Abstract
Despite the tremendous progresses toward our understanding of the mechanisms of how liver cancer was developed, the therapeutic outcomes of liver cancer in the clinic have very limited improvement within the past three decades or so. In addition, both the incidence and mortality of liver cancer worldwide are not dropping, but increasing steadily, in the last decade. Thus, it is time for us to rethink what has been wrong and how could we do better in the upcoming years, in order to achieve our goal of improving the therapeutic outcomes of patients with liver cancer in the clinic, and at the meantime, effectively reducing the incidence of liver cancer by blocking malignant transformation of hepatocytes from chronic viral infection. This is also one of the main reasons why we try to organize this special issue on primary liver cancer in the journal of Genes & Diseases. In this perspective, I will summarize the major obstacles confronted with in the prevention and management of patients with chronic hepatitis B infection and subsequent development of liver cirrhosis and liver cancer. Next, I will delineate the pitfalls and underlying mechanisms of why the current anti-viral strategies and therapeutic agents are not as effective as one expected in terms of successful reduction or prevention chronic hepatitis B infection associated liver cirrhosis and liver cancer. I will then provide my personal perspectives on potential approaches and strategies for effective prevention and management of hepatitis B-related liver cancer.
Keywords: Anti-viral therapy, Chronic inflammation, Liver cancer, Liver cirrhosis, Malignant transformation
Abbreviations: HCC, Hepatocellular carcinoma; HBV, Hepatitis B virus; OS, Overall Survival; NASH, Non-alcoholic steatohepatitis; NAFLD, Non-alcoholic fatty liver diseases; NA, Nucleos(t)ide analogue; DAA, Direct antiviral agents; AFP, alpha-fetoprotein; cccDNA, covalently closed circular DNA
Hepatocellular carcinoma (HCC) is one of the most common gastrointestinal malignancies worldwide and the second leading cause of cancer death of males in China after lung cancer.1 According to the report of GLOBOCAN's analysis on global incidence and mortality of liver cancer in 2015, there are about 841,000 new cases and 782,000 deaths worldwide.2 The number of new cases of liver cancer was 466,100 and the death toll was 422,100 in China during the same period, according to Chen et al and Zheng et al.1,3 In fact, over the past three decades, China alone contributed more than half of new cases and deaths of liver cancer worldwide.1, 2, 3 Zheng et al recently predicted that the incidence and mortality of liver cancer in China will decline by 2030, due to national wide vaccination plan of hepatitis B vaccine enacted some thirty years ago in general population, and newborns in particular.4 With the development of early detection, molecular classification, immunotherapy, molecular targeting therapy and minimally invasive surgical technology in recent years, the overall prognosis of patients with liver cancer has improved.5, 6, 7, 8, 9, 10, 11, 12 For patients with early radical resection or liver transplantation, the 5-year survival rate reached 75% or above10, 11, 12; but generally, the 5-year Overall Survival (OS) of HCC remained below 17%, and the 5-year Survival rate of HCC patients with distant metastases did not exceed 3% (Fig. 1).1, 2, 3
Figure 1.
Liver cancer incidence, mortality and 5-year overall survival.
The causes of poor prognosis of HCC are mainly due to the high intra-neoplastic heterogeneity and chemo-radio-resistance nature of liver cancer, in addition to difficulties in early diagnosis, rapid disease progression, and lack of effective target-specific drugs.8,9 Liver is the largest metabolic and detoxification organ in the body, and hepatocytes are in general tolerant to ionizing radiation and chemotherapy.13 These tissue specificities greatly affect the overall therapeutic outcomes of patients with liver cancer. Clinically, less than 20% of patients with early liver cancer could have an opportunity for radical surgery. Most patients are at the advanced stage of the disease at the diagnosis, thus they have lost the chance of surgical resection.6,8, 9, 10, 11, 12 Even within those HCC patients who received radical surgery or liver transplantation, and followed by postoperative Sorafinib adjuvant therapy, with or without radio- and chemo-therapy, the cumulative recurrence and metastasis rate within 5 years is still around 80%–100%.6,10, 11, 12, 13, 14
Therefore, the clinical treatment of patients with advanced liver cancer is still a big challenge, and new treatment modalities are still either in the pre-clinical development or undergoing early clinical trials (Fig. 2).6 At present, Sorafinib is still the only first-line molecular-targeting drug for the treatment of patients with advanced liver cancer.6,15,16 The effectiveness of treatment is only to prolong the median survival of patients for 2.8 months (median survival of the treatment group was 10.7 months, while the placebo control group was 7.9 months) in Europe, and only 2.2 months (treatment group 6.5 months, control group 4.3 months) in Asia.15,16 Due to the accompanied cirrhosis, patients with advanced HCC are in general not tolerant to traditional radiotherapy and chemotherapy, and the overall effective response rate is only 10–20%.12,13 Therefore, the US FDA has not approved any first-line chemotherapeutic agent for advanced liver cancer.7,13 Obviously, substantial breakthrough for the treatment of patients with advanced liver cancer is anticipated, but unrealistic within a foreseeable period of time. Thus, how to utilize the existing technology to develop effective measures and strategies for prevention and control of liver cancer is urgently needed for patients in the clinic.
Figure 2.
Liver Cancer Therapeutics: Current Status and Ongoing Development. Notes: The illustration demonstrates strategies for prevention and treatment of liver cancer, including: (1) HBV- or HCV- (if available in the future) based prophylaxis vaccination for the prevention of liver cancer; (2) Various forms of tumor associated antigen or specific neoantigen based vaccinations (such as DNA vaccine, peptide vaccine, dendritic cell (DC)- or tumor cell-based vaccines) with or without combination with immune checkpoint inhibitors; (3) Specific neutralizing antibodies or soluble receptor or antagonizing ligand mediated elimination of immunosuppressive cytokines; (4) Loco-regional treatment of tumors in the liver, including surgical resection, radiofrequency ablation (RFA) or high intensity focused ultrasound (Hifu), systemic or loco-regional chemo-radiotherapy, molecular-targeted therapy (MTT), trans-arterial catheter embolization (TACE) or liver transplantation; (5) Adoptive immunotherapy, including natural killers (NK), natural killer T (NKT) cells, LAK (Lymphokine-activated killer cells), cytokine-induced killer cells (CIK), or chimeric antigen receptor-modified T cells (CAR-T), CAR-NK or T cell receptor modified T cells (TCR-T); (6) Demonstrate a combination of various ways of locoregional therapy with adoptive immunotherapy for liver cancer. Additionally, chemokines, cytokines and interleukines may be used to modulate the innate immunity of the host, either alone or in combination with adoptive immunotherapy.
Causative factors associated with liver cancer vary between the more advanced western countries and developing or underdeveloped areas, such as South African or Far East and Southeast Asian states. In the developed western countries, non-alcoholic steatohepatitis (NASH) and non-alcoholic fatty liver diseases (NAFLD), occurring in the presence of obesity, diabetes and metabolic syndrome, become leading risk factors associated with liver cancer, even in the absence of cirrhosis. Accordingly, incidence of NASH and NAFLD, in parallel with overweight and obesity in both children and adults, are increasing in China, due in part to the changes in life styles and food intake in the past two decades or so.17, 18, 19, 20, 21, 22, 23, 24 According to the report by Wang et al, there are about 300 million people suffering from liver diseases in China alone.17 Each year, about 1.2 million people worldwide die from chronic HBV infection–associated complications, such as liver cirrhosis and liver cancer.17 In China, there are more than 93 million hepatitis B surface antigen (HBsAg) positive carriers and 20 million of them are patients with active chronic HBV infection who need anti-viral therapy, based on a nationwide sero-epidemiological survey performed in 2006.17 Recent report by Yang et al25 demonstrates that more than 87.5% of patients with HCC in eastern region of China are positive for HBV DNA and are likely developed on the chronic hepatitis B viral infection background. In contrast, less than ∼5% patients with liver cancer in China are positive for HCV that can also cause malignant transformation of hepatocytes.17,25 Therefore, the successful prevention of malignant transformation in HBV carriers from chronic inflammation would be the most effective way to prevent the development of HCC in China. And, prevention is and should always be more important than treatment in the control of liver cancer.
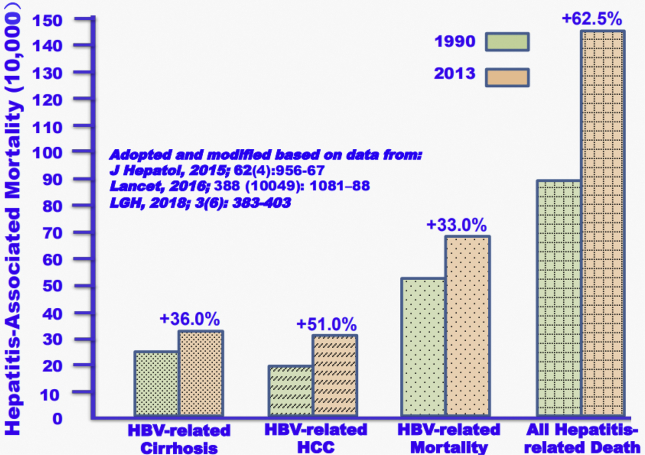
The current methods to counter the malignant transformation from benign chronic inflammation induced by HBV are mainly focused on the prevention of HBV infection and management of chronic hepatitis via anti-viral therapy after infection.26, 27, 28 Clinical epidemiological data indicate that the immune state of the HBV infected host itself is the key in determining whether the host will manifest a controllable inflammation or a chronic uncontrolled one.29, 30, 31, 32, 33 Theoretically, clinical application of immune-regulatory or immune-modulation therapy, such as the use of interferon to modulate the inflammatory process in patients with HBV infection, should be an effective approach to control the viral infection. Unfortunately, the side effects of interferon are so severe that only a small percentage of patients (10% or less) can tolerate the toxicity of IFN therapy and eventually less than 6% of those patients who had tolerated the IFN toxicity reached a clinical cure as determined by the disappearance of HBsAg.34 Over the past decade or so, due to the development of nucleos(t)ide analogue (NA) anti-viral drugs, especially new generation of NAs, such as Telbivudine or Tenofovir, application of NAs in clinic have been very effective in controlling HBV replication in HBV-infected hosts.26, 27, 28,34,35 However, the long-term outcome from clinical follow-up on NAs alone therapy or various combinations of NAs with IFN in controlling disease progression is controversial and far from satisfactory 36, 37, 38, 39, 40, 41, 42 (Fig. 3).
Figure 3.
Global Mortality and Incidence of HBV-Related Liver Cirrhosis and Cancer Notes: The bar graphic data presented here are a comparison of HBV infected population who had received no anti-viral treatment before 1990 versus those when anti-viral drugs, such as interferon and nucleos(t)ide analogues, are available after 1990 and up until 2013, worldwide.
On the one hand, application of NAs had successfully achieved sero-conversion in certain patients with chronic hepatitis B infection, but when compared to those failed to achieve sero-conversion, clinical follow-up found that there was no significant difference in the prevention of chronic viral infection-associated complications, such as liver cirrhosis and liver cancer.36, 37, 38, 39, 40, 41, 42 In addition, for NA-based therapy, there was still a risk of viral mutation, leading to drug resistance. On the other hand, even the new generation of NAs (such as Tenofovir) needs to be taken for a long period of time (or even for a life time), and patients who have undergone clinical conversion remain at risk of recurrence after discontinuation of NAs.42,43 The major reason for that is none of these NA agents could be able to directly eliminate the viral replication template, HBV cccDNA.44,45 More importantly, for those who had received either interferon and/or NAs agents, both the incidence and mortality of HBV virus infection-related complications, such as cirrhosis and liver cancer, have increased, rather than decreased, according to a global meg-data analysis of clinical patients with hepatitis, when compared with patients who did not receive any antiviral therapy before 1990.26,42,46 This line of evidence is counter-intuitive and opposed to the expected outcome of our national mid-to-long term development plan that had been implemented in China around 2007 and up to now, aimed at controlling infectious diseases, such as hepatitis, and reducing both the incidence and mortality of the viral infection - associated complications, such as cirrhosis and liver cancer.17,47 Each year, an estimated 263,000 persons in China die from HBV-related liver cancer or cirrhosis, accounting for ∼50% of HBV-related deaths worldwide.17,47 Based on the aforementioned clinic mega-data analysis 26,42,46 and outcomes from current anti-viral therapies,28,40, 41, 42, 43, 44, 45 I personally believe that, in order to achieve our goal of reducing both the incidence and mortality of viral-infection associated liver cirrhosis and liver cancer, our current anti-viral approaches and strategies are in need to be revisited. In terms of specifically blockade of the malignant transformation of chronic hepatitis B infection to HCC, the current existing anti-viral approaches are not as effective as they assumed, and thus, innovative ideas and novel strategies must be developed to circumvent the hurdles associated with current anti-viral therapy. Thereof, listed below are the three major obstacles in my view that are currently encountered in controlling chronic hepatitis B associated liver cirrhosis and malignant transformation.
Firstly, in the management of chronic hepatitis B, the current antiviral treatment strategy may not be in the right direction. Direct antiviral agents (DAA) are potent and successful in treatment of patients with HCV infection, more than 95% patients with HCV infection can get cured within 3–6 months, and in the long run, reduction of HCV associated mortality and incidence of cirrhosis and liver cancer is optimistically anticipated and will be informed by the results from future long-term follow-up of patients treated with DAA.48 As in the management of chronic hepatitis B, according to the existing therapeutic regimens with interferon or NAs, no matter it is used alone or in combination of different NAs or NAs with IFN, the rate of achieved clinic cure as determined by HBsAg disappearance, is no more than 10%,36, 37, 38, 39, 40, 41, 42 indicating that close to 90% of patients currently receiving anti-viral therapies failed to achieve clinical cure, not to mention complete cure, which means a clearance of viral replication template cccDNA in HBV infected hepatocytes.43, 44, 45 Moreover, some patients achieved HBsAg loss still has a risk of HBsAg re-conversion and viral reactivation.43 In addition, severe hepatotoxicity and nephrotoxicity of NAs and the systemic toxicity of interferon in a way may accelerate, instead of block or reduce the occurrence of liver cirrhosis.28,37, 38, 39, 40, 41, 42,45,46 According to the above clinic mage data,26,42,46 the increased incidence of liver cirrhosis in patients who had received anti-viral therapy might indirectly reflect its hepatotoxic problem in particular. Thus, the efficacy of current anti-viral agents needs to be revisited and the anti-viral strategy waits to be renovated.
Secondly, an appropriate humanized animal model that could recapitulate the pathophysiological processes, from natural HBV infection to chronic viral inflammation, then to malignant transformation, is lacking. Very recently, Li et al reported their efforts in the establishment of a humanized cirrhosis model with HBV infection in rodents.48 Based on their report, this humanized rodent model seems partially solved the problems with modelling chronic HBV infection induced cirrhosis, but it is still uncertain whether the humanized cirrhosis model can mimic chronic hepatitis B infection-associated malignant transformation. According to their observation, mice infected with HBV did not produce spontaneous hepatocellular carcinoma within 56 weeks after HBV infection. Thus, generation of a model system that could recapitulate the pathophysiological processes of human HBV infection is of paramount importance for the development of more effective and novel anti-viral agents that are able to eliminate the viral replication template cccDNA in HBV-infected hepatocytes.
Thirdly, the lack of antiviral drugs that can effectively and directly eliminate HBV cccDNA, the virus replication template, is another major obstacle we are confronted with.44,45 Data from clinical follow-up of current antiviral treatment in patients with chronic hepatitis B virus infection clearly demonstrate that the persistence of the viral replication template, HBV cccDNA, may be the major cause that leads chronic viral infection to uncontrollable malignant transformation.28,33 Although new drugs developed at Gilead are based on re-activation of the innate immunity via TLR7/9 receptor agonists, such as GS-9620, which may be able to remove cccDNA by eliminating viral infected hepatocytes,49, 50, 51, 52 theoretically, these agents themselves cannot directly or specifically eliminate cccDNA. Mechanistically, excessive activation of the body's innate immunity might come at a risk of breaking the tightly regulated networks of innate immunity and triggering an overwhelming autoimmune reaction, such as that have been observed in the administration of immune checkpoint inhibitors in patients with cancer.51, 52, 53, 54, 55, 56 Needless to say, whether the TLR7/9 receptor agonists are effective enough to specifically remove viral infected hepatocytes in HBV-infected patients is unclear. The termination of further clinical trials on TLR7/9 receptor agonists in HBV-infected patients by Gilead may be partially associated with the efficacy and toxicity of TLR7/9 receptor agonists (www.clinictrials.gov).
In summary, to achieve the goal of effectively blocking the transition from chronic benign inflammation to malignant transformation in patients with chronic hepatitis B infection, the development of a drug that can directly remove or eliminate the cccDNA, the replication template of the hepatitis B virus, in addition to re-activate the host innate immunity against viral infection, is urgently needed, and it might be the only way toward a complete cure of chronic hepatitis B infection. Strategically, development of agents that could directly target the formation of HBV cccDNA will undoubtedly pave a way for the effective prevention of chronic hepatitis B infection-associated liver cancer.43,44,57,58
As a matter of fact, in the effort to prevent and control of liver cancer, China took the lead in the screening of liver cancer patients from the high-risk groups by a quantitative detection of serum alpha-fetoprotein (AFP) in the 1970s. From the field studies, pioneers in the Chinese Alliance for the Study of Liver Cancer had formulated the seven-word policy of “control drinking water, moldy food, and hepatitis” and widely implemented it in the endemic regions with high incidence of liver cancer, such as Qidong in Jiangsu Province.59 Particularly, in the mid-1980s to the early 1990s, the nationwide vaccination plan of hepatitis B vaccine in infants had laid the foundation for the prospected decline in the incidence and mortality of liver cancer in China around 2030.4 Given the fact that there are about 1.2 million people worldwide die annually from chronic HBV infection–associated complications as stated above,17 the major strategies for prevention of hepatitis B-related liver cancer in China, at the moment, should focus mainly on the following aspects, as my personal perspective:
The first approach is to strengthen the blockade of mother-to-infant transmission of Hepatitis B virus and boost vaccination for children aged 8–12 years.
The second is to effectively improve the clinical cure rate of chronic carriers of hepatitis B virus. Efficacy of the current existing antiviral drugs (either alone or a combination of NAs with different formulations of IFN) is limited in terms of achieving a clinic cure, even though these agents are potent in repression of viral replication. In addition, application of these drugs, either alone or in various ways of combinations, is not capable of drastically improving clinic cure rate for patients with chronic hepatitis B viral infection. Although the major aim for the clinical management of patients with hepatitis B is to recover the host innate immunity against virus, currently, the criteria of most, if not all, clinical guidelines for a “complete cure” of patients with hepatitis B is the clearance of the viral replication template cccDNA.34,35,43,44 Unfortunately, it seems that we could never be able to meet the goal for a “complete cure” by the existing anti-viral agents or formulations, because none of them can directly repress the formation of cccDNA or effectively clear cccDNA from HBV-infected hepatocytes. Therefore, the development of new drugs that are able to effectively repress or block the synthesis or formation of cccDNA will not only become the ultimate solution to resolve the issues confronted with the management of patients with hepatitis B infection in the clinic, but also the most effective way and straightforward strategy in the prevention of liver cancer.
The third approach is early diagnosis and treatment of liver cancer, especially in the pre-clinical early-stage diagnosis. Recently, Wang and his co-workers have done some pioneering exploration in the detection of very early-stage HCC from at-risk populations, some 6–12 months earlier than that of current existing clinical diagnostic methodology could possibly achieve.9,60 It is expected that implementation of this new diagnostic tool may lead to a better therapeutic outcome and a higher chance of cure for some of those patients with pre-clinic early-stage liver cancer. Meanwhile, the risk of over-diagnosis and over-treatment for those patients diagnosed in the pre-clinic early-stage is a major concern in the clinic and should be guarded against in the future practice.
The fourth strategy is early prediction, surveillance, and diagnosis of intrahepatic recurrence and distant metastasis in liver cancer patients after radical therapy, including hepatic resection and liver organ transplantation. In the past three decades or so, clinicians and scholars both at home and abroad have done a lot of relevant studies, from basic mechanistic research to the bedside clinic investigation on the prediction and management of post-operative intrahepatic recurrence and distant metastasis of primary liver cancer, but the outcome turns out to be that the overall 5-year survival rate has not been dramatically improved.1,2,5,6 Up to now, intrahepatic recurrence and distant metastasis after radical therapy are still the major factors that contributed to the death of liver cancer patients. This is also one of the main reasons why we try to organize this special issue on primary liver cancer in the journal of Genes & Diseases. In addition, we choose to focus specifically on the obstacles and opportunities in the prevention and control of primary liver cancer. In this issue, authors will cover current status in clinical intervention and management of liver cancer from various angles. They may repeatedly touch on the major obstacles for a cure and counter-intuitive therapeutic outcomes in patients with advanced liver cancer, whether it is by surgical resection, or liver transplantation, or radiotherapy, or chemotherapy. As discussed in this issue, either surgery or chemo-radio-therapy sometimes may facilitate or promote, rather than prevent, intrahepatic recurrence and distant metastasis. Therefore, the future clinical management of advanced liver cancer should be focused on loco-regional control of tumors and minimally invasive surgery, rather than the conventional radical surgery or massive destructive chemo-radio-therapy aimed to eliminate all of the tumor cells. In addition, the purpose for multi-modality therapy for patients with advanced liver cancer should be deliberately focused on recovering the host anti-tumor immunity, improving, rewiring or re-formulating the altered metabolism in both the microenvironment of the tumor cells in the vicinity and the systematic macro-environment of the host body, so as to improve the life quality of patients, prolong overall survival, and eventually to achieve “a harmonious symbiosis” of the host with the tumor, which should be assumed as the ultimate goal of “cure” for patients with an advanced liver cancer.9,60, 61, 62, 63, 64, 65, 66, 67
Last, but not the least, it is important to recognize that the liver is the largest regional and embryonic innate immune organ in the body. The immune response in the liver is dominated by a large number of innate immune cells enriched in the liver, such as Kupffer cells, NK, γδT and NKT cells.29,30 The liver is in a state dominated by innate immunity. This state of innate immune dominance may also be the cellular basis for the formation of immune tolerance in the liver. As the most powerful immune-exempt organ in the body, the immune response inside the liver is usually induced by the induction of immune tolerance.29,30 This may also explains why HBV infection tends to be a persistent process that can hardly be completely eliminated for a long time. The same mechanism is also applicable to the adaptation of engrafted liver by the host after orthotopic organ transplantation, which is the least to be rejected. Unlike most other organ transplantations, liver transplantation requires the minimum of histocompatibility matching, and once transplanted successfully, will also reduce the rejection of other organ transplants from the same donor, indicating that the liver can induce a systemic tolerance based on its in situ regional tolerance. Similarly, tumor development is also based on tumor-induced immunoediting, which causes tumor cells to escape the immune surveillance and thus avoid clearance by the host immune system.68 Therefore, if there was a way to break tumor-induced immune tolerance and simultaneously revitalize host anti-tumor specific immunity, such as the development and generation of a combined immunotherapy based on tumor-specific clonal neoantigen along with immune checkpoint inhibitors, it would be possible to achieve an individualized precision immunotherapy for patients with liver cancer.68, 69, 70, 71, 72, 73, 74, 75
In conclusion, innovative technology and novel therapeutic strategies are urgently needed in order to control the progression of the diseases for patients with advanced liver cancer, and the ultimate therapeutic goals are to improve the quality of life and to prolong their overall survival time.
Conflict of interest
The Author declares no conflict of interest.
Acknowledgement
The author would like to dedicate this article to Dr. Zhao-You Tang, a great mentor, in celebrating his 90th birthday and the 50th anniversary of Liver Cancer Institute, Fudan University, in which Dr. Tang laid the cornerstone and has managed to foster new generations of clinical investigators of liver cancer in the past five decades.
Research in Yong Liao's lab was supported in part by funds from the National Natural Science Foundation of China (NSFC-81172066, NSFC-81472858) and a major research project award from the NSFC (NSFC-915291003), A Start-up Fund (08-1286-001) and the Fund for “Innovation Team on Cancer Metabolism” (2209-16) from the 2nd Affiliated Hospital, Chongqing Medical University, Chongqing, P.R. China.
Footnotes
Peer review under responsibility of Chongqing Medical University.
References
- 1.Chen W., Zheng R., Baade P.D. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. doi: 10.3322/caac.21338. [DOI] [PubMed] [Google Scholar]
- 2.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 3.Zheng R.S., Sun K.X., Zhang S.W. Report of cancer epidemiology in China, 2015. Zhonghua Zhongliu Zazhi. 2019;41:19–28. doi: 10.3760/cma.j.issn.0253-3766.2019.01.005. [DOI] [PubMed] [Google Scholar]
- 4.Zheng R., Qu C., Zhang S. Liver cancer incidence and mortality in China: temporal trends and projections to 2030. Chin J Canc Res. 2018;30:571–579. doi: 10.21147/j.issn.1000-9604.2018.06.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Miller K.D., Siegel R.L., Lin C.C. Cancer treatment and survivorship statistics. CA Cancer J Clin. 2016;66(4):271–289. doi: 10.3322/caac.21349. [DOI] [PubMed] [Google Scholar]
- 6.Zhu J., Yin T., Xu Y., Lu X.J. Therapeutics for advanced hepatocellular carcinoma: recent advances, current dilemma, and future directions. J Cell Physiol. 2019;234(8):12122–12132. doi: 10.1002/jcp.28048. [DOI] [PubMed] [Google Scholar]
- 7.Siu E.H., Chan A.W., Chong C.C., Chan S.L., Lo K.W., Cheung S.T. Treatment of advanced hepatocellular carcinoma: immunotherapy from checkpoint blockade to potential of cellular treatment. Transl Gastroenterol Hepatol. 2018;3(11):89. doi: 10.21037/tgh.2018.10.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sullivan K.M., Kenerson H.L., Pillarisetty V.G., Riehle K.J., Yeung R.S. Precision oncology in liver cancer. Ann Transl Med. 2018;6(14):285. doi: 10.21037/atm.2018.06.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Fu J., Wang H. Precision diagnosis and treatment of liver cancer in China. Cancer Lett. 2018;412:283–288. doi: 10.1016/j.canlet.2017.10.008. [DOI] [PubMed] [Google Scholar]
- 10.Dai W.C., Chan S.C., Chok K.S. Good longterm survival after primary living donor liver transplantation for solitary hepatocellular carcinomas up to 8 cm in diameter. HPB (Oxford) 2014;16:749–757. doi: 10.1111/hpb.12212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lee K.K., Kim D.G., Moon I.S., Lee M.D., Park J.H. Liver transplantation versus liver resection for the treatment of hepatocellular carcinoma. J Surg Oncol. 2010;101:47–53. doi: 10.1002/jso.21415. [DOI] [PubMed] [Google Scholar]
- 12.Kwak H.W., Park J.W., Nam B.H. Clinical outcomes of a cohort series of patients with hepatocellular carcinoma in a hepatitis B virus-endemic area. J Gastroenterol Hepatol. 2014;29:820–829. doi: 10.1111/jgh.12470. [DOI] [PubMed] [Google Scholar]
- 13.Ikeda M., Morizane C., Ueno M., Okusaka T., Ishii H., Furuse J. Chemotherapy for hepatocellular carcinoma: current status and future perspectives. Jpn J Clin Oncol. 2018;48:103–114. doi: 10.1093/jjco/hyx180. [DOI] [PubMed] [Google Scholar]
- 14.Bruix J., Takayama T., Mazzaferro V. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16:1344–1354. doi: 10.1016/S1470-2045(15)00198-9. [DOI] [PubMed] [Google Scholar]
- 15.Llovet J.M., Ricci S., Mazzaferro V. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 16.Cheng A.L., Kang Y.K., Chen Z. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25–34. doi: 10.1016/S1470-2045(08)70285-7. [DOI] [PubMed] [Google Scholar]
- 17.Wang F.S., Fan J.G., Zhang Z., Gao B., Wang H.Y. Global burden of liver disease: the major impact of China. Hepatology. 2014;60:2099–2108. doi: 10.1002/hep.27406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Michelotti G.A., Machado M.V., Diehl A.M. NAFLD, NASH and liver cancer. Nat Rev Gastroenterol Hepatol. 2013;10(11):656–665. doi: 10.1038/nrgastro.2013.183. [DOI] [PubMed] [Google Scholar]
- 19.Moriya A., Iwasaki Y., Ohguchi S. Roles of alcohol consumption in fatty liver: a longitudinal study. J Hepatol. 2015;62(4):921–927. doi: 10.1016/j.jhep.2014.11.025. [DOI] [PubMed] [Google Scholar]
- 20.Estes C., Razavi H., Loomba R., Younossi Z., Sanyal A.J. Modeling the epidemic of nonalcoholic fatty liver disease demonstrates an exponential increase in burden of disease. Hepatology. 2018;67(1):123–133. doi: 10.1002/hep.29466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dhamija E., Paul S.B., Kedia S. Non-alcoholic fatty liver disease associated with hepatocellular carcinoma: an increasing concern. Indian J Med Res. 2019;149(1):9–17. doi: 10.4103/ijmr.IJMR_1456_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Anstee Q.M., Reeves H.L., Kotsiliti E., Govaere O., Heikenwalder M. From NASH to HCC: current concepts and future challenges. Nat Rev Gastroenterol Hepatol. 2019;16(7):411–428. doi: 10.1038/s41575-019-0145-7. [DOI] [PubMed] [Google Scholar]
- 23.Berkan-Kawińska A., Piekarska A. Hepatocellular carcinoma in non-alcohol fatty liver disease - changing trends and specific challenges. Curr Med Res Opin. 2019;1 doi: 10.1080/03007995.2019.1683817. [Epub ahead of print] PubMed PMID: 31631714. [DOI] [PubMed] [Google Scholar]
- 24.Reig M., Gambato M., Man N.K. Should patients with NAFLD/NASH Be surveyed for HCC? Transplantation. 2019;103(1):39–44. doi: 10.1097/TP.0000000000002361. [DOI] [PubMed] [Google Scholar]
- 25.Yang F., Ma L., Yang Y. Contribution of hepatitis B virus infection to the aggressiveness of primary liver cancer: a clinical epidemiological study in eastern China. Front Oncol. 2019 May 21;9(370):1–10. doi: 10.3389/fonc.2019.00370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Polaris Observatory Collaborators Global prevalence, treatment, and prevention of hepatitis B virus infection in 2016: a modelling study. Lancet Gastroenterol Hepatol. 2018;3(6):383–403. doi: 10.1016/S2468-1253(18)30056-6. [DOI] [PubMed] [Google Scholar]
- 27.Locarnini S., Hatzakis A., Chen D., Lok A. Strategies to control hepatitis B: public policy, epidemiology, vaccine and drugs. J Hepatol. 2015;62:S76–S86. doi: 10.1016/j.jhep.2015.01.018. [DOI] [PubMed] [Google Scholar]
- 28.Zhang Z., Zhou Y., Yang J., Hu K., Huang Y. The effectiveness of TDF versus ETV on incidence of HCC in CHB patients: a meta analysis. BMC Canc. 2019;19(1):511. doi: 10.1186/s12885-019-5735-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Tian Z., Cao X., Chen Y., Lyu Q. Regional immunity in tissue homeostasis and diseases. Sci China Life Sci. 2016;59:1205–1209. doi: 10.1007/s11427-016-0351-y. [DOI] [PubMed] [Google Scholar]
- 30.Makarova-Rusher O.V., Medina-Echeverz J., Duffy A.G., Greten T.F. The yin and yang of evasion and immune activation in HCC. J Hepatol. 2015;62(6):1420–1429. doi: 10.1016/j.jhep.2015.02.038. [DOI] [PubMed] [Google Scholar]
- 31.Maini M.K., Gehring A.J. The role of innate Immunity in the immunopathology and treatment of HBV infection. J Hepatol. 2016;64:S60–S70. doi: 10.1016/j.jhep.2016.01.028. [DOI] [PubMed] [Google Scholar]
- 32.Bertoletti A., Ferrari C. Adaptive immunity in HBV infection. J Hepatol. 2016;64:S71–S83. doi: 10.1016/j.jhep.2016.01.026. [DOI] [PubMed] [Google Scholar]
- 33.Levrero M., Zucman-Rossi J. Mechanism of HBV-induced hepatocellular carcinoma. J Hepatol. 2016;64:S84–S101. doi: 10.1016/j.jhep.2016.02.021. [DOI] [PubMed] [Google Scholar]
- 34.European Association For The Study Of The L. EASL clinical practice guidelines: management of chronic hepatitis B virus infection. J Hepatol. 2012;57:167–185. doi: 10.1016/j.jhep.2012.02.010. [DOI] [PubMed] [Google Scholar]
- 35.Papatheodoridis G.V., Manolakopoulos S., Dusheiko G., Archimandritis A.J. Therapeutic strategies in the management of patients with chronic hepatitis B virus infection. Lancet Infect Dis. 2008;8:167–178. doi: 10.1016/S1473-3099(07)70264-5. [DOI] [PubMed] [Google Scholar]
- 36.Buster E.H., Flink H.J., Cakaloglu Y. Sustained HBeAg and HBsAg loss after long-term follow-up of HBeAg-positive patients treated with peginterferon alpha-2b. Gastroenterol. 2008;135(2):459–467. doi: 10.1053/j.gastro.2008.05.031. [DOI] [PubMed] [Google Scholar]
- 37.Sun J., Ding H., Chen G. Sustained serological and complete responses in HBeAg-positive patients treated with Peginterferon alfa-2b: a 6-year long-term follow-up of a multicenter, randomized, controlled trial in China. BMC Gastroenterol. 2019;19(1):65. doi: 10.1186/s12876-019-0981-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Marcellin P., Ahn S.H., Chuang W.L. Predictors of response to tenofovir disoproxil fumarate plus peginterferon alfa-2a combination therapy for chronic hepatitis B. Aliment Pharmacol Ther. 2016;44(9):957–966. doi: 10.1111/apt.13779. [DOI] [PubMed] [Google Scholar]
- 39.Cheng J., Wang Y., Hou J. Peginterferon alfa-2b in the treatment of Chinese patients with HBeAg-positive Chronic hepatitis B: a randomized trial. J Clin Virol. 2014;61(4):509–516. doi: 10.1016/j.jcv.2014.08.008. [DOI] [PubMed] [Google Scholar]
- 40.Sohn W., Cho J.Y., Kim J.H. Risk score model for the development of hepatocellular carcinoma in treatment-naïve patients receiving oral antiviral treatment for chronic hepatitis B. Clin and Mol Hepatol. 2017;23:170–178. doi: 10.3350/cmh.2016.0086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Papatheodoridis G.V., Lampertico P., Manolakopoulos S., Lok A. Incidence of hepatocellular carcinoma in chronic hepatitis B patients receiving nucleos(t)ide therapy: a systemic review. J Hepatol. 2010;53:348–356. doi: 10.1016/j.jhep.2010.02.035. [DOI] [PubMed] [Google Scholar]
- 42.Papatheodoridis G.V., Chan H.L., Hansen B.E., Janssen H.L.A., Lampertico P. Risk of hepatocellular carcinoma in chronic hepatitis B: assessment and modification with current antiviral therapy. J Hepatol. 2015;62:956–967. doi: 10.1016/j.jhep.2015.01.002. [DOI] [PubMed] [Google Scholar]
- 43.Bojito-Marrero L., Pyrsopoulos N. Hepatitis B and hepatitis C reactivation in the biologic era. J Clin Transl Hepatol. 2014;2:240–246. doi: 10.14218/JCTH.2014.00033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lucifora J., Protzer U. Attacking hepatitis B virus cccDNA-The holy grail to hepatitis B cure. J Hepatol. 2016;64:S41–S48. doi: 10.1016/j.jhep.2016.02.009. [DOI] [PubMed] [Google Scholar]
- 45.Levrero M., Subic M., Villeret F., Zoulim F. Perspectives and limitations for nucleo(t)side analogs in future HBV therapies. Curr Opin Virol. 2018;30:80–89. doi: 10.1016/j.coviro.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 46.Stanaway J.D., Flaxman A.D., Naghavi M. The global burden of viral hepatitis from 1990 to 2013:findings from the Global Burden of Disease Study 2013. Lancet. 2016;388:1081–1088. doi: 10.1016/S0140-6736(16)30579-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Li X., Xu W.F. China's efforts to shed its title of Leader in liver disease. Drug Discov Ther. 2007;1(2):84–85. [PubMed] [Google Scholar]
- 48.Yuan L., Jiang J., Liu X. HBV infection-induced liver cirrhosis development in dual-humanized mice with human bone mesenchymal stem cell transplantation. Gut. 2019;68(11):2044–2056. doi: 10.1136/gutjnl-2018-316091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Du K., Liu J., Broering R. Recent advances in the discovery and development of TRL ligand as novel therapeutics for chronic HBV and HIV infections. Expert Opin Drug Discov. 2018;13(7):661–670. doi: 10.1080/17460441.2018.1473372. [DOI] [PubMed] [Google Scholar]
- 50.Lucifora J., Bonnin M., Aillot L. Direct anti-viral properties of TLR ligands against HBV replication in immune-competent hepatocytes. Sci Rep. 2018;8(1):5390. doi: 10.1038/s41598-018-23525-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Niu C., Li L., Daffis S. Toll-like receptor 7 agonist GS-9620 induces prolonged inhibition of HBV via a type I interferon-dependent mechanism. J Hepatol. 2018;68(5):922–931. doi: 10.1016/j.jhep.2017.12.007. [DOI] [PubMed] [Google Scholar]
- 52.Moslehi J.J., Salem J.E., Sosman J.A., Lebrun-Vignes B., Johnson D.B. Increased reporting of fatal immune checkpoint inhibitor-associated myocarditis. Lancet. 2018;391:933. doi: 10.1016/S0140-6736(18)30533-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Johnson D.B., Balko J.M., Compton M.L. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016;375:1749–1755. doi: 10.1056/NEJMoa1609214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Tajmir-Riahi A., Bergmann T., Schmid M., Agaimy A., Schuler G., Heinzerling L. Life-threatening autoimmune cardiomyopathy reproducibly induced in a patient by checkpoint inhibitor therapy. J Immunother. 2018;41:35–38. doi: 10.1097/CJI.0000000000000190. [DOI] [PubMed] [Google Scholar]
- 55.Neilan T.G., Rothenberg M.L., Amiri-Kordestani L. Myocarditis associated with immune checkpoint inhibitors: an expert consensus on data gaps and a call to action. The Oncologist. 2018;23:874–878. doi: 10.1634/theoncologist.2018-0157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Xu F., Jin T., Zhu Y., Dai C. Immune checkpoint therapy in liver cancer. J Exp Clin Cancer Res. 2018;37:110. doi: 10.1186/s13046-018-0777-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Tang L., Zhao Q., Wu S., Cheng J., Chang J., Guo J.T. The current status and future directions of hepatitis B anti-viral drug discovery. Expert Opin Drug Discov. 2017;12(1):5–15. doi: 10.1080/17460441.2017.1255195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Guo J.T., Guo H. Metabolism and function of hepatitis B virus cccDNA: implications for the development of cccDNA-targeting anti-viral therapeutics. Antivir Res. 2015;122:91–100. doi: 10.1016/j.antiviral.2015.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.[Tang Z.Y., Yu Y.Q., editors. Primary Liver Cancer. 2nd ed. Shanghai Science &Technology Press; Shanghai: 1998. pp. 1–16. [Google Scholar]
- 60.Luo P., Yin P., Hua R. A large-scale, multicenter serum metabolite biomarker identification study for early detection of hepatocellular carcinoma. Hepatology. 2018;67(2):662–675. doi: 10.1002/hep.29561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Okusaka T., Ikeda M. Immunotherapy for hepatocellular carcinoma: current status and future perspectives. ESMO Open. 2018;3 doi: 10.1136/esmoopen-2018-000455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Waidmann O. Recent developments with immunotherapy for hepatocellular carcinoma. Expert Opin Biol Ther. 2018;18:905–910. doi: 10.1080/14712598.2018.1499722. [DOI] [PubMed] [Google Scholar]
- 63.Zhou G., Sprengers D., Boor P.P.C. Antibodies against immune checkpoint molecules restore functions of tumor-infiltrating T cells in hepatocellular carcinomas. Gastroenterology. 2017;153:1107–1119. doi: 10.1053/j.gastro.2017.06.017. e1110. [DOI] [PubMed] [Google Scholar]
- 64.Guo X., Jiang H., Shi B. Disruption of PD-1 enhanced the anti-tumor activity of chimeric antigen receptor T cells against hepatocellular carcinoma. Front Pharmacol. 2018;9:1118. doi: 10.3389/fphar.2018.01118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yu M., Luo H., Fan M. Development of GPC3-specific chimeric antigen receptor-engineered natural killer cells for the treatment of hepatocellular carcinoma. Mol Ther. 2018;26:366–378. doi: 10.1016/j.ymthe.2017.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Bi Y., Jiang H., Wang P. Treatment of hepatocellular carcinoma with a GPC3-targeted bispecific T cell engager. Oncotarget. 2017;8:52866–52876. doi: 10.18632/oncotarget.17905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Desai J.R., Ochoa S., Prins P.A., He A.R. Systemic therapy for advanced hepatocellular carcinoma: an update. J Gastrointest Oncol. 2017;8:243–255. doi: 10.21037/jgo.2017.02.01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wagner M., Koyasu S. Cancer immunoediting by innate lymphoid cells. Trends Immunol. 2019;40(5):415–430. doi: 10.1016/j.it.2019.03.004. [DOI] [PubMed] [Google Scholar]
- 69.Lu Y.C., Robbins P.F. Cancer immunotherapy targeting neoantigens. Semin Immunol. 2016;28:22–27. doi: 10.1016/j.smim.2015.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hartmaier R.J., Charo J., Fabrizio D. Genomic analysis of 63,220 tumors reveals insights into tumor uniqueness and targeted cancer immunotherapy strategies. Genome Med. 2017;9:16. doi: 10.1186/s13073-017-0408-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Jiang Y., Sun A., Zhao Y. Proteomics identifies new therapeutic targets of early-stage hepatocellular carcinoma. Nature. 2019;567(7747):257–261. doi: 10.1038/s41586-019-0987-8. [DOI] [PubMed] [Google Scholar]
- 72.Zhang X., Sharma P.K., Peter Goedegebuure S., Gillanders W.E. Personalized cancer vaccines: targeting the cancer mutanome. Vaccine. 2017;35:1094–1100. doi: 10.1016/j.vaccine.2016.05.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Li S., Yang F., Ren X. Immunotherapy for hepatocellular carcinoma. Drug Discov Ther. 2015;9:363–371. doi: 10.5582/ddt.2015.01054. [DOI] [PubMed] [Google Scholar]
- 74.Khemlina G., Ikeda S., Kurzrock R. The biology of Hepatocellular carcinoma: implications for genomic and immune therapies. Mol Cancer. 2017;16:149. doi: 10.1186/s12943-017-0712-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.da Motta Girardi D., Correa T.S., Crosara Teixeira M., Dos Santos Fernandes G. Hepatocellular carcinoma: review of targeted and immune therapies. J Gastrointest Cancer. 2018;49:227–236. doi: 10.1007/s12029-018-0121-4. [DOI] [PubMed] [Google Scholar]