Figure 5. Post-translational modifications and co-chaperones fine-tune Hsp90 chaperone machinery.
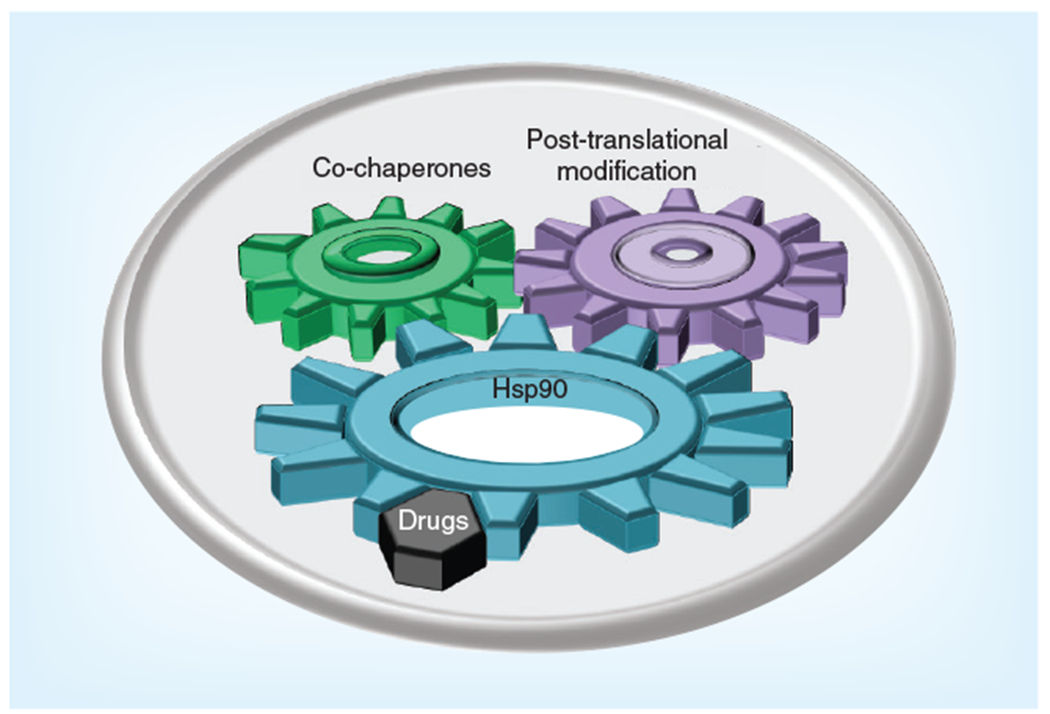
The Hsp90 chaperone cycle is regulated by the interplay between ATP binding to Hsp90 and the regulated association/dissociation of various co-chaperones. The Hsp90 chaperone machine is also regulated by a number of diverse post-translational modifications of Hsp90 and co-chaperones, including phosphorylation, S-nitrosylation, oxidation, acetylation and ubiquitination. Hsp90 inhibitors effectively displace ATP from its binding pocket in the N-domain of Hsp90, disrupt this complex mechanism and prevent chaperone cycling.
