediator of Akt survival signal in post-mitotic human neuron-like cells. Chin J Cancer. 2012;31:364–372. doi: 10.5732/cjc.012.10086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Trenti A, Zulato E, Pasqualini L, Indraccolo S, Bolego C, Trevisi L. Therapeutic concentrations of digitoxin inhibit endothelial focal adhesion kinase and angiogenesis induced by different growth factors. Br J Pharmacol. 2017;174:3094–3106. doi: 10.1111/bph.13944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Patel S. Plant-derived cardiac glycosides: Role in heart ailments and cancer management. Biomed Pharmacother. 2016;84:1036–1041. doi: 10.1016/j.biopha.2016.10.030. [DOI] [PubMed] [Google Scholar]
- 17.Zhang YZ, Chen X, Fan XX, He JX, Huang J, Xiao DK, Zhou YL, Zheng SY, Xu JH, Yao XJ, et al. Compound library screening identified cardiac glycoside digitoxin as an effective growth inhibitor of gefitinib-resistant non-small cell lung cancer via downregulation of α-tubulin and inhibition of microtubule formation. Molecules. 2016;21:374. doi: 10.3390/molecules21030374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lopez-Lazaro M. Digitoxin as an anticancer agent with selectivity for cancer cells: Possible mechanisms involved. Expert Opin Ther Targets. 2007;11:1043–1053. doi: 10.1517/14728222.11.8.1043. [DOI] [PubMed] [Google Scholar]
- 19.Lee DH, Lee CS, Kim DW, Ae JE, Lee TH. Digitoxin sensitizes glioma cells to TRAIL-mediated apoptosis by upregulation of death receptor 5 and downregulation of survivin. Anticancer Drugs. 2014;25:44–52. doi: 10.1097/CAD.0000000000000015. [DOI] [PubMed] [Google Scholar]
- 20.Liu M, Feng LX, Sun P, Liu W, Mi T, Lei M, Wu W, Jiang B, Yang M, Hu L, et al. Knockdown of apolipoprotein E enhanced sensitivity of Hep3B cells to cardiac steroids via regulating Na+/K+-ATPase signalosome. Mol Cancer Ther. 2016;15:2955–2965. doi: 10.1158/1535-7163.MCT-15-0961. [DOI] [PubMed] [Google Scholar]
- 21.Pollard BS, Suckow MA, Wolter WR, Starr JM, Eidelman O, Dalgard CL, Kumar P, Battacharyya S, Srivastava M, Biswas R, et al. Digitoxin inhibits epithelial-to-mesenchymal-transition in hereditary castration resistant prostate cancer. Front Oncol. 2019;9:630. doi: 10.3389/fonc.2019.00630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lopez-Lazaro M, Pastor N, Azrak SS, Ayuso MJ, Austin CA, Cortes F. Digitoxin inhibits the growth of cancer cell lines at concentrations commonly found in cardiac patients. J Nat Prod. 2005;68:1642–1645. doi: 10.1021/np050226l. [DOI] [PubMed] [Google Scholar]
- 23.Yang QF, Dalgard CL, Eidelman O, Jozwik C, Pollard BS, Srivastava M, Pollard HB. Digitoxin induces apoptosis in cancer cells by inhibiting nuclear factor of activated T-cells-driven c-MYC expression. J Carcinog. 2013;12:8. doi: 10.4103/1477-3163.112268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lee DH, Cheul Oh S, Giles AJ, Jung J, Gilbert MR, Park DM. Cardiac glycosides suppress the maintenance of stemness and malignancy via inhibiting HIF-1α in human glioma stem cells. Oncotarget. 2017;8:40233–40245. doi: 10.18632/oncotarget.16714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yao N, Wang C, Hu N, Li Y, Liu M, Lei Y, Chen M, Chen L, Chen C, Lan P, et al. Inhibition of PINK1/Parkin-dependent mitophagy sensitizes multidrug-resistant cancer cells to B5G1, a new betulinic acid analog. Cell Death Dis. 2019;10:232. doi: 10.1038/s41419-019-1470-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Paruthiyil S, Cvoro A, Tagliaferri M, Cohen I, Shtivelman E, Leitman DC. Estrogen receptor β causes a G2 cell cycle arrest by inhibiting CDK1 activity through the regulation of cyclin B1, GADD45A, and BTG2. Breast Cancer Res Treat. 2011;129:777–784. doi: 10.1007/s10549-010-1273-5. [DOI] [PubMed] [Google Scholar]
- 27.Deng LJ, Peng QL, Wang LH, Xu J, Liu JS, Li YJ, Zhuo ZJ, Bai LL, Hu LP, Chen WM, et al. Arenobufagin intercalates with DNA leading to G2 cell cycle arrest via ATM/ATR pathway. Oncotarget. 2015;6:34258–34275. doi: 10.18632/oncotarget.5545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Löbrich M, Shibata A, Beucher A, Fisher A, Ensminger M, Goodarzi AA, Barton O, Jeggo PA. gammaH2AX foci analysis for monitoring DNA double-strand break repair: Strengths, limitations and optimization. Cell Cycle. 2010;9:662–669. doi: 10.4161/cc.9.4.10764. [DOI] [PubMed] [Google Scholar]
- 29.Durocher D, Jackson SP. DNA-PK, ATM and ATR as sensors of DNA damage: Variations on a theme? Curr Opin Cell Biol. 2001;13:225–231. doi: 10.1016/S0955-0674(00)00201-5. [DOI] [PubMed] [Google Scholar]
- 30.Smith J, Tho LM, Xu N, Gillespie DA. The ATM-Chk2 and ATR-Chk1 pathways in DNA damage signaling and cancer. Adv Cancer Res. 2010;108:73–112. doi: 10.1016/B978-0-12-380888-2.00003-0. [DOI] [PubMed] [Google Scholar]
- 31.Kastan MB, Bartek J. Cell-cycle checkpoints and cancer. Nature. 2004;432:316–323. doi: 10.1038/nature03097. [DOI] [PubMed] [Google Scholar]
- 32.Zhao H, Piwnica-Worms H. ATR-mediated checkpoint pathways regulate phosphorylation and activation of human Chk1. Mol Cell Biol. 2001;21:4129–4139. doi: 10.1128/MCB.21.13.4129-4139.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kroemer G, Galluzzi L, Brenner C. Mitochondrial membrane permeabilization in cell death. Physiol Rev. 2007;87:99–163. doi: 10.1152/physrev.00013.2006. [DOI] [PubMed] [Google Scholar]
- 34.Deng LJ, Hu LP, Peng QL, Yang XL, Bai LL, Yiu A, Li Y, Tian HY, Ye WC, Zhang DM. Hellebrigenin induces cell cycle arrest and apoptosis in human hepatocellular carcinoma HepG2 cells through inhibition of Akt. Chem Biol Interact. 2014;219:184–194. doi: 10.1016/j.cbi.2014.06.003. [DOI] [PubMed] [Google Scholar]
- 35.Zhu YJ, Zheng B, Wang HY, Chen L. New knowledge of the mechanisms of sorafenib resistance in liver cancer. Acta Pharmacol Sin. 2017;38:614–622. doi: 10.1038/aps.2017.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ceballos MP, Rigalli JP, Cere LI, Semeniuk M, Catania VA, Ruiz ML. ABC Transporters: Regulation and association with multidrug resistance in hepatocellular carcinoma and colorectal carcinoma. Curr Med Chem. 2019;26:1224–1250. doi: 10.2174/0929867325666180105103637. [DOI] [PubMed] [Google Scholar]
- 37.Sun W, Chen X, Xie C, Wang Y, Lin L, Zhu K, Shuai X. Co-Delivery of doxorubicin and Anti-BCL-2 siRNA by pH-responsive polymeric vector to overcome drug resistance in in vitro and in vivo HepG2 hepatoma model. Biomacromolecules. 2018;19:2248–2256. doi: 10.1021/acs.biomac.8b00272. [DOI] [PubMed] [Google Scholar]
- 38.Gao DY, Lin Ts T, Sung YC, Liu YC, Chiang WH, Chang CC, Liu JY, Chen Y. CXCR4-targeted lipid-coated PLGA nanoparticles deliver sorafenib and overcome acquired drug resistance in liver cancer. Biomaterials. 2015;67:194–203. doi: 10.1016/j.biomaterials.2015.07.035. [DOI] [PubMed] [Google Scholar]
- 39.Marin JJG, Briz O, Herraez E, Lozano E, Asensio M, Di Giacomo S, Romero MR, Osorio-Padilla LM, Santos-Llamas AI, et al. Molecular bases of the poor response of liver cancer to chemotherapy. Clin Res Hepatol Gastroenterol. 2018;42:182–192. doi: 10.1016/j.clinre.2017.12.006. [DOI] [PubMed] [Google Scholar]
- 40.Prassas I, Karagiannis GS, Batruch I, Dimitromanolakis A, Datti A, Diamandis EP. Digitoxin-induced cytotoxicity in cancer cells is mediated through distinct kinase and interferon signaling networks. Mol Cancer Ther. 2011;10:2083–2093. doi: 10.1158/1535-7163.MCT-11-0421. [DOI] [PubMed] [Google Scholar]
- 41.Feng Q, Leong WS, Liu L, Chan WI. Peruvoside, a cardiac glycoside, induces primitive myeloid leukemia Cell Death. Molecules. 2016;21:534. doi: 10.3390/molecules21040534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Felth J, Rickardson L, Rosén J, Wickström M, Fryknäs M, Lindskog M, Bohlin L, Gullbo J. Cytotoxic effects of cardiac glycosides in colon cancer cells, alone and in combination with standard chemotherapeutic drugs. J Nat Prod. 2009;72:1969–1974. doi: 10.1021/np900210m. [DOI] [PubMed] [Google Scholar]
- 43.Xiao Y, Yan W, Guo L, Meng C, Li B, Neves H, Chen PC, Li L, Huang Y, Kwok HF, Lin Y. Digitoxin synergizes with sorafenib to inhibit hepatocelluar carcinoma cell growth without inhibiting cell migration. Mol Med Rep. 2017;15:941–947. doi: 10.3892/mmr.2016.6096. [DOI] [PubMed] [Google Scholar]
- 44.Nolte E, Wach S, Silva IT, Lukat S, Ekici AB, Munkert J, Müller-Uri F, Kreis W, Oliveira Simões CM, Vera J, et al. A new semisynthetic cardenolide analog 3β-[2-(1-amantadine)- 1-on-ethylamine]-digitoxigenin (AMANTADIG) affects G2/M cell cycle arrest and miRNA expression profiles and enhances proapoptotic survivin-2B expression in renal cell carcinoma cell lines. Oncotarget. 2017;8:11676–11691. doi: 10.18632/oncotarget.14644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Elbaz HA, Stueckle TA, Wang HY, O'Doherty GA, Lowry DT, Sargent LM, Wang L, Dinu CZ, Rojanasakul Y. Digitoxin and a synthetic monosaccharide analog inhibit cell viability in lung cancer cells. Toxicol Appl Pharmacol. 2012;258:51–60. doi: 10.1016/j.taap.2011.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Liu JS, Huo CY, Cao HH, Fan CL, Hu JY, Deng LJ, Lu ZB, Yang HY, Yu LZ, Mo ZX, Yu ZL. Aloperine induces apoptosis and G2/M cell cycle arrest in hepatocellular carcinoma cells through the PI3K/Akt signaling pathway. Phytomedicine. 2019;61:152843. doi: 10.1016/j.phymed.2019.152843. [DOI] [PubMed] [Google Scholar]
- 47.Bloom J, Cross FR. Multiple levels of cyclin specificity in cell-cycle control. Nat Rev Mol Cell Biol. 2007;8:149–160. doi: 10.1038/nrm2105. [DOI] [PubMed] [Google Scholar]
- 48.Nurse P. A long twentieth century of the cell cycle and beyond. Cell. 2000;100:71–78. doi: 10.1016/S0092-8674(00)81684-0. [DOI] [PubMed] [Google Scholar]
- 49.Fisher DL, Nurse P. A single fission yeast mitotic cyclin B p34cdc2 kinase promotes both S-phase and mitosis in the absence of G1 cyclins. EMBO J. 1996;15:850–860. doi: 10.1002/j.1460-2075.1996.tb00420.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Hayles J, Fisher D, Woollard A, Nurse P. Temporal order of S phase and mitosis in fission yeast is determined by the state of the p34cdc2-mitotic B cyclin complex. Cell. 1994;78:813–822. doi: 10.1016/S0092-8674(94)90542-8. [DOI] [PubMed] [Google Scholar]
- 51.Gould KL, Nurse P. Tyrosine phosphorylation of the fission yeast cdc2+ protein kinase regulates entry into mitosis. Nature. 1989;342:39–45. doi: 10.1038/342039a0. [DOI] [PubMed] [Google Scholar]
- 52.He YC, He L, Khoshaba R, Lu FG, Cai C, Zhou FL, Liao DF, Cao D. Curcumin nicotinate selectively induces cancer cell apoptosis and cycle arrest through a p53-mediated mechanism. Molecules. 2019;24:4179. doi: 10.3390/molecules24224179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Żuryń A, Litwiniec A, Safiejko-Mroczka B, Klimaszewska- Wiśniewska A, Gagat M, Krajewski A, Gackowska L, Grzanka D. The effect of sulforaphane on the cell cycle, apoptosis and expression of cyclin D1 and p21 in the A549 non-small cell lung cancer cell line. Int J Oncol. 2016;48:2521–2533. doi: 10.3892/ijo.2016.3444. [DOI] [PubMed] [Google Scholar]
- 54.Lee ST, Lee JY, Han CR, Kim YH, Jun do Y, Taub D, Kim YH. Dependency of 2-methoxyestradiol-induced mitochondrial apoptosis on mitotic spindle network impairment and prometaphase arrest in human Jurkat T cells. Biochem Pharmacol. 2015;94:257–269. doi: 10.1016/j.bcp.2015.02.011. [DOI] [PubMed] [Google Scholar]
- 55.Mak JP, Man WY, Ma HT, Poon RY. Pharmacological targeting the ATR-CHK1-WEE1 axis involves balancing cell growth stimulation and apoptosis. Oncotarget. 2014;5:10546–10557. doi: 10.18632/oncotarget.2508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Rong JJ, Hu R, Song XM, Ha J, Lu N, Qi Q, Tao L, You QD, Guo QL. Gambogic acid triggers DNA damage signaling that induces p53/p21(Waf1/CIP1) activation through the ATR-Chk1 pathway. Cancer Lett. 2010;296:55–64. doi: 10.1016/j.canlet.2010.03.016. [DOI] [PubMed] [Google Scholar]
- 57.Laiho M, Latonen L. Cell cycle control, DNA damage checkpoints and cancer. Ann Med. 2003;35:391–397. doi: 10.1080/07853890310014605. [DOI] [PubMed] [Google Scholar]
- 58.Piacente S, Masullo M, De Néve N, Dewelle J, Hamed A, Kiss R, Mijatovic T. Cardenolides from Pergularia tomentosa display cytotoxic activity resulting from their potent inhibition of Na+/K+-ATPase. J Nat Prod. 2009;72:1087–1091. doi: 10.1021/np800810f. [DOI] [PubMed] [Google Scholar]
- 59.Mijatovic T, Dufrasne F, Kiss R. Cardiotonic steroids-mediated targeting of the Na(+)/K(+)-ATPase to combat chemoresistant cancers. Curr Med Chem. 2012;19:627–646. doi: 10.2174/092986712798992075. [DOI] [PubMed] [Google Scholar]
- 60.Xu ZW, Wang FM, Gao MJ, Chen XY, Hu WL, Xu RC. Targeting the Na(+)/K(+)-ATPase alpha1 subunit of hepatoma HepG2 cell line to induce apoptosis and cell cycle arresting. Biol Pharm Bull. 2010;33:743–751. doi: 10.1248/bpb.33.743. [DOI] [PubMed] [Google Scholar]
- 61.Mijatovic T, Dufrasne F, Kiss R. Na+/K+-ATPase and cancer. Pharm Pat Anal. 2012;1:91–106. doi: 10.4155/ppa.12.3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.