Abstract
Coronavirus disease 2019 (COVID-19) pandemic has brought challenges to health and social care systems. However, the empirical use of antibiotics is still confusing. Presently, a total of 1123 patients with COVID-19 admitted to Renmin Hospital of Wuhan University was included in this retrospective cohort study. The clinical features, complications and outcomes were compared between the suspected bacterial infection and the no evidence of bacterial infection. The risk factors of mortality and the incidence of acute organ injury were analyzed. As a result, 473 patients were selected to suspected bacterial infection (SI) group based on higher white blood cell count and procalcitonin or bacterial pneumonia on chest radiography. 650 patients were selected to the no evidence of bacterial infection (NI) group. The SI group had more severely ill patients (70.2% vs. 39.8%), more death (20.5% vs. 2.2%), and more acute organ injury (40.2% vs. 11.2%). Antibiotics were found associated with improved mortality and an increased risk for acute organ injury in hospitalized patients with COVID-19. Intravenous moxifloxacin and meropenem increased the death rate in patients with suspected bacterial infection, while oral antibiotics reduced mortality in this group. Moreover, penicillin and meropenem treatments were associated with increased mortality of the patients with no evidence of bacterial infection. In conclusion, patients with suspected bacterial infection were more likely to have negative clinical outcomes than those without bacterial infection. Empirical use of antibiotics may not have the expected benefits.
Keywords: COVID-19, Bacterial infection, Mortality, Antibiotics
1. Introduction
Lower respiratory tract infections had a major impact on public health over the past two decades [1], [2]. Severe acute respiratory syndrome (SARS) [3], Middle East respiratory syndrome (MERS) [4], and Coronavirus disease 2019 (COVID-19) [5] have presented an unprecedented challenge for the healthcare community worldwide.
In previous influenza pandemics, studies have considered that bacterial infection accompanied with viral infection is an important factor affecting mortality [6]. Bacterial co‐infection and secondary bacterial infection are considered critical risk factors for the severity and mortality rates of COVID-19. The prevalence of bacterial infection was range from 14% to 100% in intensive care unit (ICU) patients [7], [8], [9]. A recent meta-analysis suggested that only 7% of COVID-19 patients were expected to have bacterial infection [10]. However, it should be noted that most patients received antibiotics before they were diagnosed with bacterial infection, which may influence the prevalence of bacterial infection. A low rate of laboratory-confirmed bacterial coinfection was observed in patients with COVID-19 in the latest studies [11], [12]. Thus, the widely accepted view was the rate of bacterial infection with COVID-19 is not high.
Using antibiotics to treat viral diseases may increase drug resistance and raise the risks of allergic reactions [13]. Herein, determining the presence of bacterial infection is essential to guide the empirical use of antibiotics, thereby reducing antibiotic abuse. On the other hand, the diagnosis of bacterial infection is sometimes challenging because clinical symptoms of infections are similar. Therefore, it may be hard to differentiate viral from bacterial infection. Raised procalcitonin observed in COVID-19 could be due either to bacterial infection [14] and non-elevated procalcitonin, which may be a good predictor of the absence of bacterial infection. Studies have reported that patients infected with other pathogens had higher white blood cell (WBC), neutrophil counts, D-dimer, C-reactive protein (CRP) and procalcitonin (PCT) levels than those of infected with SARS- coronavirus 2 (SARS-CoV-2) homogeneously [15]. The empirically use of the antibiotics was used for patients with suspected bacterial infection. However, it was unknown whether this empirically use of the antibiotics had positive outcomes.
Presently, we analyzed the biomarkers of patients admitted to Renmin Hospital of Wuhan University with COVID-19 and screened the patients with the clinical indication of suspected bacterial infection. We compared the clinical characteristics and outcomes of patients with suspected bacterial infection and patients with no bacterial infection to further explore the association between antibiotics usage and death in COVID-19.
2. Methods
Patients with COVID-19 admitted to Renmin Hospital of Wuhan University (Hubei, China) from January 26, 2020, to March 18, 2020, were reflected in the present retrospective analysis. This study was approved by the Research Ethics Commission of Renmin Hospital of Wuhan University with a waiver of informed consent (WDRY2020-K069). Demographic information and clinical records (clinical characteristics, laboratory, radiological results and treatments) of COVID-19 patients on hospital admission were extracted for retrospective analysis. Exclusion criteria: (1) patients during pregnancy and lactation. (2) Patients who received treatments less than 3 days. (3) Patients who transferred to other hospitals.
2.1. Definitions
The diagnosis of COVID-19 was confirmed as a positive result for a nasopharyngeal swab and respiratory pathogen nucleic acid test with high-throughput sequencing or real-time reverse transcriptase-polymerase chain reaction (RT-PCR).
The severity of COVID-19 was categorized as moderate and severe. The moderate type represents patients with non-pneumonia and mild to moderate pneumonia. The severe type was characterized by (1) dyspnea (respiratory rate ≥ 30/min); (2) blood oxygen saturation ≤ 93%; (3) PaO2/FiO2 ratio < 300 or lung infiltrates > 50% within 24–48 h [16]. If one of the above items was met, it was classified as severe.
For bacterial infection, the COVID-19 patients were grouped as suspected bacterial infection (SI) and no evidence of bacterial infection (NI). The patients were classified as the SI group if the any one of (1)–(4) and (5) were met: (1) Recent cough, expectoration or aggravation of original respiratory disease symptoms, and purulent sputum, with or without chest pain; (2) White blood cell count > 10 × 109/L; (3) PCT > 0.1 ng/ml; (4) Fever (body temperature is over 37 °C), (5) Chest radiography showed bacterial pneumonia. The NI group was characterized by normal white blood cell count and PCT, and the chest radiography showed viral pneumonia.
The acute organ injury was described as acute heart injury, acute kidney injury and acute liver injury newly discovered after admission. Acute kidney injury was defined on the basis of highest serum level of creatinine according to the Kidney Disease Improving Global Outcome (KDIGO) clinical practice guidelines [17]. Cardiac injury was defined as a serum level of cardiac troponin I above the 99th percentile upper baseline limit [18]. Acute liver injury was identified as an increase in alanine aminotransferase of 5 times the upper reference limit or alkaline phosphatase increase of twice the upper reference limit.
2.2. Statistical analysis
IBM SPSS Statistics (version 23.0) was adopted for statistical analysis. Categorical variables were expressed as proportions and compared between groups using the Chi-square test. Continuous data were expressed as median (interquartile range) and compared between groups using the Mann-Whitney U test. Multivariate logistic regression models were utilized to explore the risk factors associated with in-hospital death. Forest plots were employed to display logistic regression analysis results. The incidence of acute organ injury was examined with the same method of the mortality model. A 2-sided α of less than 0.05 was considered statistically significant.
3. Results
3.1. Presenting characteristics
Of 1500 COVID-19 patients, 1123 patients were included. The suspected bacterial infection (SI) group included 473 patients, and the no evidence of bacterial infection (NI) group included 650 patients (Table 1 ). Male patients accounted for 49.9% of all patients, and 60.0% of patients were in the SI group. The median age of the SI patients was 65 (IQR, 55–72), which was higher than that of the NI group patients (58, IQR, 47–67). Patients of the SI group had a higher preexisting rate of hypertension (35.5% vs. 29.7%), and diabetes disease (16.3% vs. 10.8%) (all P values < 0.05). Cardiovascular disease, cancer and COPD were not significantly different between groups. Fever and cough were the most common onset of symptoms in both groups. The SI group had a significantly higher proportion of fever (75.3% vs. 61.5%) and expiratory dyspnea (16.3% vs. 10.9%).
Table 1.
Demographic, past medical history and symptoms findings of patients on admission.
| No./total No. (%) |
||||
|---|---|---|---|---|
| Total (n = 1123) | SI(n = 473) | NI(n = 650) | P Value | |
| Male | 560 (49.9) | 284 (60.0) | 276 (42.5) | <0.001 |
| Median age (IQR) year | 61 (50–69) | 65 (55–72) | 58 (47–67) | <0.001 |
| Past Medical History | ||||
| Hypertension | 361 (32.1) | 168 (35.5) | 193 (29.7) | 0.039 |
| Coronary heart disease | 95 (8.5) | 44 (9.3) | 51 (7.8) | 0.387 |
| Other heart disease | 46 (4.1) | 26 (5.5) | 20 (3.1) | 0.048 |
| Diabetes | 147 (13.1) | 77 (16.3) | 70 (10.8) | 0.007 |
| Cancer | 40 (3.6) | 21 (4.4) | 19 (2.9) | 0.176 |
| COPD | 40 (3.6) | 17 (3.6) | 23 (3.6) | 0.963 |
| Symptom | ||||
| Fever | 756 (67.3) | 356 (75.3) | 400 (61.5) | <0.001 |
| Cough | 467 (41.6) | 200 (42.3) | 267 (41.1) | 0.685 |
| Expectoration | 53 (4.7) | 27 (5.7) | 26 (4.0) | 0.183 |
| Expiratory dyspnea | 148 (13.2) | 77 (16.3) | 71 (10.9) | 0.009 |
| Weakness | 138 (12.3) | 66 (14.0) | 72 (11.1) | 0.147 |
| Chest tightness | 106 (9.4) | 45 (9.5) | 61 (9.4) | 0.942 |
SI = suspected bacterial infection. NI = no evidence of bacterial infection. IQR = interquartile range.
3.2. Severity, treatments and complications
Among 1123 patients with COVID-19, 591 patients (52.6%) were the severe type. The suspected bacterial infection (SI) group included 332 severe patients, and the no evidence of bacterial infection (NI) group included 295 severe patients (Table 2 ). Compared with the SI group, the NI group had lower rates of antiviral therapy (79.7% vs. 93.0%), antibiotic therapy (53.5% vs. 93.9%) and glucocorticoid therapy (20.9% vs. 72.9%).
Table 2.
Severity, treatments and complications.
| No./total No. (%) |
||||
|---|---|---|---|---|
| Total (n = 1123) | SI (n = 473) | NI(n = 650) | P Value | |
| Clinical severity | ||||
| General | 532 (47.4) | 141 (29.8) | 391 (60.2) | <0.001 |
| Severe | 591 (52.6) | 332 (70.2) | 295 (39.8) | <0.001 |
| Treatment | ||||
| Antiviral | 958 (85.3) | 440 (93.0) | 518 (79.7) | <0.001 |
| Antibiotics | 792 (70.5) | 444 (93.9) | 348 (53.5) | <0.001 |
| Oral antibiotics | 385 (34.3) | 198 (41.9) | 187 (28.8) | <0.001 |
| Intravenous antibiotics | 602 (53.6) | 390 (82.5) | 212 (32.6) | <0.001 |
| Azithromycin | 63 (5.6) | 33 (7.0) | 30 (4.6) | 0.090 |
| Fluoroquinolones | 666 (59.3) | 378 (79.9) | 288 (44.3) | <0.001 |
| Levofloxacin | 77 (6.9) | 45 (9.5) | 32 (4.9) | 0.003 |
| Oral moxifloxacin | 281 (20.5) | 147 (31.1) | 134 (20.6) | <0.001 |
| Intravenous moxifloxacin | 409 (36.4) | 274 (57.9) | 135 (20.8) | <0.001 |
| Cephalosporins | 137 (12.2) | 91 (19.2) | 46 (7.1) | <0.001 |
| Ceftazidime | 83 (7.4) | 62 (13.1) | 21 (3.2) | <0.001 |
| Penicillins | 50 (4.5) | 35 (7.4) | 15 (2.3) | <0.001 |
| Carbapenems | 108 (9.6) | 95 (20.1) | 13 (2.0) | <0.001 |
| Meropenem | 77 (6.9) | 68 (14.4) | 9 (1.4) | <0.001 |
| Glucocorticoids | 481 (42.8) | 345 (72.9) | 136 (20.9) | <0.001 |
| Mechanical ventilation | 128 (11.4) | 105 (22.2) | 23 (3.5) | <0.001 |
| Outcome | ||||
| Death | 111(9.9) | 97 (20.5) | 14 (2.2) | <0.001 |
| Median period in hospital | 21 (12–35) | 27 (13–40) | 18 (11–31) | <0.001 |
| Acute organ injury | 324 (28.9) | 228(48.2) | 96 (14.8) | <0.001 |
| Myocardial injury | 175 (15.6) | 139 (29.4) | 36 (5.5) | <0.001 |
| Renal injury | 101 (9.0) | 80 (16.9) | 21 (3.2) | <0.001 |
| Liver dysfunction | 187 (16.7) | 130 (27.5) | 57 (8.8) | <0.001 |
| Electrolyte disturbance | 657 (58.5) | 378 (79.9) | 279 (42.9) | <0.001 |
| Hypoproteinemia | 196 (17.5) | 169 (35.7) | 27 (4.2) | <0.001 |
| Anemia | 504 (44.9) | 290 (61.3) | 214 (32.9) | <0.001 |
Clinical outcomes in the SI group were significantly different from those in the NI group. The mortality rate was higher in the SI group than in the NI group (20.5% vs. 2.2%). Among the SI group, the period from admission to discharge or death was 27 (13–40) days, distinctly longer than that of the NI group. The incidence of acute organ injury was higher in the SI group (48.2% vs. 14.8%). Compared to the NI group, complications were significantly more in the SI group. Electrolyte disturbance was the most common complication in both groups. The corresponding incidence of cardiac injury (29.4%), acute renal injury (16.9%), liver dysfunction (27.5%), electrolyte disturbance (79.9%), hypoproteinemia (35.7%), and anemia (61.3%) were all significantly higher in SI group (Table 2).
3.3. Risk factors for the whole
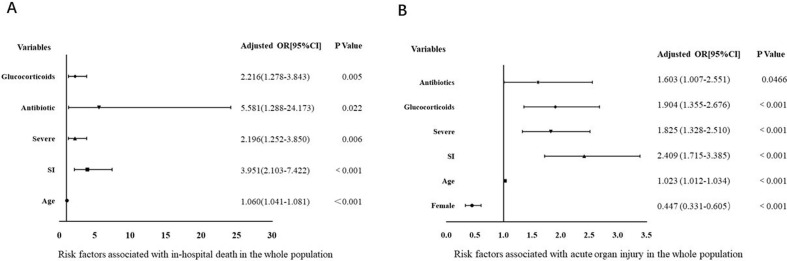
We included 1123 patients in the multivariate logistic regression model. The clinical parameters, including sex, age, comorbidity, severity, suspected bacterial infection, and treatments were incorporated into the regression models. We revealed that older age, suspected bacterial infection, severe illness, antibiotics, and glucocorticoid treatment promoted in-hospital death (Fig. 1 A). In comparison, older age, male gender, suspected bacterial infection, severe illness, antibiotic usage and glucocorticoid treatment were associated with an increased incidence of acute organ injury in patients with COVID-19 (Fig. 1B).
Fig. 1.
Forest plot of multivariate logistic regression analysis of factors associated with COVID-19.
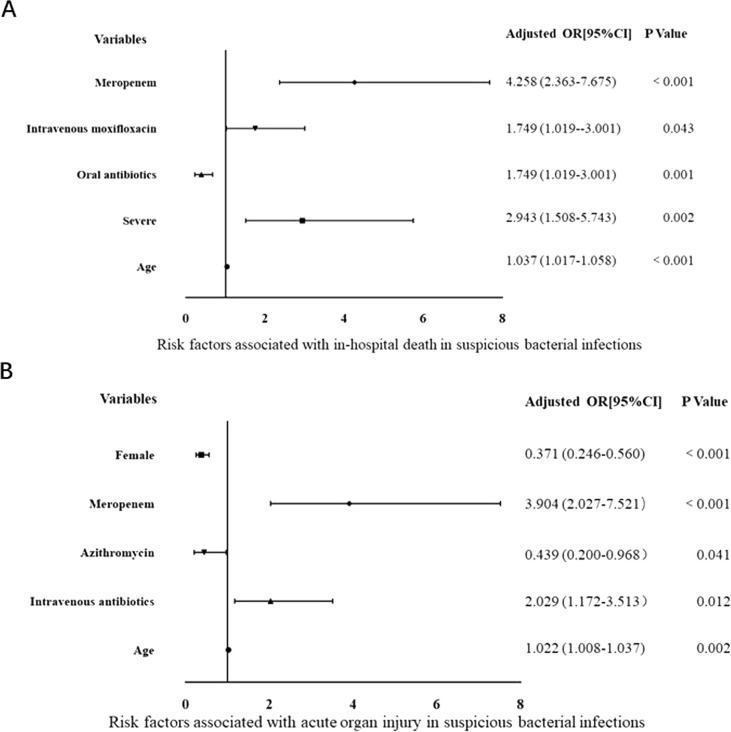
3.4. Risk factors for the suspected bacterial infection
Since different groups had significantly different outcomes, stratified analysis for the SI group and NI group was made to find the association between death and antibiotic therapy. The specific types of antibiotics were incorporated into the statistics. In the SI group, older age, severer illness, and intravenous moxifloxacin and meropenem were associated with increased risks of death. In contrast, receiving oral antibiotics enhanced mortality (Fig. 2 A). Meanwhile, older age, male gender, intravenous antibiotics and receiving meropenem were associated with the elevated incidence of acute organ injury in the SI group (Fig. 2B). On the contrary, receiving azithromycin and female gender were related to decreased acute organ injury incidence in the SI group.
Fig. 2.
Forest plot of multivariate logistic regression analysis of factors associated with suspected bacterial infection.
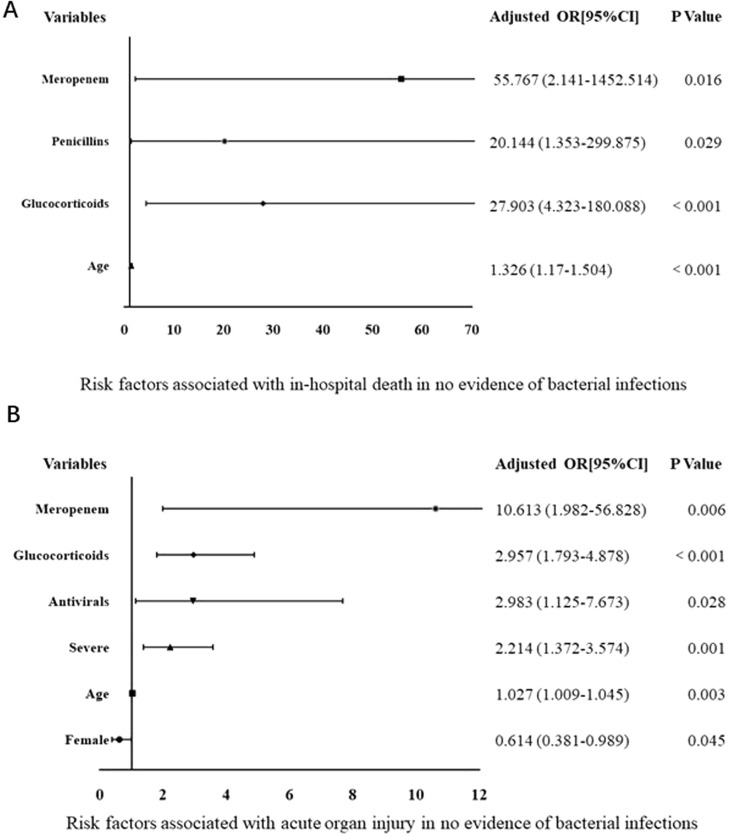
3.5. Risk factors for the no evidence of bacterial infection
For the NI group, receiving penicillin, meropenem, and glucocorticoids treatments was associated with an increased death rate (Fig. 3 A). In contrast, older age, male gender, severe illness, receiving antivirals, glucocorticoids and meropenem were linked to the increased incidence of acute organ injury (Fig. 3B).
Fig. 3.
Forest plot of multivariate logistic regression analysis of factors associated with no evidence of bacterial infection.
4. Discussion
Our results showed that patients with suspected bacterial infection were more likely to have negative clinical outcomes than those with no evidence of bacterial infection. The suspected bacterial infection group included severely ill patients, and the outcomes showed that the SI group had more death and complications. Similarly, a recent review paper suggested that a mortality rate of 10.9% (53 of 482) was observed in all cases of viral pneumonia due to secondary infections [19]. It is probably because that increased procalcitonin values are associated with a nearly 5-fold higher risk of severe SARS-CoV-2 infection [20] , and bacterial infection is likely to worsen an already poor prognosis [21]. Based on past experience, clinicians often take more drastic treatment for patients with severe illness. We can also see this trend in our research. The proportion of patients receiving antibiotics and glucocorticoids in the SI group was significantly higher than that of the NI group.
We identified several risk factors for in-hospital death of the patients with COVID-19. Receiving antibiotics was associated with higher in-hospital death. Previous studies demonstrated that the administration of multiple antibiotics did not change the outcomes of the disease [22], [23]. In our center, antibiotic therapy seemed to increase the risk of death. It’s because clinicians are more likely to use antibiotics for severe patients, who often die of SARS-CoV-2. The rate of bacterial infection is not high, but 70.5% of patients received antibiotics in our research, which means most patients would not benefit from antibiotics. Evidence suggests that very high proportion of COVID-19 patients were receiving unnecessary antibiotic treatment [24], [25], [26]. This increase in antibiotic administration can cause pressure on bacterial pathogens which lead to antibiotic resistance [27]. Yet, there is a serious problem that the presence of antimicrobial elements in the environment can stimulate antimicrobial resistance [28] and the potential consequence of the COVID-19 pandemic is the long-term propagation of antimicrobial resistance [29].
Both guidelines for COVID-19 management suggest prescribing antibiotics therapy for suspected bacterial infection [30], [31]. In the present study, we found that if laboratory tests indicated suspected bacterial infection, the empirical use of oral antibiotics might reduce mortality of patients with COVID-19. Previous studies demonstrated that procalcitonin and neutrophilic leukocytosis are valuable markers for bacterial infection. Timothy MU [32] believes that procalcitonin is not specific in differentiating viral and bacterial pneumonia. We still chose these ways because when this new virus occurred in China, there is no standard strategy. As a result, some doctors chose routine empiric antibiotic use in patients with elevated WBC or PCT. In terms of the value of PCT, there is a lot of controversies. In Meisner’s research [33], the patients with PCT value > 0.25 µg/liter were defined bacterial infection and those who had PCT value ≤ 0.25 µg/liter were defined non-bacterial. In our center, the PCT value > 0.1 µg/liter indicated the presence of bacterial infection. Hence, we chose PCT value > 0.1 µg/liter as the indicators of grouping. From our study, the WBC and procalcitonin may not be the biomarkers for distinguishing bacteria from SARS‐CoV‐2. Facing the increased WBC or procalcitonin, clinicians should not immediately choose antibiotics, especially intravenous antibiotics. Empiric antibiotic use in patients with COVID-19 will not offer considerable benefit. The reliable way is to rely on the microbiological results.
Receiving azithromycin may decrease the incidence of acute organ injury in suspected bacterial infection. The results were interesting. Azithromycin has been the focus of intense scholarly debate. The in vitro evidence suggests that azithromycin has antiviral properties [34], including against SARS‐CoV‐2. A retrospective study found that treatment with hydroxychloroquine and azithromycin reduced COVID-19-related mortality [35]. However, another retrospective cohort study of 1438 patients in New York [36] showed that treatment with hydroxychloroquine and azithromycin was not associated with lower in-hospital mortality. Kome Gbinigie [37] reported that no evidence supports the use of azithromycin for the treatment of COVID-19, unless it is used to treat bacterial super-infection. Collectively, no link was found between azithromycin and mortality, but azithromycin repressed organ damage from suspected bacterial infection. Nevertheless, the number of patients receiving azithromycin in our research is small and more prospective studies are needed to clarify the association.
For the patients with no evidence of bacterial infection, receiving penicillin and meropenem increased mortality, and meropenem elevated the incidence of acute organ injury. It was confirmed that antibiotic therapy should only be used for patients with a presentation suggestive of bacterial infection or supportive, positive microbiology [38]. Hantoushzadeh S and his colleagues [39] reported that the consumption of some antibiotics in the lack of bacterial infection could lead to undesirable outcomes. We found that meropenem was related to increased mortality and organ damage in both SI group and NI group. It means meropenem may not be the first choice for empirical antibiotic use in patients with COVID-19.
Our study presents several limitations. Firstly, due to the imperfect clinical records, there was no record of symptom changes, which limited our analysis of the clinical efficacy. Secondly, our study was retrospective, and the impact of the treatments on the patients was inferred. In addition, the number of patients and treatment drugs were limited. In the future, more rigorous case-control studies will be needed to clarify the effect of antibiotic treatment, and more work is needed on biomarkers to help identify bacterial infection.
5. Conclusion
Patients with suspected bacterial infection were more likely to have negative clinical outcomes than those with no evidence of bacterial infection. Antibiotic therapy was associated with increased mortality and most patients would not benefit from antibiotics. Empirical use of antibiotics may not have the positive outcomes as expected. We are in the pandemic of COVID-19, understanding the potential for bacterial infection is important. Detailed antimicrobial policies and guidelines should be applied and promoted in the times of COVID-19.
Authors contribution
Conceived and designed the experiments: Xuejun Jiang.
Performed the experiments: Chengyin Liu, Ying Wen, Weiguo Wan, Jingchao Lei.
Statistical analysis: Chengyin Liu.
Wrote the paper: Chengyin Liu, Ying Wen.
All authors read and approved the final manuscript.
Ethics statement
Our study was approved by the Ethics Review Board of Renmin Hospital of Wuhan University.
Data Availability Statement
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not- for- profit sectors.
CRediT authorship contribution statement
Chengyin Liu: Writing - original draft, Writing - review & editing. Ying Wen: Writing - original draft. Weiguo Wan: Investigation, Data curation. Jingchao Lei: Data curation. Xuejun Jiang: Conceptualization, Methodology.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Wang X., Zhang X., He J. Challenges to the system of reserve medical supplies for public health emergencies: reflections on the outbreak of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) epidemic in China. Biosci Trends. 2020;14(1):3–8. doi: 10.5582/bst.2020.01043. [DOI] [PubMed] [Google Scholar]
- 2.Yin Y., Wunderink R.G. MERS, SARS and other coronaviruses as causes of pneumonia. Respirology. 2018;23(2):130–137. doi: 10.1111/resp.13196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Stadler K., Masignani V., Eickmann M., et al. SARS–beginning to understand a new virus. Nat. Rev. Microbiol. 2003;1(3):209–218. doi: 10.1038/nrmicro775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chafekar A., Fielding B.C. MERS-CoV: understanding the latest human coronavirus threat. Viruses. 2018;10(2):93. doi: 10.3390/v10020093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Akande O.W., Akande T.M. COVID-19 pandemic: A global health burden. Niger. Postgrad. Med. J. 2020;27(3):147–155. doi: 10.4103/npmj.npmj_157_20. [DOI] [PubMed] [Google Scholar]
- 6.Joseph C., Togawa Y., Shindo N. Bacterial and viral infections associated with influenza. Influenza Other Respir Viruses. 2013;7(Suppl 2):105–113. doi: 10.1111/irv.12089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fu Y., Yang Q., Xu M., et al. Secondary bacterial infections in critical Ill patients with coronavirus disease 2019. Open Forum Infect. Dis. 2020;7(6) doi: 10.1093/ofid/ofaa220. ofaa220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Contou D., Claudinon A., Pajot O., et al. Bacterial and viral co-infections in patients with severe SARS-CoV-2 pneumonia admitted to a French ICU. Ann. Intensive Care. 2020;10(1):119. doi: 10.1186/s13613-020-00736-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sharifipour E., Shams S., Esmkhani M., et al. Evaluation of bacterial co-infections of the respiratory tract in COVID-19 patients admitted to ICU. BMC Infect. Dis. 2020;20(1):646. doi: 10.1186/s12879-020-05374-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lansbury L., Lim B., Baskaran V., Lim W.S. Co-infections in people with COVID-19: a systematic review and meta-analysis. J. Infect. 2020;81(2):266–275. doi: 10.1016/j.jinf.2020.05.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Youngs J., Wyncoll D., Hopkins P., Arnold A., Ball J., Bicanic T. Improving antibiotic stewardship in COVID-19: Bacterial co-infection is less common than with influenza. J. Infect. 2020;S0163-4453(20):30436–30439. doi: 10.1016/j.jinf.2020.06.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hughes S., Troise O., Donaldson H., Mughal N., Moore L.S.P. Bacterial and fungal coinfection among hospitalized patients with COVID-19: a retrospective cohort study in a UK secondary-care setting. Clin. Microbiol. Infect. 2020;26(10):1395–1399. doi: 10.1016/j.cmi.2020.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.A. Machowska, C. Stålsby Lundborg, Drivers of irrational use of antibiotics in europe. Int. J. Environ. Res. Public Health 2018; 16(1): 27. Published 2018 Dec 23. [DOI] [PMC free article] [PubMed]
- 14.Han J., Gatheral T., Williams C. Procalcitonin for patient stratification and identification of bacterial co-infection in COVID-19. Clin. Med. (Lond). 2020;20(3) doi: 10.7861/clinmed.Let.20.3.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 cases from the Chinese center for disease control and prevention. JAMA. 2020;323(13):1239–1242. doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 17.Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin. Pract. 2012;120(4):c179–c184. doi: 10.1159/000339789. [DOI] [PubMed] [Google Scholar]
- 18.Thygesen K., Alpert J.S., Jaffe A.S., et al. Fourth universal definition of myocardial infarction (2018) Glob Heart. 2018;13(4):305–338. doi: 10.1016/j.gheart.2018.08.004. [DOI] [PubMed] [Google Scholar]
- 19.Manohar P., Loh B., Nachimuthu R., Hua X., Welburn S.C., Leptihn S. Secondary bacterial infections in patients with viral pneumonia. Front. Med. (Lausanne). 2020;7:420. doi: 10.3389/fmed.2020.00420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lippi G., Plebani M. Procalcitonin in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. Clin. Chim. Acta. 2020;505:190–191. doi: 10.1016/j.cca.2020.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Crotty M.P., Akins R., Nguyen A.n., Slika R., Rahmanzadeh K., Marie H. Investigation of subsequent and co-infections associated with SARS-CoV-2 (COVID-19) in hospitalized patients. medRxiv Prepr. 2020:1–19. [Google Scholar]
- 22.Buetti N., Mazzuchelli T., Lo Priore E., et al. Early administered antibiotics do not impact mortality in critically ill patients with COVID-19 [published online ahead of print, 2020 Jun 5] J. Infect. 2020;81(2):e148–e149. doi: 10.1016/j.jinf.2020.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Du Y., Tu L., Zhu P., et al. Clinical features of 85 fatal cases of COVID-19 from Wuhan. A retrospective observational study. Am. J. Respir. Crit. Care Med. 2020;201(11):1372–1379. doi: 10.1164/rccm.202003-0543OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wei W., Ortwine J., Mang N., Joseph C., Hall B., Prokesch B. Limited role for antibiotics in COVID-19: scarce evidence of bacterial coinfection. SSRN Electronic J. 2020 doi: 10.2139/ssrn.3622388. [DOI] [Google Scholar]
- 25.Antimicrobial resistance in the age of COVID-19. Nat. Microbiol. 5(6) (2020 Jun) 779. [DOI] [PubMed]
- 26.Manohar P., Loh B., Leptihn S. Will the overuse ofantibiotics during the coronavirus pandemic accelerate antimicrobial resistanceof bacteria? Infect. Microb. Dis. 2020;2(3):87–88. doi: 10.1097/IM9.0000000000000034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Mirzaei R., Goodarzi P., Asadi M., et al. Bacterial co-infections with SARS-CoV-2. IUBMB Life. 2020 doi: 10.1002/iub.2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Usman M., Farooq M., Hanna K. Environmental side effects of the injudicious use of antimicrobials in the era of COVID-19. Sci. Total Environ. 2020;745 doi: 10.1016/j.scitotenv.2020.141053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rawson T.M., Ming D., Ahmad R., Moore L.S.P., Holmes A.H. Antimicrobial use, drug-resistant infections and COVID-19. Nat. Rev. Microbiol. 2020;18(8):409–410. doi: 10.1038/s41579-020-0395-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.COVID-19 rapid guideline: managing suspected or confirmed pneumonia in adults in the community. Available at https://www.nice.org.uk/guidance/ng165. Accessed [June 24 2020]. [PubMed]
- 31.Clinical management of COVID-19. Available at https://www.who.int/publications/i/item/clinical-management-of-covid-19. Accessed [June 24 2020].
- 32.Uyeki T.M., Bernstein H.H., Bradley J.S., et al. Clinical practice guidelines by the infectious diseases society of America: 2018 update on diagnosis, treatment, chemoprophylaxis, and institutional outbreak management of seasonal influenzaa. Clin. Infect. Dis. 2019;68(6):895–902. doi: 10.1093/cid/ciy874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Meisner M. Update on procalcitonin measurements. Ann. Lab. Med. 2014;34(4):263–273. doi: 10.3343/alm.2014.34.4.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Damle B., Vourvahis M., Wang E., Leaney J., Corrigan B. Clinical pharmacology perspectives on the antiviral activity of azithromycin and use in COVID-19. Clin. Pharmacol. Ther. 2020;108(2):201–211. doi: 10.1002/cpt.1857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Million M., Lagier J.C., Gautret P., et al. Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: a retrospective analysis of 1061 cases in Marseille, France. Travel Med. Infect. Dis. 2020;35 doi: 10.1016/j.tmaid.2020.101738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rosenberg E.S., Dufort E.M., Udo T., et al. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York State. JAMA. 2020;323(24):2493–2502. doi: 10.1001/jama.2020.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.K. Gbinigie, K. Frie, Should azithromycin be used to treat COVID-19? A rapid review. BJGP Open. 4(2) (2020) bjgpopen20X101094. Published 2020 Jun 23. [DOI] [PMC free article] [PubMed]
- 38.NICE. COVID-19 rapid guideline: antibiotics for pneumonia in adults in hospital. NICE guideline [NG173] 2020; (May):1–17. [PubMed]
- 39.Hantoushzadeh S., Norooznezhad A.H. Possible cause of inflammatory storm and septic shock in patients diagnosed with (COVID-19) Arch. Med. Res. 2020;51(4):347–348. doi: 10.1016/j.arcmed.2020.03.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data sets used and analyzed during the current study are available from the corresponding author on reasonable request.