Abstract
Cisplatin (DDP) is a commonly used chemotherapy drug; however, the side effects associated with its use, particularly acute kidney injury (AKI), limit its clinical application. Puerarin is a natural flavonoid extracted from the Chinese medical herb Radix puerariae, which has been reported to alleviate DDP-induced nephrotoxicity. However, the mechanisms underlying puerarin regulation on microRNA (miR)-31-mediated signaling pathways in AKI remain unknown. Thus, the present study aimed to investigate the function of puerarin in a DDP-induced AKI rat model via reverse transcription-quantitative PCR and western blot analyses. The results demonstrated that DDP upregulated the levels of miR-31 in a concentration-dependent manner, both in vitro and in vivo. Furthermore, DDP significantly increased blood urea nitrogen and malondialdehyde content, serum creatinine and histopathological changes, while significantly decreasing the expression levels of superoxide dismutase, catalase and glutathione S-transferase in kidney tissues. TUNEL and western blot analyses indicated that DDP increased the expression levels of apoptotic proteins and affected the Numb/Notch1 signaling pathway, which is downstream of miR-31. The effects induced by DDP were counteracted following treatment with puerarin. Taken together, the results of the present study suggest that puerarin exhibits a renal protective effect against DDP-induced AKI by upregulating miR-31 expression and inhibiting the Numb/Notch1 signaling pathway.
Keywords: puerarin, acute kidney injury, microRNA-31, Numb/Notch1, cisplatin
Introduction
Cisplatin (DDP) is recognized as one of the most effective antineoplastic drugs and is extensively used in several types of cancer, such as lung, liver, ovarian, bladder and cervical cancers (1-5). However, the clinical application of cisplatin is limited due to its toxic side effects, particularly acute kidney injury (AKI) (6). Thus, development of novel adjuvant therapeutic strategies remains critical to alleviate nephrotoxicity induced by cisplatin. Puerarin is an isoflavonoid extracted from the Chinese medical herb Radix puerariae (R. puerariae), with a structure of 7-hydroxy-3-(4-hydroxyphenyl)-1-benzopyran-4-one-8-β-D-glucopyranoside (Fig. 1) (7). Puerarin has recently attracted interest for its extensive pharmacological properties in the treatment of kidney diseases, including AKI and chronic renal injury (8-11). Furthermore, it was suggested that puerarin exerted a protective effect against DDP-induced nephrotoxicity (12). However, the molecular mechanisms underlying renal protection of puerarin in DDP-induced AKI remain unclear.
Figure 1.
Chemical structure of puerarin.
MicroRNAs (miRNAs/miRs) play a regulatory role in biological processes, including inflammation, by negatively regulating several coding genes (13). miR-31 was demonstrated to be associated with the progress of renal cell carcinoma (RCC) (14). In addition, miR-31-5p was markedly downregulated in RCC tissues compared with paired adjacent normal tissues (15). miR-31 was also found to directly target the 3'-untranslated region (UTR) sequence of the Numb gene (16). Furthermore, miR-31-5p was reported to promote the proliferation of colorectal cancer cells by targeting the Numb protein (17). Numb is considered an intrinsic determinant in Drosophila, which alters the cell fate by negatively regulating the Notch signaling pathway (18). The depletion of Numb can promote dynamin-related protein 1-mediated mitochondrial fission and exacerbate mitochondrial dysfunction and tubular cell apoptosis in DDP-induced AKI (19). Numb has also been demonstrated to alleviate puromycin aminonucleoside-induced renal proximal tubular cell apoptosis by suppressing the Notch signaling pathway (20). The present study aimed to investigate whether puerarin exerts its renoprotective function via the miR-31-mediated Numb/Notch1 signaling pathway, using a DDP-induced AKI rat model.
Materials and methods
Reagents and chemicals
Puerarin was obtained from Zhejiang Conba Pharmaceutical Co., Ltd. DDP was purchased from Qilu Pharmaceutical Co., Ltd. Other chemicals were all purchased from Beijing Solarbio Science & Technology Co., Ltd.
Cells and animals
The human renal proximal tubular epithelial (HK-2) cell line was provided by the Central aboratory, the Second Hospital of Shandong University. HK-2 cells were cultured in DMEM (4.5 g/l glucose) with 10% FBS and 1% penicillin-streptomycin at 37˚C in 5% CO2. HK-2 cells were treated with 40 µM DDP at different timepoints (0, 12, 24 and 48 h) or 100 µM puerarin.
Female Sprague-Dawley rats (age 6-8 weeks; weight 180-220 g) were purchased from the Division of Animals of Peking University (China) and housed in specific-pathogen free conditions at the animal center of the Second Hospital of Shandong University, China. The use of rats was approved by the Institutional Care and Use Committee of the Second Hospital of Shandong University [permit no. KYLL-2017 (GJ) A-0028]. The rats were disposed in compliance with the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health.
Experimental design
A total of 18 rats were randomly divided into three groups (21,22): i) Sham control group (Sham, n=6), which received intraperitoneal (i.p.) injection of 2 ml saline and intravenous (i.v.) injection of 0.1 ml saline every other day over the course of 14 days; ii) DDP group (n=6), which received i.p. injection of 20 mg/kg DDP on day 1 and iii) puerarin + DDP (Pue+DDP group, n=6), which received i.p. injection of 20 mg/kg DDP on day 1 and with i.v. injection of puerarin (50 mg/kg) every other day over the course of 14 days.
The dosage regimen was implemented according to previous studies (2,12). The weights of each rat were recorded every day during the entire experimental period, and the kidneys were assessed in g per g of body weight. On day 14, the body and kidney weight of rats were recorded, and blood was collected before the rats sacrificed by spinal dislocation after intraperitoneal injection of 4% chloral hydrate (300 mg/kg) for ~10 min. Kidney samples were removed for western blotting.
Evaluation of renal function
Blood was collected into a centrifuge tube containing heparin and then centrifuged at 500 x g for 4 min at room temperature to obtain plasma samples. An automated blood analyzer (Dimension Xpand Plus; Siemens Healthineers) and standard diagnostic kits (urea nitrogen, cat. no. EIABUN; Invitrogen; Thermo Fisher Scientific, Inc. and creatinine, cat. no. C011-1-1; Nanjing Jiancheng Bioengeneering Institute) was used to determine blood urea nitrogen (BUN) and serum creatinine (SCR) content.
Hematoxylin and eosin (HE) and TUNEL examination
Kidneys were fixed in 10% buffered formalin overnight at 4˚C, embedded in paraffin, sectioned (4-6 µm thickness) and stained with HE at room temperature for 6 min. A light microscope (CKX41; Olympus Corporation) was used to detect histopathological results at x4 magnification. Kidney sections were also used to determine apoptosis. Apoptotic cells were identified using a TUNEL assay (In Situ Cell Death Detection kit; cat. no. 11684817910; Roche; Merck KGaA). TUNEL staining was performed according to the manufacturer's instructions. Apoptotic cells were monitored by dark brown nuclear staining and counted with 100 cells from six random microscopic fields under a fluorescent microscope (x10 magnification) in each group. The values were presented as the percentage of the total number of cells.
Assessment of oxidative stress
Tissue samples were homogenized with saline solution (0.9%) at 4˚C and centrifuged at 200 x g for 30 min at room temperature. The supernatants were collected for oxidative stress detection. The activities of superoxide dismutase (SOD; SOD assay kit; cat. no. A001-3-2; Nanjing Jiancheng Bioengineering Institute) and malondialdehyde (MDA; MDA assay kit; cat. no. A003-1-1; Nanjing Jiancheng Bioengineering Institute), and the contents of glutathione S-transferase (GS; GST Fluorometric Assay kit; cat. no. KT002; AmyJet Scientific) and catalase (CA; CAT assay kit; cat. no. A007-1-1; Nanjing Jiancheng Bioengineering Institute) in kidneys were determined according to the manufacturer's protocols.
Cell transfection
HK-2 cells were plated in 6-well plates at a density of 1x105 cells/well until 80 90% confluence and were transfected with miR-31 inhibitor and negative control (NC) (both, final concentration 100 nM) using Lipofectamine™ 2000 (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol. HK-2 cells transfected with miR-NC were used as controls. Briefly, following culture for 24-48 h, the expression of Numb and Notch-1 genes were detected using reverse transcription-quantitative PCR (RT-qPCR) analysis. After 6 h of incubation at 37˚C with 5% CO2, the approximate transfection efficiency was measured, where expression of miR-31 significantly decreased in miR-31 inhibitor-treated (12.52%) or both miR-31 inhibitor and DDP (18.12%), compared with sham (97.83%) or DDP treatment (85.45%), respectively (Fig. S1). The results were calculated using fluorescence-activated cell sorting (Nikon Eclipse C1; Nikon Corporation and ImageJ; National Institutes of Health). The cells were continued to be cultured after the medium was replaced once the transfection efficiency was calculated. Total RNA was extracted from the groups at 48 h after transfection.
RT-qPCR analysis
Total miRNA from HK-2 cells and kidneys were extracted using an miRNeasy mini kit (Qiagen GmbH, 217704) accordance with the manufacturer's protocol. cDNA was reverse transcribed from miRNA using a miScript II RT kit according to the manufacturer's protocol (Invitrogen; Thermo Fisher Scientific, Inc.) at 72˚C for 10 min, 42˚C for 1 h and 72˚C for 5 min. TaqMan fluorescent probe (cat. no. 4351379; Invitrogen; Thermo Fisher Scientific, Inc.) was used for the qPCR. The following thermocycling conditions were used for the qPCR: Initial denaturation at 95˚C for 2 min and 28 cycles of 95˚C for 1 min, 53˚C for 1 min and 72˚C for 5 min. The expression of levels Numb and Notch1were quantified using the 2-ΔΔCq method (23). U6 were used as internal reference genes to calculate the mRNA miRNA expression of the target genes, respectively. All experiments were performed three times. The following primer pairs were used for the qPCR: miRNA-31 forward, 5'-CAGCTATGCCAGCATCTTG CCT-3' and miRNA-31 reverse, 5'-ATATGGAACGCTTCAC GAATT-3'; Numb forward, 5'-TTTCGAAAGTGTTGGGATT ATATAC-3' and reverse, 5'-AACTACAATAAACCAAAATC GCG-3'; Notch1 forward, 5'-GCGAGGTCAACACAGAC GAG-3' and reverse, 5'-CAGGCACTTGGCACCATTC-3'; GAPDH forward, 5'-TTCACCACCATGGAGAAGGC-3' and reverse, 5'-GGCATGGACTGTGGTCATGA-3' and U6 forward, 5'-CTCGCTTCGGCAGCACA-3' and reverse, 5'-AACGCTTCACGAATTTGCGT-3'.
Western blotting
Kidney tissues were homogenized in ice-cold suspension buffer (BioTeke Corporation) and HK-2 cells were lysed with RIPA buffer (Thermo Fisher Scientific, Inc.). The samples were centrifuged at 500 x g for 20 min at 4˚C, and protein concentration was determined using a bicinchoninic acid protein assay kit (Thermo Fisher Scientific, Inc.). The proteins (30 µg/lane) were separated using 10% SDS-PAGE and then transferred onto PVDF membranes (EMD Millipore).
Milk (5% non-fat) was used to block the non-specific binding sites at room temperature for 2 h. Subsequently, the membranes were incubated with the following primary antibodies: Bcl-2 (cat. no. 3869s; 1:1,000; Cell Signaling Technology, Inc.), Bax (cat. no. 2772s; 1:1,000; Cell Signaling Technology, Inc.), caspase-3 (cat. no. 9654s; 1:1,000; Cell Signaling Technology, Inc.), Numb (cat. no. 2761s; 1:1,000; Cell Signaling Technology, Inc.), Notch1 (cat. no. 4830s; 1:1,000; Cell Signaling Technology, Inc.) and β-actin (cat. no. 4970s; 1:1,000; Cell Signaling Technology, Inc.) overnight at 4˚C. Protein signals were all normalized to β-actin expression.
The membranes were washed thrice with TBS-10% Tween-20 at room temperature and incubated with horseradish-peroxidase conjugated anti-mouse immunoglobulin G secondary antibody (cat. no. 7076S; 1:1,100; Cell Signaling Technology, Inc.) for 2 h at room temperature. Protein signals were detected using ECL reagent (Beijing Solarbio Science & Technology Co., Ltd.), and analyzed using Gel-Pro Analyzer densitometry software (version 6.3; Media Cybernetics, Inc.). Triplicate experiments with triplicate samples were performed.
Statistical analysis
SPSS/Win 15.0 software (SPSS Inc.) was used for data analysis. Data were expressed as the mean ± standard deviation. Differences were analyzed using one-way ANOVA and Student's t-test. Tukey's post hoc test was used for pairwise comparisons. P<0.05 was considered to indicate a statistically significant difference.
Results
Puerarin suppresses mi-31 expression in DDP-induced AKI
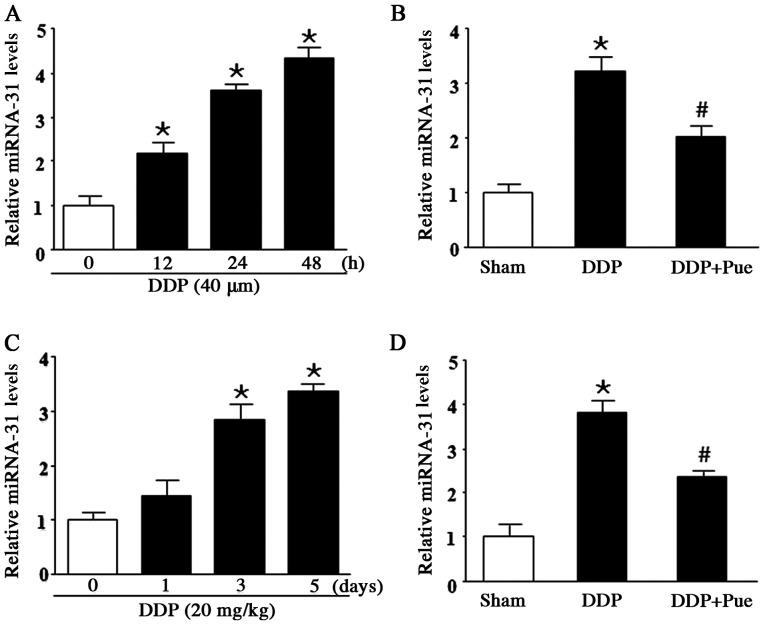
RT-qPCR was performed to determine miR-31 expression in DDP-treated HK-2 cells and SD rats. The results demonstrated that miR-31 significantly increased following treatment with DDP compared with the controls. Furthermore, treatment with 40 µM DDP for 12, 24 and 48 h increased miR-31 expression in a concentration-dependent manner (Fig. 2A). To determine the effect of puerarin on miR-31 expression in DDP-induced AKI, HK-2 cells were treated with 40 µM DDP and/or 100 µM puerarin. miR-31 expression was significantly upregulated following DDP treatment compared with controls. Furthermore, the combined treatment of DDP and puerarin significantly counteracted the increased expression levels of miR-31 expression (Fig. 2B). miR-31 expression levels were assessed in DDP-treated rats (20 mg/kg) on days 1, 3 and 5. The results demonstrated that miR-31 levels significantly increased on day 3 and 5 compared with day 0 (Fig. 2C). Conversely, combination treatment with 20 mg/kg DDP and 50 mg/kg puerarin downregulated miR-31 expression compared with DDP treatment alone (Fig. 2D).
Figure 2.
Effect of puerarin on DDP-induced miR-31 expression. (A) Treatment of HK-2 cells with 40 µM DDP for 12, 24 and 48 h increased miR-31 expression in a concentration-dependent manner. (B) Combined treatment of 40 µM DDP and 100 µM puerarin reversed the effects of DDP. (C) miR-31 expression levels significantly increased in DDP-treated rats (20 mg/kg) on days 3 and 5 while (D) combined treatment of 20 mg/kg DDP and 50 mg/kg puerarin downregulated miR-31 expression compared with DDP treatment alone. Data are presented as the mean ± standard deviation. n=6. *P<0.05 vs. sham. #P<0.05 vs. DDP. DDP, cisplatin; miR, miRNA; Pue, puerarin.
Puerarin improves DDP-induced renal injury
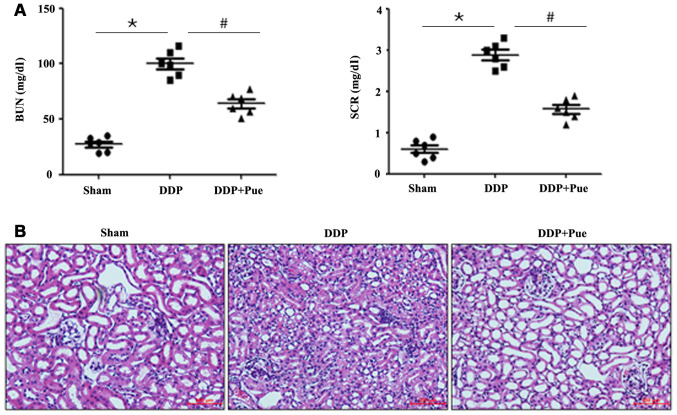
The contents of the kidney injury biomarkers (BUN) and (SCR) significantly increased in DDP-treated rats compared with controls (Fig. 3A). Conversely, combined treatment of puerarin and DDP significantly decreased BUN and SCR contents. Furthermore, a significant increase in the absolute and relative kidney weights were observed in the DDP-treated group compared with controls, whereas puerarin prevented the alterations compared with the DDP-treated group (Table I).
Figure 3.
Effect of puerarin on DDP-induced renal injury. (A) BUN and SCR contents increased in DDP-treated rats, the effects of which were reversed following treatment with puerarin. Data are presented as the mean ± standard deviation. n=6. (B) Histopathological staining was performed to detect renal changes. *P<0.05 vs. sham; #P<0.05 vs. DDP. BUN, blood urea nitrogen; SCR, serum creatinine; DDP, cisplatin; Pue, puerarin.
Table I.
Effect of DDP treatment (alone and with puerarin) on the body weight, kidney weight and kidney/body weight ratio in Sprague-Dawley rats.
| Group | Body weight (g) | Kidney weight (g) | Kidney/body weight ratio (%) |
|---|---|---|---|
| Sham | 219.5±6.12 | 1.98±0.09 | 0.91±0.12 |
| DDP | 185.2±5.83a | 2.79±0.11a | 1.51±0.21a |
| DDP+Puerarin | 200.5±3.39 | 2.25±0.13b | 1.13±0.18b |
n=6.
aP<0.05, DDP group vs. control group,
bP<0.05, DDP group vs. DDP+Puerarin group. DDP, cisplatin.
Consistent with functional assay results, HE-stained kidney sections from the control rats revealed integrated histomorphology (Fig. 3B). Treatment with DDP was associated with severe kidney injury, characterized by tubular injury, infiltration of inflammatory cells, interstitial hemorrhage and necrosis. However, combined treatment of puerarin and DDP relieved the histopathological alterations compared with DDP treatment alone. The results indicated that DDP treatment alone (20 mg/kg) was associated with distinct deterioration of renal function after 48 h.
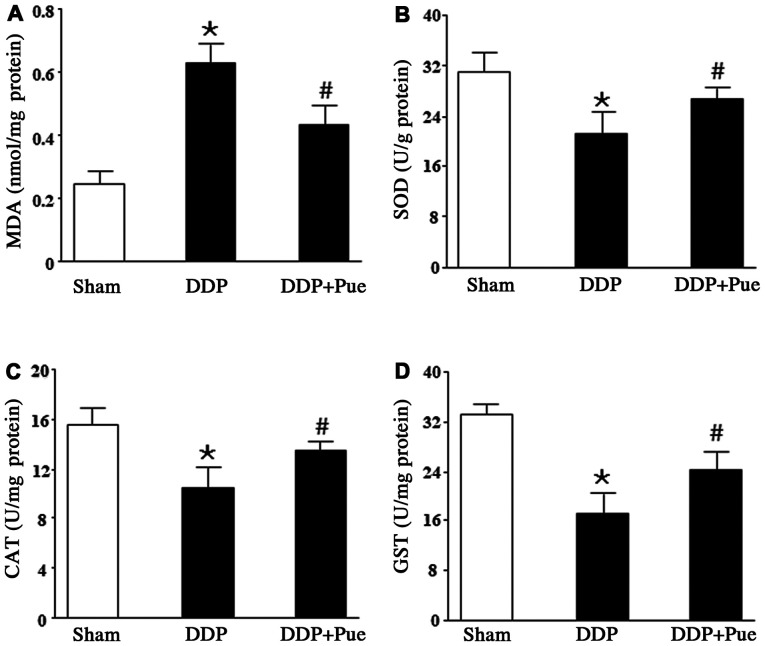
Puerarin reverses DDP-induced changes in renal antioxidants
The rats were treated with 20 mg/kg DDP, the dose which is able to induce AKI (2). As shown in Fig. 4A-D, treatment with DDP suppressed renal antioxidant defense, evidenced by significantly increased MDA content and significantly decreased expression levels of SOD, CAT and GST compared with the controls. Furthermore, combined treatment with puerarin significantly reversed the alterations in renal antioxidant activities compared with DDP treatment alone.
Figure 4.
Effect of puerarin on DDP-induced renal antioxidant alterations. (A) MDA content significantly increased following DDP treatment compared with DDP+Pue treatment. However, the activities of (B) SOD (C) CAT and (D) GST decreased in kidney tissues following DDP treatment but increased following DDP+Pue treatment. Data are presented as the mean ± standard deviation. n=6. *P<0.05 vs. sham; #P<0.05 vs. DDP. MDA, malondialdehyde; SOD, superoxide dismutase; CAT, catalase; GST, glutathione S-transferase; DDP, cisplatin; Pue, puerarin.
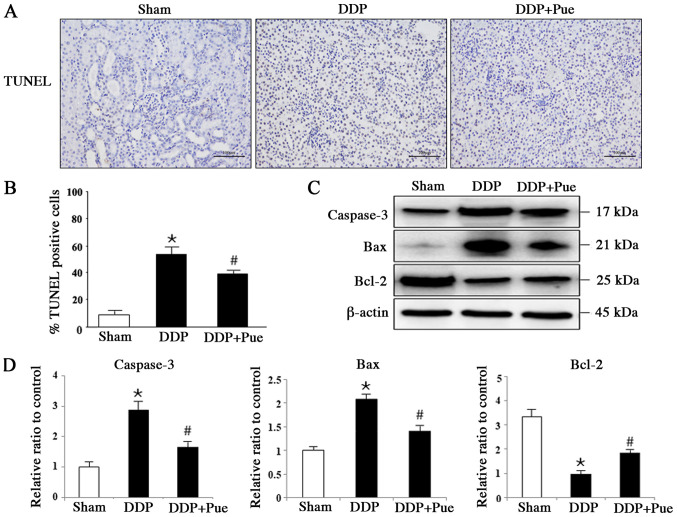
Puerarin suppresses DDP-induced apoptosis-associated protein expression
The apoptotic rates exhibited in TUNEL-stained sections from rats were detected and calculated based on six randomly selected fields (Fig. 5A and B). Treatment with DDP induced apoptosis in 56.98% of kidney cells compared with the Sham group, while combination treatment with puerarin and DDP induced apoptosis in 37.33% of cells compared with the DDP group (n=6). The expression levels of apoptosis-associated proteins in kidneys were determined via western blot analysis (Fig. 5C and D). The expression levels of cleaved caspase-3 and Bax significantly increased while Bcl-2 significantly decreased in DDP-treated rats compared with controls. Furthermore, combination treatment with puerarin was demonstrated to block DDP-induced activation of caspase-3 and Bax, and inactivation of Bcl-2 in kidney tissues.
Figure 5.
Effect of puerarin on DDP-induced expression of apoptosis-associated proteins. (A) TUNEL staining was performed to (C) determine the apoptotic rate. (B) Representative western blot image and (D) quantification of the expression levels of typical apoptotic proteins, including caspase-3, Bax and Bcl-2 were assessed in rats. Data are presented as the mean ± standard deviation. n=3. *P<0.05 vs. sham; #P<0.05 vs. DDP treatment. DDP, cisplatin; Pue, puerarin.
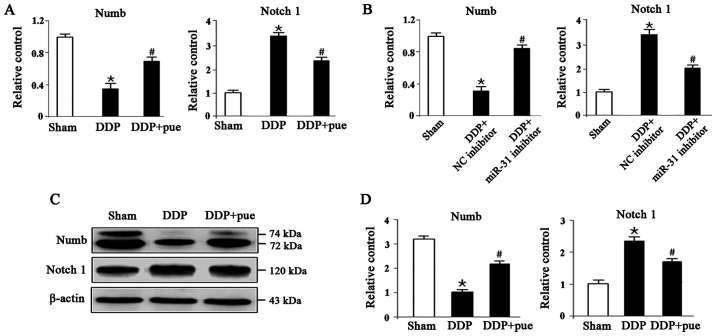
Puerarin protects against DDP-induced AKI by regulating the Numb/Notch1 signaling pathway
To determine the role of the miRNA-31-related Numb/Notch1 signaling pathway in DDP-induced AKI, the expression of Numb and Notch1 both at mRNA and protein levels we detected using RT-qPCR and western blotting, respectively, in HK-2 cells. As shown in Fig. 6A, compared with the controls, RNA levels of Numb and Notch1 significantly decreased and increased in DDP- treated cells, respectively, while the combination of DDP and puerarin showed opposite effects. The same was observed in Sham, DDP combined with NC or miR-31 inhibitor groups as shown in Fig. 6B. Fig. 6C shows the expression levels of Numb protein, which significantly decreased, while Notch1 expression increased in DDP-induced AKI compared with the controls. Furthermore, combination treatment with DDP and puerarin significantly suppressed DDP-induced Numb inhibition and Notch1 activation compared with DDP treatment alone (Fig. 6D).
Figure 6.
Effect of puerarin on DDP-induced Numb/Notch1 signaling pathway. (A and B) Reverse transcription-quantitative PCR and (C and D) western blot analysis was performed to determine the expression levels of Numb and Notch1 in HK-2 cells. Data are presented as the mean ± standard deviation. n=3. *P<0.05 vs. sham; #P<0.05 vs. DDP treatment or DDP+NC inhibitor. DDP, cisplatin; Pue, puerarin; NC, negative control.
Discussion
Puerarin is an isoflavonoid extracted from the Chinese medical herb R. puerariae, which regulates several physiological functions, including anti inflammatory and antioxidant processes (24). Recently, puerarin has been extensively used in the therapy of different types of disease, such as renal fibrosis, liver fibrosis and myocardial ischemia (25-27). Furthermore, puerarin is considered to induce beneficial effects on the treatment of different types of kidney disease, including acute renal injury and chronic renal injury (8-11). Gao et al (28) reported that puerarin provides renoprotection in rat models by altering apoptosis and autophagy. Furthermore, puerarin was demonstrated to exhibit a renal protective effect against DDP-induced nephrotoxicity by regulating the toll-like receptor 4/NF-κB signaling pathway and attenuating diabetic kidney injury via suppression of NADPH oxidase 4 expression (29,21). However, the molecular mechanisms underlying the renal protection of puerarin in DDP-induced AKI remain unclear. The results of the present study demonstrated that puerarin exerted a protective effect on DDP-induced renal injury in both HK-2 cells and rat kidneys.
In the present study, treatment with DDP notably increased miR-31 expression in a dose-dependent manner, expression levels of renal function biomarkers (BUN and SCR), MDA content and apoptosis-associated proteins (caspase-3 and Bax), while significantly decreasing the expression levels of GST, SOD and CAT antioxidants in serum, and antiapoptotic factor, Bcl-2 in kidney samples. Histopathological analysis, including HE and TUNEL demonstrated similar results. The effects induced by DDP were reversed following treatment with puerarin, suggesting that puerarin may protect DDP-induced renal injury. Furthermore, western blot analysis demonstrated that treatment with DDP decreased Numb protein expression, while increasing Notch1 protein expression in HK-2 cells. Conversely, treatment with puerarin and transfection with miR-31-mimics altered the effects on the Numb/Notch1 signaling pathway induced by DDP.
miRNAs play a regulatory role across several biological processes, such as carcinogenesis, inflammation and numerous signaling pathways including the Numb/Notch1 signaling pathway (12,13). miR-31 is associated with the progression of renal cell carcinoma via targeting cyclin-dependent kinase 1(15). Numb is confirmed as the downstream target of miR-31-5p and the overexpression of Numb inhibited the proliferation, migration, invasion and induced cell cycle arrest and apoptosis in colorectal cancers (16). In addition, the symmetric division of mouse Lewis lung carcinoma stem cells was regulated via miR-31-mediated Numb expression (30). A previous study reported that puerarin facilitated the proliferation, migration and invasion of colorectal cancer cells by targeting Numb protein (18). Numb was originally identified as an intrinsic determinant in Drosophila, affecting the cell fate by negatively altering the Notch signaling pathway (17). Numb is predominantly expressed in normal proximal tubules and detected in interstitial cells of fibrotic kidneys (31). Activation of Numb facilitated interstitial fibrosis by inducing G2/M arrest of tubular cells, while inhibition of Numb promoted dynamin-associated protein 1-mediated mitochondrial fission and exacerbates mitochondrial fragmentation and dysfunction in AKI (32). A previous study reported that Numb alleviates puromycin aminonucleoside-induced renal proximal tubular cell apoptosis by suppressing Notch signaling activity (21). For the vital function of Notch signaling in regulating the balance among cell proliferation and apoptosis that influence the progress of various organ injuries, the present study hypothesized that the negative regulation of Numb affects Notch signaling via miR-31 in DDP-induced AKI.
The present study indicated that puerarin may alleviate DDP-induced acute renal damage by suppressing miR-31 expression, resulting in enhancing Numb activation, thereby inhibiting the Notch signaling pathway. However, the present study only observed the protective effects of puerarin associated with miR-31 and Numb/ Notch 1 signaling pathway. Further studies will confirm the function of puerarin protection in DDP-induced renal injury via knockdown of Numb to verify that puerarin played its protection role via regulating miR-31/Numb/Notch pathway. Puerarin may act as a novel therapeutic candidate in the treatment of DDP-induced AKI.
Supplementary Material
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
ZW, CL, QL, JL and XL participated in the study design, performed experiments, collected and analyzed the data and prepared the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The present study was approved by the ethical requirements of the Institutional Care and Use Committee of the Second Hospital of Shandong University.
Patient consent for publication
Not applicable.
Competing interests.
The authors declare that they have no competing interests.
References
- 1.Ma G, Cheng W, Ma M. Effect of Docetaxel Combined with Cisplatin Preoperative Neoadjuvant Chemotherapy for Stage III NSCLC. J Coll Physicians Surg Pak. 2019;29:1230–1231. doi: 10.29271/jcpsp.2019.12.1230. [DOI] [PubMed] [Google Scholar]
- 2.Sultana S, Verma K, Khan R. Nephroprotective efficacy of chrysin against cisplatin-induced toxicity via attenuation of oxidative stress. J Pharm Pharmacol. 2012;64:872–881. doi: 10.1111/j.2042-7158.2012.01470.x. [DOI] [PubMed] [Google Scholar]
- 3.Chen H, Liu S, Li M, Huang P, Li X. circ_0003418 Inhibits Tumorigenesis And Cisplatin Chemoresistance Through Wnt/β-Catenin Pathway In Hepatocellular Carcinoma. OncoTargets Ther. 2019;12:9539–9549. doi: 10.2147/OTT.S229507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Liu W, Wang W, Wang X, Xu C, Zhang N, Di W. Cisplatin-stimulated macrophages promote ovarian cancer migration via the CCL20-CCR6 axis. Cancer Lett. 2020;472:59–69. doi: 10.1016/j.canlet.2019.12.024. [DOI] [PubMed] [Google Scholar]
- 5.Li Y, Zu X, Hu X, Wang L, He W. Forkhead Box R2 Knockdown Decreases Chemoresistance to Cisplatin via MYC Pathway in Bladder Cancer. Med Sci Monit. 2019;25:8928–8939. doi: 10.12659/MSM.917345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Luo X, Wei J, Yang FL, Pang XX, Shi F, Wei YX, Liao BY, Wang JL. Exosomal lncRNA HNF1A-AS1 affects cisplatin resistance in cervical cancer cells through regulating microRNA-34b/TUFT1 axis. Cancer Cell Int. 2019;19(323) doi: 10.1186/s12935-019-1042-4. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 7.Li WQ, Wu JY, Xiang DX, Luo SL, Hu XB, Tang TT, Sun TL, Liu XY. Micelles Loaded With Puerarin And Modified With Triphenylphosphonium Cation Possess Mitochondrial Targeting And Demonstrate Enhanced Protective Effect Against Isoprenaline-Induced H9c2 Cells Apoptosis. Int J Nanomedicine. 2019;14:8345–8360. doi: 10.2147/IJN.S219670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ma JQ, Ding J, Xiao ZH, Liu CM. Puerarin ameliorates carbon tetrachloride-induced oxidative DNA damage and inflammation in mouse kidney through ERK/Nrf2/ARE pathway. Food Chem Toxicol. 2014;71:264–271. doi: 10.1016/j.fct.2014.06.017. [DOI] [PubMed] [Google Scholar]
- 9.She S, Liu W, Li T, Hong Y. Effects of puerarin in STZ-induced diabetic rats by oxidative stress and the TGF-β1/Smad2 pathway. Food Funct. 2014;5:944–950. doi: 10.1039/c3fo60565e. [DOI] [PubMed] [Google Scholar]
- 10.Zhong Y, Zhang X, Cai X, Wang K, Chen Y, Deng Y. Puerarin attenuated early diabetic kidney injury through down-regulation of matrix metalloproteinase 9 in streptozotocin-induced diabetic rats. PLoS One. 2014;9(e85690) doi: 10.1371/journal.pone.0085690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang L, Lin S, Li Z, Yang D, Wang Z. Protective effects of puerarin on experimental chronic lead nephrotoxicity in immature female rats. Hum Exp Toxicol. 2013;32:172–185. doi: 10.1177/0960327112462729. [DOI] [PubMed] [Google Scholar]
- 12.Ma X, Yan L, Zhu Q, Shao F. Puerarin attenuates cisplatin-induced rat nephrotoxicity: The involvement of TLR4/NF-κB signaling pathway. PLoS One. 2017;12(e0171612) doi: 10.1371/journal.pone.0171612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang J, Ni J, Beretov J, Thompson J, Graham P, Li Y. Exosomal microRNAs as liquid biopsy biomarkers in prostate cancer. Crit Rev Oncol Hematol. 2020;145(102860) doi: 10.1016/j.critrevonc.2019.102860. [DOI] [PubMed] [Google Scholar]
- 14.He J, He J, Min L, He Y, Guan H, Wang J, Peng X. Extracellular vesicles transmitted miR-31-5p promotes sorafenib resistance by targeting MLH1 in renal cell carcinoma. Int J Cancer. 2020;146:1052–1063. doi: 10.1002/ijc.32543. [DOI] [PubMed] [Google Scholar]
- 15.Li Y, Quan J, Chen F, Pan X, Zhuang C, Xiong T, Zhuang C, Li J, Huang X, Ye J, et al. miR-31-5p acts as a tumor suppressor in renal cell carcinoma by targeting cyclin-dependent kinase 1 (CDK1) Biomed Pharmacother. 2019;111:517–526. doi: 10.1016/j.biopha.2018.12.102. [DOI] [PubMed] [Google Scholar]
- 16.Chou CH, Tu HF, Kao SY, Chiang CF, Liu CJ, Chang KW, Lin S. Targeting of miR-31/96/182 to the Numb gene during head and neck oncogenesis. Head Neck. 2018;40:808–817. doi: 10.1002/hed.25063. [DOI] [PubMed] [Google Scholar]
- 17.Zhu GF, Chu T, Ruan Z, Zhang M, Zhou M, Zhang Q, Zhang R, Wu L. Inflammation-Related MicroRNAs Are Associated with Plaque Stability Calculated by IVUS in Coronary Heart Disease Patients. J Interv Cardiol. 2019;2019(9723129) doi: 10.1155/2019/9723129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Peng H, Wang L, Su Q, Yi K, Du J, Wang Z. miR-31-5p promotes the cell growth, migration and invasion of colorectal cancer cells by targeting NUMB. Biomed Pharmacother. 2019;109:208–216. doi: 10.1016/j.biopha.2018.10.048. [DOI] [PubMed] [Google Scholar]
- 19.Liu Z, Li H, Su J, Xu S, Zhu F, Ai J, Hu Z, Zhou M, Tian J, Su Z, et al. Numb Depletion Promotes Drp1-Mediated Mitochondrial Fission and Exacerbates Mitochondrial Fragmentation and Dysfunction in Acute Kidney Injury. Antioxid Redox Signal. 2019;30:1797–1816. doi: 10.1089/ars.2017.7432. [DOI] [PubMed] [Google Scholar]
- 20.Luo Z, Mu L, Zheng Y, Shen W, Li J, Xu L, Zhong B, Liu Y, Zhou Y. NUMB enhances Notch signaling by repressing ubiquitination of NOTCH1 intracellular domain. J Mol Cell Biol. 2020;12:345–358. doi: 10.1093/jmcb/mjz088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ding X, Zhu F, Li T, Zhou Q, Hou FF, Nie J. Numb protects renal proximal tubular cells from puromycin aminonucleoside-induced apoptosis through inhibiting Notch signaling pathway. Int J Biol Sci. 2011;7:269–278. doi: 10.7150/ijbs.7.269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang Y, Wang H, Yu L, Chen J. The Puerarin improves renal function in STZ-induced diabetic rats by attenuating eNOS expression. Ren Fail. 2015;37:699–703. doi: 10.3109/0886022X.2015.1011500. [DOI] [PubMed] [Google Scholar]
- 23.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 24.Wu J, Xu L, Sun C, Zhang B, Li J, Sun J, Zhang Y, Sun D. Paeonol alleviates epirubicin-induced renal injury in mice by regulating Nrf2 and NF-κB pathways. Eur J Pharmacol. 2017;795:84–93. doi: 10.1016/j.ejphar.2016.12.005. [DOI] [PubMed] [Google Scholar]
- 25.Song Q, Zhao Y, Li Q, Han X, Duan J. Puerarin protects against iron overload-induced retinal injury through regulation of iron-handling proteins. Biomed Pharmacother. 2020;122(109690) doi: 10.1016/j.biopha.2019.109690. [DOI] [PubMed] [Google Scholar]
- 26.Zhou X, Bai C, Sun X, Gong X, Yang Y, Chen C, Shan G, Yao Q. Puerarin attenuates renal fibrosis by reducing oxidative stress induced-epithelial cell apoptosis via MAPK signal pathways in vivo and in vitro. Ren Fail. 2017;39:423–431. doi: 10.1080/0886022X.2017.1305409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li X, Zhang H, Pan L, Zou H, Miao X, Cheng J, Wu Y. Puerarin alleviates liver fibrosis via inhibition of the ERK1/2 signaling pathway in thioacetamide-induced hepatic fibrosis in rats. Exp Ther Med. 2019;18:133–138. doi: 10.3892/etm.2019.7534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gao S, Li L, Li L, Ni J, Guo R, Mao J, Fan G. Effects of the combination of tanshinone IIA and puerarin on cardiac function and inflammatory response in myocardial ischemia mice. J Mol Cell Cardiol. 2019;137:59–70. doi: 10.1016/j.yjmcc.2019.09.012. [DOI] [PubMed] [Google Scholar]
- 29.Song X, Li Z, Liu F, Wang Z, Wang L. Restoration of autophagy by puerarin in lead-exposed primary rat proximal tubular cells via regulating AMPK-mTOR signaling. J Biochem Mol Toxicol. 2017;31(e21869) doi: 10.1002/jbt.21869. [DOI] [PubMed] [Google Scholar]
- 30.Wang J, Zhou T, Sun Z, Ye T, Zhou S, Li J, Liu Y, Kong L, Tang J, Liu D, Xing HR. Zeb1 Regulates the Symmetric Division of Mouse Lewis Lung Carcinoma Stem Cells Through Numb Mediated by miR-31. Int J Biol Sci. 2018;14:1399–1410. doi: 10.7150/ijbs.27446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Jadhav S, Ajay AK, Trivedi P, Seematti J, Pellegrini K, Craciun F, Vaidya VS. RNA-binding Protein Musashi Homologue 1 Regulates Kidney Fibrosis by Translational Inhibition of p21 and Numb mRNA. J Biol Chem. 2016;291:14085–14094. doi: 10.1074/jbc.M115.713289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zhu F, Liu W, Li T, Wan J, Tian J, Zhou Z, Li H, Liu Y, Hou FF, Nie J. Numb contributes to renal fibrosis by promoting tubular epithelial cell cycle arrest at G2/M. Oncotarget. 2016;7:25604–25619. doi: 10.18632/oncotarget.8238. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets used/or analyzed during the current study are available from the corresponding author on reasonable request.