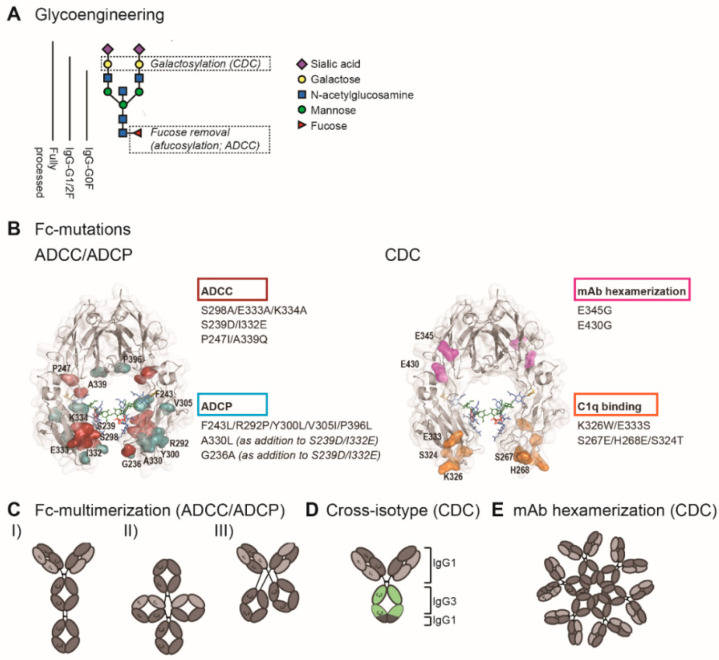
Figure 2.
Fc engineering strategies to enhance Fc-effector functions. (A) The biantennary complex glycan structure of the most frequent variants for the N-glycan of therapeutic IgG (G0F, G1F, G2F, and fully processed). Glycoengineering strategies focus on galactosylation for enhanced CDC and afucosylation for enhanced ADCC. (B) ADCC/ADCP (left) and CDC (right) enhancing point mutations depicted in the crystal structure of the Fc region (PDB:3DO3). The ADCC-enhancing mutations S298A/E333A/K334A, S239D/I3332E, and P247I/A339Q are depicted in magenta; the ADCP-enhancing mutations F234L/R292P/Y300L/V305I/P396L, A330L, and G236A are shown in blue. A330L and G236A were added to the ADCC-enhancing mutations S239D/I332E to decrease the mutation-related increase in affinity to the inhibitory receptor FcγRIIb. The CDC-enhancing mutations E345G and E430G, which induce Fc/Fc interactions and subsequent mAb hexamerization (see E), are shown in orange and K326W/E333S and S267E/H268E/S324T, which enhance C1q binding affinity, are shown in pink. (C) Schematic representation of three Fc multimerization strategies which are aimed to enhance ADCC and ADCP. (D) Cross-isotype antibody generated by replacing the CH2 and part of the CH3 domains of an IgG1 antibody with the corresponding regions of an IgG3 antibody, to increase the CDC response. (E) Schematic representation of mAb hexamerization which facilitates C1q binding and enhances CDC, and can occur upon antigen binding by IgG1 antibodies naturally or when harboring the E345G or E430g mutations.