Extended Data Figure 7: Variance in the pre-helix αG loops of KSR and RAF proteins determines selectivity for trametinib.
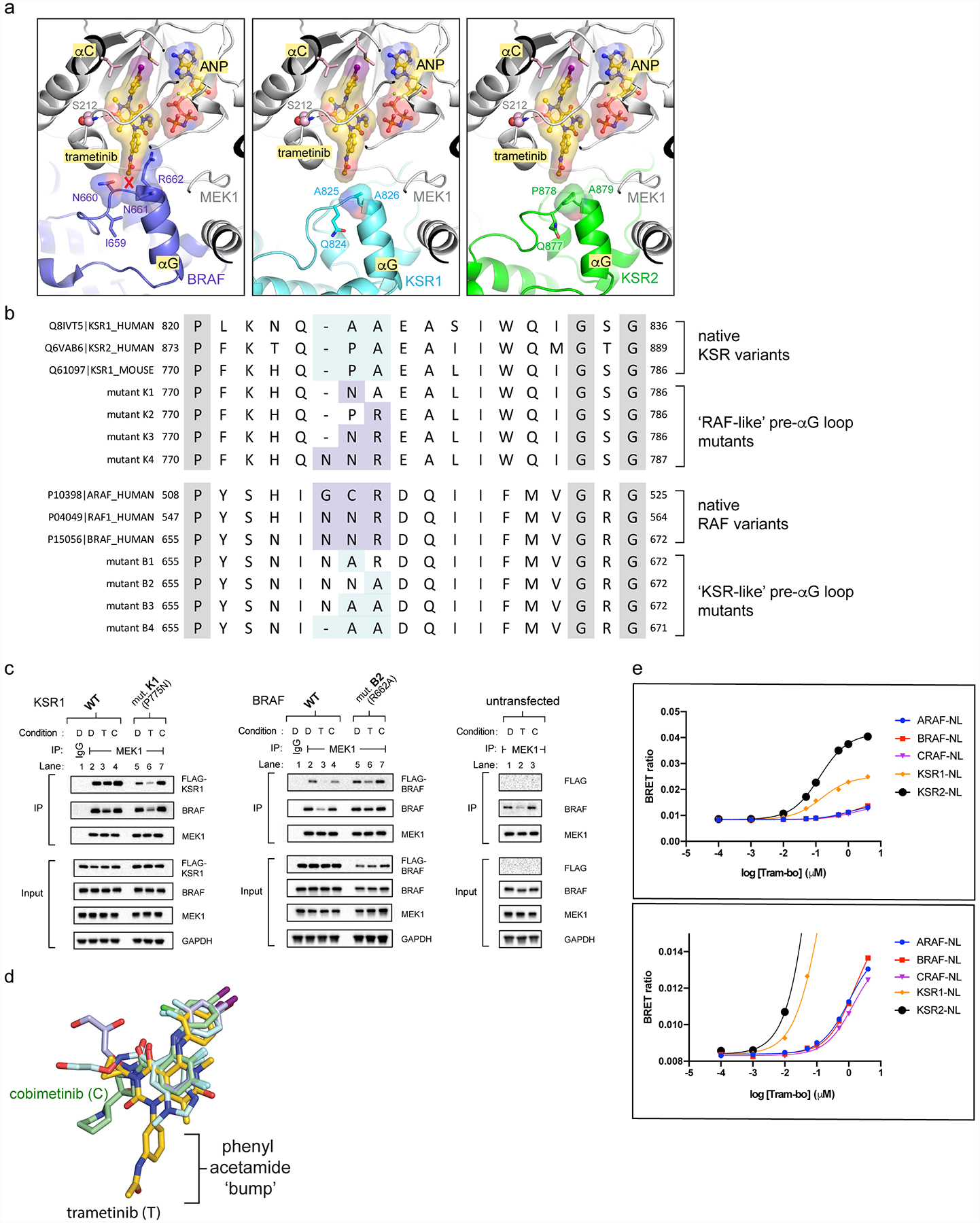
A. The pre-helix αG loop in BRAF (left; N660-N661-R662) includes an insertion and larger amino acid side chains compared to KSR1 (middle; GAP-A825-A826) and KSR2 (right; GAP-P878-A879), creating a clash with trametinib.
B. Sequence alignment highlighting conserved variations between RAF kinases and KSR pseudokinases at the trametinib-binding site. Native sequences and mutants in mouse KSR1 and human BRAF used for functional studies in main Figure 3C,D are listed. Mouse KSR1 mutants include K1 (KSR1_P775N), K2 (KSR1_A776R), K3 (KSR1_P775N/A776R), and K4 (KSR1_insertionN/P775N/A776R). Human BRAF mutants include B1 (BRAF_N661A), B2 (BRAF_R662A), B3 (BRAF_N661A/R662A), and B4 (BRAF_N660deletion/N661A/R662A).
C. IP/WB of endogenous MEK1 from lysates of HCT116 cells transfected with (left) wild-type KSR1 and mutant K1 (P775N, mouse KSR1 numbering); (middle) wild-type BRAF and mutant B2 (R662A); (right) untransfected controls. Cells were treated with DMSO (D), 200 nM trametinib (T), or 200 nM cobimetinib (C) for 1 hour prior to harvesting cells. IgG was used as a control for non-specific binding of proteins during IPs. Transfected KSR1 or BRAF were detected using an anti-FLAG antibody. All other western blot signals were detected using specific antibodies against endogenous proteins. Blots are representative of three independent experiments. We conducted side-by-side analysis of cobimetinib as a control compound that does not generate direct interfacial contacts like trametinib but displays a similar IC50 on the KSR:MEK complex. Note; compare the effects of cobimetinib addition on complex stability to the effects of trametinib in Figure 3C,D. Unlike trametinib, cobimetinib does not impact the KSR1 or BRAF mutants in terms of pulldown through endogenous MEK similar to trametinib. This data supports that the ‘bump-and-hole’ model for trametinib selectivity between KSR-bound MEK and RAF-bound MEK. Further note from Figure 3C,D that all of the tested KSR1 alleles, and also the full swaps of the pre-helix αG loops between RAF and KSR proteins, resulted in partial or complete loss of pulldown via MEK (Figure 3C,D; lanes 2 vs 10 for mutants K4 and B4), which suggests that the length and composition of interfacial residues within both KSR and RAF proteins are critical and unique determinants of binding towards MEK.
D. Overlay of four clinical MEKi highlights the phenyl acetamide group of trametinib as a unique ‘bump’ not found in the other compounds including cobimetinib.
E. BRET buildup curves with increasing concentrations of tram-bo on the indicated luciferase-tagged versions of human KSR1, KSR2, ARAF, BRAF, and CRAF/RAF1. KSR1-luc and KSR2-luc both show higher BRET ratios, and also ~10-fold tighter binding, with tram-bo relative to ARAF-luc, BRAF-luc, and CRAF-luc. Lower inset is a y-axis magnification of the top inset. Data points represent the average of two technical replicates; experiments were conducted at least three independent times with similar results.
