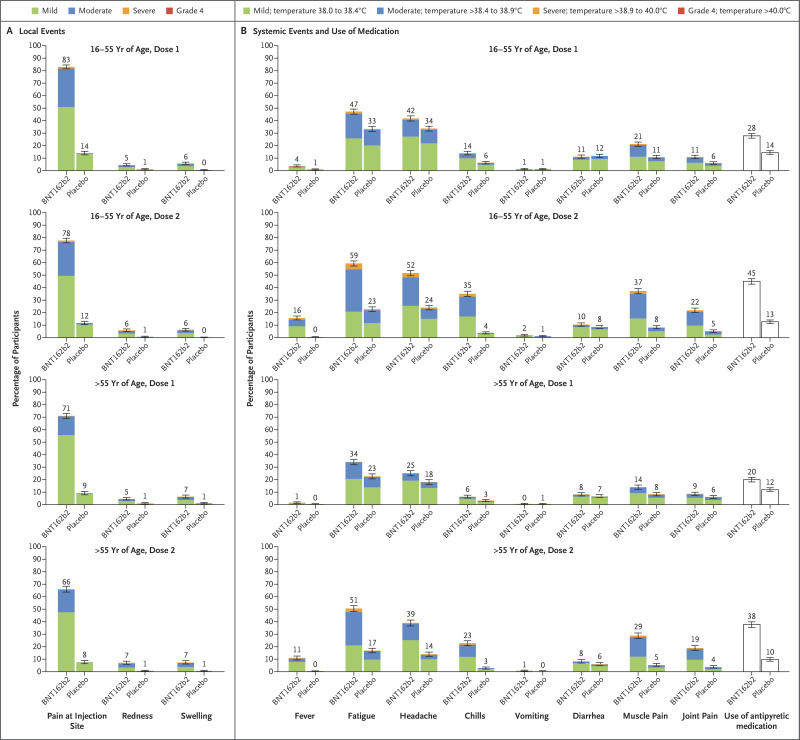
Figure 2. Local and Systemic Reactions Reported within 7 Days after Injection of BNT162b2 or Placebo, According to Age Group.
Data on local and systemic reactions and use of medication were collected with electronic diaries from participants in the reactogenicity subset (8,183 participants) for 7 days after each vaccination. Solicited injection-site (local) reactions are shown in Panel A. Pain at the injection site was assessed according to the following scale: mild, does not interfere with activity; moderate, interferes with activity; severe, prevents daily activity; and grade 4, emergency department visit or hospitalization. Redness and swelling were measured according to the following scale: mild, 2.0 to 5.0 cm in diameter; moderate, >5.0 to 10.0 cm in diameter; severe, >10.0 cm in diameter; and grade 4, necrosis or exfoliative dermatitis (for redness) and necrosis (for swelling). Systemic events and medication use are shown in Panel B. Fever categories are designated in the key; medication use was not graded. Additional scales were as follows: fatigue, headache, chills, new or worsened muscle pain, new or worsened joint pain (mild: does not interfere with activity; moderate: some interference with activity; or severe: prevents daily activity), vomiting (mild: 1 to 2 times in 24 hours; moderate: >2 times in 24 hours; or severe: requires intravenous hydration), and diarrhea (mild: 2 to 3 loose stools in 24 hours; moderate: 4 to 5 loose stools in 24 hours; or severe: 6 or more loose stools in 24 hours); grade 4 for all events indicated an emergency department visit or hospitalization. 𝙸 bars represent 95% confidence intervals, and numbers above the 𝙸 bars are the percentage of participants who reported the specified reaction.