Abstract
Background
C-reactive protein (CRP) has two natural isomers: C-reactive protein pentamer (pCRP) and C-reactive protein monomer (mCRP). The levels of CRP are significantly elevated in patients with anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV). mCRP not only activates the endothelial cells, platelets, leukocytes, and complements, but also has a proinflammatory structural subtype that can localize and deposit in inflammatory tissues. Thus, it regulates a variety of clinical diseases, such as ischemia/reperfusion (I/R) injury, Alzheimer’s disease, age-related macular degeneration, and cardiovascular disease. We hypothesized that plasma mCRP levels are related to cardiovascular disease in AAV.
Methods
In this cross-sectional study, 37 patients with AAV were assessed. Brain natriuretic peptide (BNP) and mCRP in plasma were assessed by enzyme-linked immunosorbent assay (ELISA). The acute ST-segment elevation myocardial infarction (STEMI) was diagnosed by coronary angiography, and the Gensini score calculated. Echocardiography evaluated the ejection fraction (EF%), left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), and left ventricular mass index (LVMI). Estimated glomerular filtration rate (eGFR) was calculated based on serum creatinine, age, and gender.
Results
The plasma level of mCRP in AAV was significantly higher than that in healthy volunteers (P < 0.001). Then, mCRP and CRP levels were compared with and without STEMI complications in AAV. The plasma level of mCRP was higher, but that of CRP was lower in STEMI. The plasma level of mCRP was correlated with Birmingham vasculitis activity score (BVAS), eGFR, BNP, EF%, LVEDV, LVESV, LVMI, and STEMI complications’ Gensini score in AAV; however, CRP did not correlate with BNP, EF%, LVEDV, LVESV, LVMI, and Gensini score.
Conclusions
The plasma level of mCRP was related to cardiovascular diseases in AAV patients.
Keywords: mCRP, AAV, Cardiovascular diseases, eGFR
Introduction
Anti-neutrophil cytoplasmic antibody (ANCA)-associated vasculitis (AAV) is commonly characterized by a small amount of immunoglobulin deposition and segmental vascular wall necrotic inflammation [1]. AAV patients are classified into granulomas with polyangiitis (GPA), eosinophilic granulomas with polyangiitis (EGPA), and microvascular polyangiitis (MPA), according to pathological and clinical characteristics [2]. Target antigens for AAV diseases include protease 3 (PR3) and myeloperoxidase (MPO) [3, 4]. The majority of the Chinese patients with AAV are positive for MPO-ANCA [5–7], which activates and damages glomerular endothelial cells (GEnC) in patients with AAV [7–9].
C-reactive protein (CRP) is an acute-phase protein that serves as an inflammatory response biomarker [10, 11]. During activity and relapse of AAV disease, serum CRP levels increase rapidly, followed by a decrease during remission of the disease [12, 13]. Human CRP in blood circulation consists of C-reactive protein pentamer (pCRP) and tissue-related C-reactive protein monomer (mCRP) [14]. In atherosclerosis-related experiments in mice, CRP bound to the Fc receptor CD32, while mCRP bound to another human Fc receptor subtype CD16 in neutrophils and performed function that was opposite to that of CRP [15]. Thus, FC receptor may be the key that links CRP and mCRP to AAV.
The most common pathogenesis of ST-segment elevation myocardial infarction (STEMI) is atherosclerotic fragile plaque rupture and thrombus-induced cardiac muscle cell necrosis. Also, inflammatory reactions are crucial responses [16, 17]. CRP is involved in acute myocardial infarction. mCRP is obtained from the dissociation process of necrobiosis. Moreover, mCRP explains the function of CRP, while pCRP does not promote inflammation [18]. In vitro tests have shown that mCRP induces the synthesis of neutrophil-derived chemokines, stimulates the expression of monocyte integrin [19], and promotes neutrophil adhesion to vascular endothelial cells [20].
CRP is the classical acute-phase protein named for its ability to precipitate and bind to the pneumococcal C-polysaccharide. Although the blood circulation concentration is low in healthy individuals, the level of CRP increases sharply when tissue damage and inflammation occur [21, 22]. Currently, CRP is used not only as a marker of inflammation but also as a crucial and independent predictor of atherosclerotic thrombosis (including cardiovascular events) [22].
Active AAV patients have high levels of CRP [13, 23]. Also, renal involvement is common in patients with AAV, including renal tubulointerstitial (TI) lesions [24, 25]. mCRP is an acute-phase protein in tissues or cells, wherein it might act as an antigen in acute tubular interstitium (ATIN) [25]. Serum CRP is decomposed into mCRP and induces the generation of free oxygen radicals on the surface of apoptotic and necrotic cells, resulting in inflammatory reactions [26]. It was also found that mCRP is not only related to thrombosis [27] and atherosclerotic plaque rupture but can also be concentrated in localized areas after transient myocardial ischemia and related to acute myocardial infarction (AMI) [28]. Therefore, we hypothesized that plasma mCRP levels are related to cardiovascular disease in AAV.
Methods
Collecting plasma samples
This cross-sectional study utilized plasma samples collected from 37 patients with AAV and 20 healthy volunteers. The AAV patients were diagnosed in the Department of Nephrology, Affiliated Hospital of Inner Mongolia Medical University, from October 2018 to November 2019. The patients met the criteria set at the Chapel Hill Consensus Meeting [29]. All the participants provided informed consent. Subjects with other autoimmune diseases, such as IgA nephropathy, rheumatoid arthritis, Graves’ disease, and lupus nephritis, as well as secondary and various acute infectious vasculitis, were excluded. A volume of 5 mL venous blood from each patient was withdrawn and collected in EDTA anticoagulation tubes within 24 h of admission. The supernatant was separated by centrifugation at 3000 rpm, 10 min, and stored at − 80 °C for subsequent use. Repeated freeze-thaw cycles are prohibited for this sample. The disease activity was based on the Birmingham vasculitis activity score (BVAS) in AAV patients [30]. We collected baseline patient data including status, age, gender, tobacco, alcohol, hypertension, diabetes, obesity, and biochemical indexes. These cardiovascular risk factors included triglyceride (TG), total cholesterol (Tch), low-density lipoprotein (LDL-C), high-density lipoprotein (HDL-C), CRP, and fibrinogen (Fib). The study was approved by the Ethics Committee of the Helsinki Declaration of Inner Mongolia Medical University.
Detection of mCRP by enzyme-linked immunosorbent assay (ELISA)
Plasma mCRP levels were tested by ELISA using a commercial kit (Kamaishu Biotechnology Co, Shanghai, China). The 96-well plate was coated with mCRP and blocked to reduce non-specific binding. Antibodies compete for binding to a plate or sample. This kit uses the double-antibody sandwich method to determine the level of human mCRP in the specimen. The microtiter plates were coated with purified human mCRP antibodies to make solid-phase antibodies. Then, mCRP was added to the microwells, followed by the addition of HRP-labeled mCRP antibodies to form an antibody-antigen complex, as assessed by ELISA. Subsequently, the substrate tetramethylbenzidine (TMB) was used to develop the color. The absorbance (OD value) of the colored complex was measured at 450 nm, and the human mCRP concentration in the sample was calculated by a standard curve [31].
Detection of brain natriuretic peptide (BNP) by ELISA
BNP was detected by a double-antibody sandwich immunoenzyme method, using Unicel-TM-DXI800 (Beckman, CA, USA) full-automated immunoassay instrument. The ELISA was performed, as described previously [32].
Detection of circulating blood serum creatinine (Scr), estimated glomerular filtration rate (eGFR), and CRP
Scr was determined by a rating method using a commercial kit (Beckman, CA, USA) [33]. The level of CRP was determined by immunoturbidimetry using a commercially available kit (Goldsite, Shenzhen, China) [34]. According to Scr, age, and gender, the eGFR of the patient was calculated. The modification of diet in renal disease (MDRD) formula was adapted by Chinese experts. The female eGFR calculation formula was as follows: eGFR (mL/min/1.73 m2) = 186 × Scr − 1.154 × (age) − 0.203 × 0.742 × 1.227 and male eGFR calculation formula eGFR (mL/min/1.73 m2) = 186 × Scr − 1.154 × (age) − 0.203 × 1.227 [35].
Detection of ejection fraction (EF%), left ventricular end-diastolic volume (LVEDV), left ventricular end-systolic volume (LVESV), and left ventricular mass index (LVMI)
The values of EF%, LVEDV, LVESV, left ventricular septal end-diastolic thickness (IVSTD), and left ventricular posterior wall diastolic thickness (LVPWTD) were obtained by echocardiography. Then, the value of left ventricular myocardial mass (LVM) was calculated based on LVIDD, IVSTD, and LVPWTD. Body surface area (BSA) was calculated through weight (W) and height (H).
LVM (g) = 1.04 {[(LVIDD + PWTD + IVSTD)3 − (LVIDD)3] − 13.6}.
BSA (m2) = (W0.425 × H0.725) × 0.007184 [36].
Statistical analysis
All data were statistically analyzed using SPSS19.0. All grouped data were tested for normality before further analysis. The two groups of enumeration data that conformed to the normal distribution were compared using an independent t test. If any of the groups did not match, a non-parametric rank-sum test was used. If the data fulfilled the normal distribution, it would be expressed as means (x̅) ± standard deviation (SD). The Shapiro-Wilk test, median, and interquartile range (IQR) were used to check the degree of dispersion of the data. The correlation between the two groups of data was analyzed; if both groups met the normal distribution, Pearson’s rank correlation was used, and if any group did not meet the normal distribution, Spearman’s rank correlation was utilized. Cross-tabulation χ2 test was used for gender comparison. P < 0.05 indicated statistical significance.
Results
General data from AAV patients and healthy volunteers (normal control)
All the 37 AAV patients, including 17 (45.9%) females and 20 (54.1%) males, harbored MPO-ANCA. Among the 4 AAV patients with STEMI complications, 2 (50.0%) were males and 2 (50.0%) were females, with average age 59.5 ± 14.3 years, while that of the 37 AAV patients was 58.5 ± 10.6 years. The healthy volunteers consisted of 10 (50.0%) males and 10 (50.0%) females, aged 55.3 ± 10.7 years. The comparison of age and sex between 37 AAV patient groups and healthy volunteers (n = 20, P = 0.266; n = 20, P = 0.788) did not show significant differences. Next, we used the same method to compare the age and gender of 4 patients with STEMI complications and 33 patients without STEMI complications in the AAV group. Therefore, both the AAV patient group and the normal control, as well as patients with and without STEMI complications in the AAV group, showed no significant difference in the age and sex (P > 0.05). Also, the relevant biochemical indicators were analyzed in AAV patients (Table 1).
Table 1.
General data (biochemical indexes) in AAV
Abbreviations: Scr serum creatinine, Hb hemoglobin, PTH parathyroid hormone, CTnT cardiac traponin T, CK creatine kinase, CK-MB creatine kinase isoenzyme, HDL-C high-density lipoprotein, Tch total serum cholesterol, TG triglyceride, LDL-C low-density lipoprotein
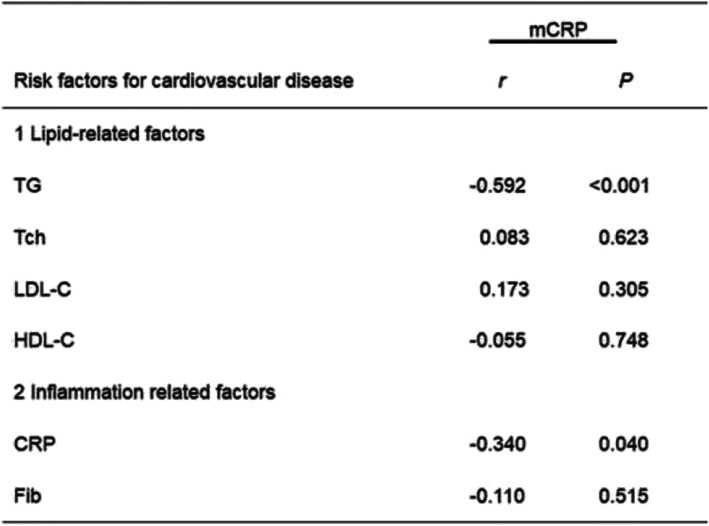
Correlation between mCRP and cardiovascular disease risk factors
The risk factors of cardiovascular diseases include status, age, gender, tobacco, alcohol, hypertension, diabetes, and obesity. Moreover, the correlation between clinical indicators of cardiovascular risk factors (lipid- and inflammation-related factors) and mCRP were compared. We found that mCRP was negatively correlated with total cholesterol (TG) and CRP (r = − 0.592, P < 0.001; r = − 0.340, P = 0.040, respectively), (Table 2).
Table 2.
Comparison of the relationship between clinical indicators of cardiovascular risk factors in AAV
Abbreviations: TG triglyceride, Tch total cholesterol, LDL-C low-density lipoprotein, HDL-C high-density lipoprotein, CRP C-reactive protein, Fib fibrinogen
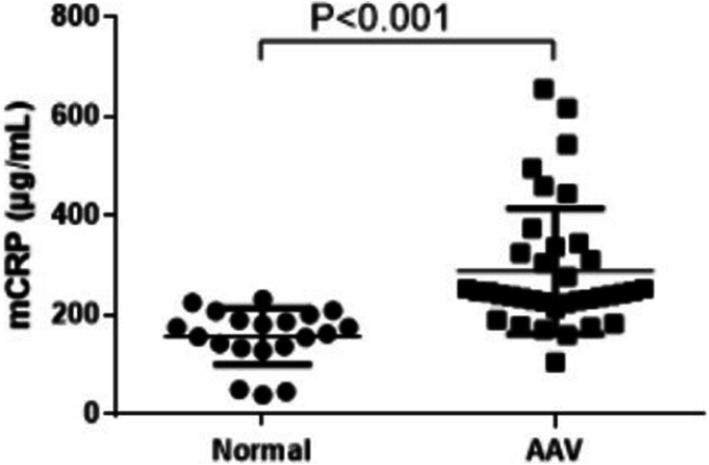
Plasma levels of mCRP were higher in AAV patients than in normal controls
The plasma level of mCRP in AAV patients was 244.12 (226.12, 331.725) μg/mL and that in the normal control was 170.0 (135.7, 199.3) μg/mL. Next, we compared the plasma levels of mCRP in AAV patients and healthy volunteers (P < 0.001) and found significant differences (Fig. 1).
Fig. 1.
Comparison of the plasma levels of mCRP in AAV patients (n = 37) and healthy volunteers (n = 20, P < 0.001)
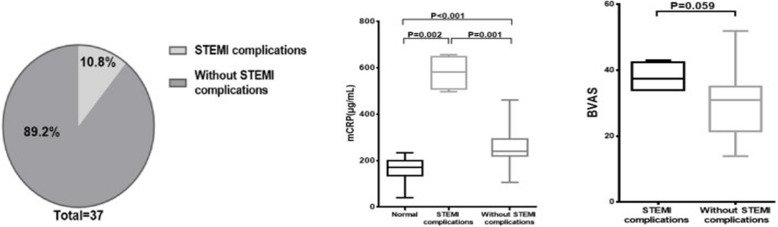
Plasma levels of mCRP were highest in patients with than in those without STEMI complications
The AAV patients were divided into groups with (n = 4) and without STEMI complications (n = 33) groups (Fig. 2a). The plasma concentration of mCRP in patients with and without STEMI complications in AAV group was 581.4 (508.7, 647.3) and 240.8 (219.2, 292.1) μg/mL, respectively, while that in normal controls was 170.0 (135.7, 199.3) μg/mL (Fig. 2b). We also compared the BVAS of patients with and without STEMI complications (Fig. 2c).
Fig. 2.
Plasma levels of mCRP were highest in STEMI complications in AAV than without STEMI complications in AAV and normal controls. a Thirty-seven AAV patients were divided into two groups: with STEMI complications (n = 4) and without STEMI complications (n = 33). b Plasma levels of mCRP in STEMI complications were highest in the three groups (with STEMI complications in AAV, without STEMI complications, normal). c Comparison of BVAS in groups with and without STEMI complications
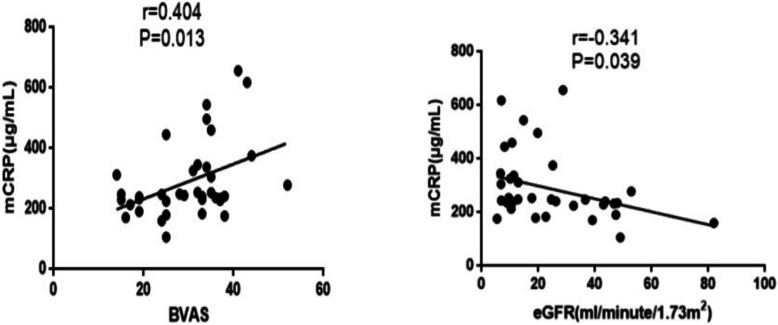
Plasma levels of mCRP were positively correlated with BVAS and Scr and negatively correlated with eGFR
BVAS in 37 AAV patients and normal controls was 29.9 ± 9.1 and 0, while Scr was 291.0 (161.5, 434.5), respectively. The eGFR was calculated based on gender, age, and Scr levels. The eGFR was 17.7 (10.1, 41.0) mL/min/1.73 m2 in AAV patients and 170.0 (135.7, 199.3) mL/min/1.73 m2 in normal controls. Thus, we analyzed the correlation between mCRP and BVAS (r = 0.404, P = 0.013, Fig. 3a) and mCRP and eGFR (r = − 0.341, P = 0.039, Fig. 3b).
Fig. 3.
Plasma levels of mCRP were compared with BVAS and eGFR in AAV. a Correlation between mCRP and BVAS. The plasma levels of mCRP were positively correlated with BVAS (r = 0.404, P = 0.013). BVAS, Birmingham vasculitis activity score. b Correlation between mCRP and eGFR. The plasma levels of mCRP were negatively correlated with eGFR (r = − 0.341, P = 0.039). eGFR, estimated glomerular filtration rate
Plasma levels of mCRP were correlated with BNP, EF%, LVEDV, LVESV, LVMI, and STEMI complications’ Gensini score, but not CRP
The Gensini score of patients with STEMI complications in AAV was 72.0 ± 15.1. The BNP level in circulating blood was 155.7 (104.4, 204.3) pg/mL, while the EF (%) levels were 63.0 ± 8.8%, LVEDV was 124.2 ± 25.3 mL, LVESV was 44.2 ± 10.7 mL, LVMI was 86.5 (50.6, 112.6), and the Gensini score of four STEMI complications was 72.0 ± 15.1 in AAV patients.
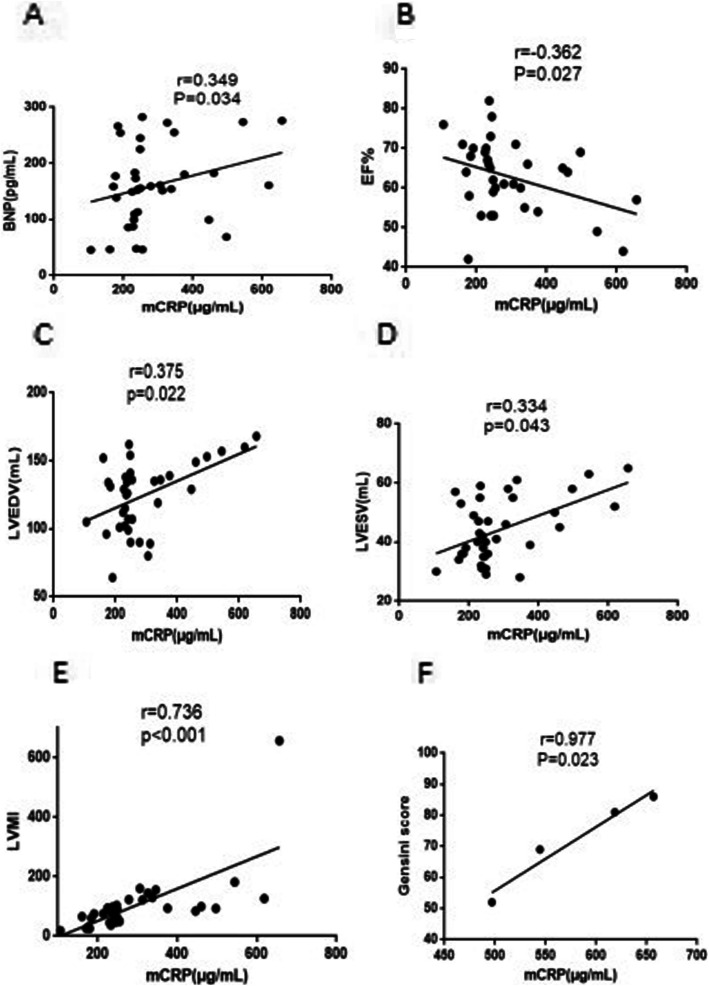
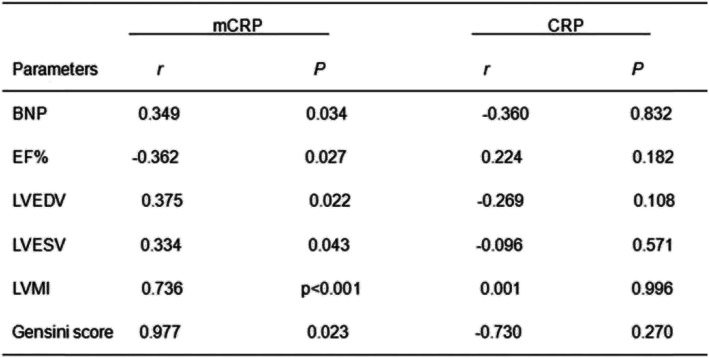
Furthermore, we speculated that the plasma levels of mCRP might be related to the patient’s heart disease, and hence, the correlation between mCRP and BNP, EF%, LVEDV, LVESV, LVMI, and Gensini score of STEMI complications was analyzed (r = 0.349, P = 0.034, Fig. 4a; r = − 0.362, P = 0.027, Fig. 4b; r = 0.375, P = 0.022, Fig. 4c, r = 0.334, P = 0.043, Fig. 4d; r = 0.736, P < 0.001, Fig. 4e; r = 0.997, P = 0.023, Fig. 4f, respectively), while CRP was not correlated (Table 3).
Fig. 4.
Plasma levels of mCRP were correlated with BNP, EF%, LVEDV, LVESV, LVMI, and Gensini score of STEMI complications in the AAV group. a Correlation between mCRP and BNP. The plasma levels of mCRP were positively correlated with BNP (r = 0.349, P = 0.034). BNP, brain natriuretic peptide. b Correlation of mCRP and EF%. The plasma levels of mCRP were negatively correlated with EF% (r = − 0.362, P = 0.027). EF%, ejection fraction. c Correlation between mCRP and LVEDV. The plasma levels of mCRP were positively correlated with LVEVD (r = 0.375, P = 0.022). LVEDV, left ventricular end-diastolic volume. d Correlation between mCRP and LVESV. The plasma levels of mCRP were positively correlated with LVESV (r = 0.334, P = 0.043). LVESV, left ventricular end-systolic volume. e Correlation between mCRP and LVMI. The plasma levels of mCRP were positively correlated with LVMI (r = 0.736, P < 0.001). LVMI, left ventricular mass index. f Correlation between mCRP and Gensini score. The plasma levels of mCRP were positively correlated with Gensini score of STEMI complications in the AAV group (r = 0.977, P = 0.02). STEMI, ST-segment elevation myocardial infarction
Table 3.
Comparison of mCRP and CRP with BNP, EF%, LVEDV, LVESV, LVMI, and Gensini score with STEMI complications
Abbreviations: BNP brain natriuretic peptide, EF% echocardiography: ejection fraction, LVEDV left ventricular end-diastolic volume, LVESV left ventricular end-systolic volume, LVMI left ventricular mass index
Discussion
Accumulating evidence has shown that CRP is pathogenic in atherosclerosis, acute myocardial infarction, cerebral infarction, and AAV disease [37]. CRP is one of the biomarkers for assessing AAV disease activity. The serum CRP concentration increased during the AAV active phase and decreased rapidly with the remission of the disease [13, 38]. mCRP can bind with complement factor H (CFH) and is more effective than pCRP in inhibiting alternative complement [39] and plays a critical role in the development of AAV [40, 41]. Xu et al. showed that MPO inhibits the binding between CFH and mCRP, thereby inhibiting the regulatory activation of alternative complements. PR3 does not bind to either pCRP or mCRP, while MPO can bind to mCRP, which blocks the binding between mCRP and CFH [41]. Therefore, the mechanism of mCRP and AAV disease is indistinguishable.
CRP can be decomposed into mCRP at high temperature, urea, or acidic microenvironment [42, 43]. mCRP activates platelets [20], monocytes [44], and endothelial cells [45]. It is a key substance that promotes inflammatory response [21, 46]. The mechanism underlying the inflammatory response of endothelial cells is caused by the interaction of mCRP with neutrophils, macrophages, and platelets [46, 47]. Furthermore, mCRP binds to CFH and directs it to the damaged part of the cell [48, 49], inactivates C3b, and limits the progression of inflammation [39]. Therefore, we speculated that the role of mCRP in the pathogenesis of AAV disease might be related to CFH.
In patients with lupus, the level of anti-mCRP autoantibodies is related to the degree of renal interstitial lesions [50], and mCRP has been reported as an autoantigen in interstitial nephritis-associated uveitis (TINU) syndrome [51]. In AAV patients, anti-mCRP antibodies may be the cause of severe TI lesions [25].
Serum CRP levels increase due to acute infection, trauma, and inflammation [52]. Thus, CRP is commonly used for the risk stratification of cardiovascular disease [53, 54], as well as a marker of generalized atherosclerosis [54, 55]. CRP levels are significantly increased in cardiovascular patients without any symptoms, obvious cardiovascular disease, unstable angina pectoris, myocardial infarction, and other diseases [22, 56]. Also, it is directly involved in the response process of acute myocardial infarction [18]. Thielle et al. demonstrated that mCRP stimulates inflammation, while pCRP has no proinflammatory effect [45]. In addition, Diehl et al. detected mCRP deposition in human atherosclerotic plaque vessels [57]. Similarly, studies on cardiovascular disease have found that mCRP is present in endothelial cells cultured in vitro and can promote inflammatory responses [20]. In the current study, 37 patients with AAV were collected and 4 patients with STEMI complications were found.
mCRP has lectin-like properties and can bind galactose-containing residues [12, 58, 59]. ANCA-induced NETs activate the platelets and then promote mCRP formation on these platelets. Subsequently, the newly generated mCRP can further enhance thrombosis and inflammatory response during platelet activation. mCRP may be a potential link between thrombosis and inflammation in AAV. Therefore, we suspected that the occurrence of STEMI complications in patients was related to mCRP [12]. AAV exhibits various echocardiographic characteristics, such as decreased systolic function, increased diastolic dysfunction, and pulmonary hypertension at the time of diagnosis [60]. The current study focused on cardiovascular disease in patients with AAV, which reflected the clinical examination of cardiovascular function by echocardiography. EF%, LVEDV, and LVESV are indicators of heart function. LVMI is associated with the mortality of patients with chronic kidney disease (CKD) and is a high-risk factor of heart failure [61]. Therefore, we analyzed the correlation between EF%, LVEDV, LVESV, LVMI, and mCRP. In this study, we proved that circulating blood levels of CRP were significantly higher in AAV patients than in healthy volunteers. Also, mCRP levels, but not CRP levels, were significantly increased in patients with STEMI complications. Next, we analyzed the correlation between mCRP levels and the Gensini score of STEMI complications and that between mCRP levels, BNP, and EF% in patients with AAV and found that the level of mCRP was positively correlated with the Gensini score and BNP and negatively correlated with EF%.
Nevertheless, the present study had some limitations. First, it was not a longitudinal study because while collecting cardiovascular clinical indicators, a large amount of experimental data were collected first, followed by comparison analysis. Finally, it was found that the mCRP level was related to the different parameters mentioned in this study. However, AAV patients have low morbidity but high mortality [12]. Most AAV patients visit our department for treatment due to abnormal renal function. The patients we collected in this study were MPO-ANCA because it is a predominant condition in Chinese AAV patients [14, 17]. Therefore, follow-up studies are imperative.
Conclusions
The level of mCRP is related not only to AAV disease but also to the occurrence of cardiovascular disease in patients with AAV. Therefore, mCRP can be used as a plasma biomarker of cardiovascular diseases in AAV.
Acknowledgements
The Affiliated Hospital of Inner Mongolia Medical University. Funding details are included above.
Abbreviations
- CRP
C-reactive protein
- pCRP
C-reactive protein pentamer
- mCRP
C-reactive protein monomer
- ANCA
Anti-neutrophil cytoplasmic antibody
- AAV
Anti-neutrophil cytoplasmic antibody-associated vasculitis
- PR3
Protease 3
- MPO
Myeloperoxidase
- Scr
Serum creatinine
- Hb
Hemoglobin
- PTH
Parathyroid hormone
- CTnT
Cardiac troponin T
- CK
Creatine kinase
- CK-MB
Creatine kinase isoenzyme
- HDL-C
High-density lipoprotein
- Tch
Total serum cholesterol
- TG
Triglyceride
- LDL-C
Low-density lipoprotein
- Fib
Fibrinogen
- ELISA
Enzyme-linked immunosorbent assay
- TMB
The substrate tetramethylbenzidine
- BNP
Brain natriuretic peptide
- STEMI
ST-segment elevation myocardial infarction
- EF%
Ejection fraction
- LVEDV
Left ventricular end-diastolic volume
- LVESV
Left ventricular end-systolic volume
- LVMI
Left ventricular mass index
- AMI
Acute myocardial infarction
- LVIDD
Left ventricular end-diastolic diameter
- IVSTD
Left ventricular septal end-diastolic thickness
- LVPWTD
Left ventricular posterior wall diastolic thickness
- LVM
Left ventricular myocardial mass
- GEnC
Glomerular endothelial cells
- TI
Tubulointerstitial
- ATIN
Acute tubulointerstitial
- eGFR
Glomerular filtration rate
- BVAS
Birmingham Vasculitis Activity Score
Authors’ contributions
Kai-Li Wu and Qing-Hui Liang conducted experiments, analyzed the data, and drafted the manuscript. Na Ding and Bo-Wei Li conducted experiments. Bin-Tao Huang participated in revising the manuscript. Jian Hao conceived the study, participated in revising the manuscript, and provided final approval to submit the version of the document. All authors read, and the report was approved.
Funding
The two funds are the China Natural Science Foundation (NO81460145) and Inner Mongolia Natural Science Foundation Project Number (2018MS08105).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
All patients provided their informed written consent. The study was approved by the Ethics Committee of the Helsinki Declaration of Inner Mongolia Medical University.
Consent for publication
Not applicable
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Kai-Li Wu and Qing-Hui Liang contributed equally to this work.
References
- 1.Jennette JC, Falk RJ. Small-vessel vasculitis. N Engl J Med. 1997;337(21):1512–1523. doi: 10.1056/NEJM199711203372106. [DOI] [PubMed] [Google Scholar]
- 2.Jennette JC, Falk R, Bacon P, et al. 2012 revised international Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheumatism. 2013;65(1):1–11. doi: 10.1002/art.37715. [DOI] [PubMed] [Google Scholar]
- 3.Segelmark M, Wieslander J. IgG subclasses of antineutrophil cytoplasm autoantibodies (ANCA) Nephrol Dialysis Transplantation. 1993;8(8):696–702. doi: 10.1093/ndt/8.8.696. [DOI] [PubMed] [Google Scholar]
- 4.Hao J, Huang Y-M, Zhao M-H, Chen M. The interaction between C5a and sphingosine-1-phosphate in neutrophils for antineutrophil cytoplasmic antibody mediated activation. Arthritis Res Therapy. 2014;16(4):R142. doi: 10.1186/ar4604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chen M, Yu F, Zhang Y, Zou W-Z, Zhao M-H, Wang H-Y. Characteristics of Chinese patients with Wegener’s granulomatosis with anti-myeloperoxidase autoantibodies. Kidney Int. 2005;68(5):2225–2229. doi: 10.1111/j.1523-1755.2005.00679.x. [DOI] [PubMed] [Google Scholar]
- 6.Li ZY, Chang DY, Zhao MH, Chen M. Predictors of treatment resistance and relapse in antineutrophil cytoplasmic antibody–associated vasculitis: a study of 439 cases in a single Chinese center. Arthritis Rheumatol. 2014;66(7):1920–1926. doi: 10.1002/art.38621. [DOI] [PubMed] [Google Scholar]
- 7.Sun X-J, Chen M, Zhao M-H. Thrombin contributes to anti-myeloperoxidase antibody positive IgG-mediated glomerular endothelial cells activation through SphK1-S1P-S1PR3 signaling. Front Immunol. 2019;10:237. doi: 10.3389/fimmu.2019.00237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nagao T, Matsumura M, Mabuchi A, et al. Up-regulation of adhesion molecule expression in glomerular endothelial cells by anti-myeloperoxidase antibody. Nephrol Dialysis Transplantation. 2007;22(1):77–87. doi: 10.1093/ndt/gfl555. [DOI] [PubMed] [Google Scholar]
- 9.Nagao T, Suzuki K, Utsunomiya K, et al. Direct activation of glomerular endothelial cells by anti-moesin activity of anti-myeloperoxidase antibody. Nephrol Dialysis Transplantation. 2011;26(9):2752–2760. doi: 10.1093/ndt/gfr032. [DOI] [PubMed] [Google Scholar]
- 10.Ten Oever J, Netea MG, Kullberg B-J. Utility of immune response-derived biomarkers in the differential diagnosis of inflammatory disorders. J Infect. 2016;72(1):1–18. doi: 10.1016/j.jinf.2015.09.007. [DOI] [PubMed] [Google Scholar]
- 11.Coster D, Wasserman A, Fisher E, et al. Using the kinetics of C-reactive protein response to improve the differential diagnosis between acutebacterial and viral infections. Infection. 2020;48:241–8. [DOI] [PubMed]
- 12.Xu P-C, Lin S, Yang X-W, et al. C-reactive protein enhances activation of coagulation system and inflammatory response through dissociating into monomeric form in antineutrophil cytoplasmic antibody-associated vasculitis. BMC Immunology. 2015;16(1):10. doi: 10.1186/s12865-015-0077-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kälsch A-I, Csernok E, Münch D, et al. Use of highly sensitive C-reactive protein for followup of Wegener’s granulomatosis. J Rheumatol. 2010;37(11):2319–2325. doi: 10.3899/jrheum.100302. [DOI] [PubMed] [Google Scholar]
- 14.Shrive AK, Cheetham GM, Holden D, et al. Three dimensional structure of human C-reactive protein. Nat Struct Biol. 1996;3(4):346–354. doi: 10.1038/nsb0496-346. [DOI] [PubMed] [Google Scholar]
- 15.Yeh ETH. A new perspective on the biology of C-reactive protein. Circ Res. 2005;97:609–11. [DOI] [PubMed]
- 16.Nakajima T, Yamazaki K. Periodontal disease and risk of atherosclerotic coronary heart disease. Odontology. 2009;97(2):84–91. doi: 10.1007/s10266-009-0104-9. [DOI] [PubMed] [Google Scholar]
- 17.Papathanasiou AI, Pappas KD, Korantzopoulos P, et al. An epidemiologic study of acute coronary syndromes in northwestern Greece. Angiology. 2004;55(2):187–194. doi: 10.1177/000331970405500211. [DOI] [PubMed] [Google Scholar]
- 18.Ridker PM. C-reactive protein and the prediction of cardiovascular events among those at intermediate risk: moving an inflammatory hypothesis toward consensus. J Am Coll Cardiol. 2007;49(21):2129–2138. doi: 10.1016/j.jacc.2007.02.052. [DOI] [PubMed] [Google Scholar]
- 19.Khreiss T, József L, Potempa LA, Filep JG. Loss of pentameric symmetry in C-reactive protein induces interleukin-8 secretion through peroxynitrite signaling in human neutrophils. Circ Res. 2005;97(7):690–697. doi: 10.1161/01.RES.0000183881.11739.CB. [DOI] [PubMed] [Google Scholar]
- 20.Eisenhardt SU, Habersberger J, Murphy A, et al. Dissociation of pentameric to monomeric C-reactive protein on activated platelets localizes inflammation to atherosclerotic plaques. Circ Res. 2009;105(2):128–137. doi: 10.1161/CIRCRESAHA.108.190611. [DOI] [PubMed] [Google Scholar]
- 21.Wu Y, Potempa LA, El Kebir D, Filep JG. C-reactive protein and inflammation: conformational changes affect function. Biol Chem. 2015;396(11):1181–1197. doi: 10.1515/hsz-2015-0149. [DOI] [PubMed] [Google Scholar]
- 22.Boncler M, Wu Y, Watala C. The multiple faces of C-reactive protein—physiological and pathophysiological implications in cardiovascular disease. Molecules. 2019;24(11):2062. doi: 10.3390/molecules24112062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chen S-F, Wang F-M, Li Z-Y, Yu F, Chen M, Zhao M-H. The functional activities of complement factor H are impaired in patients with ANCA-positive vasculitis. Clin Immunol. 2017;175:41–50. doi: 10.1016/j.clim.2016.11.013. [DOI] [PubMed] [Google Scholar]
- 24.Zonozi R, Niles JL, Cortazar FB. Renal involvement in antineutrophil cytoplasmic antibody–associated vasculitis. Rheum Dis Clin. 2018;44(4):525–543. doi: 10.1016/j.rdc.2018.06.001. [DOI] [PubMed] [Google Scholar]
- 25.Lin Z-S, Liu X-L, Cui Z, et al. Acute tubulointerstitial nephritis with germinal centers in antineutrophil cytoplasmic antibody-associated vasculitis: a case report and literature review. Medicine. 2019;98(48):e18178. doi: 10.1097/MD.0000000000018178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Wang H-W, Sui S-F. Dissociation and subunit rearrangement of membrane-bound human C-reactive proteins. Biochem Biophys Res Commun. 2001;288(1):75–79. doi: 10.1006/bbrc.2001.5733. [DOI] [PubMed] [Google Scholar]
- 27.Molins B, Peña E, de la Torre R, Badimon L. Monomeric C-reactive protein is prothrombotic and dissociates from circulating pentameric C-reactive protein on adhered activated platelets under flow. Cardiovasc Res. 2011;92(2):328–337. doi: 10.1093/cvr/cvr226. [DOI] [PubMed] [Google Scholar]
- 28.Wang J, Tang B, Liu X, et al. Increased monomeric CRP levels in acute myocardial infarction: a possible new and specific biomarker for diagnosis and severity assessment of disease. Atherosclerosis. 2015;239(2):343–349. doi: 10.1016/j.atherosclerosis.2015.01.024. [DOI] [PubMed] [Google Scholar]
- 29.Schreiber A, Xiao H, Jennette JC, Schneider W, Luft FC, Kettritz R. C5a receptor mediates neutrophil activation and ANCA-induced glomerulonephritis. J Am Soc Nephrol. 2009;20(2):289–298. doi: 10.1681/ASN.2008050497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Luqmani R, Bacon P, Moots R, et al. Birmingham vasculitis activity score (BVAS) Dim system necrotizinig vasculitis. QJM Int J Med. 1994;87(11):671–678. [PubMed] [Google Scholar]
- 31.Tan Y, Yu F, Qu Z, et al. Modified C-reactive protein might be a target autoantigen of TINU syndrome. Clin J Am Soc Nephrol. 2011;6(1):93–100. doi: 10.2215/CJN.09051209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mayo D, Colletti JE, Kuo D. Brain natriuretic peptide (BNP) testing in the emergency department. J Emerg Med. 2006;31(2):201–210. doi: 10.1016/j.jemermed.2005.08.022. [DOI] [PubMed] [Google Scholar]
- 33.World Health Organization. Use of anticoagulants in diagnostic laboratory investigations. World Health Organization. 2002. p. 1–62.
- 34.Rosen M. C-reactive protein: a marker of infection, inflammation, tissue damage and malignancy. Diag Clin Testing. 1990;28:18–22. [Google Scholar]
- 35.Ma Y-C, Zuo L, Chen J-H, et al. Modified glomerular filtration rate estimating equation for Chinese patients with chronic kidney disease. J Am Soc Nephrol. 2006;17(10):2937–2944. doi: 10.1681/ASN.2006040368. [DOI] [PubMed] [Google Scholar]
- 36.Mostfa SA. Impact of obesity and surgical weight reduction on cardiac remodeling. Indian Heart J. 2018;70(Suppl3):S224–8. [DOI] [PMC free article] [PubMed]
- 37.Gill R, Kemp JA, Sabin C, Pepys MB. Human C-reactive protein increases cerebral infarct size after middle cerebral artery occlusion in adult rats. J Cereb Blood Flow Metab. 2004;24(11):1214–1218. doi: 10.1097/01.WCB.0000136517.61642.99. [DOI] [PubMed] [Google Scholar]
- 38.Xu P-C, Hao J, Yang X-W, D-y C, Chen M, M-h Z. C-reactive protein enhances the respiratory burst of neutrophils-induced by antineutrophil cytoplasmic antibody. Mol Immunol. 2012;52(3–4):148–154. doi: 10.1016/j.molimm.2012.05.012. [DOI] [PubMed] [Google Scholar]
- 39.Mihlan M, Blom AM, Kupreishvili K, et al. Monomeric C-reactive protein modulates classic complement activation on necrotic cells. FASEB J. 2011;25(12):4198–4210. doi: 10.1096/fj.11-186460. [DOI] [PubMed] [Google Scholar]
- 40.Hao J, Meng L-Q, Xu P-C, et al. p38MAPK, ERK and PI3K signaling pathways are involved in C5a-primed neutrophils for ANCA-mediated activation. PLoS ONE. 2012;7:e38317. [DOI] [PMC free article] [PubMed]
- 41.Xu P-c, Z-y L, Yang X-w, M-h Z, Chen M. Myeloperoxidase influences the complement regulatory function of modified C-reactive protein. Innate Immunity. 2014;20(4):440–448. doi: 10.1177/1753425913508164. [DOI] [PubMed] [Google Scholar]
- 42.Potempa LA, Maldonado BA, Laurent P, Zemel ES, Gewurz H. Antigenic, electrophoretic and binding alterations of human C-reactive protein modified selectively in the absence of calcium. Mol Immunol. 1983;20(11):1165–1175. doi: 10.1016/0161-5890(83)90140-2. [DOI] [PubMed] [Google Scholar]
- 43.Kresl JJ, Potempa LA, Anderson BE. Conversion of native oligomeric to a modified monomeric form of human C-reactive protein. Int J Biochem Cell Biol. 1998;30(12):1415–1426. doi: 10.1016/S1357-2725(98)00078-8. [DOI] [PubMed] [Google Scholar]
- 44.Braig D, Nero TL, Koch H-G, et al. Transitional changes in the CRP structure lead to the exposure of proinflammatory binding sites. Nat Commun. 2017;8(1):1–19. doi: 10.1038/ncomms14188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Thiele JR, Habersberger J, Braig D, et al. Dissociation of pentameric to monomeric C-reactive protein localizes and aggravates inflammation: in vivo proof of a powerful proinflammatory mechanism and a new anti-inflammatory strategy. Circulation. 2014;130(1):35–50. doi: 10.1161/CIRCULATIONAHA.113.007124. [DOI] [PubMed] [Google Scholar]
- 46.Caprio V, Badimon L, Di Napoli M, et al. pCRP-mCRP dissociation mechanisms as potential targets for the development of small-molecule anti-inflammatory chemotherapeutics. Front Immunol. 2018;9:1089. doi: 10.3389/fimmu.2018.01089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eisenhardt SU, Thiele JR, Bannasch H, Stark GB, Peter K. C-reactive protein: how conformational changes influence inflammatory properties. Cell Cycle. 2009;8(23):3885–3892. doi: 10.4161/cc.8.23.10068. [DOI] [PubMed] [Google Scholar]
- 48.Mihlan M, Stippa S, Józsi M, Zipfel P. Monomeric CRP contributes to complement control in fluid phase and on cellular surfaces and increases phagocytosis by recruiting factor H. Cell Death Differentiation. 2009;16(12):1630–1640. doi: 10.1038/cdd.2009.103. [DOI] [PubMed] [Google Scholar]
- 49.O'Flynn J, van der Pol P, Dixon KO, Prohászka Z, Daha MR, van Kooten C. Monomeric C-reactive protein inhibits renal cell-directed complement activation mediated by properdin. Am J Physiol-Renal Physiol. 2016;310(11):F1308–F1316. doi: 10.1152/ajprenal.00645.2014. [DOI] [PubMed] [Google Scholar]
- 50.Tan Y, Yu F, Yang H, Chen M, Fang Q, Zhao M-H. Autoantibodies against monomeric C-reactive protein in sera from patients with lupus nephritis are associated with disease activity and renal tubulointerstitial lesions. Hum Immunol 2008;69(12):0–844. [DOI] [PubMed]
- 51.Tan Y, Yu F, Qu Z, Su T, Zhao MH. Modified C-reactive protein might be a target autoantigen of TINU syndrome. Clin J Am Soc Nephrol Cjasn. 2011;6(1):93–100. doi: 10.2215/CJN.09051209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pepys MB, Hirschfield GM. C-reactive protein: a critical update. J Clin Invest. 2003;111(12):1805–1812. doi: 10.1172/JCI200318921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ridker PM. A test in context: high-sensitivity C-reactive protein. J Am Coll Cardiol. 2016;67(6):712–723. doi: 10.1016/j.jacc.2015.11.037. [DOI] [PubMed] [Google Scholar]
- 54.Li Y, Zhong X, Cheng G, et al. Hs-CRP and all-cause, cardiovascular, and cancer mortality risk: a meta-analysis. Atherosclerosis. 2017;259:75–82. doi: 10.1016/j.atherosclerosis.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 55.Elias-Smale SE, Kardys I, Oudkerk M, Hofman A, Witteman JC. C-reactive protein is related to extent and progression of coronary and extra-coronary atherosclerosis; results from the Rotterdam study. Atherosclerosis. 2007;195(2):e195–e202. doi: 10.1016/j.atherosclerosis.2007.07.006. [DOI] [PubMed] [Google Scholar]
- 56.Ridker PM. Clinical application of C-reactive protein for cardiovascular disease detection and prevention. Circulation. 2003;107(3):363–369. doi: 10.1161/01.CIR.0000053730.47739.3C. [DOI] [PubMed] [Google Scholar]
- 57.Diehl EE, Haines GK, Radosevich JA, Potempa LA. Immunohistochemical localization of modified C-reactive protein antigen in normal vascular tissue. Am J Med Sci. 2000;319(2):79–83. doi: 10.1016/S0002-9629(15)40692-5. [DOI] [PubMed] [Google Scholar]
- 58.Lee RT, Lee YC. Carbohydrate ligands of human C-reactive protein: binding of neoglycoproteins containing galactose-6-phosphate and galactose-terminated disaccharide. Glycoconj J. 2006;23(5–6):317–327. doi: 10.1007/s10719-006-6173-x. [DOI] [PubMed] [Google Scholar]
- 59.Köttgen E, Hell B, Kage A, Tauber R. Lectin specificity and binding characteristics of human C-reactive protein. J Immunol. 1992;149(2):445–453. [PubMed] [Google Scholar]
- 60.Ahn SSPE, Jung SM, Song JJ, Park YB, Lee SW. Echocardiographic features in patients with ANCA-associated vasculitis within 3 months before and after diagnosis. Clin Rheumatol. 2017;36(12):2751–2759. doi: 10.1007/s10067-017-3868-2. [DOI] [PubMed] [Google Scholar]
- 61.Tripepi G, D'Arrigo G, Mallamaci F, et al. Prognostic values of left ventricular mass index in chronic kidney disease patients. Nephrol Dial Transplant. 2019;1–8. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.