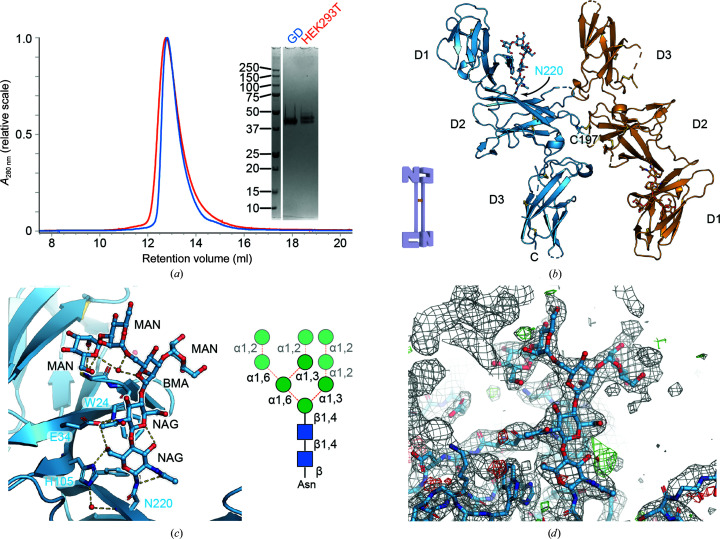
Figure 3.
Structural analysis of IL-12B derived from HEK293 GD. (a) Comparison of HEK293T and HEK293 GD-derived IL-12B; the inset shows SDS–PAGE analysis of the purified proteins under reducing conditions. (b) Cartoon representation of the IL-12B homodimer formed through crystallographic symmetry (a twofold crystallographic axis is present at Cys197). (c) Close-up of the immature N-linked glycan present on Asn220 and its extensive interactions with the neighboring protein. The inset shows a schematic representation of the glycan; the mannose residues that could not be readily identified in the electron density are partially transparent. (d) Representative electron density around the glycan at position Asn220. Density at the top left belongs to a symmetry mate. The 2mF o − DF c electron-density map is shown as a gray mesh. Residual positive and negative mF o − DF c electron-density maps (contoured at ±3σ) are shown in green and red, respectively.