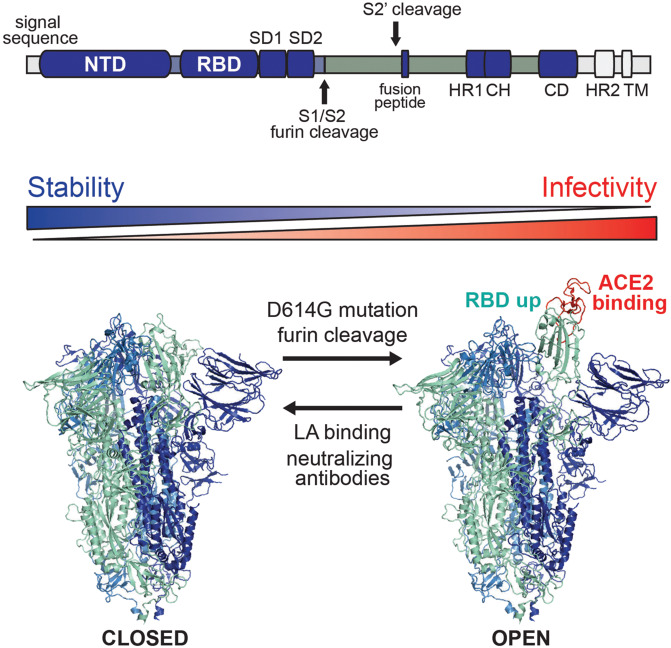
Fig. 1. SARS-CoV-2 spike prefusion structure in a dynamic equilibrium between closed and open states.
Above: Domain arrangement of SARS-CoV-2 S glycoprotein. S1 comprises a signal sequence, NTD, RBD, SD1 and SD2. S2 comprises a second cleavage site upstream of the fusion peptide, HR1, CH, CD, HR2, the TM domain and cytoplasmic C-terminus. NTD, N-terminal domain; RBD, receptor-binding domain; SD1, subdomain 1; SD2, subdomain 2; S2’, second protease cleavage site; HR1, heptad repeat 1; CH, central helix; CD, connector domain; HR2, heptad repeat 2; TM, transmembrane domain. Cleavage sites are marked with arrow. Below: Closed (PDB ID: 6zb58) and open (PDB ID: 6zgg3) conformations of S are in a dynamic equilibrium which is shifted by mutations, proteolytic cleavage, binding of LA or antibodies. The open conformations, with one or more RBDs up, expose the RBM (colored in red) required for ACE2 binding and subsequent host cell infection.