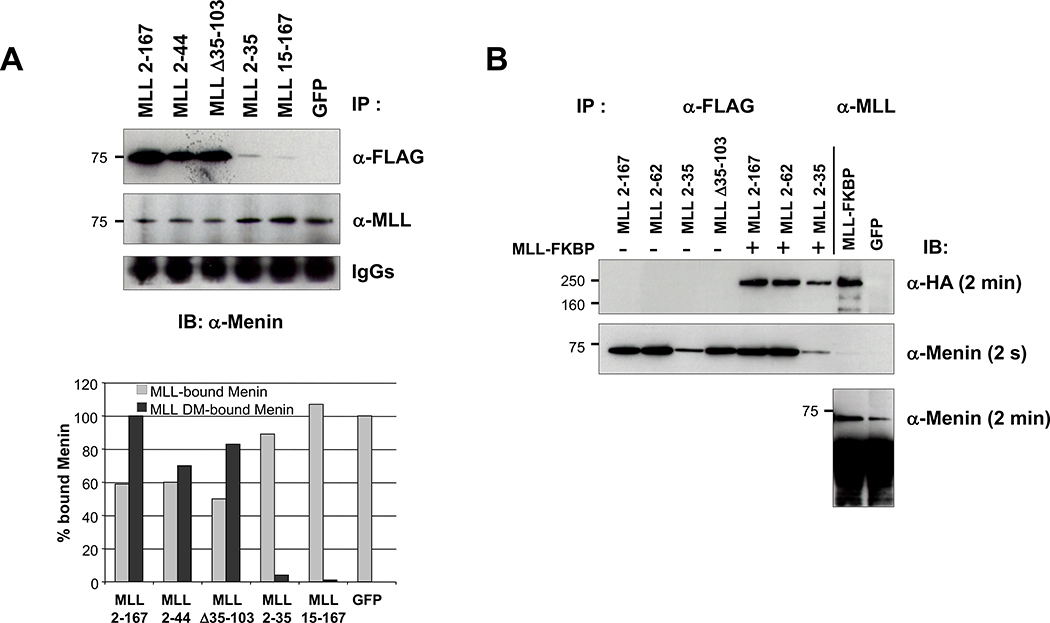
Figure 4.
Competition of MLL deletion fragments for menin binding with endogenous MLL. A, equal amounts of transfected cell extracts from GFP-positive–sorted cells were immunoprecipitated with M2 (anti-FLAG) or MLL612 (anti-MLL) antibodies, and then probed with anti-menin antibody. The representative Western blot was quantitatively determined by the ImageJ program (bottom), and the results presented as a percentage of endogenous MLL-bound menin relative to MSCV-GFP (gray columns) and as a percentage of MLL deletion mutant–bound menin relative to MSCV-MLL2–167 (black columns). B, MLL deletion mutants binding with cotransfected hemagglutinin-tagged MLL-FKBP into 293 cells. After transfections, cell extracts were immunoprecipitated with anti-FLAG and anti-MLL antibodies and Western blot analyzed with anti-hemagglutinin and anti-menin. MLL deletion mutants and MLL-FKBP coimmunoprecipitate through their association with menin. Immunoprecipitations for each MLL fragment were done at least twice (some immunoprecipitations were shown twice for figure consistency).