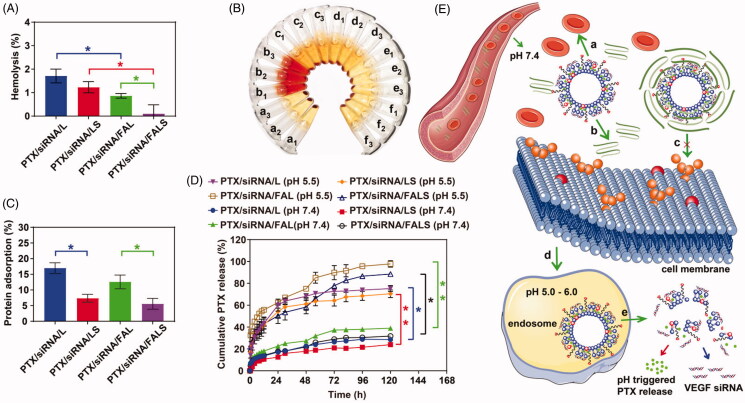
Figure 3.
In vitro simulated blood circulation and release behavior of the nanoparticles. (A) Hemolysis assays of nanoparticles after 6 h incubation at 37 °C. (B) Images of the hemolysis of RBCs produced by nanoparticles. a1-a3: negative control (PBS) group, b1–b3: positive control (water) group, c1–c3: PTX/siRNA/L, d1–d3: PTX/siRNA/LS, e1–e3: PTX/siRNA/FAL, f1-f3: PTX/siRNA/FALS. (C) BSA adsorption on nanoparticles after 6 h incubation at 37 °C. (D) In vitro PTX release of nanoparticles at 37 °C (pH = 7.4 and pH = 5.5). (E) Schematic illustration of the targeted delivery and controlled release mechanism of nanoparticles. In a biological environment, nanoparticles were expected to directly contact with red blood cells, leading to the hemolysis (a), and nanoparticles might interact with protein, resulting the protein adsorption of nanoparticles (b). The protein adsorption highly depends on the surface structure of nanoparticles. The high protein adsorption of nanoparticles might prevent exposure of the targeting ligand, resulting a low cell uptake (c). In contrast, a low protein adsorption of nanoparticles would ensure the successful folate receptor-mediated uptake (d), and then nanoparticles could response to the decreased pH in the endosomes, the encapsulated drug would be efficiently released (e).