Abstract
Unlike their peripheral nervous system counterparts, the capacity of central nervous system neurons and axons for regeneration after injury is minimal. Although a myriad of therapies (and different combinations thereof) to help promote repair and recovery after spinal cord injury (SCI) have been trialed, few have progressed from bench-top to bedside. One of the few such therapies that has been successfully translated from basic science to clinical applications is electrical stimulation (ES). Although the use and study of ES in peripheral nerve growth dates back nearly a century, only recently has it started to be used in a clinical setting. Since those initial experiments and seminal publications, the application of ES to restore function and promote healing have greatly expanded. In this review, we discuss the progression and use of ES over time as it pertains to promoting axonal outgrowth and functional recovery post-SCI. In doing so, we consider four major uses for the study of ES based on the proposed or documented underlying mechanism: (1) using ES to introduce an electric field at the site of injury to promote axonal outgrowth and plasticity; (2) using spinal cord ES to activate or to increase the excitability of neuronal networks below the injury; (3) using motor cortex ES to promote corticospinal tract axonal outgrowth and plasticity; and (4) leveraging the timing of paired stimuli to produce plasticity. Finally, the use of ES in its current state in the context of human SCI studies is discussed, in addition to ongoing research and current knowledge gaps, to highlight the direction of future studies for this therapeutic modality.
Keywords: axonal outgrowth, axonal regeneration, neuromodulation, neuroplasticity plasticity, SCI, treatment
Introduction
Neurons in the central nervous system (CNS) are unable to regenerate their axons following injury. This inability is based on various factors, including the presence of growth inhibitory factors in CNS myelin and at the lesion site, a lack of trophic support, and an age-related reduction in the intrinsic neuronal capacity to grow.1,2 Considering the debilitating effects of CNS injuries, approaches to enable repair are desperately needed. One method to promote and possibly direct axonal outgrowth, to re-establish lost connections, and to restore function is electrical stimulation (ES), an area of research that began nearly a century ago.3,4 These early experiments demonstrated the wide range of effects that ES can have on neurons, including promoting neurite outgrowth. The ability of ES to generate an electric field (EF) in the target tissue to influence neuronal outgrowth in vitro was subsequently explored in animal models of spinal cord injury (SCI).5,6 We are defining EF as the electrical field created across tissue between an anode and a cathode. Although ES will generally generate an EF, the intention of ES frequently goes beyond the creation of an EF and is intended to increase the excitability of the target neurons to increase spiking or to enable enhanced responses to afferent or descending inputs (i.e., to bring them closer to the threshold for firing). Since the publication of these seminal articles,5,6 the use of ES to restore function after SCI has expanded considerably, and ES is currently used for a variety of purposes. Recently, ES has become more prominent in the field of SCI, as it is clinically used to activate or facilitate the activation of neuronal networks below a lesion rather than to promote axonal outgrowth.
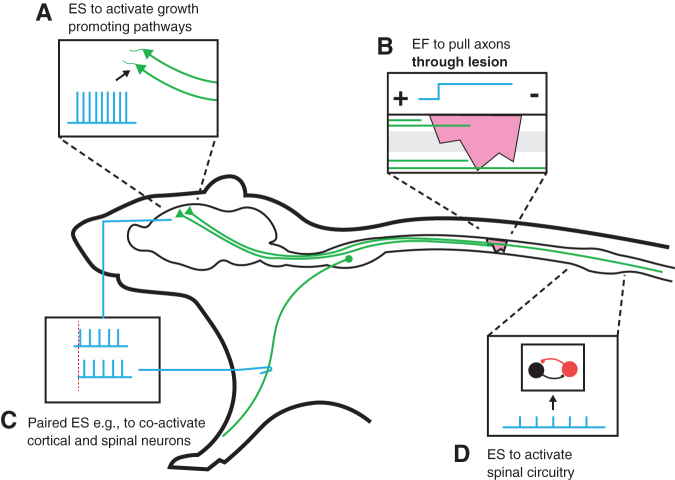
The intent of this review is to provide an overview of the major areas of ES that are used to treat SCI and to clarify their differential purposes and approaches. Considering the vast literature for each of the different areas, our objective is not to provide a comprehensive review for each of them, but rather to provide an overview and clarify their relationship to each other. For this purpose, we consider four major uses for the study of ES based on the proposed or documented underlying mechanism, which are summarized in Figure 1: (1) using ES to introduce an EF at the site of injury to promote axonal outgrowth and plasticity; (2) using spinal cord ES to activate or to increase the excitability of neuronal networks below the injury; (3) using motor cortex (MCX) ES to promote corticospinal tract (CST) axonal outgrowth and plasticity; and (4) leveraging the timing of paired cortical and peripheral stimuli to produce plasticity.
FIG. 1.
The difference in the approach and location of electrical stimulation following spinal cord injury of the four applications discussed in this review are summarized. (A) Cortical stimulation to activate growth promoting cellular pathways with the intention to promote axonal outgrowth (i.e., collateral sprouting and regeneration). (B) Using electrical fields across the spinal lesion to promote axonal growth (i.e., regeneration) through the lesion. (C) Paired stimulation (here cortex and a peripheral nerve) to strengthen synaptic connections by coactivating pre- and post-synaptic neurons. (D) Electrical stimulation to activate (or facilitate activation) of spinal circuitry below the level of the injury.
This review will focus on ES to enhance spinal circuit functions, the strength and efficacy of spared motor pathways, and the ultimately motor recovery after SCI. For each approach, we address the questions of the mechanisms recruited and the neural target engaged by the stimulation. We describe animal behavioral studies demonstrating efficacy in motor recovery after injury. Further, putative mechanisms for repair of the CST, a key pathway for movement in humans and many animals, and improved recovery of motor function, will be discussed. These mechanisms serve as the basis for translating ES in animals with SCI to humans. Each of the studies discussed in this review is based on similar, but different, ES protocols and stimulation parameters (and therefore potentially different mechanisms). Because of this, direct comparison among the different studies is difficult. Also, the animal and injury models used in experiments from each study vary substantially (a list of which, including ES parameters and major results, can be seen in Appendix Table A1).
Promoting Spinal Neuronal Outgrowth with Weak EFs Applied to the Lesioned Tissue
Spinal cord ES was initially explored to target axons that were damaged after SCI in order to bias the direction and possibly extent of neurite outgrowth. Evidence suggests that spinal cord direct current ES creates a relatively static EF. Importantly, SCI in lamprey was reported to rapidly create an EF at the injury site, likely based on Na+ and Ca++ influx, possibly important for the regenerative response of the neurons.7 This is similar to what is seen in wound healing, where injury-induced EFs have been said to direct cell migration and growth8,9 and developmental biology, where neural tube formation can greatly be affected by ES.10,11 Various cellular changes caused by an EF have been discussed, including asymmetrical distribution of charged protein/channels, redistribution of the actin cytoskeleton, changes in the localization and expression of focal adhesion proteins, and nchanges in the Rho pathways,12,13 and a major factor linking these effects seems to depend on voltage-gated ion channels, and possibly flow of Ca++ ions.14
Although ES and the generation of EFs for promoting outgrowth dates back nearly a century,3,4 its application to SCI in animals has been much more recent.5,15–24 With a few exceptions,22–24 the majority of early studies examining the rationale behind the use of ES post-SCI in animal models have emanated from the laboratory of Borgens and colleagues.5,7,15–21 It should be noted that in these early studies, Borgens and colleagues used both static direct current (DC) and alternating or repetitive ES. Because of the potential damaging effects of applied DC, studies generally used very low currents (on the order of 10s of microamperes in guinea pigs, and 600 μA in dogs) that generally do not activate spinal circuits or muscle.25–27 Only recently have experimental and computational studies begun to calibrate the EF values necessary for producing biological/biophysical effects. The extent to which microampere-level body surface currents will generate sufficient fields in the spinal cord needs to be validated by direct measurements or informed by predictive modeling. In this way, we will be able to evaluate if this form of stimulation weakly modulates neuronal membrane potential to bias network function, as proposed for transcranial DC stimulation.28
Much of the work from Borgens' in-vivo studies in injured spinal cords has been based on the findings that neurons respond to EFs in vitro. EFs alter local ion gradients, which is thought to encourage growth of lesioned axons. The idea that axons demonstrate outgrowth toward a cathode and regress from an anode in vitro has been around for quite some time.29,30 In an in-vitro experiment in which EF polarity was reversed, McCaig found that there was an asymmetrical response in neurite outgrowth with the cathode-facing outgrowth being faster than anode-facing regression).31 This suggested that neurite outgrowth could be achieved in opposite directions by reversing the EF polarity. The Borgens group applied these ideas to several of their own models of SCI. Here, we will discuss their use of epidural ES to produce an EF at the site of SCI in order to promote axonal outgrowth into the lesion. However, this is not to be mistaken for epidural ES or intraspinal micro-stimulation below the SCI, approaches used to excite local circuitry and/or central pattern generators to evoke or facilitate functional movement patterns. This approach has been studied in humans and animals, respectively, and will be discussed later in this article in the context of the clinical applicability of ES and EFs.
One of the first studies to examine the effects of an imposed EF on spinal cord axon regeneration was performed in a complete spinal cord transection lamprey model.5 In this experiment, larval lamprey underwent ES in the form of 10 μA DC applied across the spinal cord lesion for 5 or 6 days. A strength of this model for elucidating the mechanisms by which applied fields can promote axon outgrowth is that stimulation was delivered directly to the spinal cord using indwelling Ringer-based electrodes. This ensured delivery of current to the injury site and a reasonable estimate of current value. Following the stimulation-induced enhancement of their constitutive capacity for axonal outgrowth, Mauthner cell axons were dye filled to reveal a significantly higher number of axonal processes growing into and across the lesion in stimulated versus sham animals. Intracellular recordings demonstrated action potentials crossing the lesion.5 Subsequent studies investigated ES after complete and partial transection in a guinea pig model. These studies showed increased axonal growth in the experimental groups into, but not across the lesion site.17,18 Although these studies did not include behavioral data, a follow-up study demonstrated functional recovery in a group of electrically stimulated guinea pigs when the EF was applied rostral to the site of the lesion.16 Moreover, further experimentation with DC ES to produce a static field in awake dogs with complete paraplegia and implantation of an ES pack promoted moderate functional recovery.21
In the dog model, an oscillating field stimulator (OFS) was used based on the idea that alternating the polarity of an EF can result in neuronal outgrowth in opposite directions. It was reasoned that alternating the polarity might promote outgrowth of ascending sensory fibers in one direction of current flow, and outgrowth of descending motor fibers in the other. Whereas polarity-dependent effects with DC ES have been reported,6,29–31 oscillating ES may directly activate axons that can trigger upregulation of growth-promoting signaling pathways in neurons.
The success of early studies of ES of the spinal cord led to the first clinical trial in humans in 2005.32 This was a phase 1a study and consisted of an OFS device implanted in 10 complete SCI patients (level C5-T10). The device was placed within 18 days of injury with the electrodes spanning one segment on either side of the level of injury in an extra-spinal location (no contact with any neural elements). The device was left implanted for 15 weeks, after which time it was removed and its function tested. Outcome measures from this study included ongoing neurological examination by an unblinded study neurologist, surgeon, and research nurse using the American Spinal Injury Association (ASIA) Impairment Scale (AIS), somatosensory evoked potentials (SSEPs), and the Visual Analog Scale (VAS) for pain at baseline, 6 weeks, 6 months, and 1 year post-implantation. The trial concluded that the surgical procedure and OFS implant were well tolerated and safe, and resulted in improvement in all parameters assessed. This resulted in approval of further patient recruitment, in which an additional four patients were included and compared with 14 historical control patients. Results from this expanded study were subsequently filed in a report to the Securities and Exchange Commission by the owner of the technology.33 Patients treated with OFS showed marked improvement in all domains assessed, with the exception of motor recovery, a significant limitation to the trial. However, as pointed out by Tator, much of the enthusiasm from this preliminary and seminal clinical work is tempered by the unblinded nature of the assessments, the small number of patients included, minor to moderate neurological recovery, lack of a control group, and lack of a peer-reviewed publication.34,35
ES to Increase Excitability or Directly Activate Spinal Neuronal Networks below the Level of Injury
The second distinct mechanism of action for ES following SCI is to increase the excitability of neuronal networks in the spinal cord or directly activate distinct neuronal motor pools. In contrast to experiments in which the creation of an electric field was the intent, the applied currents are higher and are not applied at the lesion site, but rather below the level of the injury (Figure 1). This field of research is now commonly referred to as “neuromodulation”; however, historically, neuromodulation in the field of motor control was not limited to ES. Research on neuromodulation was pioneered in various in-vivo animal models and in in-vitro preparations. The first form of neuromodulation described is likely based on research in decerebrate cats in which stimulation of the mesencephalic locomotor region (MLR), a small region in the brainstem, could initiate and modulate stepping. Interestingly, the higher the intensity of this stimulation, the faster that animals would walk, even changing their gate from walking to trotting and ultimately to galloping.36 In order to explore spinal pattern generating networks for locomotion in isolation and to learn how to imitate their activity, isolated spinal cords (in vitro) were utilized as well as adult cats37–39 and rats40,41 with a complete thoracic spinal cord transection. Various groundbreaking in-vitro experiments with spinal cords from neonatal rats not only showed that pattern-generating networks can produce rhythmic output without descending motor and sensory input, but also demonstrated the powerful effects of peptides, such as dopamine or serotonin, in triggering and modulating network activity.42,43
The seminal work of Barbeau and Rossignol44,45 explored the role of monoamines and noradrenergic and dopaminergic drugs in cats with spinal transections. These animals were trained to step on a treadmill, and represented an optimal model to study the spinal networks in vivo. They found that neither serotonergic nor dopaminergic agonists successfully induced locomotion, which was, however, achieved with noradrenergic precursors and the agonist clonidine. Further, Barbeau and Rossignol reported that serotonergic agonists and precursors modulated the stepping pattern, suggesting that these drugs increased excitability of the spinal networks.44,45 These studies pioneered the field of chemical neuromodulation after SCI, and laid out the path for various possible treatment approaches. One of these approaches was to implant serotonin-producing cells into the spinal cord of rats with complete spinal transections to compensate for the lack of descending serotonergic innervation, and thus to enhance the excitability of spinal networks.46,47 In both cases, this approach was reported to restore coordinated hindlimb movements in the animals.
Resulting from years of studies in cat locomotion, another powerful neuromodulatory factor that has been frequently employed, but rarely reported, is cutaneous stimulation or exteroceptive stimuli. This approach of gentle mechanical stimulation of the perineal area in cats was regularly employed to initiate walking. However, this has rarely been considered for clinical translatability.48–50
Currently, the term “neuromodulation” has been somewhat misused, as it is generally referred to as ES-induced modulation. Considering the current excitement regarding ES-induced neuromodulation, it is surprising to learn that this approach has been around for many years, and originated in the field of pain control.51 Another fact is that ES of spinal networks to enhance motor function was performed in humans long before it was considered in animal models of SCI. In 1980, Sherwood and colleagues explored epidural stimulation to enhance motor function in individuals with upper motoneuron disorders,52 and in 1998 it was shown for the first time that lumbosacral epidural stimulation can facilitate rhythmic motor activity in functionally complete SCI.53 The human trials and possible mechanism of where stimulation exerts its modulatory effects will be discussed in more details in subsequent sections of the review.
In animals, epidural stimulation was frequently used as a tool to explore pattern- generating networks in the spinal cord. These studies showed that placing electrodes above the cervical or thoracic spinal cord could trigger quadrupedal or bipedal locomotion, respectively, in cats.54 Various later studies in cats39 and rats41 have bolstered knowledge about the use of epidural stimulation in animal models and evolved as treatments for SCI.55,56 More recently, studies have focused on combining epidural stimulation of spinal circuitry with other treatment approaches, including training and pharmacological neuromodulation.41,56–60 These studies have been summarized in detail in excellent reviews, and the reader is referred to these articles for specific details on the stimulation parameters.61–64
Considering that the specificity of neuromodulatory epidural stimulation is limited, and the result is typically a subthreshold stimulation increasing neuronal excitability, approaches to stimulate spinal networks directly and in a more targeted fashion have also been explored. For example, a somewhat different approach is to use epidural stimulation to produce a spatiotemporal activation configuration that produces a locomotor pattern. This approach to epidural stimulation has been pioneered by Courtine and collaborators, by whom a stimulation paradigm was developed to activate muscle synergies to improve locomotion.65 This approach has then been combined with a closed-loop brain–spine interface, in which it was reported to restore weight-bearing locomotion in non-human primates66,67 and rats.68
Another approach for a more targeted stimulation of specific neuronal populations is intraspinal micro-stimulation, which requires the direct implantation of electrodes into the spinal cord (enabling more precise targeting of interneuron and motoneuron pools). The cost of increased invasiveness from this approach comes with the benefit that targeted stimulation can evoke specific and sequential movements such as leg extension or flexion,69,70 preferentially by recruiting fatigue-resistant muscle fibers.71 With a large number of electrodes and appropriate orchestration of the stimulation at different spinal locations, overground stepping with propulsive and supportive forces could be elicited in anesthetized cats, often for very extended periods of time.72
An interesting vision for the future is to control such intraspinal stimulation with cortical activity, to control movements while bypassing the injured spinal cord.73–75 The value of intraspinal stimulation has also been explored at the cervical (C4-C5) level to evoke forelimb function in rats following a severe, lateralized contusion injury76 and in spinally intact primates,77 from which a variety of hand and arm movements were elicited. However, the question remains whether the highly complex stimulation patterns required to control arm and hand function can be achieved using this approach. Similarly, intraspinal stimulation was recently reported to promote diaphragm activity.78 In these elegant studies, medullary respiratory input in rats with cervical hemisection was utilized to trigger cervical intraspinal stimulation to activate the diaphragm.
Less invasive than either intraspinal or epidural stimulation is transcutaneous stimulation, in which electrodes can be easily applied and removed (reviewed in Mayr and coworkers79). Similar to epidural stimulation, this field was also pioneered in studies on pain, in which transcutaneous nerve and dorsal column stimulation were reported as effective modalities for pain treatment.80,81 The ability of transcutaneous stimulation to evoke and modulate spinal reflex function reliably was demonstrated in various studies, and made it an attractive approach to treat and assess SCI.82,83 It was suggested that transcutaneous stimulation was able to activate large diameter afferents in both dorsal84,85 and, depending on location, ventral roots.86–88 Consequently, transcutaneous stimulation has been used as a tool to assess supraspinal and spinal connectivity,89 and has already produced promising and persistent results in treating individuals with cervical SCI when combined with training.90 Recently it was reported that even a single session of transcutaneous stimulation in individuals with incomplete SCI had excitatory effects on spinal and inhibitory effects on cortical circuitry, possibly contributing to recovery.91
Clinical studies
A major advance in translating spinal stimulation to humans with SCI followed the success of the animal studies showing efficacy of spinal cord epidural stimulation to enhance the excitability of spinal cord networks below the level of injury and promote locomotor function. Studies from the Harkema and Edgerton groups use epidural stimulation applied via a 16-electrode array over spinal cord segments L1-S1 during standing training following motor complete SCI (AIS A or B). After successful implantation of their epidural stimulator in one AIS B patient resulted in limited weight-bearing and locomotor-like leg movements, they found that additional SCI patients implanted (AIS A, n = 2; AIS B, n = 2) displayed full weight-bearing ability during ES, as well as stepping-like patterns of movement when ES parameters were optimized for locomotion.92–94 Whereas these participants were able to voluntarily move their lower extremities in the presence of epidural stimulation, this capacity was lost when the stimulator was turned off.
Stimulation and training
In a study by Gill and coworkers,95 epidural stimulation was combined with locomotor training over 43 weeks in an individual with a chronic thoracic SCI. The use of an interleaved stimulation paradigm allowed bilateral leg movements, and was seen as a key to the success of the stimulation. The overall recovery was remarkable and ranged from independent treadmill walking to overground walking with an assistive device (walker). This study was an impressive demonstration that a lesion originally considered complete allowed for voluntary movements when epidural stimulation was applied. In another study from the Harkema group, it was found that individuals were able to achieve some degree of independent standing and trunk stability for a limited period of time with stimulation and intense training (standing with/without assistance 1 h/day, 5 days/week for a total of 80–82 sessions; stepping with body weight support on treadmill 1 h/day, 5 days/week for a total 80–84 sessions). Interestingly, the study also investigates the effects of task-specific rehabilitative training, akin to earlier cat studies by the Edgerton group,57,58,96–98 providing insight into the mechanism behind the improved motor function that they observed with ES. As a part of their study protocol described, the four individuals were trained to stand in combination with ES, and subsequently trained to step in combination with a different set of ES parameters optimized for evoking locomotor electromyographic (EMG) patterns. They found that this subsequent step training impaired three out of four individuals' ability to stand.99 Similar task-specific training impairing the ability to perform alternative motor tasks has been reported in cats and rats with complete SCI.100,101 More recent studies have also reported the efficacy of ES in promoting recovery beyond motor function. More specifically, promising findings on the beneficial role of ES on cardiovascular function, bowel/bladder continence, and sexual function in SCI individuals has increased awareness on other ES-based therapies.102–105 Although many questions remain such as the underlying mechanism for ES-based therapies, the generalizability, and larger, longer-term follow-up results from such studies, the preliminary studies mentioned represent a promising step forward in the treatment and recovery potential for SCI.
Mechanisms of recovery
The mechanism behind the motor recovery facilitated by epidural stimulation is not yet resolved. One plausible explanation is that the ES-induced modulation of interneuron and motor neuron excitability distal to the SCI may allow afferent input to enhance and optimize neural activity within the spinal cord. This idea is supported by studies using computer simulations and behavioral experiments66,106,107 that suggest that it is large diameter afferent fibers that are being stimulated by the ES. This stimulation of afferent proprioceptive fibers likely occurs in the dorsal roots, thereby activating segmental interneurons and motoneurons.107,108 This is consistent with the task-specificity of training, as the afferent information and neural activation patterns differ between standing and stepping (continuous bilateral activation of extensors for standing and a rhythmic flexor-extensor pattern for stepping). It is also consistent with the fact that epidural stimulation parameters must be tailored to individuals in order to produce weight bearing.94 Further, sufficient EMG activity for weight bearing in lower limb muscles has not been induced by epidural stimulation in a sitting position, and requires the sensory information from standing.
It is worth noting that studies investigating the use of spinal epidural electrical stimulation in humans, as well as in animals, have not reported any significant harm related to the use of ES. That is to say, no neurological deterioration of intact limbs, or increase in pain spasticity/spasms have been observed in the stimulated cohort.18,20,21 Although this may also be related to the small number of SCI patients having undergone ES implantation for the purposes outlined in this article, the lack of significant adverse events and its importance should not be underappreciated. This is especially true in light of the fact that the full effects of increasing excitability of spinal cord circuits post-SCI are as of yet unknown, and the mechanisms have yet to be fully elucidated. It is only through the rigorous protocols and studies such as those outlined here that progress will be made in ES-based treatment applications for SCI.
Phasic Electrical Stimulation to Increase Neuronal Axon Growth Promoters and Intracellular Signaling
The third mechanism for ES after SCI is to increase intracellular signaling in stimulated neurons to upregulate axon growth-promoting factors and regeneration-associated genes in injured and spared neurons. Different from the approaches discussed previously regarding the stimulation, ES after SCI is generally applied to the cell body of the long-axon projection neurons. As such, in this case, the intention is to repair injured corticospinal axons by targeting their cell bodies in the cortex by ES (see Figure 1).
We distinguish two forms of axonal outgrowth: regeneration and sprouting. Axonal regeneration is defined as outgrowth of an axotomized neuron from its terminal, lesioned end at the site of injury, whereas axon sprouting is defined as outgrowth from spared axons or axotomized neurons proximal to the site of injury.109 Mature neurons have a reduced intrinsic capacity for axonal outgrowth that severely hampers both axonal regeneration and sprouting.110 The principal goal of phasic ES is to enhance this axon growth capacity and, in turn, produce structural, and possibly genetic, changes that are durable and can persist after the stimulator is turned off. Phasic ES can be distinguished from subjecting injured neurons to very weak currents and EFs, in that the former are expected to activate the neuron to produce action potentials or to raise excitability close to threshold. The latter on the other hand may bias transmembrane functions and Ca++ influx in more subtle ways.
The use of phasic ES triggers action potentials (as evidenced by c-Fos expression and downstream EMG responses) at and rostral from the site of SCI (such as in upper motor neurons upstream of the injury or downstream sensory neurons).111,112 Here, the objective of ES is to engage mechanisms, including elevating cyclical adenosine monophosphate (cAMP), the growth factor brain derived neurotrophic factor (BDNF) and mammalian target of rapamycin (mTOR), and Janus kinases/Signal transducer and activator of transcription proteins (Jak/Stat) associated signaling, to promote axonal outgrowth. These ideas and their link to ES of the CNS and its descending pathways have been gathered initially from the peripheral nervous system and the growth-promoting effects of a peripheral nerve conditioning lesion. Further, studies have identified that the reduced axon regenerative capacity of mature neurons can be enhanced, like that of immature neurons, by manipulating neuronal activity and signaling.
Peripheral conditioning lesions
Conditioning lesions consist of injuring the peripheral branch of a sensory axon from the dorsal root ganglion (DRG) followed by the CNS branch in a delayed fashion (for example, 7 days later). In the absence of a conditioning lesion, the CNS axon branch fails to regenerate; it cannot penetrate the dorsal root entry zone.113 However, by first lesioning the peripheral branch, increased axonal outgrowth into the central lesion is observed.114–116 The first, peripheral lesion, primes the DRG neurons and upregulates regeneration/growth associated gene expression so that when their central branches are lesioned there is regenerative capacity. The initial lesion upregulates cAMP, which, as discussed earlier, is thought to contribute to the enhanced regenerative capacity.
Interestingly, in a combined conditioning lesion and ES experiment, peripheral nerve ES was shown to increase axonal outgrowth of the injured nerve's central projection into a central spinal cord lesion and increase cAMP levels to the same extent as a conditioning lesion.113 The mechanism by which increased axonal outgrowth after ES, similar to a conditioning lesion, is thought to be mediated partially through cAMP. In fact, injecting cAMP into the DRG can mimic the effect seen by the conditioning lesion.117 It has been shown that cortical and spinal cAMP levels decrease following SCI,118 and that these levels can be partially restored with rehabilitative training.119
A brief period (1 h) of phasic electrical stimulation has been used to promote the long-term process of motor axon regeneration and muscle targeting by Gordon and colleagues.113,120,121 In this model, ES has been shown to increase the expression of BDNF and its receptor tyrosine receptor kinase B (TrkB).122 BDNF is a multifaceted neurotrophin whose breadth of effects is beyond the scope of this review; however, a full review can be found elsewhere.123 Among its many actions, BDNF has been shown to stimulate axon growth via the extracellular signal regulated kinase (ERK) pathway.123 Based on these findings, akin to a peripheral conditioning lesion, increased cAMP and BDNF levels resulting in increased axonal outgrowth represents a plausible and potential mechanism for ES-induced axonal outgrowth.
ES to upregulate CNS axon growth signaling
Martin and colleagues have applied epidural ES to CST axons in the medullary pyramid or epidural ES to the MCX, the principal source of CST axons, to promote CST axonal sprouting of spared CST fibers in the spinal cord.25,124–127 This is a very different approach from that of other studies discussed, and was based on earlier findings from the Martin group that CST ES promoted axon sprouting during development, whereas CST inactivation retarded outgrowth.26,128–132 The stimulation pulse pattern, which is a phasic high-frequency burst (45 ms duration; 333 Hz; 6 h) in mature animals elevates mTOR and Jak/Stat signaling.111 Both pathways are downregulated during development, as axons lose their intrinsic capacity to regenerate, and are subsequently upregulated with ES. Stimulation for 10 days in intact rats produced significant sprouting of ipsilateral CST axons, which was used as an outgrowth assay. Interestingly, blocking mTOR signaling with rapamycin during the stimulation period eliminated all stimulation-dependent outgrowth.111 By contrast, blocking Jak/Stat signaling with AG490 did not affect axonal outgrowth, but prevented stimulation-dependent CST pre-synaptic site formation. Genetic upregulation of these signaling pathways has been shown to promote CST axon regeneration, as well as sprouting.133–135
Using their CST ES model, the Martin group demonstrated that after a pyramidal tract lesion, which eliminates all CST fibers from one hemisphere and almost all projecting to the contralateral spinal cord, ES of the intact CST or MCX for 10 days causes significant ipsilateral CST sprouting into the denervated side.25,126 Importantly, stimulation-dependent CST sprouting is able to complement injury-dependent outgrowth: although both injury and ES promote axonal sprouting in rats, the effects are, in fact, additive.25 Rats undergoing stimulation and pyramidotomy had a significantly increased stepping-error rate on the horizontal ladder task compared with the rats with injury only. Further, by the end of the post-operative testing period, there was no significant difference between the stimulation group and their error rate before injury.125,126,136 Errors remained elevated in the unstimulated group. The errors that did occur in the stimulation group were comparable with their baseline errors, arguing against behavioral compensation or adaptation.136 To investigate whether or not the stimulated MCX was responsible for the observed behavioral effects, muscimol (a GABAA agonist) was injected into the stimulated MCX. This resulted in the reversal of the behavioral improvements seen in the stimulated rats compared with controls. Once the muscimol effects had worn off, the rats' motor deficits once again improved to baseline.124,136 These experiments demonstrate that the ipsilateral MCX is a viable target for ES in order to promote recovery following SCI. In this model, restoration of function may be mediated by the uninjured CST and/or indirect cortical projections to the segmental circuits.
These studies raise an interesting question about which anatomical pathways to stimulate in order to maximize recovery of motor function: the undamaged or the damaged pathways. The studies described utilized unilateral lesions and stimulated the undamaged pathway to promote sprouting to the ipsilateral, denervated, side of the spinal cord. The CST in primates has been shown to have a higher number of midline crossing branches than the CST in rodents,137,138 making spared ipsilateral CST fibers an optimal target for promoting recovery in primates after an incomplete lesion. Further, microstimulation experiments investigating the use of the uninjured CST have shown that at higher current thresholds, cortical stimulation (to affect the uninjured CST) can generate similar movements to those seen contralaterally.127 Following injury to the CST, efforts to improve functional outcome and recovery have traditionally centered on trying to repair damaged connections or preserve spared connections from the injured CST. Alternatively, treatment can be aimed at strengthening and expanding connections from the uninjured CST so that it assumes control of both sides of the body.
For a midline cervical SCI, the CSTs from both hemispheres are damaged. To begin to address translational repair strategies, the Martin laboratory has applied stimulation bilaterally in a rat C4 midline contusion model.112 As could be expected from using such a different model, the proposed mechanism by which ES promotes axonal outgrowth is also substantially different from that described previously with EFs and epidural stimulation below the level of injury. In the rat, the majority of CST axons are located centrally and are eliminated after a contusion injury. What remain are sparse CST axons in the lateral and ventral white matter columns. To address weakened CST projections, the Martin group modified the pyramidal tract ES protocol in two ways. First, they developed a patterned ES based on the theta burst stimulation protocol (TBS) using transcranial magnetic stimulation (TMS) in humans to promote long-term potentiation (LTP) of motor-evoked potentials (MEPs).139 Thirty minutes of TBS strongly facilitates evoked motor responses, similar to LTP. Second, when MCX ES is combined with concurrent cathodal DC stimulation of the cervical spinal cord (termed trans-spinal DC stimulation [tsDCS]), which strongly potentiates MCX-evoked muscle responses.112,140 It was reasoned that cathodal tsDCS would facilitate the actions of the MCX ES.
Ten days of daily stimulation, beginning 1 week after a C4 contusion injury, produced significant sprouting of spared CST axons below the injury, as well as above the injury.112 This was associated with improvement in manipulation and walking (horizontal ladder task) skills compared with the unstimulated group. Interestingly, the ES protocol promoted rostral sprouting that was very robust and primarily involved the axotomized dorsal CST, which contains most of the CST axons. Considering the various studies of the Borgens laboratory showing axonal outgrowth in the presence of EFs, the tsDCS may have also promoted sprouting of damaged CST axons rostral to the injury. The benefit of this dual stimulation protocol (intermittent TBS [iTBS] in MCX and tsDCS of the cervical spinal cord) on motor improvement after C4 contusion in rats was replicated by an independent laboratory, using the Martin laboratory experimental protocol.141
Injury-dependent sprouting has also been shown to correlate with functional recovery after SCI.142,143 The expectation is that with ES, the growing connections may be more effective than without ES. ES after SCI appears to boost injury-dependent sprouting and, importantly, helps to refine new connections to increase their potential for becoming stronger and more functional.
More recent work examined the effects of cortical ES of the CST prior to its injury in rats,144 in which the (to be) injured CST underwent a conditioning ES (in contrast to the uninjured CST being stimulated in the pyramidal tract lesion studies published by Martin and colleagues).25,124–127 The results showed that the ES increased collateralization and reduced axonal dieback depending on the ES frequency employed (20 Hz vs. 333 Hz). A more clinically relevant approach recently reported comparable results and enhanced functional recovery when the cortical ES of the lesioned CST was performed subsequent to its spinal lesioning.145
Whereas the stimulated cortex is responsible for behavioral improvement, at least after pyramidal tract lesioning,124,136 many questions remain. For example, what capacity do extrapyramidal tracts display for ES-induced outgrowth and recovery? Many of the studies discussed have pertained to the CST; however, in another study exploring the effect of ES on the rubrospinal tract, compared with the control group, ES failed to result in BDNF upregulation, or an enhanced growth response.146 Another issue is the role that CST sprouting caudal to the injury plays in behavioral recovery versus sprouting at sites rostral to the injury. In addition to spinal CST sprouting, MCX ES increases outgrowth of corticofugal axons into the forelimb area of the red nucleus and into the medullary reticular formation.125 The relative amount of new connections in the brainstem and rostral spinal cord that each contributes to the improvement of motor impairments remains unknown. In addition, although increased axon varicosity density correlates with increased axon density, the role of these new axon growth terminals has not yet been established. Whether they are synapsing onto motoneurons or interneurons, or play an excitatory role or an inhibitory role for example, has yet to be determined.
To summarize, studies in which peripheral nerves are stimulated show that ES increases cAMP and BDNF to enable both enhanced muscle target selection peripherally after motoneuron peripheral axotomy, and sensory fiber central branch regeneration through the dorsal root entry zone after injury. Moreover, studies of central neurons of the CST show that ES elevates mTOR and Jak/Stat intracellular signaling, which is necessary for axonal outgrowth and synapse formation. It remains to be determined how best to target the disparate axon growth-regulating mechanisms to restore function after SCI. Further, using different lesion models, axonal outgrowth and some recovery of motor behavior has been demonstrated after ES of damaged and undamaged CST projections.
Pairing Stimulation To Promote Transmission In Specific Pathways
The fourth mechanism for ES after SCI is to leverage the timing of paired stimulation to produce plasticity. The idea is based on work from Hebb proposing that synaptic plasticity occurs when pre- and post-synaptic neurons are active together within a narrow time window.147 This process is frequently referred to as “fire together, wire together.” This wiring (or synaptic adaptation) can naturally occur when multiple neurons that are active simultaneously connect to a common target and can thus activate it. In vitro, this can be recapitulated where it has been demonstrated that synaptic strength can be amplified by precisely timing pre-synaptic activity and depolarization of the post- synaptic cell.148
Paired associative stimulation (peripheral and MCX stimulation) has been used for some time.149–155 The approach of paired associative stimulation or paired pulse stimulation can be used to potentiate either voluntary movements or the motor response to another stimulus. The idea is that if the timing is correct, the neural consequences of the two stimuli will interact in adaptive ways and, one hopes, lead to persistent forms of plasticity. Such stimulation can use a single site (e.g., MCX ES) or two different sites (e.g., MCX and a peripheral nerve or muscle). The interstimulus interval (ISI) is typically adjusted so that a peripheral stimulus activates the cortex at the time that the MCX stimulus is triggered, implying a cortical locus for plasticity. Of particular interest is when the ISI enables facilitation at the spinal cord level, implying plasticity at the spinal level.150,151 This convergence of descending and afferent signaling may be on common post-synaptic interneurons or common neural populations.151 Unlike in rodents where the CST does not make corticomotoneuronal connections, in the human, convergence for the CST would additionally be expected to be on motoneurons, which may make this modality more impactful than signal convergence occurring solely on interneurons.
In an example of dual stimulation at a single cortical site, Perez and colleagues used a TMS pulse interval based on the timing of the normal pattern of MCX-evoked activation of the CST, which recruited strong temporal summation in cervical SCI subjects.156 This resulted in stronger MEPs and, remarkably, the participants were able to perform basic motor tasks (involving hand dexterity) better after the therapy. The time intervals are tailored to the timing of synaptic events evoked by an MCX stimulus. If the stimulus mimics a phasic movement control signal, then the synaptic events associated with that signal are partially replicated with the paired stimulation protocol. Importantly, if alternate timing is used, there is no MEP plasticity or improvement in motor function. This is an example of a stimulation protocol that can be coupled with motor training to enhance the efficacy of rehabilitation. For two stimulation sites, such as motor cortex and the periphery (see Figure 1), the interstimulus interval can be adjusted so that the evoked responses from stimulation of both sites converge at the spinal level.
Following their work in rodents and primates, Courtine and colleagues57 applied epidural stimulation to activate motoneurons via proprioceptive circuits in the dorsal roots in individuals with chronic severe SCI. This technology was applied to selected roots in a timed manner triggered by residual movements while the participants were walking in a gravity assist device.157 This approach resulted in immediate recovery of locomotor function, and over a few months, enabled recovery of voluntary control of originally paralyzed muscles even without stimulation. Underlying mechanisms in this scenario was an increase in motoneuron excitability.
An important new development is to substitute one “stimulus” in a paired-stimulation protocol for a naturally occurring neural event, such as a recorded action potential, local field potential, or EMG response. In primate studies, spontaneous EMG responses or cortical activity have been used to trigger spinal cord microstimulation to strengthen the ventral spinal cord connections mediating the EMG response.158,159 After cervical injury, this approach can lead to improvement of motor function for 3 additional weeks without stimulation.160 Further, the detailed research on electrical activation of spinal circuitry has been rapidly expanding to include closed-loop brain–spinal cord interfaces that were reported to be able to restore weight-bearing locomotion in non-human primates66,67 and rats.68
Approaches that optimize the timing of ES events may lead to greater strengthening of the targeted connections than other approaches. However, as timing is critical to strengthening plasticity, the wrong timing can lead to much weaker connections. For example, in models of spike-timing dependent plasticity (STDP), the stimulus that is being used to strengthen the motor response must precede the response over a critical time window for strengthening to occur. If it follows the response, again within a critical time window, the response is weakened. This is an important concern for translation of approaches that make use of STDP. Similarly, the selective reinforcement via STDP of spared motor pathways and their functional response after injury during the rehabilitative process assumes that the response is adaptive. If indeed adaptive, the use of STDP technology and mechanisms would support improved motor control if strengthened.
Synthesis and Future Directions
ES offers itself as a straightforward treatment approach and is currently explored primarily for neuromodulation to excite neuronal networks directly. It is important to recognize that as rehabilitative training may not be feasible in the setting of acute trauma, ES may be an alternative if both it and rehabilitative training work via complementary mechanisms. ES can be applied during the acute injury period when rehabilitation is not possible, in order to begin to repair damaged neural networks.
As discussed, ES to promote recovery after SCI is more than simply increasing the excitability of the spinal cord neural networks caudal to the injury. An entire body of literature exists supporting the notion that different modalities of ES can help promote and direct axonal outgrowth. We are now beginning to understand some of the cellular mechanisms that underlie the capacity of ES to promote axonal outgrowth. Animal studies show that more active inputs to neurons in spinal motor circuits, and the spinal neurons themselves, are able to secure more and stronger connections than their less active counterparts reflecting activity-dependent synaptic competition.26,124 If this is similar to the normal establishment of synaptic connections, where use-dependent processes and training strengthen connections that support function and (in the case of development) eliminate unnecessary connections, it helps to explain how ES-assisted rehabilitation could lead to stronger and more effective connections. Choice of which pathway to stimulate is particularly critical with ES because of the novel activity-dependent competitive interactions that ES may produce; stimulation of the wrong pathway could be detrimental to recovery, akin to what is seen in task-specific training. Given our limited understanding of the role of different motor pathways in everyday movements (apart from the CST) and the importance of afferent input in facilitating movement, we have even less of an understanding of which pathways are best promoted after injury.
It is important to understand how ES can be applied to the chronically injured person. Whereas it is generally hypothesized that there is an early time window for axon growth into and through a lesion site, activity-dependent CST axon sprouting may occur weeks after injury. With scar formation after SCI, there likely will be a time window in which electrical stimulation needs to be implemented to promote axonal growth at and through the injury site, and behavioral recovery. Nevertheless, spinal ES in chronically injured people may lead to outgrowth of spared axons below the lesion. Intriguingly, the limited reports of some improvement being observed after persistent stimulation in chronic SCI individuals, as well as after the stimulator is turned off, may be the result of the durable structural plasticity induced by ES.
There are many challenges to translate ES-based approaches to clinical practice. Among these is better target engagement by the stimulation. This assumes that we know what the target is, so that we can optimize electrode placement. Imaging-based finite element method (FEM) modeling is a way to standardize electrode placement and reduce between-study/subject variability, leading to an individualized, patient-specific approach to ES. Dosing, such as for pharmaceutical interventions, will also have to be carefully worked out. Nevertheless, these challenges can be addressed with further animal research in which biological parameters can be rigorously examined in order to translate these minimally invasive approaches to people with SCI, both acutely and chronically.
Acknowledgment
Material in this review was submitted in the thesis of Andrew S. Jack to fulfill the requirements of a Master's in Neuroscience at the University of Alberta.
Appendix Table A1.
Overview of the Use of Electrical Simulation following Spinal Cord Injury
| Study | Animal and injury model | Electrical stimulation model | Parameters | Experimental groups | Outcome (histological and behavioral) |
|---|---|---|---|---|---|
| Borgens, Roederer and Cohen (1981) | -Model: ammocoete larva of the lampre, transection model of injury | -5-6 days | -Intensity: 10uA, constant current -Electrode: saline bridge as wick electrode across the transected spinal cord |
-ES with transection (n = 11) -Sham ES and transection (n = 13) |
-Electrophysiological: Action potentials (APs) conducted in both directions across lesion in ES group, no APs in sham group -Histological: active growth in and across the lesion more common in ES-treated group |
| Borgens, Blight et al. (1986) | -Model: Hartley female guinea pigs -Injury: Low-thoracic dorsal transection |
-Implanted direct current (DC) ES (epidural) across the SCI for mean 51 days | -Power supply: DC, 9V (3x3V in series) LiMn battery -Electrodes: Ag/AgCl on either side of the SCI |
-ES: 1uA (n = 4) -ES: 5uA (n = 6) -ES: 10uA (n = 11) -Sham ES (n = 11) |
-Histological: ES showed more regeneration into scar; 10uA group extended into spinal cord below the lesion |
| Wallace, Tator et al. (1987) | -Model: Wistar female rats -Injury: T6-7 extradural clip compression (125g for 1min) |
-Implanted continuous DC ES (epidural) across the SCI for 15-20 weeks | -Voltage applied: <340mV (minimum for muscle twitch) -Electromagnetic field: 460kHz -Power supply: DC 9V ES group -Frequency: 10Hz -Electrodes: Pt on each side of the SCI (cathode proximal and anode distal) |
-Sham ES with SCI (n = 10) -ES with SCI: <340mV (n = 10) |
-Behavioural: 1. Inclined plane: no difference between groups -Neurophysiological: no difference between groups -Histological: no difference in regeneration, scarring, or lesions between groups -Adverse Events: 9/20 with single electrode dislodgement; 6/20 both electrodes dislodged; no changes in EM field based on position/location of rat |
| Maiman, Myklebust et al. (1987) | -Model: cats -Injury: T8 contusion with 20g weight drop from 25cm |
-Intermittent (i.e., not continuous) DC ES monopolar below and bipolar (epidural) across the SCI, 3-5 months post-SCI examining effects on spasticity | -Intensity: <1.0mA -Frequency: 100Hz for 25ms -Electrodes: Pt |
-Multiple trials in same experiment (n = 14): -Monopolar ES 100Hz for 25ms with negative electrode below SCI -Monopolar ES 25Hz for 25ms with negative electrode below SCI -Bipolar ES 100Hz for 25ms with negative electrode below SCI |
-Behavioural: 100Hz group had less intense spasms than 25Hx group; Bipolar ES across SCI less effective than monopolar ES -Histological: gliosis and cyst formation at SCI site in all groups, with no changes with electrode use |
| Politis and Zanakis (1988) | -Model: Wistar female rats -Injury: T8 contusion with 350g weight drop from 1cm against ventral plate |
-Implanted continuous DC ES (epidural) across the SCI for 3 weeks | -Intensity: 3uA -Power supply: 1.5V zinc/alkaline/silver oxide battery -Electrodes: 90/10 Pt:Ir attached to epidural pad |
-SCI only (n = 6) -No ES or SCI (normal) (n = 4) -ES with SCI anode rostral (n = 6) -ES with SCI cathode rostral (n = 6) |
-Behavioural: 1. Hindlimb use: no difference between ES groups, both better than control 2. Inclined plane: Cathode rostral group better than anode rostral (p < 0.05), which were better than SCI only (p < 0.05) -Histological: Cathode rostral group had more neurofilament stained axons than other groups |
| Dimitrijevic, Gerasimenko and Pinter (1998) | -Model: Humans -Injury: complete (AIS A), chronic (>1yr) SCI |
-Quadripolar epidural stimulation at vertebral level T11-L1 | -Frequency: 25-60 Hz, 0.2-0.5ms -Intensity: 5-9V |
n = 6, all participants received stimulation | -Rhythmic, alternating stance and swing phases of the lower extremities were elicited with stimulation at the L2 segment |
| Borgens, Toombs et al (1999) | -Model: dogs (various species) -Injury: complete thoracic SCI dogs after intervertebral disk herniation or trauma |
-Implanted OFS (epidural) across the SCI for 6 months | -Intensity: 600μA from constant current stimulator alternating polarity every 15min -Power supply: DC, 3.6V Li battery -Electrodes: Pt:Ir on either side of the SCI |
-ES: 600uA (n = 20) -Sham OFS (n = 14) |
-Behavioural (6-month follow-up): 1. Superficial pain: more recovery in OFS group (p = 0.0002) 2. Deep pain: no difference between groups (p = 0.11) 3. Proprioception and ambulation: no difference between groups (p = 0.43 and 0.22, respectively) 4. Combined score: more improvement in OFS group (p = 0.047) -Neurophysiological: No difference between groups (p = 0.13) -Adverse Effects: no |
| Moriarty, Borgens et al. (2001) | -Model: Sprague-Dawley female rats -Injury: T10 dorsal penetrating SCI |
-Implanted oscillating field stimulation (OFS) (epidural) across the SCI for 4 weeks | -Intensity: 40μA alternating polarity every 15min -Power supply: DC, 3V Li Cell -Electrodes: 90:10 Pt:Ir on either side of the SCI |
-ES: OFS (n = 7; 4 replaced due to stimulator malfunction) -Sham OFS |
-Histological: OFS decreased astrocytes in SCI region |
| Gerasimenko, Makarovskii and Nikitin (2002) | -Model: Humans -Injury: complete (AIS A), chronic (>1yr) SCI |
-Bipolar epidural stimulation from T10-L2 | -Frequency: 1-120 Hz -Intensity: 1–20 V |
n = 8, all participants received stim | -In most cases, L2 stimulation evoked unilateral stepping. Bilateral stepping in three subjects. |
| Herman, He et al. (2002) | -Model: Human -Injury: C5/6 (AIS C), 3.5 years post-SCI |
A pair of Pisces-Quadplus electrodes were implanted over the upper lumbar enlargement | -Long pulse durations (eg, 0.8 msec) were essential -Frequency: 20-60 Hz, found to be comparatively less sensitive -Intensity: above sensory threshold (sense of `parasthesia or vibration') but below that causing motor contraction |
n = 1 -the participant received partial weight bearing training prior to electrode implantation and again afterwards in combination with stimulation |
-Combination of training and stimulation resulted in a well-organized, smoother stepping pattern at higher treadmill rates and self-supported body weight, considerable improvement in endurance and speed during over-ground walking and reduced sense of effort -After four months of over-ground training with stimulation, the subject could ambulate 270 m |
| Gerasimenko, Nikitin and Lavrov (2003) | -Model: cats -Injury: decerebration followed by spinalization at T10-12 |
-Unipolar epidural stimulation from L1-S1 | -Electrodes: spring electrode with disc -Frequency: 1-100 Hz -Intensity: 10-200 uA, pulse duration 0.1msec |
n = 8, all animals received stim | -Locomotor activity occurred with stimulation between L4 and L5 |
| Fujiki, Kobayashi et al. (2004) | -Model: Sprague-Dawley female rats -Injury: T7 hemisection |
Intermittent bipolar ES (epidural) across the SCI, 24h prior to lesioning and repeated every 24h for 7 days | -Voltage applied: double voltage threshold for motor evoked potential -Frequency: 500Hz, 10 pulses/train every 10 seconds for 2h (720 trains) |
-ES with no SCI (n = 3) -ES with SCI surviving 6h (n = 3) -ES with SCI surviving 24h (n = 3) -ES with SCI surviving 1 week (n = 6) -ES with SCI surviving 3 weeks (n = 3) -ES with SCI surviving 8 weeks (n = 6) |
-Histological: ES groups had increased GFAP staining (diffusely and focally at electrode site) 1. 24h post-SCI: ES with SCI group had less necrosis, hemorrhage and neutrophilic infiltration with smaller lesion /cavity 2. 1st and 3rd week post-SCI: ES with SCI group had smaller lesion/cavity, increased GFAP, vimentin, and GAP-43 staining 3. 8th week: no difference in lesion size/cavity between groups |
| Brus-Ramer, Carmel et al. (2007) | -Model: Sprague-Dawley female rats -Injury: unilateral pyramidotomy (PTx) transection contralateral to ES |
-Intermittent constant current bipolar ES (epidural) on intact pyramid (contralateral to the lesion) for 6h daily for 10 days beginning post-operative day 1 | -Intensity: minimum required for forelimb contraction (35-120μA) -Power supply: constant current stimulator -Frequency: 333Hz for 45ms every 2seconds -Electrodes: stainless steel implanted at the time of PTx |
-Sham ES with no PTx (n = 9) -PTx only (n = 8) -ES only (n = 7) -ES and PTx (n = 8) |
-Neurophysiological: lower activation threshold for ES+PTx group (strong ipsilateral motor activation; the sum of effects of PTx alone and ES alone were equal to the ES+PTx group -Histological: ES with PTx group had greatest increase in CST axon terminations (equal to sum effects of PTx alone and ES alone), axon length, and axon terminal varicosity density in ipsilateral grey matter; ES and PTx group had greatest topographical outgrowth in the ventral grey matter, contralateral CST axons re-crossing midline, and contralateral grey matter CST axon density |
| Hentall, Burns (2009) | -Model: Sprague-Dawley (ES experiment) and Fisher (neurophysiology experiment) female rats -Injury: thoracic contusion (neurophysiology experiment) via impactor (4mm tip causing 3kdyn force for 20ms displacing cord 0.95mm) and T8 contusion (ES experiment) via 10g weight (2mm diameter) drop from 12.5mm |
-Implanted intermittent DC ES (to NRM) every 5 minutes for 12 h every day beginning 30min-1 h after SCI (stimulator implanted within 60min of SCI or 5-7 days prior) | -Intensity: 30uA -Power supply: 3V (2 x1.5V in series) silver oxide battery -Frequency: 8Hz every 5 minutes for 12h every day for approximately 3d (mean battery life 3.2d) -Electrodes: Tungsten cathode and stainless steel anode |
-SCI only (n = 6) -ES and no SCI (n = 4) -No ES or SCI (normal) (n = 6) -Sham ES with SCI (n = 13) -ES with SCI (n = 14) |
-Behavioural: 1. BBB score: no difference between groups from having implants 2. Tail-flick test: Prolonged latencies 7-15 days after implant in ES group 3. Von Frey allodynia test: ES with SCI group had less forepaw (hindpaw, no difference) and allodynia than sham ES with SCI, which had more allodynia than normal group in forepaw and hindpaw -Neurophysiological: Normal group had more neutral cells than SCI only group and less spontaneous firing from on-cells and off-cells than ES with SCI group; sham ES with SCI rats had weaker on- and off-cell responses to noxious stimuli above lesion than normal rats -Histological: More myelination in ES with SCI, but no difference in lesion size/cavity or NeuN staining, though less GFAP |
| Li, Brus-Ramer et al. (2010) | -Model: Sprague-Dawley female rats -Injury: unilateral PTx transection contralateral to ES |
-Intermittent constant-current bipolar ES (epidural) on intact pyramid for 6h every day for 10 days beginning post-operative day 1 | -Intensity: minimum required for forelimb contraction (35-120uA) -Power supply: constant current stimulator -Frequency: 333Hz for 45ms every 2seconds -Electrodes: stainless steel implanted at time of PTx |
- ES and PTx (n = 4) -Sham ES and PTx (n = 4) |
-Histological: ES with PTx group had more BrdU+ cells (proliferating) in dCST, BrdU+ cells apposing axons, OPCs, mature proliferating OLs, and OPCs differentiating into OLs; No difference in proliferating astrocytes or endothelial cells between groups |
| Carmel, Berrol et al. (2010) | -Model: Sprague-Dawley female rats -Injury: unilateral PTx contralateral to cortical ES |
-Intermittent constant-current bipolar ES (epidural) on intact CST motor cortex (contralateral to the lesion) for 6h every day for 10 days beginning post-operative day 1 | -Intensity: minimum required for forelimb contraction (1.1-1.8mA) -Power supply: constant current stimulator -Frequency: 333Hz for 45ms every 2seconds -Electrodes: stainless steel implanted >1 week prior to PTx |
-ES and PTx (n = 5) -Sham ES with PTx (n = 5) |
-Behavioural: 1. Horizontal ladder test: ES and PTx group had improvement of affected forelimb over time and returned to baseline scores; Sham ES had no improvement of forelimb with worsening from baseline; difference between groups at days 20 and 30; ES with PTx group had reduction in all error types beginning at day. -Histological: ES with PTx group had increased axon density in ipsilateral grey matter, overall length of axons with a similar topographic distribution, had dorsal horn outgrowth (muscle and cutaneous receptor terminations), motor laminae outgrowth but not superficial dorsal laminae (nociceptive afferent terminations) |
| Carmel, Kimura et al. (2013) | -Model: Sprague-Dawley female rats -Injury: unilateral PTx contralateral to ES |
-Intermittent constant-current bipolar ES (epidural) on intact CST motor cortex (contralateral to the lesion) for 6h every day for 10 days beginning post-operative day 1 | -Intensity: minimum required for forelimb contraction (1.1-1.8mA) -Power supply: constant current stimulator -Frequency: 333Hz, 45ms, for 0.2ms duration every 2 seconds -Electrodes: stainless steel implanted >1 week prior to PTx |
-ES with PTx -PTx alone -Total: n = 17 |
-Histological: no difference in cortical cellular architecture or GFAP staining between groups due to implants 1. Spinal Cord: ES with PTx group had greater total axon length in ipsilateral grey matter 2. Cuneate nuclei: ES with PTx group had greater total axon length in ipsilateral and contralateral grey matter; 3. Parvocellular nuclei: ES with PTx group had greater total axon length in ipsilateral and contralateral grey matter; 4. Magnocellular nuclei: ES with PTx group had greater total axon length in ipsilateral and contralateral grey matter; ES increases growth bilaterally with similar topographical distribution (thus greatest outgrowth in red nucleus that restore function) -Number of axons re-crossing midline in SC greater in ES with PTx. |
| Carballosa-Gonzalez, Vitores et al. (2014) | -Model: Sprague-Dawley female rats -Injury: T8 contusion via 10g weight (2mm diameter) drop from 12.5mm |
-Single session ES (to nucleus raphe magnus (NRM)) every 5minutes for 2h starting 72h after SCI | -Intensity: 30uA -Frequency: 8Hz every 5 minutes for 2h with a pulse width of 1ms -Electrodes: Tungsten |
-Sham ES and SCI (n = 4) -No ES or SCI (normal) (n = 4) -ES with SCI (n = 4) CREB/PKA and pCREB/pPKA experiment -No ES or SCI (normal) (n = 7) -No ES with SCI (n = 8) -ES with SCI (n = 7) |
-Histological: ES with SCI group had higher cAMP levels in cervical, thoracic and lumbar tissue with return to near-normal levels; pimozide use reduced cAMP levels those in SCI (ES failed to increase it afterward) -PKA and CREB higher in SCI animals with ES reversing this increase -pPKA/PKA and pCREB/CREB reduced after SCI and increased with ES |
| Carmel, Kimura et al. (2014) | -Model: Sprague-Dawley female rats -Injury: unilateral PTx contralateral to ES |
Intermittent constant-current bipolar ES (epidural) on intact CST motor cortex (contralateral to the lesion) for 6h every day for 10 days beginning 8 weeks after PTx | -Intensity: motor threshold (minimum required for forelimb contraction; 0.9-1.7mA) -Frequency: 333Hz, 0.2ms biphasic pulse, 45ms duration, every 2 seconds -Electrodes: stainless steel implanted 5 weeks after PTx -Power supply: constant current stimulator |
-ES with PTx (n = 5) -Sham ES+PTx (n = 5) |
-Behavioural: 1. Horizontal ladder test: ES with PTx group had more improvement with errors back to baseline at week 11; muscimol use in ES with PTx resulted in transient re-emergence of errors with no change in subtypes of errors (ES after chronic PTx improves recovery through intact M1 to impaired forelimb connections) |
| Fujiki, Kobayashi et al. (2004) | -Model: Sprague-Dawley female rats -Injury: unilateral hemisection |
-ES (at T7 level of spinal cord) 24 hrs before injury, immediately following SCI and then every 24hrs for 7 days | -Intensity: 2x threshold voltage of spinal cord evoked potential -Frequency: 500 Hz, 10 pulses/train, at an inter-train interval of 10 sec -Duration: 2 h -Electrode: bipolar flexible wire |
-Preconditioning stimulation, no SCI (n = 3) -SCI only with survival time: 6hr (n = 3), 24hr (n = 3), 1 week (n = 6), 3 weeks (n = 3), 8 weeks (n = 6) -ES and SCI with survival time: 6hr (n = 3), 24hr (n = 3), 1 week (n = 6), 3 weeks (n = 3), 8 weeks (n = 6) |
-Histological: Upregulation of glial fibrillary acidic protein (GFAP) and vimentin immunoreactivity were increased at 1 week after injury in the rats treated with electrical stimulation -Preconditioning electrical stimulation of the spinal cord activated reactive astrocytes, then significantly attenuated edema, progressive necrosis, and cavitation, especially in the secondary cavity lesions |
| Ichiyama, Gerasimenko et al. (2005) | -Model: Sprague-Dawley female rats -Injury: transection (T7-T9) |
-Continuous epidural stimulation at 2, 3 or 4 sites between T12 and L6, 2-3 weeks post-SCI | - Frequency: 30, 40 or 50 Hz, 200 us duration rectangular pulses - Intensity: 0-13 V |
n = 6, all animals received stim | -Bilateral hindlimb locomotor activity was evoked most often with epidural stimulation at 40–50 Hz applied at L2. Providing >5% body weight support was necessary for locomotion |
| Shapiro, Borgens, et al. (2005) | -Model: Humans -Injury: complete motor and sensory SCI between C5 and T10, no transection demonstrated on MRI |
-Oscillating field stimulator (OFS) implanted within 18 days of SCI and explanted at 15 weeks -Three electrodes were placed above and below level of injury |
-Unit had an onboard oscillator timed for 15-minute intervals and delivered a field of 500 to 600 mV/mm and a current density of 42.4 mAmp/mm2 to each electrode |
-10 participants received OFS. -One participant lost to follow up after 6 months | -No complications at insertion of OFS -Significant improvement in pinprick sensation and motor status at 1 year post-SCI, as compared to baseline |
| Lavrov, Gerasimenko et al. (2006) | -Model: Sprague-Dawley female rats -Injury: transection T8 |
-Epidural stimulation at S1 spinal cord level | During Reflex Testing: - single stimuli, duration 0.5ms - Frequency: 0.2, 1, 3 and 5 Hz -Intensity: between 0.5 and 10 V During locomotion: - Continuous stimulation -Frequency: 40 Hz (25 ms pulse interval) and a pulse duration of 0.2 ms |
Total n = 9, recorded from 4 rats pre-transection and 7 rats following transection |
Epidurally-induced (S1) spinal cord reflexes: -The late response was abolished following transection and reappeared after 3 weeks and increased, whereas the middle response was facilitated and progressively increased following transection -Behavioural: restoration of stepping coincided with reappearance of the late response |
| Ichiyama, Courtine et al. (2008) | -Model: Sprague-Dawley female rats -Injury: transection T9 |
-Continuous epidural stimulation at L2 throughout step trainings (7d/week, 30 min/session for 6 weeks) |
-Frequency: 40 Hz with 200 us duration rectangular pulses |
-Non-injured (n = 10) - SCI and step trained (n = 5) - SCI and no step training (n = 5) |
-Histological: significantly lower number of FOS+ neurons in trained versus nontrained rats throughout lumbosacral segments after 1 hr of stepping -Behavioural: trained rats had higher and longer steps, narrower base of support at stance, and lower variability in EMG parameters |
| Lavrov, Courtine et al. (2008) | -Model: Sprague-Dawley female rats -Injury: transection (T8) and unilateral deafferentation (T12-S1) |
-Epidural stimulation at S1 spinal cord level | During Reflex Testing: -single stimuli, duration 0.5ms -Frequency: 0.2, 1, 3 and5 Hz -Intensity: between 0.5 and 10 V During locomotion: -Continuous stim -Frequency: 40 Hz (25 ms pulse interval) and a pulse duration of 0.2 ms |
-Control (n = 5) -Transection and deafferentation (n = 4) |
-All animals were able to generate stepping-like patterns on a moving treadmill on the non-deafferented, but not deafferented, side from 3 to 7 weeks after surgery when facilitated by epidural stimulation -Spinal-cord-evoked potentials were observed on both sides, although middle (monosynaptic) and late (long latency) responses were more prominent on the non-deafferented side. |
| Courtine, Gerasimenko et al. (2009) | -Model: Sprague-Dawley female rats -Injury: transection (T7) |
-L2 and/or S1 epidural stimulation |
-Frequency: 40–50 Hz -Intensity: 1–4 V rectangular pulses (0.2 ms duration) |
n = 8, trained for 8 weeks beginning 8 days post-transection n = 6, trained for 3 weeks n = 7, untrained |
-Combinations of serotonergic agonists and epidural electrical stimulation were able to acutely transform spinal networks from nonfunctional to highly functional and adaptive states as early as 1 week after transection -Behavioural: full weight bearing treadmill locomotion following SCI was observed |
| Harkema, Gerasimenko et al. (2011) | -Model: Human -Injury: C7/T1 absent voluntary motor function and partial preservation of sensation below T1 (AIS B) |
-Tonic epidural stimulation of L1-S1 spinal cord segments with a 16-electrode array | Within a session, stimulation duration ranged from 40 to 120 min -Intensity: 0.5 to 10.0 V -Frequency: 5 to 40 Hz with either a 210 μs or 450 μs pulse width |
n = 1 -The participant received 170 locomotor training sessions (body-weight supported treadmill training) over 26 months prior to electrode implantation |
-Epidural stimulation enabled full weight-bearing standing with assistance provided only for balance for 4.25 min -7 months after implantation, the patient recovered supraspinal control of some leg movements, but only during epidural stimulation. |
| Wenger, Mourad et al. (2014) | -Model: Lewis female rats -Injury: transection (T7) |
-Closed-loop neuromodulation system including epidural stimulation at L2 and S1 spinal cord level | -Frequency, amplitude, and pulse width of EES were adjusted to cover the entire range of values that promoted functional movements -Frequency: 20 to 90Hz |
-Characterization of relationships between EES frequency and gait pattern modulation (n = 5) -Real-time control tested daily over a period of 4 weeks, starting 5 weeks post-lesion (n = 3) |
-Optimized a neuromodulation in real time to achieve high-fidelity control of leg kinematics during locomotion in rats -established a technological platform with embedded control policies that integrated robust movement feedback and feed-forward control loops in real time -Animals with complete spinal cord injury performed more than 1000 successive steps without failure, and were able to climb staircases with precision |
| Rejc, Angeli and Harkema (2015) | -Model: Human -Injury: motor complete (AIS A or B) above T10, chronic (>1yr) SCI |
-A 16-electrode array with a wide-field electrode configuration with cathodes positioned caudally was implanted over the spinal cord segments L1-S1 | -Intensity: near-motor threshold stimulation amplitude that did not directly elicit lower limb movements -Frequency: 25 Hz |
n = 4 After implantation all participants underwent 80 sessions of stand training with stimulation (1 hour, 5 sessions per week) |
-Standing with the least amount of assistance was achieved with individual-specific stimulation parameters, which promoted overall continuous EMG patterns in the lower limb muscles -Stimulation parameters optimized for one individual resulted in poor standing and additional need of external assistance for hip and knee extension in the other participants. -Negligible EMG activity of lower limb muscles by epidural stimulation during sitting |
| Capogrosso, Milekovic et al. (2016) |
-Model: male rhesus monkeys (Macaca mulatta) -Injury: approximately two-thirds of the dorsoventral extent of the spinal cord at T7 |
-Implanted intracortical electrode array in the leg area of the motor cortex and with a spinal cord stimulation system composed of a spatially selective epidural implant (at T13–L1 vertebrae) and a pulse generator | Mapping leg muscles: -Single pulses of cathodic monopolar, charge-balanced stimulation (0.3 ms, 1 Hz) During locomotion: -Frequency: 30–80 Hz -Intensity: 1.5–3.9 V |
Total n = 9 Of those, n = 2 received SCI |
-Development and validation of selective spinal cord simulation protocols was possible -Behavioural: stimulation restored weight-bearing locomotion of the paralyzed leg on a treadmill and over ground |
| Rejc, Angeli et al. (2017) | -Model: Human -Injury: chronic motor complete SCI (AIS A or B) above T10, chronic (>1yr) SCI |
-A 16-electrode array was implanted over the spinal cord segments L1-S1 | -Intensity: 0.1V to 5 V -Frequency: 21-31 Hz to induce a rhythmic EMG pattern |
n = 4 After implantation all participants underwent 80 sessions of stand training with stimulation (1 hour, 5 sessions per week) followed by 80 sessions of step training with stimulation (1 hour, 5 sessions per week) |
-Step training performed after a bout of stand training substantially impaired standing ability in three of the four individuals -Poorer standing ability was associated with more variable EMG patterns that alternated EMG bursts and longer periods of negligible activity in most of the muscles -Stand and step training with epidural stimulation were not sufficient to improve motor function for standing without stimulation |
| Rejc, Angeli et al. (2017) | -Model: Human -Injury: motor complete (AIS B) C7 SCI |
-A 16-electrode array was implanted over the spinal cord segments L1-S1 | -Stimulation parameters are variable, depending on the training (standing, stepping or voluntary movement) | n = 1 Data collected over 4.1 years, individual participated in a number of activity-based interventions in combination with epidural stimulation |
-Progressive recovery of voluntary leg movement and standing without epidural stimulation in an individual with chronic, motor complete SCI over a number of years of activity-based interventions utilizing stimulation configurations customized for the different motor tasks that were specifically trained (standing, stepping, volitional leg movement) |
| Lo, Kuan et al. (2017) | -Model: Sprague-Dawley female rats -Injury: transection (T8) |
-Custom epidural 18-electrode array over lumbosacral segments | Non-transected, awake rat: monophasic stimulus -Frequency: 0.5 Hz -Intensity: 0.3 mA Rats with transection: bi-phasic stimulus - Frequency: 40 Hz, 0.2 ms pulse width -Intensity: 0.7 mA |
-Neurologically intact rat (n = 1) -Spinal cord transection (n = 1) |
Wireless, fully integrated system-on-a-chip was shown to record EMG while providing epidural stimulation to facilitate standing and stepping following spinal cord transection |
| Jack, Hurd et al (2017) | -Model: Lewis female rats -Injury: C4 dorsal lateral quadrant |
Single session ES (to forelimb motor cortex) for 30min immediately before SCI | -Electrodes: two tungsten electrodes (5 μm diameter, 0.1 MΩ impedance) inserted to depth of 1.5mm. Frequency 20 and 333Hz -Power supply: constant current stimulator |
-Sham (n = 10) -SCI only (n = 10) -SCI +20Hz (n = 14) -SCI +333Hz (n = 14) |
Behavioral: Montoya staircase test showed no difference between stimulated or SCI only groups Histological: No difference in axonal regeneration was between groups. Increased axonal collateralization was found in ES333 animals compared with controls |
| Zareen et al. (2018) | -Model: Sprague-Dawley female rats -Injury: bilateral moderate C4 contusion |
Bilateral intermittent theta burst (iTBS) ES (epidural) of motor cortex for 30 minutes every day for 10 days beginning 7 weeks after SCI combined with concurrent continuous cathodal transspinal direct current stimulation | -Intensity iTBS: 75% motor threshold -Intensity of spinal cord stimulation: 1.5 mA |
-SCI only (n = 10) -ES+SCI (n = 9) |
Histological: no between-group lesion size difference; enhanced CST outgrowth below and above the lesion in stimulation group. Behavioral: 23% significant improvement in horizontal ladder task with stimulation compared with non-significant 7% improvement injury nly; Significant improvement in IBB task scores in stimulation group (5.77 to 6.79) and non-significant change in SCI only group (5.05 to 5.42). |
| Batty, Torres-Espín et al. (2020) | -Model: Lewis female rats -Injury: C4 dorso-lateral quadrant lesion |
-Single session ES (to forelimb motor cortex) for 30min immediately following SCI | -Intensity: 1.1x motor threshold (0.6-1.3mA) -Frequency: 333Hz, 0.2ms biphasic pulse, every 0.5 seconds -Electrodes: two tungsten electrodes (5 μm diameter, 0.1 MΩ impedance) inserted to depth of 1.5mm -Power supply: constant current stimulator |
SCI and ES and upper extremity rehabilitative training (n = 11) -SCI and no ES and rehabilitative training (n = 9) |
-Behavioural: Single pellet reaching: ES group showed increased functional recovery compared to no ES group -Histological: 1. Significant increase in sprouting of corticospinal tract collaterals above the lesion site in ES versus control 2. No difference in regeneration through lesion site between groups |
Funding Information
K.F. was supported through grants from the International Spinal Research Trust (ISRT) (Res: 0011173) and the Canadian Institutes of Health Research (CIHR) (Res: 0011338). J.M. was supported through grants from the National Institutes of Health (NIH) (2R01-NS064004) and the New York State Spinal Cord Injury Research Board (NYS SCIRB) (C30606GG; C31291GG)
Author Disclosure Statement
No competing financial interests exist.
References
- 1. Burnside E.R., and Bradbury E.J. (2014). Manipulating the extracellular matrix and its role in brain and spinal cord plasticity and repair. Neuropathol. Appl. Neurobiol. 40, 26–59 [DOI] [PubMed] [Google Scholar]
- 2. Fawcett J.W., Schwab M.E., Montani L., Brazda N., and Muller H.W. (2012). Defeating inhibition of regeneration by scar and myelin components. Handb. Clin. Neurol. 109, 503–522 [DOI] [PubMed] [Google Scholar]
- 3. Ingvar S. (1920). Reaction of cells to the galvanic current in tissue cultures. Proc. Soc. Exp. Biol. Med., 17, 198–199 [Google Scholar]
- 4. Williams S. (1936). A study of the reactions of growing embryonic nerve fibers to the passage of direct electric current through the surrounding medium. Anat. Rec., 64, 56–57 [Google Scholar]
- 5. Borgens R.B., Roederer E., and Cohen M.J. (1981). Enhanced spinal cord regeneration in lamprey by applied electric fields. Science 213, 611–617 [DOI] [PubMed] [Google Scholar]
- 6. Roederer E., Goldberg N.H., and Cohen M.J. (1983). Modification of retrograde degeneration in transected spinal axons of the lamprey by applied DC current. J. Neurosci. 3, 153–160 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Borgens R.B., Jaffe L.F., and Cohen M.J. (1980). Large and persistent electrical currents enter the transected lamprey spinal cord. Proc. Natl. Acad. Sci. U. S. A. 77, 1209–1213 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Hinkle L., McCaig C.D., and Robinson K.R. (1981). The direction of growth of differentiating neurones and myoblasts from frog embryos in an applied electric field. J. Physiol. 314, 121–135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Zhao M., Song B., Pu J., Wada T., Reid B., Tai G., Wang F., Guo A., Walczysko P., Gu Y., Sasaki T., Suzuki A., Forrester J.V., Bourne H.R., Devreotes P.N., McCaig C.D., and Penninger J.M. (2006). Electrical signals control wound healing through phosphatidylinositol-3-OH kinase-gamma and PTEN. Nature 442, 457–460 [DOI] [PubMed] [Google Scholar]
- 10. Hotary K.B., and Robinson K.R. (1994). Endogenous electrical currents and voltage gradients in Xenopus embryos and the consequences of their disruption. Dev. Biol. 166, 789–800 [DOI] [PubMed] [Google Scholar]
- 11. Shi R., and Borgens R.B. (1995). Three-dimensional gradients of voltage during development of the nervous system as invisible coordinates for the establishment of embryonic pattern. Dev. Dyn. 202, 101–114 [DOI] [PubMed] [Google Scholar]
- 12. Baer M.L., and Colello R.J. (2016). Endogenous bioelectric fields: a putative regulator of wound repair and regeneration in the central nervous system. Neural Regen. Res. 11, 861–864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Rajnicek A.M., Foubister L.E., and McCaig C.D. (2006). Temporally and spatially coordinated roles for Rho, Rac, Cdc42 and their effectors in growth cone guidance by a physiological electric field. J. Cell Sci. 119, 1723–1735 [DOI] [PubMed] [Google Scholar]
- 14. Haan N., and Song B. (2014). Therapeutic application of electric fields in the injured nervous system. Adv. Wound Care (New Rochelle) 3, 156–165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Borgens R.B., Blight A.R., and McGinnis M.E. (1987). Behavioral recovery induced by applied electric fields after spinal cord hemisection in guinea pig. Science 238, 366–369 [DOI] [PubMed] [Google Scholar]
- 16. Borgens R.B., Blight A.R., and McGinnis M.E. (1990). Functional recovery after spinal cord hemisection in guinea pigs: the effects of applied electric fields. J. Comp. Neurol. 296, 634–653 [DOI] [PubMed] [Google Scholar]
- 17. Borgens R.B., Blight A.R., and Murphy D.J. (1986). Axonal regeneration in spinal cord injury: a perspective and new technique. J. Comp. Neurol 250, 157–167 [DOI] [PubMed] [Google Scholar]
- 18. Borgens R.B., Blight A.R., Murphy D.J., and Stewart L. (1986). Transected dorsal column axons within the guinea pig spinal cord regenerate in the presence of an applied electric field. J. Comp. Neurol. 250, 168–180 [DOI] [PubMed] [Google Scholar]
- 19. Borgens R.B., and Bohnert D.M. (1997). The responses of mammalian spinal axons to an applied DC voltage gradient. Exp. Neurol. 145, 376–389 [DOI] [PubMed] [Google Scholar]
- 20. Borgens R.B., Toombs J.P., Blight A.R., McGinnis M.E., Bauer M.S., Widmer W.R., and Cook J.R. Jr (1993). Effects of applied electric fields on clinical cases of complete paraplegia in dogs. Restor. Neurol. Neurosci. 5, 305–322 [DOI] [PubMed] [Google Scholar]
- 21. Borgens R.B., Toombs J.P., Breur G., Widmer W.R., Waters D., Harbath A.M., March P., and Adams L.G. (1999). An imposed oscillating electrical field improves the recovery of function in neurologically complete paraplegic dogs. J. Neurotrauma 16, 639–657 [DOI] [PubMed] [Google Scholar]
- 22. Politis M.J., and Zanakis M.F. (1988). Short term efficacy of applied electric fields in the repair of the damaged rodent spinal cord: behavioral and morphological results. Neurosurgery 23, 582–588 [DOI] [PubMed] [Google Scholar]
- 23. Wallace M.C., Tator C.H., and Gentles W.M. (1987). Effect of alternating current stimulation of the spinal cord on recovery from acute spinal cord injury in rats. Surg. Neurol. 28, 269–276 [DOI] [PubMed] [Google Scholar]
- 24. Wallace M.C., Tator C.H., and Piper I. (1987). Recovery of spinal cord function induced by direct current stimulation of the injured rat spinal cord. Neurosurgery 20, 878–884 [DOI] [PubMed] [Google Scholar]
- 25. Brus–Ramer M., Carmel J.B., Chakrabarty S., and Martin J.H. (2007). Electrical stimulation of spared corticospinal axons augments connections with ipsilateral spinal motor circuits after injury. J. Neurosci. 27, 13,793–13,801 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Martin J.H. (2005). The corticospinal system: from development to motor control. Neuroscientist 11, 161–173 [DOI] [PubMed] [Google Scholar]
- 27. Meng Z., Li Q., and Martin J.H. (2004). The transition from development to motor control function in the corticospinal system. J. Neurosci. 24, 605–614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Giordano J., Bikson M., Kappenman E.S., Clark V.P., Coslett H.B., Hamblin M.R., Hamilton R., Jankord R., Kozumbo W.J., McKinley R.A., Nitsche M.A., Reilly J.P., Richardson J., Wurzman R., and Calabrese E. (2017). Mechanisms and effects of transcranial direct current stimulation. Dose Response 15, 1559325816685467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Jaffe L.F., and Poo M.M. (1979). Neurites grow faster towards the cathode than the anode in a steady field. J. Exp. Zool. 209, 115–128 [DOI] [PubMed] [Google Scholar]
- 30. McCaig C.D. (1990). Nerve branching is induced and oriented by a small applied electric field. J. Cell Sci. 95 ( Pt 4), 605–615 [DOI] [PubMed] [Google Scholar]
- 31. McCaig C.D. (1987). Spinal neurite reabsorption and regrowth in vitro depend on the polarity of an applied electric field. Development 100, 31–41 [DOI] [PubMed] [Google Scholar]
- 32. Shapiro S., Borgens R., Pascuzzi R., Roos K., Groff M., Purvines S., Rodgers R.B., Hagy S., and Nelson P. (2005). Oscillating field stimulation for complete spinal cord injury in humans: a phase 1 trial. Journal of neurosurgery. Spine 2, 3–10 [DOI] [PubMed] [Google Scholar]
- 33. Walters B.C. (2010). Oscillating field stimulation in the treatment of spinal cord injury. PM R 2, S286–291 [DOI] [PubMed] [Google Scholar]
- 34. Tator C.H. (2005). Phase 1 trial of oscillating field stimulation for complete spinal cord injury in humans. J Neurosurg. Spine 2, 1–2 [DOI] [PubMed] [Google Scholar]
- 35. Tator C.H. (2006). Review of treatment trials in human spinal cord injury: issues, difficulties, and recommendations. Neurosurgery 59, 957–982 [DOI] [PubMed] [Google Scholar]
- 36. Mori S., Matsuyama K., Kohyama J., Kobayashi Y., and Takakusaki K. (1992). Neuronal constituents of postural and locomotor control systems and their interactions in cats. Brain Dev. 14 Suppl., S109–120 [PubMed] [Google Scholar]
- 37. Iwahara T., Wall P.T., Garcia-Rill E., and Skinner R.D. (1991). Stimulation-induced setting of postural muscle tone in the decerebrate rat. Brain Res. 557, 331–335 [DOI] [PubMed] [Google Scholar]
- 38. Gerasimenko Y.P., Makarovskii A.N., and Nikitin O.A. (2002). Control of locomotor activity in humans and animals in the absence of supraspinal influences. Neurosci. Behav. Physiol. 32, 417–423 [DOI] [PubMed] [Google Scholar]
- 39. Gerasimenko Y.P., Avelev V.D., Nikitin O.A., and Lavrov I.A. (2003). Initiation of locomotor activity in spinal cats by epidural stimulation of the spinal cord. Neurosci. Behav. Physiol. 33, 247–254 [DOI] [PubMed] [Google Scholar]
- 40. Lavrov I., Gerasimenko Y.P., Ichiyama R.M., Courtine G., Zhong H., Roy R.R., and Edgerton V.R. (2006). Plasticity of spinal cord reflexes after a complete transection in adult rats: relationship to stepping ability. J. Neurophysiol. 96, 1699–1710 [DOI] [PubMed] [Google Scholar]
- 41. Ichiyama R.M., Gerasimenko Y.P., Zhong H., Roy R.R., and Edgerton V.R. (2005). Hindlimb stepping movements in complete spinal rats induced by epidural spinal cord stimulation. Neurosci. Lett. 383, 339–344 [DOI] [PubMed] [Google Scholar]
- 42. Barriere G., Mellen N., and Cazalets J.R. (2004). Neuromodulation of the locomotor network by dopamine in the isolated spinal cord of newborn rat. Eur. J. Neurosci. 19, 1325–1335 [DOI] [PubMed] [Google Scholar]
- 43. Kjaerulff O., and Kiehn O. (2001). 5-HT modulation of multiple inward rectifiers in motoneurons in intact preparations of the neonatal rat spinal cord. J. Neurophysiol. 85, 580–593 [DOI] [PubMed] [Google Scholar]
- 44. Barbeau H., and Rossignol S. (1990). The effects of serotonergic drugs on the locomotor pattern and on cutaneous reflexes of the adult chronic spinal cat. Brain Res. 514, 55–67 [DOI] [PubMed] [Google Scholar]
- 45. Barbeau H., and Rossignol S. (1991). Initiation and modulation of the locomotor pattern in the adult chronic spinal cat by noradrenergic, serotonergic and dopaminergic drugs. Brain Res. 546, 250–260 [DOI] [PubMed] [Google Scholar]
- 46. Ribotta M.G., Provencher J., Feraboli-Lohnherr D., Rossignol S., Privat A., and Orsal D. (2000). Activation of locomotion in adult chronic spinal rats is achieved by transplantation of embryonic raphe cells reinnervating a precise lumbar level. J. Neurosci. 20, 5144–5152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Slawinska U., Miazga K., Cabaj A.M., Leszczynska A.N., Majczynski H., Nagy J.I., and Jordan L.M. (2013). Grafting of fetal brainstem 5-HT neurons into the sublesional spinal cord of paraplegic rats restores coordinated hindlimb locomotion. Exp. Neurol. 247, 572–581 [DOI] [PubMed] [Google Scholar]
- 48. Alluin O., Delivet-Mongrain H., and Rossignol S. (2015). Inducing hindlimb locomotor recovery in adult rat after complete thoracic spinal cord section using repeated treadmill training with perineal stimulation only. J. Neurophysiol. 114, 1931–1946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Delivet-Mongrain H., Leblond H., and Rossignol S. (2008). Effects of localized intraspinal injections of a noradrenergic blocker on locomotion of high decerebrate cats. J. Neurophysiol. 100, 907–921 [DOI] [PubMed] [Google Scholar]
- 50. Rossignol S., Martinez M., Escalona M., Kundu A., Delivet-Mongrain H., Alluin O., and Gossard J.P. (2015). The “beneficial” effects of locomotor training after various types of spinal lesions in cats and rats. Prog. Brain Res. 218, 173–198 [DOI] [PubMed] [Google Scholar]
- 51. Gildenberg P.L. (2006). History of electrical neuromodulation for chronic pain. Pain Med. 7, 7–13 [Google Scholar]
- 52. Sherwood A.M., Sharkey P.C., and Dimitrijevic M.R. (1980). Biomedical engineering specifications for epidural spinal cord stimulation to augment motor performance. Int. Rehabil. Med. 2, 62–67 [DOI] [PubMed] [Google Scholar]
- 53. Dimitrijevic M.R., Gerasimenko Y., and Pinter M.M. (1998). Evidence for a spinal central pattern generator in humans. Ann. N. Y. Acad. Sci. 860, 360–376 [DOI] [PubMed] [Google Scholar]
- 54. Iwahara T., Atsuta Y., Garcia-Rill E. and Skinner R.D. (1992). Spinal cord stimulation-induced locomotion in the adult cat. Brain Res. Bull. 28, 99–105 [DOI] [PubMed] [Google Scholar]
- 55. Gerasimenko Y.P., Ichiyama R.M., Lavrov I.A., Courtine G., Cai L., Zhong H., Roy R.R., and Edgerton V.R. (2007). Epidural spinal cord stimulation plus quipazine administration enable stepping in complete spinal adult rats. J. Neurophysiol. 98, 2525–2536 [DOI] [PubMed] [Google Scholar]
- 56. Lavrov I., Courtine G., Dy C.J., van den Brand R., Fong A.J., Gerasimenko Y., Zhong H., Roy R.R., and Edgerton V.R. (2008). Facilitation of stepping with epidural stimulation in spinal rats: role of sensory input. J. Neurosci. 28, 7774–7780 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Courtine G., Gerasimenko Y., van den Brand R., Yew A., Musienko P., Zhong H., Song B., Ao Y., Ichiyama R.M., Lavrov I., Roy R.R., Sofroniew M.V., and Edgerton V.R. (2009). Transformation of nonfunctional spinal circuits into functional states after the loss of brain input. Nat. Neurosci. 12, 1333–1342 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Ichiyama R.M., Courtine G., Gerasimenko Y.P., Yang G.J., van den Brand R., Lavrov I.A., Zhong H., Roy R.R., and Edgerton V.R. (2008). Step training reinforces specific spinal locomotor circuitry in adult spinal rats. J. Neurosci. 28, 7370–7375 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Shah P.K., and Gerasimenko Y. (2016). Multi-site spinal stimulation strategies to enhance locomotion after paralysis. Neural. Regen. Res. 11, 1926–1927 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Shah P.K., Gerasimenko Y., Shyu A., Lavrov I., Zhong H., Roy R.R., and Edgerton V.R. (2012). Variability in step training enhances locomotor recovery after a spinal cord injury. Eur. J. Neurosci. 36, 2054–2062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Young W. (2015). Electrical stimulation and motor recovery. Cell Transplant. 24, 429–446 [DOI] [PubMed] [Google Scholar]
- 62. Gerasimenko Y., Roy R.R., and Edgerton V.R. (2008). Epidural stimulation: comparison of the spinal circuits that generate and control locomotion in rats, cats and humans. Exp. Neurol. 209, 417–425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Calvert J.S., Grahn P.J., Zhao K.D., and Lee K.H. (2019). Emergence of epidural electrical stimulation to facilitate sensorimotor network functionality after spinal cord injury. Neuromodulation 22, 244–252 [DOI] [PubMed] [Google Scholar]
- 64. Calvert J.S., Grahn P.J., Strommen J.A., Lavrov I.A., Beck L.A., Gill M.L., Linde M.B., Brown D.A., Van Straaten M.G., Veith D.D., Lopez C., Sayenko D.G., Gerasimenko Y.P., Edgerton V.R., Zhao K.D., and Lee K.H. (2019). Electrophysiological guidance of epidural electrode array implantation over the human lumbosacral spinal cord to enable motor function after chronic paralysis. J. Neurotrauma 36, 1451–1460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Wenger N., Moraud E.M., Gandar J., Musienko P., Capogrosso M., Baud L., Le Goff C.G., Barraud Q., Pavlova N., Dominici N., Minev I.R., Asboth L., Hirsch A., Duis S., Kreider J., Mortera A., Haverbeck O., Kraus S., Schmitz F., DiGiovanna J., van den Brand R., Bloch J., Detemple P., Lacour S.P., Bezard E., Micera S., and Courtine G. (2016). Spatiotemporal neuromodulation therapies engaging muscle synergies improve motor control after spinal cord injury. Nat. Med. 22, 138–145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Capogrosso M., Milekovic T., Borton D., Wagner F., Moraud E.M., Mignardot J.B., Buse N., Gandar J., Barraud Q., Xing D., Rey E., Duis S., Jianzhong Y., Ko W.K., Li Q., Detemple P., Denison T., Micera S., Bezard E., Bloch J., and Courtine G. (2016). A brain–spine interface alleviating gait deficits after spinal cord injury in primates. Nature 539, 284–288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Wenger N., Moraud E.M., Raspopovic S., Bonizzato M., DiGiovanna J., Musienko P., Morari M., Micera S., and Courtine G. (2014). Closed–loop neuromodulation of spinal sensorimotor circuits controls refined locomotion after complete spinal cord injury. Sci. Transl. Med. 6, 255ra133. [DOI] [PubMed] [Google Scholar]
- 68. Lo Y.K., Kuan Y.C., Culaclii S., Kim B., Wang P.M., Chang C.W., Massachi J.A., Zhu M., Chen K., Gad P., Edgerton V.R., and Liu W. (2017). A fully integrated wireless soc for motor function recovery after spinal cord injury. IEEE Trans. Biomed. Circuits Syst. 11, 497–509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Mushahwar V.K., Aoyagi Y., Stein R.B. and Prochazka A. (2004). Movements generated by intraspinal microstimulation in the intermediate gray matter of the anesthetized, decerebrate, and spinal cat. Can. J. Physiol. Pharmacol. 82, 702–714 [DOI] [PubMed] [Google Scholar]
- 70. Mushahwar V.K., Gillard D.M., Gauthier M.J., and Prochazka A. (2002). Intraspinal micro stimulation generates locomotor-like and feedback-controlled movements. IEEE Trans. Neural Syst. Rehabil. Eng. 10, 68–81 [DOI] [PubMed] [Google Scholar]
- 71. Bamford J.A., Putman C.T., and Mushahwar V.K. (2005). Intraspinal microstimulation preferentially recruits fatigue-resistant muscle fibres and generates gradual force in rat. J. Physiol. 569, 873–884 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Holinski B.J., Mazurek K.A., Everaert D.G., Toossi A., Lucas-Osma A.M., Troyk P., Etienne-Cummings R., Stein R.B., and Mushahwar V.K. (2016). Intraspinal microstimulation produces over-ground walking in anesthetized cats. J. Neural Eng. 13, 056016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Collinger J.L., Foldes S., Bruns T.M., Wodlinger B., Gaunt R., and Weber D.J. (2013). Neuroprosthetic technology for individuals with spinal cord injury. J. Spinal Cord Med. 36, 258–272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Collinger J.L., Kryger M.A., Barbara R., Betler T., Bowsher K., Brown E.H., Clanton S.T., Degenhart A.D., Foldes S.T., Gaunt R.A., Gyulai F.E., Harchick E.A., Harrington D., Helder J.B., Hemmes T., Johannes M.S., Katyal K.D., Ling G.S., McMorland A.J., Palko K., Para M.P., Scheuermann J., Schwartz A.B., Skidmore E.R., Solzbacher F., Srikameswaran A.V., Swanson D.P., Swetz S., Tyler-Kabara E.C., Velliste M., Wang W., Weber D.J., Wodlinger B., and Boninger M.L. (2014). Collaborative approach in the development of high-performance brain–computer interfaces for a neuroprosthetic arm: translation from animal models to human control. Clin. Transl. Sci. 7, 52–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Collinger J.L., Wodlinger B., Downey J.E., Wang W., Tyler-Kabara E.C., Weber D.J., McMorland A.J., Velliste M., Boninger M.L., and Schwartz A.B. (2013). High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 381, 557–564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Kasten M.R., Sunshine M.D., Secrist E.S., Horner P.J., and Moritz C.T. (2013). Therapeutic intraspinal microstimulation improves forelimb function after cervical contusion injury. J. Neural Eng. 10, 044001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Moritz C.T., Lucas T.H., Perlmutter S.I., and Fetz E.E. (2007). Forelimb movements and muscle responses evoked by microstimulation of cervical spinal cord in sedated monkeys. J. Neurophysiol. 97, 110–120 [DOI] [PubMed] [Google Scholar]
- 78. Mercier L.M., Gonzalez-Rothi E.J., Streeter K.A., Posgai S.S., Poirier A.S., Fuller D.D., Reier P.J., and Baekey D.M. (2017). Intraspinal microstimulation and diaphragm activation after cervical spinal cord injury. J. Neurophysiol. 117, 767–776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Mayr W., Krenn M., and Dimitrijevic M.R. (2016). Epidural and transcutaneous spinal electrical stimulation for restoration of movement after incomplete and complete spinal cord injury. Curr. Opin. Neurol. 29, 721–726 [DOI] [PubMed] [Google Scholar]
- 80. Hauglie-Hanssen E., Sem-Jacobsen C.W., and Styri O.B. (1975). Proceedings: Dorsal column and transcutaneous electrical stimulation for intractable pain. Acta Neurochir. (Wien) 31, 263–264 [PubMed] [Google Scholar]
- 81. Kirsch W.M., Lewis J.A. and Simon R.H. (1975). Experiences with electrical stimulation devices for the control of chronic pain. Med. Instrum. 9, 217–220 [PubMed] [Google Scholar]
- 82. Minassian K., Persy I., Rattay F., Dimitrijevic M.R., Hofer C., and Kern H. (2007). Posterior root–muscle reflexes elicited by transcutaneous stimulation of the human lumbosacral cord. Muscle Nerve 35, 327–336 [DOI] [PubMed] [Google Scholar]
- 83. Kitano K., and Koceja D.M. (2009). Spinal reflex in human lower leg muscles evoked by transcutaneous spinal cord stimulation. J. Neurosci. Methods 180, 111–115 [DOI] [PubMed] [Google Scholar]
- 84. Ladenbauer J., Minassian K., Hofstoetter U.S., Dimitrijevic M.R., and Rattay F. (2010). Stimulation of the human lumbar spinal cord with implanted and surface electrodes: a computer simulation study. IEEE Trans. Neural Syst. Rehabil. Eng. 18, 637–645 [DOI] [PubMed] [Google Scholar]
- 85. Danner S.M., Hofstoetter U.S., Ladenbauer J., Rattay F., and Minassian K. (2011). Can the human lumbar posterior columns be stimulated by transcutaneous spinal cord stimulation? A modeling study. Artif. Organs 35, 257–262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Sayenko D.G., Rath M., Ferguson A.R., Burdick J.W., Havton L.A., Edgerton V.R., and Gerasimenko Y.P. (2019). Self-assisted standing enabled by non-invasive spinal stimulation after spinal cord injury. J. Neurotrauma 36, 1435–1450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Roy F.D., Bosgra D., and Stein R.B. (2014). Interaction of transcutaneous spinal stimulation and transcranial magnetic stimulation in human leg muscles. Exp. Brain Res. 232, 1717–1728 [DOI] [PubMed] [Google Scholar]
- 88. Wu Y.K., Levine J.M., Wecht J.R., Maher M.T., LiMonta J.M., Saeed S., Santiago T.M., Bailey E., Kastuar S., Guber K.S., Yung L., Weir J.P., Carmel J.B., and Harel N.Y. (2020). Posteroanterior cervical transcutaneous spinal stimulation targets ventral and dorsal nerve roots. Clin. Neurophysiol. 131, 451–460 [DOI] [PubMed] [Google Scholar]
- 89. Sayenko D.G., Atkinson D.A., Mink A.M., Gurley K.M., Edgerton V.R., Harkema S.J., and Gerasimenko Y.P. (2018). Vestibulospinal and corticospinal modulation of lumbosacral network excitability in human subjects. Front. Physiol. 9, 1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Inanici F., Samejima S., Gad P., Edgerton V.R., Hofstetter C.P., and Moritz C.T. (2018). Transcutaneous electrical spinal stimulation promotes long-term recovery of upper extremity function in chronic tetraplegia. IEEE Trans. Neural Syst. Rehabil. Eng. 26, 1272–1278 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Benavides F.D., Jo H.J., Lundell H., Edgerton V.R., Gerasimenko Y. and Perez M.A. (2020). Cortical and subcortical effects of transcutaneous spinal cord stimulation in humans with tetraplegia. J. Neurosci. 40, 2633–2643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Harkema S., Gerasimenko Y., Hodes J., Burdick J., Angeli C., Chen Y., Ferreira C., Willhite A., Rejc E., Grossman R.G., and Edgerton V.R. (2011). Effect of epidural stimulation of the lumbosacral spinal cord on voluntary movement, standing, and assisted stepping after motor complete paraplegia: a case study. Lancet 377, 1938–1947 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Angeli C.A., Edgerton V.R., Gerasimenko Y.P., and Harkema S.J. (2014). Altering spinal cord excitability enables voluntary movements after chronic complete paralysis in humans. Brain 137, 1394–1409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Rejc E., Angeli C., and Harkema S. (2015). Effects of lumbosacral spinal cord epidural stimulation for standing after chronic complete paralysis in humans. PLoS One 10, e0133998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Gill M.L., Grahn P.J., Calvert J.S., Linde M.B., Lavrov I.A., Strommen J.A., Beck L.A., Sayenko D.G., Van Straaten M.G., Drubach D.I., Veith D.D., Thoreson A.R., Lopez C., Gerasimenko Y.P., Edgerton V.R., Lee K.H., and Zhao K.D. (2018). Neuromodulation of lumbosacral spinal networks enables independent stepping after complete paraplegia. Nat. Med. 24, 1677–1682 [DOI] [PubMed] [Google Scholar]
- 96. Edgerton V.R., Courtine G., Gerasimenko Y.P., Lavrov I., Ichiyama R.M., Fong A.J., Cai L.L., Otoshi C.K., Tillakaratne N.J., Burdick J.W., and Roy R.R. (2008). Training locomotor networks. Brain Res. Rev. 57, 241–254 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Edgerton V.R. and Roy R.R. (2002). Paralysis recovery in humans and model systems. Curr. Opin. Neurobiol. 12, 658–667 [DOI] [PubMed] [Google Scholar]
- 98. Edgerton V.R., Tillakaratne N.J., Bigbee A.J., de Leon R.D. and Roy R.R. (2004). Plasticity of the spinal neural circuitry after injury. Annu. Rev. Neurosci. 27, 145–167 [DOI] [PubMed] [Google Scholar]
- 99. Rejc E., Angeli C.A., Bryant N. and Harkema S.J. (2017). Effects of stand and step training with epidural stimulation on motor function for standing in chronic complete paraplegics. J. Neurotrauma 34, 1787–1802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Bigbee A.J., Crown E.D., Ferguson A.R., Roy R.R., Tillakaratne N.J., Grau J.W., and Edgerton V.R. (2007). Two chronic motor training paradigms differentially influence acute instrumental learning in spinally transected rats. Behav. Brain Res. 180, 95–101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Hodgson J.A., Roy R.R., de Leon R., Dobkin B., and Edgerton V.R. (1994). Can the mammalian lumbar spinal cord learn a motor task? Med. Sci. Sports Exerc. 26, 1491–1497 [PubMed] [Google Scholar]
- 102. Pettigrew R.I., Heetderks W.J., Kelley C.A., Peng G.C., Krosnick S.H., Jakeman L.B., Egan K.D., and Marge M. (2017). Epidural spinal stimulation to improve bladder, bowel, and sexual function in individuals with spinal cord injuries: a framework for clinical research. IEEE Trans. Biomed. Eng. 64, 253–262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Harkema S.J., Legg Ditterline B., Wang S., Aslan S., Angeli C.A., Ovechkin A., and Hirsch G.A. (2018). Epidural spinal cord stimulation training and sustained recovery of cardiovascular function in individuals with chronic cervical spinal cord injury. JAMA Neurol. 75, 1569–1571 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Harkema S.J., Wang S., Angeli C.A., Chen Y., Boakye M., Ugiliweneza B., and Hirsch G.A. (2018). Normalization of blood pressure with spinal cord epidural stimulation after severe spinal cord injury. Front. Hum. Neurosci. 12, 83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Herrity A.N., Williams C.S., Angeli C.A., Harkema S.J., and Hubscher C.H. (2018). Lumbosacral spinal cord epidural stimulation improves voiding function after human spinal cord injury. Sci. Rep. 8, 8688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Gerasimenko Y.P., Lavrov I.A., Courtine G., Ichiyama R.M., Dy C.J., Zhong H., Roy R.R., and Edgerton V.R. (2006). Spinal cord reflexes induced by epidural spinal cord stimulation in normal awake rats. J. Neurosci. Methods 157, 253–263 [DOI] [PubMed] [Google Scholar]
- 107. Moraud E.M., Capogrosso M., Formento E., Wenger N., DiGiovanna J., Courtine G., and Micera S. (2016). Mechanisms underlying the neuromodulation of spinal circuits for correcting gait and balance deficits after spinal cord injury. Neuron 89, 814–828 [DOI] [PubMed] [Google Scholar]
- 108. Rattay F., Minassian K., and Dimitrijevic M.R. (2000). Epidural electrical stimulation of posterior structures of the human lumbosacral cord: 2. quantitative analysis by computer modeling. Spinal Cord 38, 473–489 [DOI] [PubMed] [Google Scholar]
- 109. Dietz V., and Fouad K. (2014). Restoration of sensorimotor functions after spinal cord injury. Brain 137, 654–667 [DOI] [PubMed] [Google Scholar]
- 110. Spencer T., and Filbin M.T. (2004). A role for cAMP in regeneration of the adult mammalian CNS. J. Anat. 204, 49–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Zareen N., Dodson S., Armada K., Awad R., Sultana N., Hara E., Alexander H., and Martin J.H. (2018). Stimulation-dependent remodeling of the corticospinal tract requires reactivation of growth-promoting developmental signaling pathways. Exp. Neurol. 307, 133–144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Zareen N., Shinozaki M., Ryan D., Alexander H., Amer A., Truong D.Q., Khadka N., Sarkar A., Naeem S., Bikson M., and Martin J.H. (2017). Motor cortex and spinal cord neuromodulation promote corticospinal tract axonal outgrowth and motor recovery after cervical contusion spinal cord injury. Exp. Neurol. 297, 179–189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Udina E., Furey M., Busch S., Silver J., Gordon T., and Fouad K. (2008). Electrical stimulation of intact peripheral sensory axons in rats promotes outgrowth of their central projections. Exp. Neurol. 210, 238–247 [DOI] [PubMed] [Google Scholar]
- 114. Filbin M.T. (2003). Myelin-associated inhibitors of axonal regeneration in the adult mammalian CNS. Nat. Rev. Neurosci. 4, 703–713 [DOI] [PubMed] [Google Scholar]
- 115. Neumann S., and Woolf C.J. (1999). Regeneration of dorsal column fibers into and beyond the lesion site following adult spinal cord injury. Neuron 23, 83–91 [DOI] [PubMed] [Google Scholar]
- 116. Richardson P.M., and Issa V.M. (1984). Peripheral injury enhances central regeneration of primary sensory neurones. Nature 309, 791–793 [DOI] [PubMed] [Google Scholar]
- 117. Neumann S., Bradke F., Tessier-Lavigne M., and Basbaum A.I. (2002). Regeneration of sensory axons within the injured spinal cord induced by intraganglionic cAMP elevation. Neuron 34, 885–893 [DOI] [PubMed] [Google Scholar]
- 118. Pearse D.D., Pereira F.C., Marcillo A.E., Bates M.L., Berrocal Y.A., Filbin M.T., and Bunge M.B. (2004). cAMP and Schwann cells promote axonal growth and functional recovery after spinal cord injury. Nat. Med. 10, 610–616 [DOI] [PubMed] [Google Scholar]
- 119. Krajacic A., Ghosh M., Puentes R., Pearse D.D., and Fouad K. (2009). Advantages of delaying the onset of rehabilitative reaching training in rats with incomplete spinal cord injury. Eur. J. Neurosci. 29, 641–651 [DOI] [PubMed] [Google Scholar]
- 120. Al-Majed A.A., Neumann C.M., Brushart T.M., and Gordon T. (2000). Brief electrical stimulation promotes the speed and accuracy of motor axonal regeneration. J. Neurosci. 20, 2602–2608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Brushart T.M., Hoffman P.N., Royall R.M., Murinson B.B., Witzel C., and Gordon T. (2002). Electrical stimulation promotes motoneuron regeneration without increasing its speed or conditioning the neuron. J. Neurosci. 22, 6631–6638 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Al-Majed A.A., Brushart T.M., and Gordon T. (2000). Electrical stimulation accelerates and increases expression of BDNF and trkB mRNA in regenerating rat femoral motoneurons. Eur. J. Neurosci. 12, 4381–4390 [PubMed] [Google Scholar]
- 123. Weishaupt N., Blesch A., and Fouad K. (2012). BDNF: the career of a multifaceted neurotrophin in spinal cord injury. Exp. Neurol. 238, 254–264 [DOI] [PubMed] [Google Scholar]
- 124. Carmel J.B., and Martin J.H. (2014). Motor cortex electrical stimulation augments sprouting of the corticospinal tract and promotes recovery of motor function. Front. Integr. Neurosci. 8, 51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Carmel J.B., Kimura H., Berrol L.J., and Martin J.H. (2013). Motor cortex electrical stimulation promotes axon outgrowth to brain stem and spinal targets that control the forelimb impaired by unilateral corticospinal injury. Eur. J. Neurosci. 37, 1090–1102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126. Carmel J.B., Berrol L.J., Brus-Ramer M., and Martin J.H. (2010). Chronic electrical stimulation of the intact corticospinal system after unilateral injury restores skilled locomotor control and promotes spinal axon outgrowth. J. Neurosci. 30, 10,918–10,926 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Brus-Ramer M., Carmel J.B., and Martin J.H. (2009). Motor cortex bilateral motor representation depends on subcortical and interhemispheric interactions. J. Neurosci. 29, 6196–6206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Salimi I., and Martin J.H. (2004). Rescuing transient corticospinal terminations and promoting growth with corticospinal stimulation in kittens. J. Neurosci. 24, 4952–4961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Salimi I., Friel K.M., and Martin J.H. (2008). Pyramidal tract stimulation restores normal corticospinal tract connections and visuomotor skill after early postnatal motor cortex activity blockade. J. Neurosci. 28, 7426–7434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Friel K.M., and Martin J.H. (2005). Role of sensory-motor cortex activity in postnatal development of corticospinal axon terminals in the cat. J. Comp. Neurol. 485, 43–56 [DOI] [PubMed] [Google Scholar]
- 131. Martin J.H., and Lee S.J. (1999). Activity-dependent competition between developing corticospinal terminations. Neuroreport 10, 2277–2282 [DOI] [PubMed] [Google Scholar]
- 132. Martin J.H., Kably B. and Hacking A. (1999). Activity-dependent development of cortical axon terminations in the spinal cord and brain stem. Exp. Brain Res. 125, 184–199 [DOI] [PubMed] [Google Scholar]
- 133. Lang C., Bradley P.M., Jacobi A., Kerschensteiner M., and Bareyre F.M. (2013). STAT3 promotes corticospinal remodelling and functional recovery after spinal cord injury. EMBO Rep. 14, 931–937 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Jin D., Liu Y., Sun F., Wang X., Liu X., and He Z. (2015). Restoration of skilled locomotion by sprouting corticospinal axons induced by co-deletion of PTEN and SOCS3. Nat. Commun. 6, 8074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Bareyre F.M., Garzorz N., Lang C., Misgeld T., Buning H., and Kerschensteiner M. (2011). In vivo imaging reveals a phase-specific role of STAT3 during central and peripheral nervous system axon regeneration. Proc. Natl. Acad. Sci. U. S. A. 108, 6282–6287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Carmel J.B., Kimura H., and Martin J.H. (2014). Electrical stimulation of motor cortex in the uninjured hemisphere after chronic unilateral injury promotes recovery of skilled locomotion through ipsilateral control. J. Neurosci. 34, 462–466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Brosamle C., and Schwab M.E. (2000). Ipsilateral, ventral corticospinal tract of the adult rat: ultrastructure, myelination and synaptic connections. J. Neurocytol. 29, 499–507 [DOI] [PubMed] [Google Scholar]
- 138. Rosenzweig E.S., Brock J.H., Culbertson M.D., Lu P., Moseanko R., Edgerton V.R., Havton L.A., and Tuszynski M.H. (2009). Extensive spinal decussation and bilateral termination of cervical corticospinal projections in rhesus monkeys. J. Comp. Neurol. 513, 151–163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139. Huang Y.Z., Edwards M.J., Rounis E., Bhatia K.P., and Rothwell J.C. (2005). Theta burst stimulation of the human motor cortex. Neuron 45, 201–206 [DOI] [PubMed] [Google Scholar]
- 140. Song W., Truong D.Q., Bikson M., and Martin J.H. (2015). Transspinal direct current stimulation immediately modifies motor cortex sensorimotor maps. J. Neurophysiol. 113, 2801–2811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Yang Q., Ramamurthy A., Lall S., Santos J., Ratnadurai-Giridharan S., Lopane M., Zareen N., Alexander H., Ryan D., Martin J.H., and Carmel J.B. (2019). Independent replication of motor cortex and cervical spinal cord electrical stimulation to promote forelimb motor function after spinal cord injury in rats. Exp. Neurol. 320, 112,962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Girgis J., Merrett D., Kirkland S., Metz G.A., Verge V., and Fouad K. (2007). Reaching training in rats with spinal cord injury promotes plasticity and task specific recovery. Brain 130, 2993–3003 [DOI] [PubMed] [Google Scholar]
- 143. Vavrek R., Girgis J., Tetzlaff W., Hiebert G., and Fouad K. (2006). BDNF promotes connections of corticospinal neurons onto spared descending interneurons in spinal cord injured rats. Brain 129, 1534–1545 [DOI] [PubMed] [Google Scholar]
- 144. Jack A.S., Hurd C., Forero J., Nataraj A., Fenrich K., Blesch A., and Fouad K. (2018). Cortical electrical stimulation in female rats with a cervical spinal cord injury to promote axonal outgrowth. J. Neurosci. Res. 96, 852–862 [DOI] [PubMed] [Google Scholar]
- 145. Batty N.J., Torres-Espin A., Vavrek R., Raposo P., and Fouad K. (2019). Single-session cortical electrical stimulation enhances the efficacy of rehabilitative motor training after spinal cord injury in rats. Exp. Neurol. 324, 113136. [DOI] [PubMed] [Google Scholar]
- 146. Harvey P.J., Grochmal J., Tetzlaff W., Gordon T., and Bennett D.J. (2005). An investigation into the potential for activity-dependent regeneration of the rubrospinal tract after spinal cord injury. Eur. J. Neurosci. 22, 3025–3035 [DOI] [PubMed] [Google Scholar]
- 147. Hebb D.O. (1949). Organization of Behavior. John Wiley & Sons: New York [Google Scholar]
- 148. Dan Y., and Poo M.M. (2004). Spike timing-dependent plasticity of neural circuits. Neuron 44, 23–30 [DOI] [PubMed] [Google Scholar]
- 149. Carson R.G., and Kennedy N.C. (2013). Modulation of human corticospinal excitability by paired associative stimulation. Front. Hum Neurosci. 7, 823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Cortes M., Thickbroom G.W., Valls-Sole J., Pascual-Leone A., and Edwards D.J. (2011). Spinal associative stimulation: a non-invasive stimulation paradigm to modulate spinal excitability. Clin. Neurophysiol. 122, 2254–2259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Leukel C., Taube W., Beck S., and Schubert M. (2012). Pathway-specific plasticity in the human spinal cord. Eur. J. Neurosci. 35, 1622–1629 [DOI] [PubMed] [Google Scholar]
- 152. Stefan K., Kunesch E., Cohen L.G., Benecke R. and Classen J. (2000). Induction of plasticity in the human motor cortex by paired associative stimulation. Brain 123, Pt. 3, 572–584 [DOI] [PubMed] [Google Scholar]
- 153. Suppa A., Quartarone A., Siebner H., Chen R., Di Lazzaro V., Del Giudice P., Paulus W., Rothwell J.C., Ziemann U., and Classen J. (2017). The associative brain at work: Evidence from paired associative stimulation studies in humans. Clin. Neurophysiol. 128, 2140–2164 [DOI] [PubMed] [Google Scholar]
- 154. Taylor J.L., and Martin P.G. (2009). Voluntary motor output is altered by spike-timing-dependent changes in the human corticospinal pathway. J. Neurosci. 29, 11,708–11,716 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Tolmacheva A., Makela J.P., and Shulga A. (2019). Increasing the frequency of peripheral component in paired associative stimulation strengthens its efficacy. Sci. Rep. 9, 3849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Long J., Federico P., and Perez M.A. (2017). A novel cortical target to enhance hand motor output in humans with spinal cord injury. Brain 140, 1619–1632 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Wagner F.B., Mignardot J.B., Le Goff-Mignardot C.G., Demesmaeker R., Komi S., Capogrosso M., Rowald A., Seanez I., Caban M., Pirondini E., Vat M., McCracken L.A., Heimgartner R., Fodor I., Watrin A., Seguin P., Paoles E., Van Den Keybus K., Eberle G., Schurch B., Pralong E., Becce F., Prior J., Buse N., Buschman R., Neufeld E., Kuster N., Carda S., von Zitzewitz J., Delattre V., Denison T., Lambert H., Minassian K., Bloch J., and Courtine G. (2018). Targeted neurotechnology restores walking in humans with spinal cord injury. Nature 563, 65–71 [DOI] [PubMed] [Google Scholar]
- 158. Zimmermann J.B., and Jackson A. (2014). Closed-loop control of spinal cord stimulation to restore hand function after paralysis. Front. Neurosci. 8, 87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Nishimura Y., Perlmutter S.I., Eaton R.W. and Fetz E.E. (2013). Spike-timing-dependent plasticity in primate corticospinal connections induced during free behavior. Neuron 80, 1301–1309 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. McPherson J.G., Miller R.R., and Perlmutter S.I. (2015). Targeted, activity-dependent spinal stimulation produces long-lasting motor recovery in chronic cervical spinal cord injury. Proc. Natl. Acad. Sci. U. S. A. 112, 12193–12198 [DOI] [PMC free article] [PubMed] [Google Scholar]