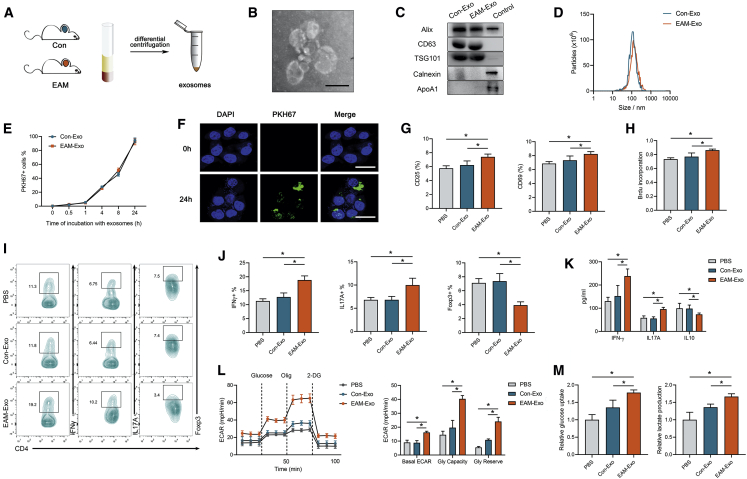
Figure 1.
Characterization and Functional Validation of Serum Exosomes from Mice with Experimental Autoimmune Myocarditis (EAM) and Controls
(A) Exosomes were isolated from the serum of mice subjected to complete Freund’s adjuvant (CFA) and PBS without MyHC-α stimulation (control group, Con-Exo) or those immunized with MyHC-α (EAM-Exo). (B) Representative electron micrograph of isolated exosomes. Scale bar, 100 nm. (C) Representative blots of exosomal marker proteins (Alix, CD63, TSG101) and contamination proteins (Calnexin, ApoA1) in exosomes purified from the serum of EAM mice and those of the control group. CD4+ T cell lysates were used as a control for Alix, CD63, TSG101, and Calnexin, whereas cell lysates containing recombinant ApoA1 were used as a control for ApoA1. (D) Representative results of nanoparticle tracking analysis demonstrating size distributions of exosomes. (E and F) CD4+ T cells were cultured in the presence or absence (control) of PKH67-labeled exosomes (green). (E) CD4+ T cells incubated with PKH67-labeled exosomes for indicated times were analyzed by flow cytometry for exosome uptake. (F) After 24 h, the uptake of exosomes was confirmed by confocal imaging. Nuclei were stained with DAPI (blue). Scale bars, 20 μm. (G) CD25 and CD69 expression on CD4+ T cells treated with PBS, Con-Exo, or EAM-Exo. (H) Proliferation of T cells, as measured by BrdU, following stimulation with anti-CD3/CD28 antibodies. (I) The numbers of Th1 (CD4+ IFN-γ+), Th17 (CD4+ IL-17A+), and Treg (CD4+ Foxp3+) were assessed by flow cytometry. (J) The percentages of Th1, Th17, and Treg were measured. (K) The levels of cytokines, including IFN-γ, IL-17A, and IL-10 in CD4+ T cells, were measured by ELISA. (L) The extracellular acidification rate (ECAR) was measured over time from basal levels and followed by glucose, oligomycin (Olig), and 2-deoxy-D-glucose (2-DG) addition. Basal ECAR, glycolytic capacity, and glycolytic reserve levels were calculated. (M) Cellular glucose uptake and lactate production were measured after exosome addition. Data are presented as the means ± SEM (n = 6); ∗p < 0.05.