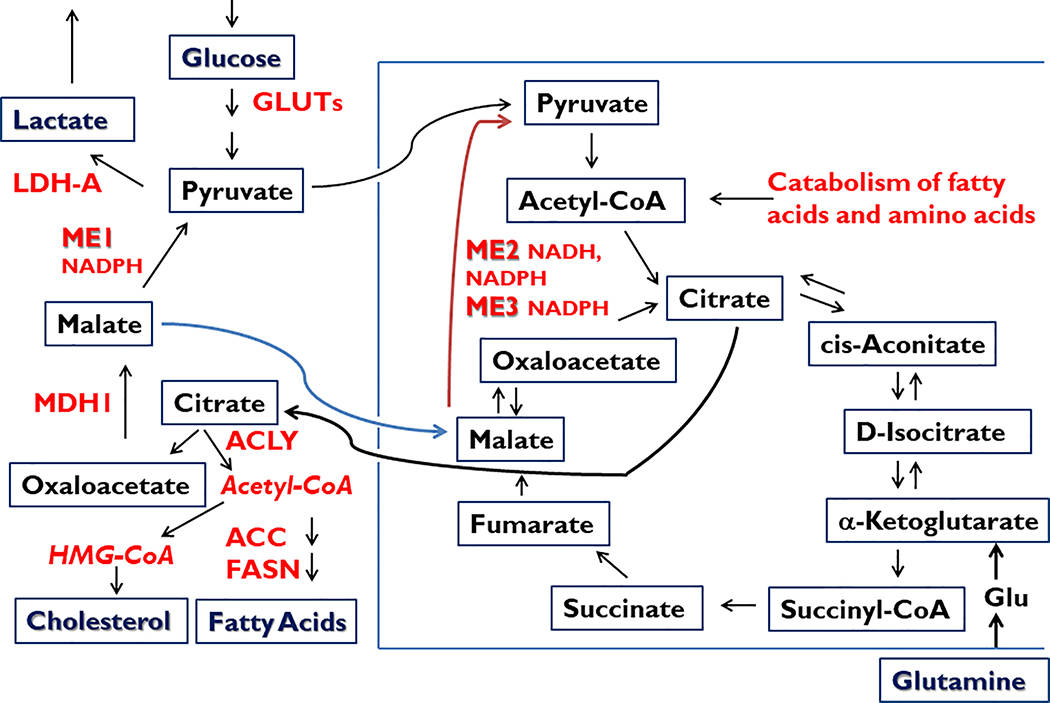
Figure 1.
Central role of malic enzymes (ME) in normal and cancer cell metabolism. Malic enzymes located within cytoplasmic (ME1) and mitochondrial (ME2, ME3) compartments catalyze the oxidative decarboxylation of malate to pyruvate, while concomitantly storing reducing equivalents in the form of NADPH (ME1, ME2, ME3) or NADH (ME2). NADPH serves in multiple biochemical capacities such as in countering ROS (via glutathione and thioredoxin pathways), contributing to ROS signaling (via NOX/DUOX family members), and providing reductant capacity for the biosynthesis of fatty acids and cholesterol, among many other metabolic pathways (Goodman et al. 2018). Besides being a major source of cytosolic NADPH, ME1 participates in a pathway by which excess mitochondrial Acetyl-CoA is transferred to cytosol (in the form of citrate; via the pyruvate-malate cycle) for incorporation into cholesterol and fatty acids. ME1 also participates in the Warburg effect (aerobic glycolysis, secretion of lactate) that is characteristic of many cancer cells and tumors. Lactate may serve as a paracrine energy source for tumor cells. ACC, acetyl-CoA carboxylase; ACLY, ATP citrate lyase; FASN, fatty acid synthase; Glu, Glutamate; GLUTs, glucose transporters; LDH-A, lactate dehydrogenase A; MDH1, malate dehydrogenase 1.