Table 3.
Methods for site-specific incorporation of an azide handle onto an antibody.
| Approach | Scheme a | Click Chemistry | Drug-Linker b | Reported DAR (Expected DAR) | Ref. |
|---|---|---|---|---|---|
| Glycan Remodeling | 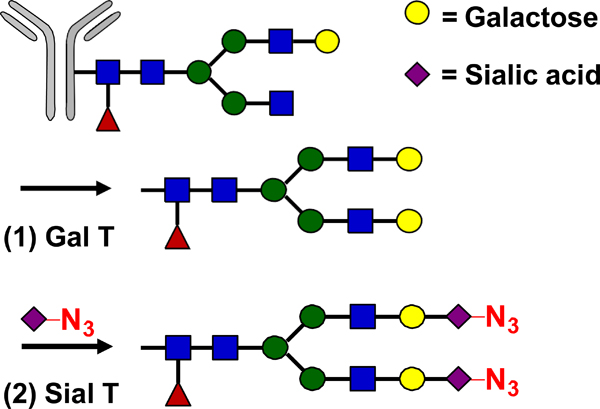 |
SPAAC | DIBO-Dox | 4.5 (4) | [31] |
| Glycan Remodeling | 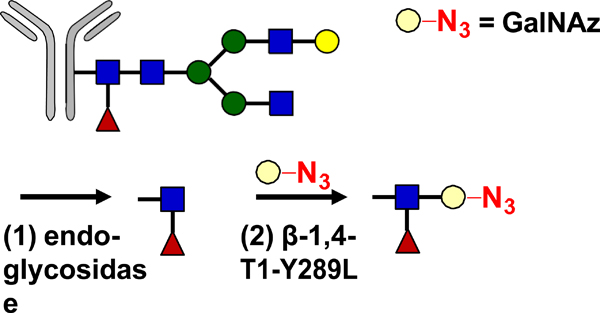 |
SPAAC | BCN-Dox BCN-MMAE BCN-MMAF BCN-maytansine BCN DUMSA | >1.9 (2) | [34] |
| CuAAC | Alkyne-PBD | >1.9 (2) 3.8 (4) | [37] | ||
| Unnatural amino acid (UAA) mutagenesis | 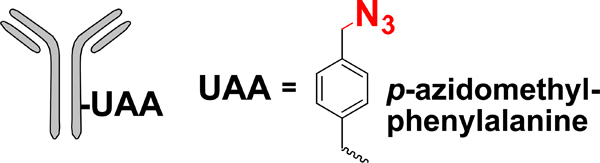 |
SPAAC | DBCO-MMAF | 1.2–1.9 (2) | [32] |
| UAA mutagenesis | 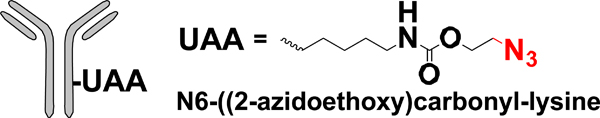 |
CuAAC SPAAC | Alkyne-PBD Alkyne-AF BCN-AF | 1.8 ->1.9 (2) | [35] |
| Enzymatic modification of peptide tag | 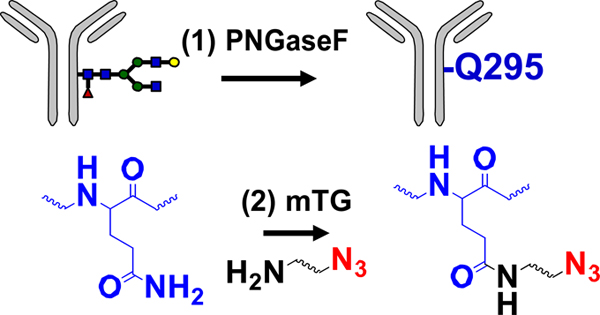 |
SPAAC | DBCO-MMAE | >1.9 (2) | [33] |
| Enzymatic modification of peptide tag | 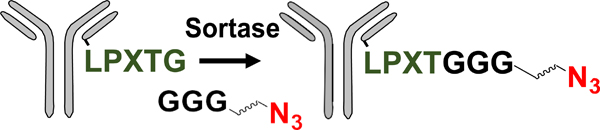 |
SPAAC | DBCO-MMAE | 3.3 (4) | [48] |
GalNAz: azido-modified N-acetyl-D-galactosamine; PNGase F: N-glycosidase F.
DIBO: dibenzocyclooctyne; BCN: bicyclo[6.1.0]nonyne; DUMSA: duocarmycin SA; PBD: pyrrolobenzodiazepine dimer; DBCO: dibenzoazacyclooctyne. Regarding the drug-linkers listed, only the functional group and the drug are specified. The linker connecting these two moieties may vary.
