Abstract
Angiostrongylus cantonensis, a rat lungworm, is one of the leading causes of eosinophilic meningitis in humans. Infection in humans occurs by the ingestion of intermediate hosts, undercooked paratenic hosts or contaminated vegetables and fruits by mucus from infected molluscs. This zoonosis is widespread in tropical and subtropical areas of Southeast Asia, it has also been reported in the Pacific Islands as well as in other regions of Americas. In Brazil, human cases of angiostrongyliasis have been reported since 2007 in Southeast, Northeast, and South regions. In January 2011, we collected a batch of 30 Belocaulus willibaldoi slides in a neighborhood of São Paulo city (Parque Fernanda). Six of them were used for identifying species, and the others (24) were used in parasitological tests through digestion in peptic solution and then larvae isolation by the Rugai method. A total of 250 larvae were obtained and they had morphological traits of Angiostrongylus spp. Later, four Golden hamsters (Mesocricetus auratus) were infected with 38 larvae that allowed the recovery of young worms from the brain and lungs of rodents on the 21st and 30th day of infection. In this same neighborhood we captured rodents (Rattus norvegicus) that, after necropsy led us to recovery of 22 adult worms in the pulmonary arteries (14 males and 8 females) in May 2011. The larvae and worms obtained from natural infection were evaluated by morphological and morphometric parameters, as well as biological behavior patterns and molecular profile. All methodologies identified the parasite as Angiostrongylus cantonensis. In this way, we report for the first time, the natural infection by A. cantonensis in intermediate (B. willibaldoi) and definitive (R. norvegicus) hosts in a new urban region of Brazil.
Keywords: Microbiology, Molecular biology, Zoology, Angiostrongyliasis, Angiostrongylus cantonensis, Belocaulus willibaldoi, Eosinophilic meningitis, Rattus norvegicus
Microbiology; Molecular Biology; Zoology; Angiostrongyliasis; Angiostrongylus cantonensis; Belocaulus willibaldoi; eosinophilic meningitis; Rattus norvegicus.
1. Introduction
Angiostrongylus cantonensis (Chen, 1935) is a parasite of the pulmonary arteries and right ventricle of rats. A. cantonensis was initially described as Pulmonema cantonensis Chen (1935), a new genus, from specimens recovered from the lungs of naturally infected rats collected in Canton, China (Chen, 1935). This parasite has been found in the Pacific Islands, Asia, Australia, Africa, Central America, North America (Cross, 1987; Wang et al., 2008) and more recently in South America (Caldeira et al., 2007; Solórzano Álava et al., 2014).
Definitive hosts A. cantonensis include various rat species, mainly in the genus Rattus, wich become infected by ingesting intermediate or paratenic hosts containing third stage A. cantonensis larvae (Wallace and Rosen, 1965, 1969; Cross and Chen, 2007; Chen et al., 2011; Yong and Eamsobana, 2013). Terrestrial and aquatic mollusc species serve as intermediate hosts (Caldeira et al., 2007; Carvalho et al., 2012; Kim et al., 2014). Humans are accidentally infected by A. cantonensis when they eat raw, undercooked snails, slugs, paratenic hosts (e.g. shrimps, crabs, lizards, frogs and land planarians) or contaminated vegetables and fruits by mucus from infected molluscs (Acha and Szyfres, 2003). After ingestion, the nematode larvae are released into the intestine, invade the intestinal wall entering the circulation, passing through the liver, heart, lungs and finally reach the brain where they become fourth-stage larvae (Hughes et al., 2003; Murphy and Johnson, 2013). Nevertheless, larvae do not develop and usually die from one to two months causing pathogenic changes (Hughes et al., 2003; Lindo et al., 2004). Third stage (L3) larvae may be found in the brain and cerebrospinal fluid (CSF) within one or three weeks after infection (Cross, 1987; Wilkins et al., 2013). Clinical manifestations, disease severity and incubation period are directly associated with the number of larvae ingested and degree of granulomatous inflammatory response (Cross, 1987; Lindo et al., 2004).
Among the variety of these gastropods identified worldwide, A. cantonensis is known to infect species in more than 34 families, some of which are more efficient than others (Kim et al., 2014). In Brazil, there are records of terrestrial molluscs naturally infected by A. cantonensis in the Southeast (Caldeira et al., 2007; Maldonado Júnior et al., 2010), Northeast (Thiengo et al., 2010; Ramos de Souza et al., 2018) and North (Moreira et al., 2013). The first two cases of eosinophilic meningitis in the country were reported by Caldeira in 2007 (Caldeira et al., 2007) in the city of Cariacica (Espírito Santo). Both of them were caused by ingestion of raw veronicelids. These are cosmopolitan terrestrial slugs in tropical and subtropical areas that, along with snails, have been identified as pests in gardens and urban gardens facilitating the contact of these molluscs with definitive and accidental hosts including humans (Thomé et al., 2006). In view of the above, the Instituto Adolfo Lutz (IAL) has been promoting actions of malacological surveillance in urban areas of the State of São Paulo since November 2008, in order to document natural infection by nematode larvae in snails and slugs in these environments.
In this work we describe the natural infection by A. cantonensis in intermediate Belocaulus willibaldoi (Ohlweiler et al., 2009) and definitive (Rattus norvegicus, Berkenhout, 1769) hosts in a new urban region of Brazil. In order to analysis this natural infection, young and adult worms of A. cantonensis were maintained in Golden hamster (Mesocricetus auratus, Waterhouse, 1839) and Wistar rats (R. norvegicus), respectively is reported.
2. Material and methods
2.1. Ethics statement
All procedures performed with rodents in this study was performed according to guidelines of the Sociedade Brasileira de Ciência em Animais de Laboratório/Colégio Brasileiro de Experimentação Animal (SBCAL/COBEA) and the institutional review board of the Ethics Committee of the Instituto Adolfo Lutz approved this study (CEUA-IAL/Pasteur 042012).
2.2. Study area
The study area is located at coordinates 23°40′05.89 ″S, 49°47′26.66″W, in the Parque Fernanda neighborhood, São Paulo (Figure 1). It is a residential extension of 5000 m2, in the extreme south of the city, being part of the district of Capão Redondo that, together with the districts of Campo Limpo and Vila Andrade, has an approximate population of 650,000 inhabitants, urbanization rate of 100% and average population density of 17.486,65 hab/km2 (Brasil, 2020).
Figure 1.
Localization of the studied area: Parque Fernanda neighborhood, São Paulo, Brazil (latitude 23°40′05.89 ″S, longitude 49°47′26.66″W). The arrow indicates the area of snail and rodent collections naturally infected by Angiostrongylus cantonensis captured from January to May 2011. Source: adapted from São Paulo MesoMicroMunicip.svg, Abreu, R. L.
2.3. Mollusc and metastrongylid identification
The 30 veronicelids (B. willibaldoi) collected in the region were referred to Núcleo de Enteroparasitas, IAL and identified according to Ohlweiler et al. (2009). Six of them were used for species identification and 24 were digested in pool. The molluscs were euthanized, cut into small fragments, incubated for four hours at 37 °C in pepsin solution (4 mg%) and hydrochloric acid (0.7%). The larvae were concentrated by the method of Rugai (Rugai et al., 1954). The tissue of the slugs digested in pool were transferred to a container with gauze folded in two with opening facing downwards in a sedimentation cup containing dechlorinated water heated to 42 °C for two hours and used for confirmation of natural infection. Larvae retrieved alive were identified and classified morphologically according to the criteria of Ash (Ash, 1970). Only L3 larvae were transferred to a plate excavated using a Carl Zeiss stereomicroscope (Stemi 500). After this, four male Golden hamsters (M. auratus) were infected with 38 L3 larvae/animal by gavage. Another fraction of L3 larvae was fixed in alcohol 70% for morphometric evaluation. Next, the images of fixed larvae were obtained by using a light microscope (Jenaval) and analyzed in AxioVision 4.8 software, according to the morphological parameters described by Ash (Ash, 1970). The third part was frozen at -20 °C for molecular identification.
Experimentally infected rodents were kept in cages, with wood shavings, water and feed at the IAL Animal Experimentation Center. Hamsters were observed until the appearance of signs compatible with neurological infection (Martin et al., 1983). Then, they were euthanized and necropsied for nematode identification from Central Nervous System (CNS) and pulmonary arteries. The parasites were frozen at -20 °C for molecular identification.
2.4. Rat and metastrongylids identification
Cage-type traps with springs were positioned near the burrows or in areas where traces of rodents were sighted cohabiting with molluscs. The captured animals were referred to Núcleo de Enteroparasitas (IAL) for identification and evaluation of natural infection by metastrongylids. For collection of fresh feces, rodents were kept overnight in individualized cages, with galvanized grid floors on paper moistened with water.
After infection confirmation by larvae recovery in feces, determined by Rugai methodology (Rugai et al., 1954) animals were euthanized and necropsied for nematode investigation in the CNS and in the pulmonary arteries. Part of the parasites were frozen at -20 °C for molecular analysis and the remainder fixed in AFA solution (acetic acid, formaldehyde and alcohol), clarified in lactophenol and photographed. The characteristics used for identification followed the recommendations of Chen and Maldonado (Chen, 1935; Maldonado Júnior et al., 2010).
First stage larvae (L1) in rodent feces were used in the experimental infection of Biomphalaria glabrata (Say, 1818) to isolate the parasite in the laboratory. After 45 days, L3 larvae extracted from these planorbids were used to infect albino rats (Wistar) by gavage. The isolate was registered in IAL collection a Village strain and has been kept in the laboratory with successive passages through B. glabrata and Wistar rats.
2.5. Molecular identification of Angiostrongylus
2.5.1. DNA extraction
Samples of L1 and L3 larvae; young and adult worms were analyzed by two molecular methods to metastrongylid species confirmation. Before performing the DNA extraction, larvae and worms were placed in 1.5 mL-tubes, crushed and digested, in a lysis buffer (Tris–HCl, 10 mM, pH 8.0; EDTA 10 mM; SDS, 0.5%; N-laurilsarcozil, 0.01%; proteinase K, 100 μg/mL). The larvae were ruptured by shaking with glass beads in a single cycle of 50 oscillations/sec for five minutes with the aid of TissueLyser LT (QIAGEN®) equipment. The mixtures were incubated in water bath at 56 °C until tissues were completely lysed (20 min). Then, DNA was extracted by QIAamp DNA Mini Kit (Qiagen), according to the manufacturer's instructions. DNA concentrations and purity were determined by the ratio of O.D. at 260 and 280nm in a NanoDrop ND1000 (Thermo Scientific).
2.5.2. PCR
A. cantonensis or Angiostrongylus costaricensis (Moreira and Céspedes, 1971) determination was performed by conventional PCR (cPCR). Additionally, results were confirmed by Real Time PCR (qPCR). The primer set used in cPCR was NC1/NC2 (5′ ACGTCTGGTTCAGGGTTGTT 3′) and (5′ TTAGTTTCTTTTCCTCCGCT3′), which amplify a fragment from a specific region of the internal transcribed spacer two (ITS2) in rDNA (Gasser et al., 1993; Caldeira et al., 2003). DNA samples and controls plus 25pmol of each primer were added to Go Taq Green Master Mix (Promega). The PCR mix (12.5μL) was composed of 1 unit of Taq DNA polymerase, 10mM Tris-HCl, pH8.5; 50mM KCl; 1.5mM MgCl2; and 200mM of each of each dNTP. Each amplification run contained a negative control (ultrapure water) and two positive controls. One was DNA extracted from A. cantonensis (Hawaii strain) kindly provided by Central Control Disease (Atlanta, USA). The second was DNA extracted from A. costaricensis from the Crissiumal/RS strain, maintained in IAL. Thermal cycling was done in a Veriti®-96 Well Thermal Cycler/Applied Biosystems under the following conditions: initial denaturation at 94°C/5min; 30 cycles where denaturation occurs at 94°C/60 s, annealing at 58°C/60sec and extension at 72°C/60sec; and final extension at 72 °C/10min. PCR products were electrophoresed in 2% agarose gel, stained with ethidium bromide and visualized under UV illumination.
For qPCR, the primer set was AcantITS that amplify the ITS1 region of A. cantonensis as template (Qvarnstrom et al., 2010). The design included the forward (5′TTCATGGATGGCGAACTGATAG3′) and reverse (5′GCGCCCATTGAAACATTATACTT3′) primers; and a hybridization probe (5′ATCGCATATCTACTATACGCATGTGACACCTG3′) labeled with FAM (6-carboxyfluorescein) and BHQ1 (Black Hole Quencher 1) at the 5′ and 3′ ends, respectively. The reactions were performed in final volume of 20 μL. DNA samples (3 μL of DNA until 100 ng/μL), were added to reaction mixture containing 10 μL of 2X TaqMan Universal PCR Master Mix and the primer set (18 μM of forward and reverse primers plus 5 μM of hydrolysis probe). Amplifications were performed in an Agilient AriaMax Real-time PCR System using the following thermal profile: 2 min, 50 °C, and 95 °C for 10 min. Next, 40 cycles were performed at 95 °C for 15 s and 60 °C for 1 min. Results were automatically determined in equipment and expressed by cycle threshold value (CT), which indicated the quantity of target gene at which the fluorescence exceeds a preset threshold.
3. Results
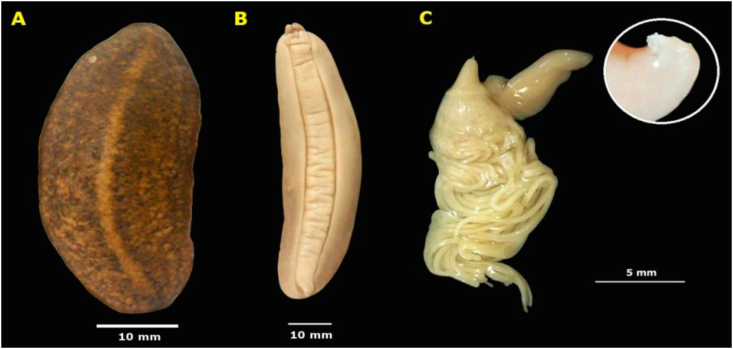
In January 2009, during an investigation we found slugs of the Sarasinula linguaeformis (Semper, 1885) containing larvae with morphological characteristics of metastrongylid (Mota et al., 2009) in a neighborhood in the southern end of São Paulo city (Parque Fernanda), whose localization is shown in Figure 1. After this finding, the collection of molluscs in the region intensified and in January 2011, the natural infection by A. cantonensis in B. willibaldoi (Veronicellidae) slugs was confirmed. The characteristics included the external morphology of the slugs (Figures 2A and 2B), as well as the male reproductive organ with penis robust, with no spathe, with a small base and glans with a wide basal region narrowing toward the apical extremity (asymmetrical arrow shaped penis). Glans with digitiform margin and small projections in the form of tubercles in the anterior region (Figure 2C). The penial gland presents a short and conical or long papilla with a terminal mammilla and 18–26 sinuous tubules (Figure 2C). Six specimens of B. willibaldoi were deposited in November 2018 in the malacological collection of Museum of Zoology of Universidade de São Paulo (USP- State of São Paulo, Brazil) - catalog number MZUSP 143479.
Figure 2.
Specimens of Belocaulus willibaldoi naturally infected by larvae L3 of the Angiostrongylus cantonensis, collected in January 2011, in a residential area in Parque Fernanda neighborhood, São Paulo, Brazil. A: dorsal view; B: ventral view; C: reproductive organ male (penial gland and penis) in detail tubercle row.
A batch of 24 pooled digested slugs yielded 250 larvae showing filiform body, cuticle with grooves, rounded anterior extremity, long buccal capsule, excretory pore located in the middle region of the esophagus, being long and with a claviform bulb (Figure 3A), in addition to genital primordia in the posterior third of the intestine and pointed tail (Figure 3B) (Ash, 1970) (See Table 1).
Figure 3.
Light microscopy of L3 larvae of Angiostrongylus cantonensis isolated from Belocaulus willibaldoi. A: Detail of the anterior extremity of the larva showing buccal capsule (CB), esophagus (E) and excretory pore (EP); B: detail of the posterior extremity of the larva showing the pointed tail (PT), genital primordium (PG) and anus (A).
Table 1.
Morphometry of L3 larvae (N = 30) isolated from Belocaulus willibaldoi naturally infected with Angiostrongylus cantonensis.
| Measures | Mean ± SD (μm) | Interval (μm) |
|---|---|---|
| Total body length | 421.00 ± 24.86 | 377.73–461.05 |
| Width at esophagus - intestine junction | 22.38 ± 1.62 | 18.04–25.96 |
| Length of esophagus | 153.88 ± 10.66 | 135.30–173.21 |
| Distance from the excretory pore to the anterior region | 75.15 ± 7.34 | 58.12–93.03 |
| Distance from genital primordium to tail | 149.22 ± 11.25 | 125.20–171.64 |
| Length of genital primordium | 32.93 ± 2.63 | 28.31–39.08 |
| Distance from anus to tail | 32.93 ± 2.63 | 28.31–39.08 |
Four Golden hamsters (M. auratus) were infected experimentally with 38 L3 larvae/animal (isolated from B. willibaldoi slugs naturally infected). After 21 days (Figure 4A), two of them had 16 young worms in CNS. They presented severe meningeal alterations with areas of hemorrhage and encephalic mass destruction due to the presence of young worms (Figure 4B). The other two golden hamsters were euthanized on 30th day post infection and had 12 young worms in their pulmonary arteries (Figures 4C, 4D and 4E). They also had paralysis of the hind limbs, blindness and intense anorexia. However, there was no evidence of release of L1 larvae in the feces of these rodents. Non-embryonated eggs were found in lung tissue of necropsied rodents in 30th day of infection (Figure 4F).
Figure 4.
Experimental infection in M. auratus with of A. cantonensis larvae L3 isolated from B. willibaldoi. (A): Weakened hamster after 21 days of infection; B: Young worm of A. cantonensis in hamster brain after 21 days of infection causing hemorrhagic focal points; C: Young worm of A. cantonensis in the hamster lung with 30 days of infection; D: Young male of A. cantonensis isolated from hamster lung with 30 days of infection; E: Young female of A. cantonensis isolated from hamster lung with 30 days of infection; F: Non-embryonated eggs in hamster lung tissue on the 30th day of infection.
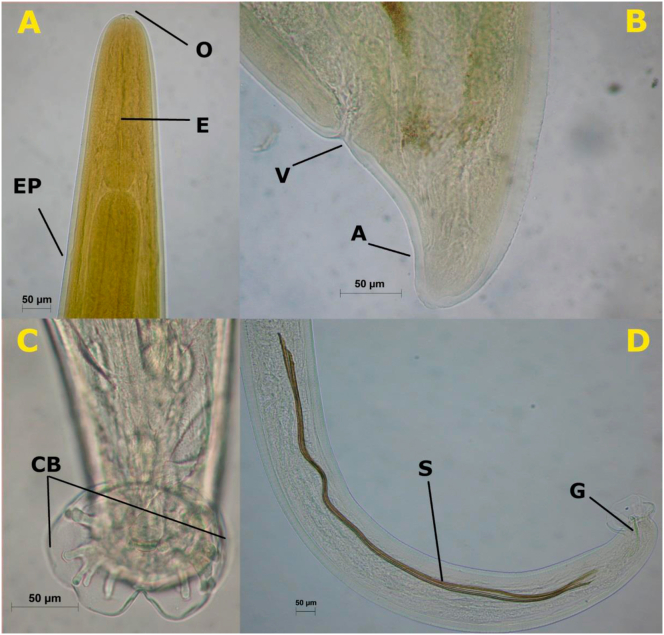
Signs of cohabitation between molluscs and rodents were also observed during the inspection in the area, due to the presence of shells of gnawed snails and rodent feces near their habitats. This led us to search for and to capture definitive hosts in Parque Fernanda neighborhood. The capture of R. norvegicus in May 2011 resulted in a female with pulmonary infection caused by 14 worms (ten males and four females) and a male with eight nematodes (four males and four females). Morphological analysis revealed that adult worms had a thin, tapered body at the anterior end in both sexes; a simple oral opening; a claviform esophagus, and an excretory pore located in the posterior region of the esophagus (Figure 5A). In females, the vulva was located near the anus at the posterior end of the body, with a transverse opening and a small protrusion adjacent to the anterior labium (Figure 5B). The males had a small, bilobed and slightly asymmetric copulatory bursa, where each side presented a lower ventro-ventral ray than the ventro-lateral, which, although having a common origin, bifurcated in the distal third and does not reach the bursal margin. The lateral rays originated from the same trunk, the anterolateral being the smallest of them and the medio-lateral one the longest, reaching the edge of the bursa, while the posterolateral ray presented half the length of the mid-side. The outer-dorsal ray was separated from the lateral, but resembled in length the posterolateral ray. The dorsal ray was short, wide and trifurcated (Figure 5C). The males also had a pair of long, thin spicules, which were oriented at the outlet of the sheath by a curved gubernaculum (Figure 5D). The results of measurements of the morphological parameters of the adult worms collected from the rodents naturally infected, as well as the experimental cycle from B. glabrata infection are shown in Table 2.
Figure 5.
Light microscopy of adult worms of A. cantonensis morphology isolated from R. norvegicus in São Paulo/Brazil (urban area) in May 2011 A: anterior extremity with oral opening (O), esophagus (E) and excretory pore (EP); B: female posterior end with vulva (V) and anus (A); C: male posterior end showing the copulatory bursa (CB); D: male posterior end with spicules (S) and gubernaculum (G).
Table 2.
Morphometry of Angiostrongylus cantonensis adult worms in Rattus norvegicus: natural isolated and experimental infection.
| Isolates (Mean) (mm) |
Sex | N | TBL | W | EL | EBL | DPA | LS | MS | G | DVT | DAC |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Natural | M | 3 | 20.45 | 0.30 | 0.29 | 0.03 | 0.37 | 1.21 | 1.19 | 0.11 | - | - |
| F | 2 | 28.44 | 0.46 | 0.31 | 0.03 | 0.43 | - | - | - | 0.23 | 0.06 | |
| Experimental | M | 7 | 19.36 | 0.28 | 0.31 | 0.03 | 0.43 | 1.27 | 1.22 | 0.12 | - | - |
| F | 12 | 25.60 | 0.43 | 0.34 | 0.03 | 0.37 | - | - | - | 0.22 | 0.06 |
TBL, Total Body Length; W, Width of the body; EL, Esophagus Length; EBL, Esophagus Base Length; DPA, Distance from Excretory Pore to Anterior Region; LS, Larger spicules; MS, Minor spicules; G, Gubernaculum; DVT, Distance from Vagina to Tail; DAT, Distance from Anus to Tail.
The molecular analyzes were performed comparing the bands profiles of samples obtained from natural and experimental hosts. The standard strains A. costaricensis and A. cantonensis were used for control. According Caldeira et al. (2003) the primer set NC1/NC2 amplifies an ITS2 region, varying in size, according Angiostrongylus species. For A. costaricensis and Angiostrongylus vasorum (Baillet, 1866), the primer set amplifies a 600 bp PCR product and a 650 bp for A. cantonensis. The (Figure 6A) shows the 650 bp amplified PCR products for A. cantonensis from studied samples. These results were also confirmed in qPCR for A. cantonensis (Figure 6B).
Figure 6.
(A) Amplified cPCR products were analyzed in electrophoresis in 1.5% agarose gel. MM, 100-bp ladder (the lowest band shown is 100 bp); line 1, negative control; line 2, A. costaricensis (standard strain); line 3, A. cantonensis (standard strain); line 4, B. willibaldoi, L3 larvae (naturally infected); line 5, young worms collected from hamster experimentally infected; line 6, L1 larvae purified of feces from the 1stR. norvegicus; line 7, worms collected from lungs from the 1stR. norvegicus; line 8, L1 larvae purified of feces from the 2ndR. norvegicus; line 9, worms collected from lungs from the 2ndR. norvegicus. (B) qPCR for A. cantonensis using AcantITS primer set with a hydrolysis probe FAM dye-labeled. Results are shown cycle threshold (CT) obtained of each DNA concentration.
4. Discussion
Since 2007, when the first human case of eosinophilic meningitis caused by A. cantonensis was recorded in Brazil, in the State of Espírito Santo, reports of the disease have increased. Currently, the parasite is registered in 13 Brazilian states infecting humans, rodents and molluscs (Ramos de Souza et al., 2018; Barbosa et al., 2020). In this last group, Veronicelids family gathers strictly terrestrial slugs, with around 100 species widely distributed in the tropical and subtropical regions the Americas, Africa, Asia and Oceania (Barker, 2001). The genus Belocaulus has only two species, Belocaulus angustipes (Heynemann, 1885) and B. willibaldoi, which are distributed in Argentina, Brazil, Uruguay, Paraguay, Honduras and south United States (Thomé, 1989; Caballero et al., 1991; Simone, 2006; Ohlweiler et al., 2009). In Brazil, B. willibaldoi has already been recorded in the states of Rio Grande do Sul, Santa Catarina, Minas Gerais and São Paulo (Ohlweiler et al., 2009).
This study describes the first record of natural infection of B. willibaldoi by A. cantonensis larvae, which presented morphology and morphometry compatible with third stage larvae (L3), according to Ash (1970), Thiengo et al. (2010) and Guerino et al. (2017). The species was confirmed by qPCR, expanding the list of intermediate hosts of this parasite. The ability of A. cantonensis to infect native or introduced species of terrestrial molluscs was demonstrated by a research conducted in the islands of Hawaii that showed the importance of molluscs as intermediate hosts for this parasite with high rates of infection (Kim et al., 2014). In Brazil, eight species of terrestrial molluscs and one from freshwater were found naturally infected by A. cantonensis in 11 states (Morassutti et al., 2014; Ramos de Souza et al., 2018; Orico et al., 2019; Barbosa et al., 2020).
In addition, it was possible to observe the parasites' tropism to the brain of hamsters on the 21st day of infection and pulmonary tropism on the 30th day. It was not possible to obtain L1 larvae in the feces of infected animals, due to their early death, probably due to the number of larvae used in the infection. The study by Ishii et al. (1980) corroborates this observation, showing that obtainment of L1 larvae from M. auratus was possible with the inoculation of a maximum of five larvae in each animal. Another study, conducted by Martin et al. (1983), demonstrated that infections with more than 10 L3 larvae of A. cantonensis in hamsters cause major damage to brain and lung tissues and are lethal to these animals. The experimental infection in M. auratus indicated the biological behavior of the parasite was compatible with A. cantonensis and provided evolutionary forms to molecular tests.
The possibility of natural infection in synanthropic rodents was already expected, in the city of São Paulo, mainly in R. norvegicus and Rattus rattus (Linnaeus, 1758). There is a wide distribution of these animals in this region, as described in the study by Masi et al. (2010), which showed a rodent infestation rate of 23.1%. In addition, there are previous records of natural infection of molluscs by A. cantonensis in different municipalities in the State of São Paulo (Carvalho et al., 2012; Guerino et al., 2017; Orico et al., 2019). In the present study, laboratory isolation of the A. cantonensis strain was possible from the natural infection of R. norvegicus captured in the south zone of the city of São Paulo for the first time. In Brazil, natural infection by this nematode was reported in R. rattus and R. norvegicus in Pará State (Moreira et al., 2013), R. norvegicus in Rio de Janeiro State (Simões et al., 2011, 2014) and in the port area of the city of Porto Alegre, Rio Grande do Sul State (Cognato et al., 2013).
The presence of terrestrial molluscs living close to rodents and related to A. cantonensis infection, as B. willibaldoi and R. norvegicus in São Paulo city, highlights the need for surveillance mechanisms in order to avoid nematode spread throughout São Paulo territory. The finding of A. cantonensis definitive hosts and naturally infected intermediates in urban areas of Brazil reinforces the need to form specialized technical groups focused on scientific studies of the aspects that affect population dynamics of rodents and molluscs against the risk of transmission in the country (Morassutti et al., 2014; Ramos de Souza et al., 2018; Orico et al., 2019; Barbosa et al., 2020).
All methodologies led to the identification of A. cantonensis specie; in this way, this is the first report of natural infection by A. cantonensis in B. willibaldoi in Brazil and in R. norvegicus in the city of São Paulo.
In addition, it was possible to establish the experimental life cycle of A. cantonensis for use in laboratory tests, also enabling new malacological studies at IAL to be carried out to investigate the transmission potential of this parasite to other mollusc species in different regions of the State of São Paulo.
Declarations
Author contribution statement
Dan Jessé Gonçalves da Mota, Leyva Cecilia Vieira de Melo: Performed the experiments; Analyzed and interpreted the data; Wrote the paper.
Vera Lúcia Pereira-Chioccola, Pedro Luiz Silva Pinto: Conceived and designed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data; Wrote the paper.
Ricardo Gava: Performed the experiments; Analyzed and interpreted the data; Contributed reagents, materials, analysis tools or data.
Funding statement
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Competing interest statement
The authors declare no conflict of interest.
Additional information
No additional information is available for this paper.
References
- Acha P.N., Szyfres B. third ed. Pan American Health Organization; 2003. Zoonoses and Communicable Diseases Common to Man and Animals; p. 395. [Google Scholar]
- Ash L.R. Diagnostic morphology of the third-stage larvae of Angiostrongylus cantonensis, Angiostrongylus vasorum, Angiostrongylus abstrusus and (Nematoda: Metastrongyloidea) Anafilaroides rostratus. J. Parasitol. 1970;56(2):249–253. [PubMed] [Google Scholar]
- Barbosa T.A. Infection by Angiostrongylus cantonensis in both humans and the snail Achatina (Lissachatina) fulica in the city of Macapá, in the Amazon region of Brazil. Mem. Inst. Oswaldo Cruz. 2020;115:1–4. doi: 10.1590/0074-02760200115. http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0074-02762020000100409&lng=en&nrm=iso e200115. Available from: [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barker G.M. Gastropods on land: phylogeny, diversity and adaptive morphology. In: Barker G.M., editor. The Biology of Terrestrial Molluscs. CABI Publishing; Hamilton, New York: 2001. p. 558. [Google Scholar]
- Brasil . 2020. Prefeitura da Cidade de São Paulo: Histórico Campo Limpo, 2020.http://www.prefeitura.sp.gov.br/cidade/secretarias/subprefeituras/campo_limpo/historico/index.php?p=131 [Google Scholar]
- Caballero R., Thomé J.W., Andrews K.L., Rueda A. Babosas de Honduras (Soleolifera: Veronicellidae): Biologıa, Ecologıa, Distribution, Descripcion, Importancia Economica, y Claves para su Identificación. CEIBA. 1991;32(2):107–126. [Google Scholar]
- Caldeira R.L. First record of molluscs naturally infected with Angiostrongylus cantonensis (Chen, 1935) (Nematoda: Metastrongylidae) in Brazil. Mem. Inst. Oswaldo Cruz. 2007;102(7):887–889. doi: 10.1590/s0074-02762007000700018. [DOI] [PubMed] [Google Scholar]
- Caldeira R.L. Molecular differentiation of Angiostrongylus costaricensis, A. cantonensis, and A. vasorum by polymerase chain reaction-restriction fragment length polymorphism. Mem. Inst. Oswaldo Cruz. 2003;98(8):1039–1043. doi: 10.1590/s0074-02762003000800011. Epub 2004 Mar 9. [DOI] [PubMed] [Google Scholar]
- Carvalho O.S. Angiostrongylus cantonensis (Nematode: Metastrongyloidea) in molluscs from harbour areas in Brazil. Mem. Inst. Oswaldo Cruz. 2012;107(6):740–746. doi: 10.1590/s0074-02762012000600006. [DOI] [PubMed] [Google Scholar]
- Chen D., Zhang Y., Shen H., Wei Y., Huang D. Epidemiological survey of Angiostrongylus cantonensis in the west-central region of Guangdong Province, China. Parasitol. Res. 2011;109:305–314. doi: 10.1007/s00436-011-2255-1. [DOI] [PubMed] [Google Scholar]
- Chen H.T. A new pulmonary nematode of rats, Pulmonema cantonensis n g , n sp from Canton. Ann. Parasitol. Hum. Comp. 1935;13:312–317. [Google Scholar]
- Cognato B.B., Morassutti A.L., da Silva A.A., Graeff-Teixeira C. First report of Angiostrongylus cantonensis in Porto Alegre, Rio Grande do Sul, southern Brazil. Rev. Soc. Bras. Med. Trop. 2013;46(5):664–665. doi: 10.1590/0037-8682-0073-2013. [DOI] [PubMed] [Google Scholar]
- Cross J.H. Public health Importance of Angiostrongylus cantonensis it relatives. Parasitol. Today. 1987;3(12):367–369. doi: 10.1016/0169-4758(87)90242-0. [DOI] [PubMed] [Google Scholar]
- Cross J.H., Chen E.R. Angiostrongyliasis. In: Murrell K.D., Fried B., editors. Food-borne Parasitic Zoonoses: Fish and Plant-Borne Parasites. Springer Science + Business Media, LLC; New York: 2007. pp. 263–290. [Google Scholar]
- Espírito-Santo M.C., Pinto P.L.S., Mota D.J.G., Gryschek R.C. The first case of Angiostrongylus cantonensis eosinophilic meningitis diagnosed in the city of Sao Paulo, Brazil. Rev. Inst. Med. Trop. Sao Paulo. 2013;55(2):129–132. doi: 10.1590/s0036-46652013000200012. [DOI] [PubMed] [Google Scholar]
- Gasser R.B., Chilton N.B., Hoste H., Beveridge I. Rapid sequencing of rDNA from single worms and eggs of parasitic helminths. Nucleic Acids Res. 1993;21(10):2525–2526. doi: 10.1093/nar/21.10.2525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guerino L.R. Prevalence and distribution of Angiostrongylus cantonensis (Nematoda, Angiostrongylidae) in Achatina fulica (Mollusca, Gastropoda) in Baixada Santista, São Paulo, Brazil. Rev. Soc. Bras. Med. Trop. 2017;50(1):92–98. doi: 10.1590/0037-8682-0316-2016. [DOI] [PubMed] [Google Scholar]
- Hughes P.A., Magnet A.D., Fishbain J.T. Eosinophilic meningitis: a case series report and review of the literature. Mil. Med. 2003;168(10):817–821. [PubMed] [Google Scholar]
- Ishii A.I., Kino H., Fujio Y., Sano M. Experimental ligth infection of Angiostrongylus cantonensis in hamsters. Int. J. Zoonoses. 1980;7(2):120–124. [PubMed] [Google Scholar]
- Kim J.R., Hayes K.A., Yeung N.W., Cowie R.H. Diverse gastropod hosts of Angiostrongylus cantonensis, the rat lungworm, globally and with a focus on the Hawaiian Islands. PloS One. 2014;13(2) doi: 10.1371/journal.pone.0094969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindo J.F., Escoffrey C.T., Reid B., Codrington G., CunninghamMyrie C., Eberhard M.L. Fatal autochthonous eosinophilic meningitis in a Jamaican child caused by Angiostrongylus cantonensis. Am. J. Trop. Med. Hyg. 2004;70:425–428. [PubMed] [Google Scholar]
- Maldonado Júnior A. First report of Angiostrongylus cantonensis (Nematoda: Metastrongylidae) in Achatina fulica (Mollusca: Gastropoda) from Southeast and south Brazil. Mem. Inst. Oswaldo Cruz. 2010;105(7):938–941. doi: 10.1590/s0074-02762010000700019. [DOI] [PubMed] [Google Scholar]
- Martin O.P., de La Torre P.G., Gonzalez M.L. Infestación mortal de hámsteres (Mesocrisetos aureatus) por Angiostrongylus cantonensis (Chen, 1935) Rev. Cubana Med. Trop. 1983;35:238–241. [PubMed] [Google Scholar]
- Masi E. Socioeconomic and environmental risk factors for urban rodent infestation in São Paulo, Brazil. J. Pest. Sci. 2010;83(3):231–241. https://link.springer.com/article/10.1007%2Fs10340-010-0290-9 [Google Scholar]
- Morassutti A.L., Thiengo S.C., Fernandez M., Sawanyawisuth K., Graeff- Teixeira C. Eosinophilic meningitis caused by Angiostrongylus cantonensis: an emergent disease in Brazil. Mem. Inst. Oswaldo Cruz. 2014;109(4):399–407. doi: 10.1590/0074-0276140023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreira V.L. Endemic angiostrongyliasis in the Brazilian Amazon: natural parasitism of Angiostrongylus cantonensis in Rattus rattus and R. norvegicus, and sympatric giant African land snails, Achatina fulica. Acta Trop. 2013;125(1):90–97. doi: 10.1016/j.actatropica.2012.10.001. [DOI] [PubMed] [Google Scholar]
- Mota D.J.G., Kawano T., Pinto P.L.S. Anais do I Encontro Latino-Americano em Helmintoses- Pesquisa, Educação e Serviço para a Saúde Pública. Teresópolis; Rio de Janeiro: 2009. Ocorrência de larvas de metastrongilídeos em Achatina (Lissachatina) fulica e Sarasinula linguaeformis em duas áreas urbanas do Estado de São Paulo. [Google Scholar]
- Murphy G.S., Johnson S. Clinical aspects of eosinophilic meningitis and meningoencephalitis caused by Angiostrongylus cantonensis, the rat lungworm. Hawai‘i J. Med. Public Health. 2013;72(Supplement 2):35–40. [PMC free article] [PubMed] [Google Scholar]
- Ohlweiler F.P., Mota D.J.G., Gomes S.R. A new species of Belocaulus (Gastropoda: Veronicellidae) from southern and southeastern Brazil. Nautilus. 2009;123(2):34–42. [Google Scholar]
- Orico L.D., Barbosa C.M., Luca L.R., Soares R.M., Gregori F. Angiostrongylus cantonensis and Ancylostoma caninum detection in snails of São Paulo City (2016-2017), Brazil. Ars Vet. 2019;35(3):115–119. [Google Scholar]
- Qvarnstrom Y. Improved molecular detection of Angiostrongylus cantonensis in mollusks and other environmental samples with a species-specific internal transcribed spacer 1-based TaqMan assay. Appl. Environ. Microbiol. 2010;(15):5287–5289. doi: 10.1128/AEM.00546-10. Epub 2010 Jun 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramos-de-Souza J. First records of molluscs naturally infected with Angiostrongylus cantonensis (Nematoda: Metastrongyloidea) in Sergipe State, Northeastern Brazil, including new global records of natural intermediate hosts. Rev. Inst. Med. Trop. S Paulo. 2018;60 e51:1–7. doi: 10.1590/S1678-9946201860051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rugai E., Mattos T., Brisola A. Nova técnica para isolar larvas de nematóides das fezes - modificação do método de Baermann. Rev. Inst. Adolfo. Lutz. 1954;14:5–8. http://www.ial.sp.gov.br/resources/insituto-adolfo-lutz/publicacoes/rial/50/rial-141_1954/a195.pdf [PubMed] [Google Scholar]
- Simões R.O. Endemic angiostrongyliasis in Rio de Janeiro, Brazil. Emerg. Infect. Dis. 2011;17:1331–1333. doi: 10.3201/eid1707.101822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simões R.O. A longitudinal study of Angiostrongylus cantonensis in an urban population of Rattus norvegicus in Brazil: the influences of seasonality and host features on the pattern of infection. Parasites Vectors. 2014;100(7):2–8. doi: 10.1186/1756-3305-7-100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simone L.R. EGB; São Paulo: 2006. Land and Freshwater Molluscs of Brazil: an Illustrated Inventory on the Brazilian Malacofauna, Including Neighbor Regions of the South America, Respect to the Terrestrial and Freshwater Ecosystems; p. 390. [Google Scholar]
- Solorzano Álava F.S. Angiostrongylus cantonensis: un parásito emergente en Ecuador. Rev. Cubana Med. Trop. 2014;66(1):20–33. [Google Scholar]
- Thiengo S.C. The giant African snail Achatina fulica as natural intermediate host of Angiostrongylus cantonensis in Pernambuco, northeast Brazil. Acta Trop. 2010;115(3):194–199. doi: 10.1016/j.actatropica.2010.01.005. [DOI] [PubMed] [Google Scholar]
- Thomé J.W., Gomes S.R., Picanço J.B. USEB; Pelotas: 2006. Guia ilustrado: Os caracóis e as lesmas dos nossos bosques e jardins; p. 123. [Google Scholar]
- Thomé J.W. Annotated and illustrated preliminary list of Veronicellidae of the Antilles, and central and North America. J. Med. Appl. Malacol. 1989;(1):11–28. [Google Scholar]
- Wallace G.D., Rosen L. Studies on eosinophilic meningitis: I. Observations on the geographic distribution of Angiostrongylus cantonensis in the Pacific area and its prevalence in wild rats. Am. J. Epidemiol. 1965;81:52–62. doi: 10.1093/oxfordjournals.aje.a120497. [DOI] [PubMed] [Google Scholar]
- Wallace G.D., Rosen L. Studies on eosinophilic meningitis. V. Molluscan hosts of Angiostrongylus cantonensis on Pacific islands. Am. J. Trop. Med. Hyg. 1969;18:206–216. [PubMed] [Google Scholar]
- Wang Q.P. Human angiostrongyliasis. Lancet Infect. Dis. 2008;8:621–630. doi: 10.1016/S1473-3099(08)70229-9. [DOI] [PubMed] [Google Scholar]
- Wilkins P.P. The current status of laboratory diagnosis of Angiostrongylus cantonensis infections in humans using serologic and molecular methods. Hawai‘i J. Med. Public Health. 2013;72(6 Suppl. 2):55–57. [PMC free article] [PubMed] [Google Scholar]
- Yong H.S., Eamsobhana P. Definitive rodent hosts of the rat lungworm Angiostrongylus cantonensis. Raffles Bull. Zool. 2013;29:111–115. [Google Scholar]