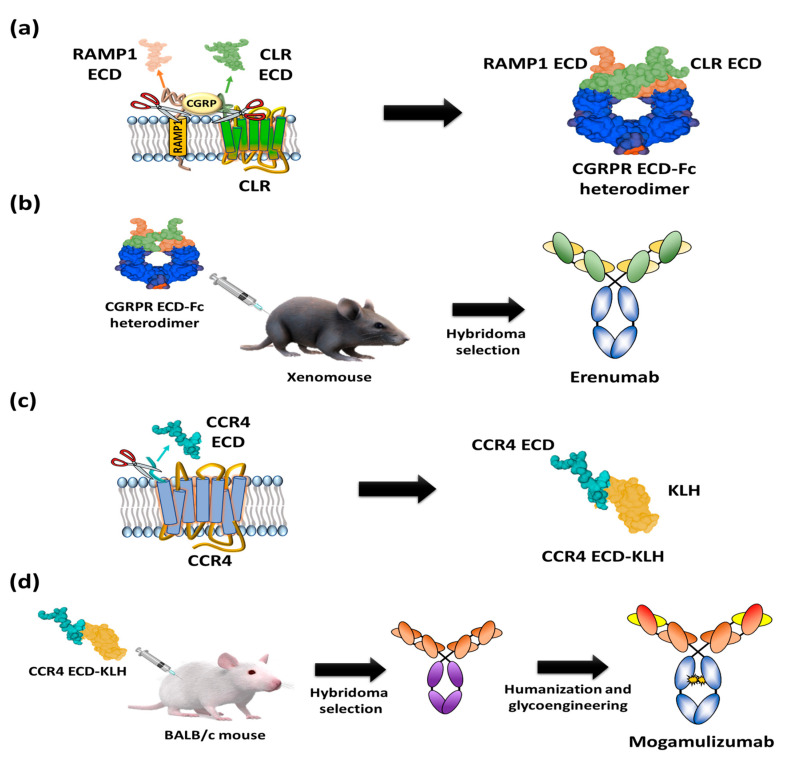
Figure 4.
Schematic diagrams of antigen preparation and overall procedure for discovery of US FDA-approved anti-GPCR antibody. (a) A heterodimer CGRPR Fc protein prepared by Fc fusion constructs of N-terminal extracellular regions of CLR and ectodomain of RAMP1. (b) Immunization of heterodimeric CGRPR Fc proteins into XenoMouse hybridoma selection to isolate erenumab. (c) The N-terminal region of CCR4 (28 amino acids: N2-C29) fused with keyhole limpet hemocyanin (KLH). (d) Injection of the N-terminal region of CCR4-KLH into BALB/c mice and hybridoma selection, humanization, and defucosylation of N-inked glycans of Fc to generate mogamulizumab.