Abstract
Myeloid cells are components of the innate immune system that represent the first line of defense. Tissue damage, associated with pathological conditions such as infection, cancer or autoimmunity, leads to the exposure of the intracellular content to the extracellular environment. Myeloid cells detect ligands exposed or released by dead cells through specific receptors that signal for a diversity of responses. Inflammatory responses triggered by myeloid cells after sensing tissue injury can contribute to resolution of the damage. The signaling response following dead-cell sensing by myeloid cells can contribute either to an inflammatory or a regulatory response. We review herein some representative examples of how myeloid cells react to the recognition of cell death during specific tissue damage contexts. A deep understanding of the cellular and molecular mechanisms underlying these processes would allow to improve therapeutical interventions in pathologies associated with tissue damage.
Current Opinion in Immunology 2021, 68:34–40
This review comes from a themed issue on Innate immunity
Edited by Carla V Rothlin, Vijay A Rathinam and Sourav Ghosh
For a complete overview see the Issue and the Editorial
Available online 7th October 2020
https://doi.org/10.1016/j.coi.2020.08.006
0952-7915/© 2020 Elsevier Ltd. All rights reserved.
Introduction
The structural integrity of a tissue may be compromised due to an infection, trauma or any other inflammatory process. Because of this damage, tissue homeostasis is altered, impacting on its performance. At the cellular level, the injury can trigger Accidental Cell Death (ACD) such as necrosis or Regulated Cell Death (RCD) mechanisms such as pyroptosis or necroptosis [1,2]. In any case, the ultimate consequence of all these phenomena is cell death and the exposure and release of intracellular content to the extracellular environment.
All mammalian tissues are surveilled by myeloid cells including monocytes, macrophages, granulocytes and dendritic cells (DCs). This myeloid compartment patrols the surroundings, ready to sense and react against both infectious and sterile challenges. These cells express germline-encoded pattern recognition receptors (PRRs) that allow the recognition of molecular traits emerging during those insults. These factors are defined as Pathogen-Associated Molecular Patterns (PAMPs) when coming from infectious agents or Damage-Associated Molecular Patterns (DAMPs) if they are of self-origin [3]. DAMPs (also referred to as alarmins) are intracellular components either cytosolic or nuclear, which are thus protected within the plasma membrane, being exposed or released upon tissue damage [4].
The relevance of DAMPs sensing was first proposed by Polly Matzinger in the ‘Danger theory’ [4]. It allowed to explain how immune responses are triggered in the apparent absence of microbial insults, by recognizing these endogenous danger signals. Interestingly, the consequences of this recognition can be a double-edge sword with either inflammatory or regulatory/tolerogenic outcomes. Herein, we will review some examples of the outcomes of tissue damage recognition, and how we could harness these processes to achieve clinical benefits in conditions associated with tissue damage, such as infection, cancer or autoimmunity.
Inflammatory sensing of tissue damage
Sensing of tissue damage can trigger potent inflammatory responses that can induce immunity. This inflammatory cell death has been particularly characterized in the context of antitumor treatments and is mediated by myeloid cells such as DCs [5]. Local chemotherapies and radiotherapies in cancer kill tumor cells based on their high metabolic and proliferative activities but can also lead to abscopal effects that result in shrinkage of distant tumors, implicating a systemic anti-tumor response. Certain forms of tumor cell death may expose or release DAMPs that are sensed by infiltrating myeloid cells, triggering an inflammatory response against the tumor. This process may overcome the immunosuppressive microenvironment generated within tumors to evade immunosurveillance. Therefore, there is an intense effort to identify and boost cancer therapies that drive immunogenic cell death because of the double benefit of these treatments, with many approaches reaching clinical trials [6•].
DNA sensing
The StimulaTor of INterferon Genes (STING) is a sensor of cyclic-GAMP produced by cGAS upon binding to intracellular DNA. STING activation stimulates the production of type I interferon (IFN-I) [7]. Notably, IFN-I is required for efficient antitumor CD8+ T cell responses [8,9]. In this sense, STING is critical for inducing inflammatory responses against grafted tumors, as STING-deficient mice lack tumor-specific cytotoxic T cells and are unable to control tumor growth [10••]. The driver of these responses is the STING-mediated recognition of tumor DNA by myeloid cells, where STING signaling promotes tumor antigen cross-presentation [10••,11••]. Supporting the protective role of DNA release associated with tumor cell death during radiotherapy, the efficacy of irradiation-based treatments in immunogenic tumors relies on the STING-dependent IFN-I production by tumor infiltrating myeloid CD11c+ cells [12]. Furthermore, chemotherapies also activate the STING pathway, inducing features of immunotherapy response such as PD-L1 expression [13]. These results support the use of STING agonist as cancer therapeutics to activate antitumor immune responses [14,15].
During tissue injury, mitochondrial DNA (mtDNA) is also released extracellularly as a pro-inflammatory DAMP [16] that is primarily recognized by the endosomal Toll-Like Receptor 9 (TLR9) upon phagocytosis by myeloid cells [17]. The relevance of extracellular mtDNA recognition by myeloid cells through TLR9 has been shown in pathologic conditions as diverse as Inflammatory Bowel Disease (IBD) [18], Systemic Inflammatory Response Syndrome (SIRS) [17] or tissue injury due to endotracheal intubation [19]. In addition, upon access of mtDNA to the cytosol, intracellular receptors such as STING [20] or the NLRP3 inflammasome [21] can also sense mtDNA. These data illustrate how, beyond the proposed therapeutical applications for STING agonists in cancer immunotherapy, the intracellular recognition of cell-free nucleic acids released during tissue damage may show a dark side: chronic inflammation and autoimmunity [22].
In fact, the inflammatory capacity of DNA sensing is tightly regulated during homeostasis, in order to maintain cytosolic self-DNA levels under the threshold of receptor activation in myeloid cells, preventing the occurrence of autoimmunity. Thus, a number of cytoplasmic nucleases are in charge of the cleavage and clearance of unwelcome cytoplasmic DNA, such as TREX1, DNaseII, RNaseH2 and SAMHD1 [23]. The accumulation of cytosolic DNA due to loss-of-function mutations of these enzymes leads to the development of autoimmune type I interferonopathies [24•]. For instance, Aicardi–Goutières syndrome (AGS) is a systemic inflammatory disease with severe neurological manifestations that is caused by TREX1 mutations, with TREX1-deficient mice showing a similar pro-inflammatory phenotype [25]. Interestingly, a concomitant cGAS deletion in these mice reverts the autoimmune disease [25], indicating the relevance of DNA sensing and the potential therapeutic application of its inhibition for treating these type of pathologies.
Nuclear proteins
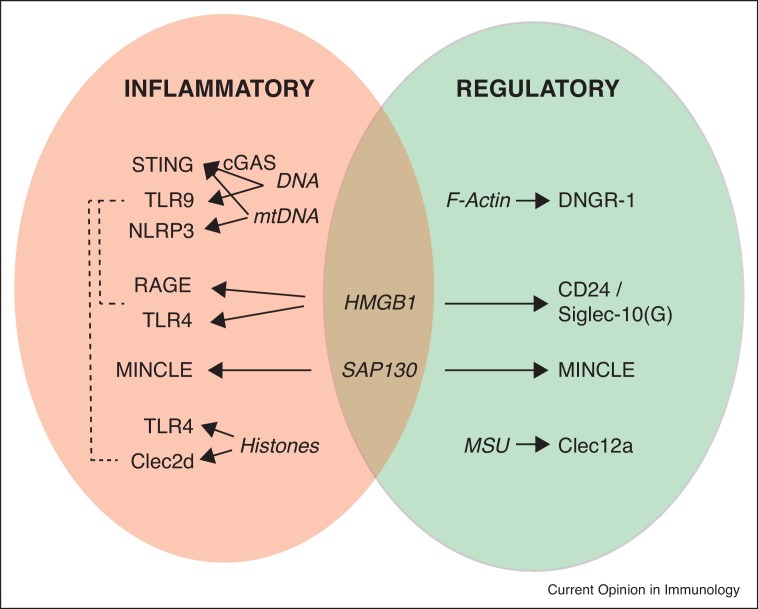
DNA is not the only pro-inflammatory trait that released during tissue injury (Figure 1 , left oval). High mobility group B1 (HMGB1) is a DNA-binding nuclear protein with important structural roles in the assembly of nucleoprotein complexes [26]. But HMGB1 can also promote autophagy when translocated to the cytosol [27]. Importantly, necrotic cells release HMGB1 to the extracellular medium while apoptotic cells do not [28], indicating a specific role of HMGB1 as a DAMP during tissue damage conditions. This alarmin is recognized by different PRR expressed in myeloid cells such as the Receptor for Advanced Glycation End products (RAGE) or the TLR2 and TLR4. HMGB1 recognition activates downstream signaling pathways to promote the production of proinflammatory TNFα, IL-1/-6 or CXCL2, among others [29] and can also promote antigen processing and cross-presentation when binding to TLR4 [30]. Mirroring what happens for DNA recognition, HMGB1 inflammatory sensing can be exploited therapeutically but can also show detrimental effects [31]. For instance, docetaxel chemotherapy induces the release of HMGB1 from dying tumor cells in non-small cell lung cancer (NSCLC) patients, which, in turn, activates infiltrating myeloid CD11c+ cells [32]. However, a detrimental pro-inflammatory activity of HMGB1 during conditions of tissue damage has been described in a number of pathologies such as hepatic obstruction [33], spinal cord injury [34] or LPS-induced lung injury [35]. This has motivated the study of the HMGB1 inhibitor glycyrrhizin to improve the outcome of tissue damage-related pathologies such as stroke [36] or radiation-induced acute lung injury [37]. In addition, activated myeloid cells actively secrete HMGB1 [38], establishing an auto/paracrine feedback-loop to reinforce the pro-inflammatory reaction to tissue damage. This circuit is relevant in Chronic Obstruction Pulmonary Disease (COPD). In this pathology, the congested lung tissue releases HMGB1. Of note, patients suffering COPD cannot clear efficiently the ubiquitous fungal pathogen Aspergillus fumigatus, which induces the production of further HMGB1 by myeloid alveolar macrophages, perpetuating the inflammatory state [39].
Figure 1.
Inflammatory and regulatory functions of myeloid receptors and DAMPs compiled in this review. Upon tissue damage conditions, dead cells release DAMPs (in italic in the chart) that can be recognized by receptors expressed in myeloid cells. This recognition triggers either inflammatory (left oval) or regulatory (right oval) responses. Some of these receptors can work simultaneously (dashed lines) as dying cells do not release isolated DAMPs but a mix of them. Of note, some of these DAMPs such as HMGB1 or SAP130 can trigger both inflammatory and regulatory responses depending on the context.
Another nucleoprotein released during accidental cell death is the spliceosome-associated protein 130 (SAP130). This DAMP binds to the C-type Lectin Receptor (CLR) MINCLE/CLEC4E expressed by myeloid cells, promoting the recruitment of neutrophils to the site of tissue damage [40]. The pro-inflammatory role of MINCLE in recognition of endogenous ligands (including SAP130 [40] and β-glucosyl-ceramide [41]) has been described in a number of tissue-injury related conditions such as subarachnoid hemorrhage [42], alcohol-induced hepatic injury [43], Crohn’s disease [44], multiple sclerosis [45] and acute kidney injury [41].
Nuclear histones are also alarmins released during tissue damage conditions occurring upon a number of acute critical illnesses [46]. Histones are recognized in the extracellular environment by myeloid cells, mostly through TLR4 [47], leading to pro-inflammatory immune responses. These responses can be further boosted in certain conditions. Streptococcus pyogenes infection generates necrotizing lesions of soft tissues, where histones are released as DAMPs. At the same time, the bacteria produce the Streptococcal Inhibitor of Complement (SIC) virulence factor that binds to histones. Histones, similar to anti-microbial peptides, show some anti-bacterial activity [48,49], which is dampened by SIC binding [50]; however, the pro-inflammatory activity of histones and their associated DNA is boosted upon SIC binding compared with free extracellular histones [50]. Interestingly, the pro-inflammatory activity of histones is also boosted when they are recognized in concert with other DAMPs.
Combined pro-inflammatory sensing of nuclear DAMPs
Although the triggering of some immunogenic cell death programs such as necroptosis or pyroptosis is well regulated at the molecular level [1], the eventual consequence of this death processes is the disruption of the cell integrity and the release of a massive ‘cocktail’ of intracellular matter. In this scenario, it is expected that a mix of DAMPs, rather than a single one, drive immunogenic responses. This is the case, for instance, for the already introduced inflammatory effect of HMGB1 and DNA sensing by myeloid cells. Both intracellular components contribute separately to inflammation upon cardiac ischemia-reperfusion through RAGE and TLR9, respectively [51]. However, under similar settings (hepatic ischemia-reperfusion), TLR4 is the critical receptor for HMGB1 recognition with an equivalent contribution of TLR9 by sensing DNA bound to histones [52].
But these DAMPs can also work in a cooperative manner. During autoimmune responses in lupus, there is a mutual interaction between HMGB1 and DNA, as complexes of both components augment the binding of HMGB1 to RAGE, boosting in turn TLR9-mediated inflammatory responses [53]. Binding of nucleic acids to HMGBs may be a precondition for the recognition by and activation of TLR, RLR and cytosolic nucleic-acid receptors [54]. In fact, this mechanism driven by HMGB1 has been proposed to play a role in the severe pulmonary inflammation occurring at late stages of COVID-19 [55]. In a similar way, histones released upon cell death are recognized by the CLR CLEC2D, transferring them to endosomal compartments where histones-bound DNA triggers TLR9-mediated inflammatory responses [56••].
Regulation of inflammation by sensing tissue damage
During most of the conditions described before, the recognition of DAMPs by myeloid cells triggers direct inflammatory responses by activating signaling pathways downstream certain PRRs. However, the sensing of tissue injury is not always inflammatory but can rather regulate inflammation (Figure 1, right oval). Under these circumstances, the sensing of tissue damage through specific sensors dampens inflammatory responses triggered by heterologous receptors in the same cell. These regulatory responses to tissue damage can dampen the recruitment of pro-inflammatory populations such as monocytes or neutrophils that may contribute to tissue damage.
This is the case for the detection of filamentous actin (F-Actin) by the CLR DNGR-1/CLEC9A [57,58], reviewed in Ref. [59]. F-Actin is sheltered intracellularly in healthy cells, but exposed to the extracellular medium when the cell membrane is disrupted. F-Actin recognition by DNGR-1 mediates cross-presentation of dead cells-associated antigens following Vaccinia virus (VACV) or Herpes Simplex Virus (HSV) infection [60, 61, 62]. However, under both sterile (caerulein-induced pancreatitis) and infectious (systemic Candida albicans) conditions running with massive local tissue damage, DNGR-1 expressed in dendritic cells dampens the infiltration of neutrophils that contribute to immunopathology. Thus, upon those conditions, DNGR-1-deficient mice show exacerbated tissue injury due to an overwhelming presence of infiltrating neutrophils. Mechanistically, DNGR-1 engagement activates the phosphatase SHP-1; this phosphatase inhibits the activation of heterologous receptors expressed in the same dendritic cell, receptors which are involved in the ignition of pro-inflammatory responses [63••].
Of note, the detrimental effect found in the absence of DNGR-1 was not coupled to an increase in pathogen burden during C. albicans infection [63••]. This effect is reminiscent of the ‘disease tolerance’ process. This protective mechanism implies the activation of regulatory immune responses during infections, in order to reduce self-inflicted immune tissue damage without affecting pathogen load [64•].
Paradoxical regulatory responses of tissue damage receptors
The negative regulation of neutrophil infiltration under sterile tissue damage conditions was also described for another myeloid CLR, CLEC12A/CLL-1 [1]. In this case, monosodium urate (MSU) crystals are the recognized DAMPs. Thus, CLEC12A-deficient mice showed elevated peritoneal neutrophil numbers after injecting dead cells, or in the thymus after sublethal total body irradiation [65•]. However, depending on the context, the immune consequences of tissue injury recognition by myeloid cells and their receptors can be paradoxical. For instance, antibody-mediated blockade of CLEC12A reduced the myeloid infiltration into the central nervous system during an experimental multiple sclerosis model [66]. This apparently contradictory role for CLEC12A in different settings may be explained by its amplifying role of type-I interferon (IFN-I) responses [67]. Therefore, the balance between regulatory and pro-inflammatory responses after sensing tissue damage can be tuned depending on the predominant immune response [68].
Thus, some DAMPs, mostly thought to be pro-inflammatory can also trigger regulatory responses (Figure 1, overlapped ovals). This is the case for the recognition of SAP130 by MINCLE, previously defined as pro-inflammatory. However, pancreatic ductal adenocarcinoma cells release SAP130 by a necroptosis-mediated mechanism. The recognition of this nucleoprotein by MINCLE in this tumor setting contributes to an immunosuppressive program in cancer infiltrating myeloid cells, favoring tumor growth. In fact, MINCLE ligation by exogenous Trehalose-6,6-dibehenate (TDB) administration recapitulates this immunosuppressive phenotype, accelerating pancreatic oncogenesis [69••]. This divergent inflammatory outcome may be related to a diverse signaling pathway triggered downstream the receptor under different conditions. Indeed, MINCLE ligation by a ligand derived from Leishmania major couples the receptor to SHP-1 signaling, triggering an inhibitory ITAM configuration that regulates activating signals triggered by heterologous receptors [70].
Sensing of HMGB1 can also have regulatory properties. This alarmin binds to the membrane-anchored glycoprotein CD24; this complex is recognized by Siglec-10 in the mouse or its human homolog Siglec-G, delivering regulatory signals that dampen the pro-inflammatory activity of HMGB1 in dendritic cells. Thus, in a model of acetaminophen (AAP)-induced liver necrosis, both CD24-deficient and Siglec-10-deficient mice show exacerbated HMGB1-mediated inflammation and worse survival than wild-type counterparts [71]. Interestingly, the Siglec-G-mediated regulatory signals in response to HMGB1 may also regulate T cells, modulating the severity of graft-versus-host disease (GVHD) [72]. Therefore, to have a whole picture of the inflammatory response triggered after the sensing of tissue damage, it is also important to understand the composition of the immune infiltrate and their coordinated responses.
Conclusions
Tissue injury conditions lead to massive release of different DAMPs. As discussed in here, the recognition of this tissue damage by myeloid cells can generate both pro-inflammatory and regulatory responses. Understanding the molecular mechanisms that govern these differential responses is critical to harness them in pathological situations such as cancer, chronic inflammation or autoimmunity. There are still open questions on what determines the activating or regulatory outcome; Is it conditioned by the PRR expression profile of surrounding cells? How do all those signals integrate? In addition, some of these tissue damage conditions are a consequence of infections, where pathogens also provide PAMPs, increasing the complexity of the response. The unbiased global study while, at the same time, we dissect the diversity at the single cell level may increase our understanding of this fascinating field of tissue damage sensing.
Conflict of interest statement
Nothing declared.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
CRediT authorship contribution statement
Carlos del Fresno: Conceptualization, Writing - original draft, Writing - review & editing. David Sancho: Conceptualization, Writing - review & editing, Funding acquisition.
Acknowledgements
We thank all members of the David Sancho laboratory at CNIC for scientific discussions. Carlos del Fresno is supported by AECC Foundation (INVES192DELF). Work in the DS laboratory is funded by the CNIC; by Ministerio de Ciencia, Innovación e Universidades (MICINN), Agencia Estatal de Investigación and Fondo Europeo de Desarrollo Regional (FEDER) (SAF2016-79040-R and PID2019-108157RB); by Comunidad de Madrid (B2017/BMD-3733 Immunothercan-CM); by FIS-Instituto de Salud Carlos III, MICINN and FEDER (RD16/0015/0018-REEM); by Acteria Foundation; by Atresmedia (Constantes y Vitales prize) and by Fundació La Marató de TV3 (201723). The CNIC is supported by the Instituto de Salud Carlos III (ISCIII), the MICINN and the Pro CNIC Foundation, and is a Severo Ochoa Center of Excellence (SEV-2015-0505).
References
- 1.Del Fresno C., Cueto F.J., Sancho D. Current Topics in Microbiology and Immunology. Springer; Berlin Heidelberg: 2019. Sensing tissue damage by myeloid C-type lectin receptors. [DOI] [PubMed] [Google Scholar]
- 2.Yan G., Elbadawi M., Efferth T. Multiple cell death modalities and their key features (Review) World Acad Sci J. 2020;2:39–48. doi: 10.3892/wasj.2020.40. [DOI] [Google Scholar]
- 3.Shekarian T., Valsesia-Wittmann S., Brody J., Michallet M.C., Depil S., Caux C., Marabelle A. Pattern recognition receptors: immune targets to enhance cancer immunotherapy. Ann Oncol. 2017;28:1756–1766. doi: 10.1093/annonc/mdx179. [DOI] [PubMed] [Google Scholar]
- 4.Matzinger P. The danger model: a renewed sense of self. Science (80-) 2002;296:301–305. doi: 10.1126/science.1071059. [DOI] [PubMed] [Google Scholar]
- 5.Lamberti M.J., Nigro A., Mentucci F.M., Rumie Vittar N.B., Casolaro V., Dal Col J. Dendritic cells and immunogenic cancer cell death: a combination for improving antitumor immunity. Pharmaceutics. 2020;12:256. doi: 10.3390/pharmaceutics12030256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6•.Vanmeerbeek I., Sprooten J., De Ruysscher D., Tejpar S., Vandenberghe P., Fucikova J., Spisek R., Zitvogel L., Kroemer G., Galluzzi L. Trial watch: chemotherapy-induced immunogenic cell death in immuno-oncology. Oncoimmunology. 2020;9 doi: 10.1080/2162402X.2019.1703449. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper describes current trends in the preclinical and clinical development of immunogenic cell death-eliciting chemotherapy as partner for immunotherapy.
- 7.Ablasser A., Chen Z.J. CGAS in action: expanding roles in immunity and inflammation. Science (80-) 2019;363 doi: 10.1126/science.aat8657. [DOI] [PubMed] [Google Scholar]
- 8.Fuertes M.B., Kacha A.K., Kline J., Woo S.R., Kranz D.M., Murphy K.M., Gajewski T.F. Host type I IFN signals are required for antitumor CD8+ T cell responses through CD8α+ dendritic cells. J Exp Med. 2011;208:2005–2016. doi: 10.1084/jem.20101159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Diamond M.S., Kinder M., Matsushita H., Mashayekhi M., Dunn G.P., Archambault J.M., Lee H., Arthur C.D., White J.M., Kalinke U. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med. 2011;208:1989–2003. doi: 10.1084/jem.20101158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10••.Woo S.R., Fuertes M.B., Corrales L., Spranger S., Furdyna M.J., Leung M.Y.K., Duggan R., Wang Y., Barber G.N., Fitzgerald K.A. STING-dependent cytosolic DNA sensing mediates innate immune recognition of immunogenic tumors. Immunity. 2014;41:830–842. doi: 10.1016/j.immuni.2014.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper shows that the STING pathway is a major immune sensor of cancer cells, driving CD8+ T cell priming against tumors.
- 11••.Ahn J., Xia T., Rabasa Capote A., Betancourt D., Barber G.N. Extrinsic phagocyte-dependent STING signaling dictates the immunogenicity of dying cells. Cancer Cell. 2018;33:862–873.e5. doi: 10.1016/j.ccell.2018.03.027. [DOI] [PMC free article] [PubMed] [Google Scholar]; This work reveals that engulfed cells containing cytosolic double-stranded DNA species (viral or synthetic) or cyclic di-nucleotides stimulate antigen presenting cells via extrinsic STING signaling, to promote antigen cross-presentation.
- 12.Deng L., Liang H., Xu M., Yang X., Burnette B., Arina A., Li X.D., Mauceri H., Beckett M., Darga T. STING-dependent cytosolic DNA sensing promotes radiation-induced type I interferon-dependent antitumor immunity in immunogenic tumors. Immunity. 2014;41:843–852. doi: 10.1016/j.immuni.2014.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Della Corte C.M., Sen T., Gay C.M., Ramkumar K., Diao L., Cardnell R.J., Rodriguez B.L., Stewart C.A., Papadimitrakopoulou V.A., Gibson L. STING pathway expression identifies NSCLC with an immune-responsive phenotype. J Thorac Oncol. 2020;15:777–791. doi: 10.1016/j.jtho.2020.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ghaffari A., Peterson N., Khalaj K., Vitkin N., Robinson A., Francis J.A., Koti M. Sting agonist therapy in combination with pd-1 immune checkpoint blockade enhances response to carboplatin chemotherapy in high-grade serous ovarian cancer. Br J Cancer. 2018;119:440–449. doi: 10.1038/s41416-018-0188-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Flood B.A., Higgs E.F., Li S., Luke J.J., Gajewski T.F. STING pathway agonism as a cancer therapeutic. Immunol Rev. 2019;290:24–38. doi: 10.1111/imr.12765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Piantadosi C.A. Mitochondrial DNA, oxidants, and innate immunity. Free Radic Biol Med. 2020;152:455–461. doi: 10.1016/j.freeradbiomed.2020.01.013. [DOI] [PubMed] [Google Scholar]
- 17.Zhang Q., Raoof M., Chen Y., Sumi Y., Sursal T., Junger W., Brohi K., Itagaki K., Hauser C.J. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature. 2010;464:104–107. doi: 10.1038/nature08780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kubiritova Z., Radvanszky J., Gardlik R. Cell-free nucleic acids and their emerging role in the pathogenesis and clinical management of inflammatory bowel disease. Int J Mol Sci. 2019;20 doi: 10.3390/ijms20153662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Puyo C.A., Earhart A., Staten N., Prince O.A., Haug C., Kollef M., Awad M. Endotracheal intubation results in acute tracheal damage induced by mtDNA/TLR9/NF-κB activity. J Leukoc Biol. 2019;105:577–587. doi: 10.1002/JLB.5A0718-254RR. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.West A.P., Khoury-Hanold W., Staron M., Tal M.C., Pineda C.M., Lang S.M., Bestwick M., Duguay B.A., Raimundo N., MacDuff D.A. Mitochondrial DNA stress primes the antiviral innate immune response. Nature. 2015;520:553–557. doi: 10.1038/nature14156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Shimada K., Crother T.R., Karlin J., Dagvadorj J., Chiba N., Chen S., Ramanujan V.K., Wolf A.J., Vergnes L., Ojcius D.M. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity. 2012;36:401–414. doi: 10.1016/j.immuni.2012.01.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Crowl J.T., Gray E.E., Pestal K., Wolkman H.E., Stetson D.B. Intracellular nucleic acid detection in autoimmunity. Annu Rev Immunol. 2017;35:313–336. doi: 10.1146/annurev-immunol-051116-052331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou R., Xie X., Li X., Qin Z., Wei C., Liu J., Luo Y. The triggers of the cGAS-STING pathway and the connection with inflammatory and autoimmune diseases. Infect Genet Evol. 2020;77 doi: 10.1016/j.meegid.2019.104094. [DOI] [PubMed] [Google Scholar]
- 24•.Motwani M., Pesiridis S., Fitzgerald K.A. DNA sensing by the cGAS–STING pathway in health and disease. Nat Rev Genet. 2019;20:657–674. doi: 10.1038/s41576-019-0151-1. [DOI] [PubMed] [Google Scholar]; Great review on the molecular components and regulation of the cGAS-STING pathway, and its implications in human inflammatory diseases and cancer.
- 25.Gray E.E., Treuting P.M., Woodward J.J., Stetson D.B. Cutting edge: cGAS is required for lethal autoimmune disease in the Trex1-deficient mouse model of Aicardi–Goutières syndrome. J Immunol. 2015;195:1939–1943. doi: 10.4049/jimmunol.1500969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Thomas J.O. HMG 1 and 2: architectural DNA-binding proteins. Biochem Soc Trans. 2001:395–401. doi: 10.1042/bst0290395. [DOI] [PubMed] [Google Scholar]
- 27.Tang D., Kang R., Livesey K.M., Cheh C.W., Farkas A., Loughran P., Hoppe G., Bianchi M.E., Tracey K.J., Zeh H.J. Endogenous HMGB1 regulates autophagy. J Cell Biol. 2010;190:881–892. doi: 10.1083/jcb.200911078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scaffidi P., Misteli T., Bianchi M.E. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418:191–195. doi: 10.1038/nature00858. [DOI] [PubMed] [Google Scholar]
- 29.Li G., Liang X., Lotze M.T. HMGB1: the central cytokine for all lymphoid cells. Front Immunol. 2013;4 doi: 10.3389/fimmu.2013.00068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Apetoh L., Ghiringhelli F., Tesniere A., Obeid M., Ortiz C., Criollo A., Mignot G., Maiuri M.C., Ullrich E., Saulnier P. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med. 2007;13:1050–1059. doi: 10.1038/nm1622. [DOI] [PubMed] [Google Scholar]
- 31.Gorgulho C.M., Romagnoli G.G., Bharthi R., Lotze M.T. Johnny on the spot-chronic inflammation is driven by HMGB1. Front Immunol. 2019;10 doi: 10.3389/fimmu.2019.01561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Haruna M., Hirata M., Iwahori K., Kanazawa T., Yamamoto Y., Goto K., Kawashima A., Morimoto-Okazawa A., Funaki S., Shintani Y. Docetaxel upregulates HMGB1 levels in non-small cell lung cancer. Biol Pharm Bull. 2020;43:399–403. doi: 10.1248/bpb.b19-00702. [DOI] [PubMed] [Google Scholar]
- 33.Huang Z., Chen M., Wei M., Lu B., Wu X., Wang Z., Ji L. Liver inflammatory injury initiated by DAMPs-TLR4-MyD88/TRIF-NFκB signaling pathway is involved in monocrotaline-induced HSOS. Toxicol Sci. 2019;172:385–397. doi: 10.1093/toxsci/kfz193. [DOI] [PubMed] [Google Scholar]
- 34.Sun L., Li M., Ma X., Zhang L., Song J., Lv C., He Y. Inhibiting high mobility group box-1 reduces early spinal cord edema and attenuates astrocyte activation and aquaporin-4 expression after spinal cord injury in rats. J Neurotrauma. 2019;36:421–435. doi: 10.1089/neu.2018.5642. [DOI] [PubMed] [Google Scholar]
- 35.Meng L., Li L., Lu S., Li K., Su Z., Wang Y., Fan X., Li X., Zhao G. The protective effect of dexmedetomidine on LPS-induced acute lung injury through the HMGB1-mediated TLR4/NF-κB and PI3K/Akt/mTOR pathways. Mol Immunol. 2018;94:7–17. doi: 10.1016/j.molimm.2017.12.008. [DOI] [PubMed] [Google Scholar]
- 36.Chen H., Guan B., Wang B., Pu H., Bai X., Chen X., Liu J., Li C., Qiu J., Yang D. Glycyrrhizin prevents hemorrhagic transformation and improves neurological outcome in ischemic stroke with delayed thrombolysis through targeting peroxynitrite-mediated HMGB1 signaling. Transl Stroke Res. 2019;11:967–982. doi: 10.1007/s12975-019-00772-1. [DOI] [PubMed] [Google Scholar]
- 37.Zheng L., Zhu Q., Xu C., Li M., Li H., Yi P.Q., Xu F.F., Cao L., Chen J.Y. Glycyrrhizin mitigates radiation-induced acute lung injury by inhibiting the HMGB1/TLR4 signalling pathway. J Cell Mol Med. 2020;24:214–226. doi: 10.1111/jcmm.14703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gardella S., Andrei C., Ferrera D., Lotti L.V., Torrisi M.R., Bianchi M.E., Rubartelli A. The nuclear protein HMGB1 is secreted by monocytes via a non-classical, vesicle-mediated secretory pathway. EMBO Rep. 2002;3:995–1001. doi: 10.1093/embo-reports/kvf198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zhang P., Xin X., Fang L., Jiang H., Xu X., Su X., Shi Y. HMGB1 mediates Aspergillus fumigatus-induced inflammatory response in alveolar macrophages of COPD mice via activating MyD88/NF-κB and syk/PI3K signalings. Int Immunopharmacol. 2017;53:125–132. doi: 10.1016/j.intimp.2017.10.007. [DOI] [PubMed] [Google Scholar]
- 40.Yamasaki S., Ishikawa E., Sakuma M., Hara H., Ogata K., Saito T. Mincle is an ITAM-coupled activating receptor that senses damaged cells. Nat Immunol. 2008;9:1179–1188. doi: 10.1038/ni.1651. [DOI] [PubMed] [Google Scholar]
- 41.Tanaka M., Saka-Tanaka M., Ochi K., Fujieda K., Sugiura Y., Miyamoto T., Kohda H., Ito A., Miyazawa T., Matsumoto A. C-type lectin Mincle mediates cell death-triggered inflammation in acute kidney injury. J Exp Med. 2020;217 doi: 10.1084/jem.20192230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.He Y., Xu L., Li B., Guo Z.N., Hu Q., Guo Z., Tang J., Chen Y., Zhang Y., Tang J. Macrophage-inducible C-type lectin/spleen tyrosine kinase signaling pathway contributes to neuroinflammation after subarachnoid hemorrhage in rats. Stroke. 2015;46:2277–2286. doi: 10.1161/STROKEAHA.115.010088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kim J.W., Roh Y.S., Jeong H., Yi H.K., Lee M.H., Lim C.W., Kim B. Spliceosome-associated protein 130 exacerbates alcohol-induced liver injury by inducing NLRP3 inflammasome–mediated IL-1β in mice. Am J Pathol. 2018;188:967–980. doi: 10.1016/j.ajpath.2017.12.010. [DOI] [PubMed] [Google Scholar]
- 44.Gong W., Guo K., Zheng T., Fang M., Xie H., Li W., Hong Z., Ren H., Gu G., Wang G. Preliminary exploration of the potential of spliceosome-associated protein 130 for predicting disease severity in Crohn’s disease. Ann N Y Acad Sci. 2020;1462:128–138. doi: 10.1111/nyas.14240. [DOI] [PubMed] [Google Scholar]
- 45.N’Diaye M., Brauner S., Flytzani S., Kular L., Warnecke A., Adzemovic M.Z., Piket E., Min J.H., Edwards W., Mela F. C-type lectin receptors Mcl and Mincle control development of multiple sclerosis-like neuroinflammation. J Clin Invest. 2020;130:838–852. doi: 10.1172/JCI125857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cheng Z., Abrams S.T., Alhamdi Y., Toh J., Yu W., Wang G., Toh C.H. Circulating histones are major mediators of multiple organ dysfunction syndrome in acute critical illnesses. Crit Care Med. 2019;47:E677–E684. doi: 10.1097/CCM.0000000000003839. [DOI] [PubMed] [Google Scholar]
- 47.Xu J., Zhang X., Monestier M., Esmon N.L., Esmon C.T. Extracellular histones are mediators of death through TLR2 and TLR4 in mouse fatal liver injury. J Immunol. 2011;187:2626–2631. doi: 10.4049/jimmunol.1003930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Westman J., Papareddy P., Dahlgren M.W., Chakrakodi B., Norrby-Teglund A., Smeds E., Linder A., Mörgelin M., Johansson-Lindbom B., Egesten A. Extracellular histones induce chemokine production in whole blood ex vivo and leukocyte recruitment in vivo. PLoS Pathog. 2015;11 doi: 10.1371/journal.ppat.1005319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Westman J., Smeds E., Johansson L., Mörgelin M., Olin A.I., Malmström E., Linder A., Herwald H. Treatment with p33 curtails morbidity and mortality in a histone-induced murine shock model. J Innate Immun. 2014;6:819–830. doi: 10.1159/000363348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Westman J., Chakrakodi B., Snäll J., Mörgelin M., Madsen M.B., Hyldegaard O., Neumann A., Frick I.M., Norrby-Teglund A., Björck L. Protein SIC secreted from Streptococcus pyogenes forms complexes with extracellular histones that boost cytokine production. Front Immunol. 2018;9 doi: 10.3389/fimmu.2018.00236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tian Y., Charles E.J., Yan Z., Wu D., French B.A., Kron I.L., Yang Z. The myocardial infarct-exacerbating effect of cell-free DNA is mediated by the high-mobility group box 1–receptor for advanced glycation end products–Toll-like receptor 9 pathway. J Thorac Cardiovasc Surg. 2019;157:2256–2269.e3. doi: 10.1016/j.jtcvs.2018.09.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Huang H., Tohme S., Al-Khafaji A.B., Tai S., Loughran P., Chen L., Wang S., Kim J., Billiar T., Wang Y. Damage-associated molecular pattern-activated neutrophil extracellular trap exacerbates sterile inflammatory liver injury. Hepatology. 2015;62:600–614. doi: 10.1002/hep.27841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Tian J., Avalos A.M., Mao S.Y., Chen B., Senthil K., Wu H., Parroche P., Drabic S., Golenbock D., Sirois C. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat Immunol. 2007;8:487–496. doi: 10.1038/ni1457. [DOI] [PubMed] [Google Scholar]
- 54.Yanai H., Ban T., Wang Z., Choi M.K., Kawamura T., Negishi H., Nakasato M., Lu Y., Hangai S., Koshiba R. HMGB proteins function as universal sentinels for nucleic-acid-mediated innate immune responses. Nature. 2009;462:99–103. doi: 10.1038/nature08512. [DOI] [PubMed] [Google Scholar]
- 55.Andersson U., Ottestad W., Tracey K.J. Extracellular HMGB1: a therapeutic target in severe pulmonary inflammation including COVID-19? Mol Med. 2020;26:42. doi: 10.1186/s10020-020-00172-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56••.Lai J.J., Cruz F.M., Rock K.L. Immune sensing of cell death through recognition of histone sequences by C-type lectin-receptor-2d causes inflammation and tissue injury. Immunity. 2020;52:123–135.e6. doi: 10.1016/j.immuni.2019.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper shows that CLEC2D binds to histones released upon necrotic cell death and facilitates the interaction of histones-bound DNA with the endosomal receptor TLR9.
- 57.Ahrens S., Zelenay S., Sancho D., Hanč P., Kjær S., Feest C., Fletcher G., Durkin C., Postigo A., Skehel M. F-actin is an evolutionarily conserved damage-associated molecular pattern recognized by DNGR-1, a receptor for dead cells. Immunity. 2012;36:635–645. doi: 10.1016/j.immuni.2012.03.008. [DOI] [PubMed] [Google Scholar]
- 58.Zhang J.-G., Czabotar P.E., Policheni A.N., Caminschi I., Wan S.S., Kitsoulis S., Tullett K.M., Robin A.Y., Brammananth R., van Delft M.F. The dendritic cell receptor Clec9A binds damaged cells via exposed actin filaments. Immunity. 2012;36:646–657. doi: 10.1016/j.immuni.2012.03.009. [DOI] [PubMed] [Google Scholar]
- 59.Cueto F.J., del Fresno C., Sancho D. DNGR-1, a dendritic cell-specific sensor of tissue damage that dually modulates immunity and inflammation. Front Immunol. 2020;10 doi: 10.3389/fimmu.2019.03146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sancho D., Joffre O.P., Keller A.M., Rogers N.C., Martínez D., Hernanz-Falcón P., Rosewell I., Reis e Sousa C. Identification of a dendritic cell receptor that couples sensing of necrosis to immunity. Nature. 2009;458:899–903. doi: 10.1038/nature07750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Iborra S., Izquierdo H.M., Sancho D. Th DC receptor DNGR-1 mediates cross-priming of CTLs during vaccinia virus infection in mice. J Clin Invest. 2012;122:1628–1643. doi: 10.1172/JCI60660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Zelenay S., Keller A.M., Whitney P.G., Schraml B.U., Deddouche S., Rogers N.C., Schulz O., Sancho D., Reis E., Sousa C. The dendritic cell receptor DNGR-1 controls endocytic handling of necrotic cell antigens to favor cross-priming of CTLs in virus-infected mice. J Clin Invest. 2012;122:1615–1627. doi: 10.1172/JCI60644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63••.del Fresno C., Saz-Leal P., Enamorado M., Wculek S.K., Martínez-Cano S., Blanco-Menéndez N., Schulz O., Gallizioli M., Miró-Mur F., Cano E. DNGR-1 in dendritic cells limits tissue damage by dampening neutrophil recruitment. Science (80-) 2018;362:351–356. doi: 10.1126/science.aan8423. [DOI] [PubMed] [Google Scholar]; This work reveals that sensing of tissue damage via DNGR-1 in cDC1s inhibits proinflammatory signaling through heterologous receptors and prevents infiltration of neutrophils and subsequent immunopathology.
- 64•.Soares M.P., Teixeira L., Moita L.F. Disease tolerance and immunity in host protection against infection. Nat Rev Immunol. 2017;17:83–96. doi: 10.1038/nri.2016.136. [DOI] [PubMed] [Google Scholar]; This is an excellent review on the topic of how disease tolerance relies on tissue damage control mechanisms without impact on pathogen load.
- 65•.Neumann K., Castiñeiras-Vilariño M., Höckendorf U., Hannesschläger N., Lemeer S., Kupka D., Meyermann S., Lech M., Anders H.J., Kuster B. Clec12a is an inhibitory receptor for uric acid crystals that regulates inflammation in response to cell death. Immunity. 2014;40:389–399. doi: 10.1016/j.immuni.2013.12.015. [DOI] [PubMed] [Google Scholar]; This paper demonstrates that CLEC12A is a regulatory MSU receptor that controls noninfectious inflammation in response to cell death, with implications for autoimmunity and inflammatory diseases.
- 66.Di S., Singh N.P., Ginwala R., Huang X., Philip R., Nagarkatti M., Nagarkatti P., Neumann K., Ruland J., Andrews A.M. Antibody blockade of CLEC12A delays EAE onset and attenuates disease severity by impairing myeloid cell CNS infiltration and restoring positive immunity. Sci Rep. 2017;7 doi: 10.1038/s41598-017-03027-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Li K., Neumann K., Duhan V., Namineni S., Hansen A.L., Wartewig T., Kurgyis Z., Holm C.K., Heikenwalder M., Lang K.S. The uric acid crystal receptor Clec12A potentiates type I interferon responses. Proc Natl Acad Sci U S A. 2019;116:18544–18549. doi: 10.1073/pnas.1821351116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.del Fresno C., Iborra S., Saz-Leal P., Martínez-López M., Sancho D. Flexible signaling of Myeloid C-type lectin receptors in immunity and inflammation. Front Immunol. 2018;9 doi: 10.3389/fimmu.2018.00804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69••.Seifert L., Werba G., Tiwari S., Giao Ly N.N., Alothman S., Alqunaibit D., Avanzi A., Barilla R., Daley D., Greco S.H. The necrosome promotes pancreatic oncogenesis via CXCL1 and Mincle-induced immune suppression. Nature. 2016;532:245–249. doi: 10.1038/nature17403. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper describes a regulatory role for the interaction of MINCLE with SAP-130, promoting macrophage-induced adaptive immune suppression and favoring pancreatic ductal adenocarcinoma progression.
- 70.Iborra S., Martínez-López M., Cueto F.J., Conde-Garrosa R., Del Fresno C., Izquierdo H.M., Abram C.L., Mori D., Campos-Martín Y., Reguera R.M. Leishmania uses Mincle to target an inhibitory ITAM signaling pathway in dendritic cells that dampens adaptive immunity to infection. Immunity. 2016;45:788–801. doi: 10.1016/j.immuni.2016.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Chen G.-Y., Tang J., Zheng P., Liu Y. CD24 and Siglec-10 selectively repress tissue damage-induced immune responses. Science (80-) 2009;323:1722–1725. doi: 10.1126/science.1168988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Toubai T., Rossi C., Oravecz-wilson K., Zajac C., Liu C., Braun T., Fujiwara H., Wu J., Sun Y., Brabbs S. Siglec-G represses DAMP-mediated effects on T cells. JCI Insight. 2017;2:1–15. doi: 10.1172/jci.insight.92293. [DOI] [PMC free article] [PubMed] [Google Scholar]