Fig. 1.
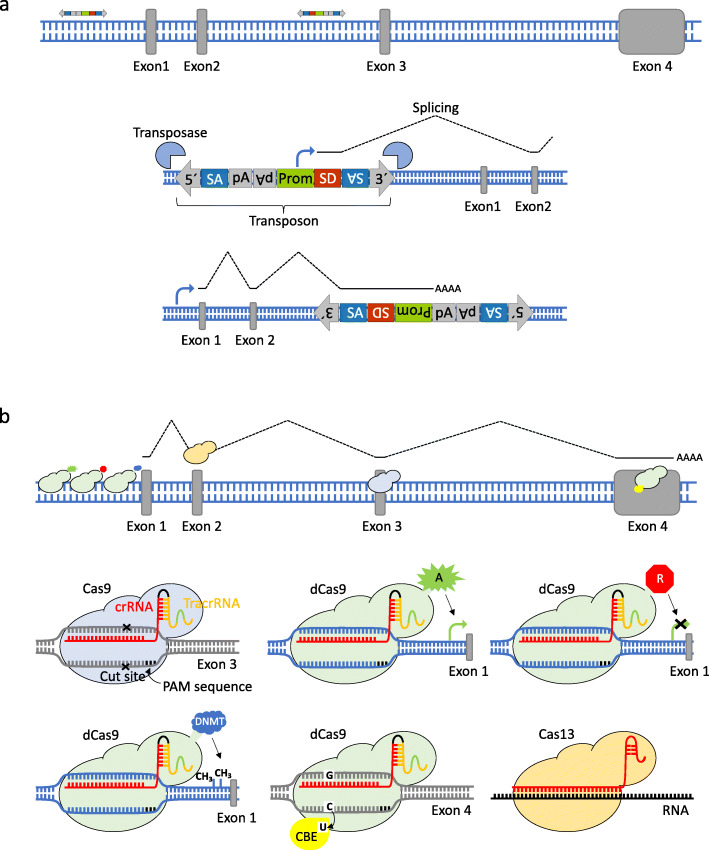
Transposon mutagenesis and CRISPR-induced genetic and epigenetic alterations. a Sleeping beauty (SB) and piggyBac (PB) transposons carry a promoter upstream of a splice donor (SD) sequence that can lead to overexpression of downstream genes. This is equivalent to activation of proto-oncogenes. Transposons also contain splice acceptors (SAs) followed by bidirectional polyadenylation (polyA) signals to cause termination of transcripts that arise when inserted into the body of a gene (primarily in introns). This is important for inactivating tumor suppressor genes (TSGs). b CRISPR-Cas9 is a two component system: the single guide RNA (sgRNA) directs the Cas9 nuclease to a complementary sequence in the genome where Cas9 will induce a double-strand break (DSB). The target genomic locus must be followed by a 5′-NGG-3′ motif (protospacer adjacent motif, PAM) for Cas9 to function. DSBs are resolved by non-homologous end-joining, or by homology directed repair (HDR) in the presence of a DNA repair template, which can be exploited to introduce precise genetic modifications or exogenous sequences. Other variants of the Cas nuclease have been engineered to function in mammalian cells, including a nuclease-deficient Cas9 (dCas9) fused to various effector domains: a transcriptional activator (A) for activation of gene expression, a transcriptional repressor (R) to knockdown gene expression [20], epigenetic effectors such as DNA methyltransferases (DNMT) for methylation, and base editors such as cytosine base editors (CBE) for generating point mutations