Abstract
We report a patient with COVID-19 infection presenting with acute pancreatitis. The diagnosis of pancreatitis was based on laboratory as well as radiological evidence, and all the usual etiologies were ruled out. The temporal association with COVID-19 is strongly suggestive of novel coronavirus induced pancreatic injury.
Keywords: Acute pancreatitis, COVID 19
Introduction
Novel coronavirus (SARS-CoV-2) infection (COVID-19), in a short span of 9 months, has affected more than 25 million people worldwide with close to a million deaths already. The most common presentation of this disease is as respiratory tract infection. Little is known about the effect of COVID-19 on other organ systems. Here, we report a patient with COVID-19 that presented with acute pancreatitis.
Case report
A 29-year-old man presented with an acute diffused abdominal pain of five days duration that radiated to the back and progressively worsened. He had a low grade fever that responded to antipyretics. Two days prior to presentation, he also developed a worsening dyspnoea. He had no cough, joint pains, and bowel or bladder disturbances. He was not an alcoholic and had no co-morbidities. No similar episodes were experienced in the past. On examination, he was hemodynamically stable but had tachypnoea (36 breaths per minute). Auscultation of the chest revealed bilateral basal wheeze and there was abdominal tenderness which was maximal in the umbilical region.
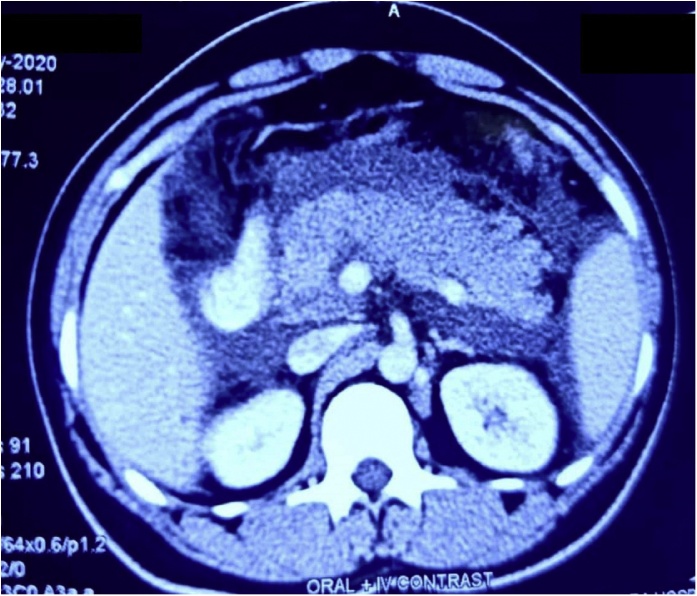
Laboratory evaluation revealed leucocytosis (17.8 × 103 cells/μL), mild transaminase elevation (AST: 88 IU/L, ALT: 34 IU/L), hypoalbuminemia (2.84 g/dL) and decreased serum calcium (6.64 mg/dL). Serum triglyceride level was normal (84 mg/dL). CRP (146 mg/L) and ESR (61 mm/first hour) were elevated. A diagnosis of acute pancreatitis was made as amylase (2861 IU/L) and lipase levels (1650 IU/L) were elevated to more than 20 and 30 times the normal, respectively. Ultrasonography revealed bulky pancreas with irregular oedematous margins along with some peripancreatic fluid and no evidence of common bile duct (CBD) calculi. These were confirmed in a subsequent CT scan of the abdomen (Fig. 1). Also, the patient’s initial blood gas analyses demonstrated hypoxemia with respiratory alkalosis and a compensatory metabolic acidosis. Chest radiograph showed left middle zone and bilateral lower zone consolidation. Nasopharyngeal swab for COVID-19 real-time PCR was positive.
Fig. 1.
CT Scan of the Abdomen showing Swollen Pancreas.
The patient was treated with intravenous meropenem and supportive care. He was given nothing by mouth for four days after which repeat amylase and lipase levels showed a reducing trend. Fever persisted for four more days even as the dyspnoea subsided. He was advanced to an oral diet and was satisfactorily discharged later.
Discussion
Apart from the obvious respiratory symptoms, nausea, vomiting and diarrhoea have commonly been noted in COVID-19. According to one meta-analysis on patients with COVID-19, as many as 50 % had detectable SARS-CoV-2 RNA in stool samples and 18 % had gastrointestinal symptoms. [1] Acute pancreatitis in COVID-19 has rarely been reported.
In our patient, COVID-19 presented as acute pancreatitis along with respiratory distress (PaO2/FiO2: 213). The diagnosis of pancreatitis in COVID-19 was based on laboratory and radiological evidence. The usual etiologies for acute pancreatitis including CBD calculi, alcohol abuse, drugs, trauma, hypertriglyceridemia and hypercalcemia were ruled out. Infectious pancreatitis is an entity mostly caused by viruses, like hepatotropic viruses, mumps, CMV, coxsackie B virus, HIV, varicella-zoster and influenza A. Previously, during the Severe Acute Respiratory Syndrome (SARS) epidemic of 2003, SARS-CoV was found to infect many visceral organs apart from lungs. [2] Angiotensin converting enzyme 2 (ACE2), the entry receptor for SARS-CoV, has been shown to be expressed in lungs, kidneys, gastrointestinal tract, pancreas and other tissues. [3] Recent analyses have revealed that SARS-CoV-2 strains have conserved most amino-acid residues including those essential for receptor binding, suggesting that they may also be targeting the same entry receptor [4]. This provides a plausible explanation to the involvement of pancreas in COVID-19.
There have been a few case reports of acute pancreatic injury in COVID-19. Hadi and his colleagues commented upon two COVID-19 cases with severe disease having significant pancreatitis. [5] Both had significantly elevated pancreatic enzyme levels and at least one of them had ultrasonographic evidence of pancreatic inflammation. Another case of COVID-19, reported by Anand and others, was found to have pancreatitis on CT evaluation for her abdominal pain [6]. A study by Liu and colleagues of 64 patients with severe COVID-19 reported that 17.9 % and 16.4 % had increased amylase and lipase levels, respectively. [7] But, of the 13 such patients, only 5 patients (7.46 %) showed pancreatic changes on CT imaging. Similarly, Wang et al. also found 17 % of their patients (n = 52) with COVID-19 to have evidence of pancreatic injury [8]. However, it is to be noted that the pancreatic injury in these cases was based on modestly elevated pancreatic enzyme levels.
Although no causal relationship has been established, a temporal association between pancreatitis and COVID-19 is strongly indicative of SARS-CoV-2-induced injury. It is worth noting that most of these cases, as also ours, have been reported in moderate or severe disease. This seemingly suggests that the pathophysiology of pancreatitis could be based on systemic inflammatory response rather than a direct cytopathological effect. We also wish to bring forth the difficulty in grading the severity of pancreatitis using traditional scoring systems which depend on assessment of resultant organ failure. It is difficult to attribute organ dysfunctions to pancreatitis when there is concomitant pneumonitis and widespread systemic inflammation. Nevertheless, our report highlights the need to thoroughly investigate COVID-19 patients with abdominal pain for pancreatitis and treat them accordingly.
Authorship statement
Dr Nikhil Kumar Patnaik, helped in writing the manuscript.
Dr Atul Gogia, Writing, conceptualizing and management of patient.
Dr Atul Kakar, Conceptualizing and management of patient.
Source of support
None.
Declaration of Competing Interest
The authors report no declarations of interest.
References
- 1.Cheung K.S., Hung I.F.N., Chan P.P.Y., Lung K.C., Tso E., Liu R. Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from the Hong Kong cohort: systematic review and meta-analysis. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.03.065. April 3: S0016-5085(20)30448-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ding Y., He L., Zhang Q., Huang Z., Che X., Hou J. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS-CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J Pathol. 2004;203(2):622–630. doi: 10.1002/path.1560. June. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang J.K., Lin S.S., Ji X.J., Guo L.M. Binding of SARS coronavirus to its receptor damages islets and causes acute diabetes. Acta Diabetol. 2010;47(3):193–199. doi: 10.1007/s00592-009-0109-4. September. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jaimes J.A., André N.M., Chappie J.S., Millet J.K., Whittaker G.R. Phylogenetic analysis and structural modeling of SARS-CoV-2 spike protein reveals an evolutionary distinct and proteolytically sensitive activation loop. J. Mol. Biol. 2020;432(10):3309–3325. doi: 10.1016/j.jmb.2020.04.009. May. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hadi A., Werge M., Kristiansen K.T., Pedersen U.G., Karstensen J.G., Novovic S. Coronavirus disease-19 (COVID-19) associated with severe acute pancreatitis: case report on three family members. Pancreatology. 2020;20(4):665–667. doi: 10.1016/j.pan.2020.04.021. June. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Anand E.R., Major C., Pickering O., Nelson M. Acute pancreatitis in a COVID-19 patient. Br. J. Surg. 2020;107(7):e182. doi: 10.1002/bjs.11657. June. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu F., Long X., Zhang B., Zhang W., Chen X., Zhang Z. ACE2 expression in pancreas may cause pancreatic damage after SARS-CoV-2 infection. Clin. Gastroenterol. Hepatol. 2020 doi: 10.1016/j.cgh.2020.04.040. April 22: S1542-3565(20)30537-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang F., Wang H., Fan J., Zhang Y., Wang H., Zhao Q. Pancreatic injury patterns in patients with COVID-19 pneumonia. Gastroenterology. 2020 doi: 10.1053/j.gastro.2020.03.055. April 1: S0016-5085(20)30409-1. [DOI] [PMC free article] [PubMed] [Google Scholar]