Abstract
Objectives
To estimate the effectiveness of endoscopic screening programme in reducing incidence and mortality of upper gastrointestinal cancer in high risks areas of China.
Design
This multicentre population-based cohort study was conducted in six areas in China from 2005 to 2015. All permanent residents aged 40 to 69 years were identified as target subjects. We refer to those who were invited for screening collectively as the invited group. Of these, we classify those who were invited and undertook endoscopic screening as the screened group and those who were invited but did not accept screening as the non-screened group. Target subjects who were not invited to the screening were assigned to the control group. The effectiveness of the endoscopic screening and screening programme were evaluated by comparing reductions in incidence and mortality from upper gastrointestinal cancer in the screened and invited group with control group.
Results
Our cohort analysis included 637 500 people: 299 483 in the control group and 338 017 in the invited to screening group, 113 340 (33.53%) of whom were screened eventually. Compared with subjects in the control group, upper gastrointestinal cancer incidence and mortality decreased by 23% (relative risk (RR)=0.77, 95% CI 0.74 to 0.81) and 57% (RR=0.43, 95% CI 0.40 to 0.47) in the screened group, respectively, and by 14% (RR=0.86, 95% CI 0.84 to 0.89) and 31% (RR=0.69, 95% CI 0.66 to 0.72) in the invited group, respectively.
Conclusion
Among individuals aged 40 to 69 years in high risk areas of upper gastrointestinal cancer, one-time endoscopic screening programme was associated with a significant decrease in upper gastrointestinal cancer incidence and mortality.
Keywords: oesophageal cancer, gastric cancer, endoscopy, screening
Significance of this study.
What is already known on this subject?
Upper gastrointestinal cancer is one of the most common cancers worldwide and half of all new cases occurred in China.
Developed countries such as Japan and South Korea have launched national screening programmes for gastric cancer, proving that endoscopies are a cost-effective screening method for gastric cancer.
Population-based endoscopic screening has been adopted as one of the national screening programmes in some high risk areas in China since 2005, but previous research to evaluate the screening effectiveness for oesophageal cancer was based predominately on small-sample sized, single-centre studies. There are no multicentre studies evaluating the effectiveness of the endoscopic screening programme for upper gastrointestinal cancer, including oesophageal and gastric cancer.
What are the new findings?
One-time endoscopic screening programme is effective in the prevention of upper gastrointestinal cancer in individuals aged 40 to 69 years in high risk areas in China.
Endoscopic screening is effective for all types of upper gastrointestinal cancers, and the effectiveness varies among oesophageal cancer, non-cardia gastric cancer and cardia gastric cancer.
Significance of this study.
How might it impact on clinical practice in the foreseeable future?
Our findings provide evidence for the optimisation and implementation of an endoscopic screening programme for upper gastrointestinal cancer in China.
The screening programme in high risk areas in China will provide practical evidence for other developing countries with a heavy burden of upper gastrointestinal cancer.
Introduction
Upper gastrointestinal cancer including oesophageal cancer and gastric cancer is the third most common cancer worldwide and three quarters of all new cases occurred in developing countries.1 In China, although population-based studies have shown an improvement in the overall 5-year survival rate for oesophageal and gastric cancer from 20.9% and 27.4% to 30.3% and 35.1%, respectively, in the past decade,2 they remain the second most common cancer and the fifth leading cause of years of life lost.3 4
Prognosis of upper gastrointestinal cancer depends largely on disease stage at diagnosis. The survival rate is less than 10% when diagnosed at an advanced stage but is as high as 85% if detected at an earlier stage.5 Endoscopic screening can potentially prevent upper gastrointestinal cancers by early diagnosis and early treatment and has been widely adopted in screening programmes. Developed countries such as Japan and South Korea have launched nationwide endoscopic screening programmes,6 7 while developing countries such as China and Iran conduct endoscopic screening only in high risk areas due to a larger cancer burden, capabilities of local doctors and availability of technology.8–11 The current evidence from large population studies has confirmed that endoscopic screening is a cost-effective screening method for gastric cancer,12 while the evidence for the effectiveness of oesophageal cancer screening are based predominately on small-sample sized, single-centre observational studies.8 9 Furthermore, two ongoing randomised controlled trials are limited to intermediate outcomes due to insufficient follow-up time.13 14
In China, since the launch of National Key Public Health Projects in 2005, endoscopic screenings for upper gastrointestinal cancer have been performed in more than 110 high risk areas throughout the country. However, there are no multicentre studies evaluating the effectiveness of the endoscopic screening programme for upper gastrointestinal cancer.
Therefore, we undertook a population-based, multicentre cohort study in high risk areas with upper gastrointestinal cancer in China where endoscopic screening has been carried out for more than 10 years. We investigated incidence and mortality to evaluate the effectiveness of endoscopic screening for both oesophageal cancer and gastric cancers.
Methods
Study design and participants
This was a multicentre population-based cohort study based on the screening programme in high risk areas of upper gastrointestinal cancer in China. Six centres (see online supplementary figure S1) were selected based on the following criteria: (1) pilot areas that had started upper gastrointestinal cancer screenings in 2005 or 2006, (2) diverse economical-geographical regions in China, (3) having cancer registration data continuously submitted and included in Cancer Incidence in Five Continents (CI5) or China Cancer Registry Annual Report, (4) little migration and relatively stable.
gutjnl-2019-320200supp001.pdf (923.5KB, pdf)
We used the household registration system at local police stations to identify all permanent residents aged between 40 and 69 years in selected centres (covering the entire county or city) as the target population.
Exposure and measurement
The screening programme used a village as a unit of each centre. Villages with a heavier burden of upper gastrointestinal cancer are chosen for screening. The target subjects were informed through various forms such as broadcasting and brochures during enrolment. According to the name list provide by police stations, the village doctors ensured that the target subjects were notified by telephone or by visiting. Those who had no alarm symptoms and history of cancer, were mentally and physically competent were enrolled an endoscopic examination after singing an informed consent.
All endoscopic examinations and therapies were conducted by well-trained doctors at local hospitals according to the guidelines for cancer screening and early diagnosis and treatment in China.15 In brief, after completion of the informed consent process, participants were provided premedication with 100 mL of warm water containing 80 mg of simethicone and/or 20 000 units of pronase and 1 g of sodium bicarbonate at 40°C before the examination. A local anaesthetic (5 mL of 1% lidocaine by mouth for 5 min) was used for sedation. Participants were placed in the left lateral position, and the entire oesophagus and stomach were visually examined including careful examination of the lesser curvature of the cardia with retroflexion. Lugol’s iodine (1.2%) staining was used to stain the full length of the oesophagus. Indigo carmine (0.2%) staining was used for auxiliary observation of the stomach if suspicious lesions were found. Biopsies were taken from all unstained focal lesions for oesophagus and any suspicious lesions in cardia and stomach. Biopsy specimens were fixed in 10% buffered formalin, embedded in paraffin, cut in 5 µm sections and stained with haematoxylin and eosin. Two pathologists independently read the biopsy slides and any disagreements were resolved through discussion with a third pathologist.
When early lesions were histologically diagnosed, participants were recalled to the clinic to receive appropriate treatments according to the lesions’ severity. For low grade intraepithelial neoplasia, endoscopic follow-up was required in 3 to 5 years. For high grade intraepithelial neoplasia or intramucosal carcinomas, endoscopic mucosal resection and/or endoscopic submucosal dissection treatments were used. For submucosal cancers and advanced oesophageal cancers, therapies included radiotherapy, and other conventional treatments. Re-examination was conducted every 6 months in the first 3 years after the first therapy.
A uniform questionnaire was administered by trained interviewers for all participants who underwent endoscopic examination to collect exposure information. Of the non-screened group, 20% of participants from the same village as those in the screened group were invited to complete the same questionnaire.
Outcomes
We matched all cohort populations with the cancer registration database and death surveillance database to obtain information on cancer incidence and all-cause mortality, and cross-referenced to medical insurance databases and medical records from local hospitals. All participants were followed for events that occurred through 31 December 2015.
The primary outcomes in this study were incidence and mortality caused by upper gastrointestinal cancer, consisting of oesophageal cancer, non-cardia gastric cancer and cardia gastric cancer. The secondary outcomes were incidence and mortality as a result of all-site cancer and all-cause mortality. International Classification of Diseases (10th revision) codes were used throughout, in which oesophageal cancer included codes C15, non-cardia gastric cancer included codes C16.1 to C16.9 and cardia gastric cancer included codes C16.0.
Statistical analyses
The cut-off for enrolment for this analysis was 31 December 2012 to ensure at least 3 years of follow-up although they were continually recruited to date. We refer to those who were invited for screening collectively as the invited to screening group. Of these, we classify those who were invited and undertook endoscopic screening as the screened group and those who were invited but did not accept screening as the non-screened group (hereby referred to as the invited, screened and non-screened groups, respectively). Those in the same region as the screened and non-screened groups that were not invited for screening were in the control group. The first primary cancer diagnosis was used for cases with multiple cancers. Time to disease occurrence or death was calculated from cohort entry date. In the screened group, cohort entry date was defined as date of screening. Similarly, cohort entry date of the non-screened group was estimated based on screening date of those enrolled in the same village. In the control group, since there was no invitation and therefore no specific date for enrolment, the date of study entry was defined as the starting date in each centre. All time-to-event data were censored by end of follow-up or death.
Rates are presented as average incidence rates per 100 000 person-years. Relative risk (RRs) were used to estimate the effectiveness of endoscopic screening (having endoscopic examination) or screening programme (being invited to screening) by comparing the screened group or the invited to screening group with the control group.16 Age is determined by the age at cohort entry. Both rates and relative risks were standardised by age, sex and centre. Risk factors were compared between screened and non-screened groups based on the survey data. The number needed to screen (NNS) to prevent one incident case or one death due to each cancer type were calculated as the inverse of the absolute risk difference in cumulative event proportions between two study groups.17 The Nelson-Aalen method was used for the calculation of cumulative hazard. Subgroup analyses were done by sex and age group (40 to 49 years, 50 to 59 years and 60 to 69 years) to compare screening effectiveness of each type of upper gastrointestinal cancer. A sensitivity analysis was also performed by excluding one centre each time and recalculating the relative risks for upper gastrointestinal cancer. In addition, we compared all outcomes between screened and non-screened groups in a propensity-matched cohort analysis based on factors including area, sex, age at entry and cohort entry year.18 All statistical analyses were completed using Stata 14. A significance level of 0.05 and two-sided tests were used throughout.
Patient and public involvement
Patients were not involved in research design or the outcome measures. No patients were asked to advise on interpretation or writing up of results. Our findings will be disseminated through providing scientific evidence for optimisation and updating of screening programmes.
Results
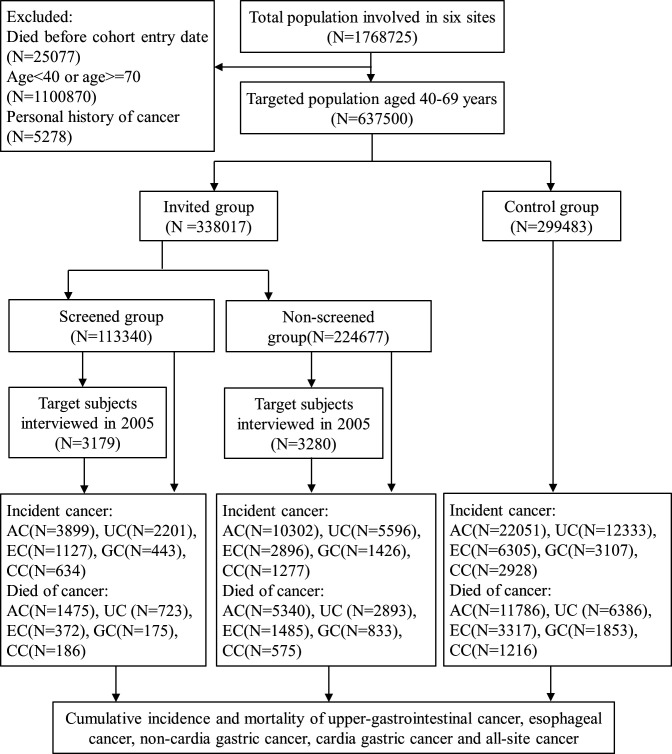
A total of 637 500 subjects aged 40 to 69 were identified from a population of 1 768 725 and were included in the final analysis (figure 1). There were 338 017 participants invited to screening, with 113 340 (33.5% compliance) who underwent the endoscopic examination and interview. The control group consisted of 299 483 individuals who were not invited for screening. Table 1 summarises the characteristics of the subjects according to the groups. The median follow-up time for all those invited for screening was 5.4 years due to the long length of enrolment, while those in the control group had a median follow-up time of 10 years.
Figure 1.
Cohort profile. AC;all-site cancer; CC; cardiagastric cancer; EC, oesophagealcancer; GC; non-cardiagastric cancer; UC, uppergastrointestinal cancer.
Table 1.
Baseline characteristics of study population
| Characteristic | Invited to screening group | Control group (n=299 483) | ||
| Screened (n=113 340) | Not screened (n=224 677) | Total (n=338 017) | ||
| Sex (%) | ||||
| Men | 51 080 (45.07) | 115 879 (51.58) | 166 959 (49.39) | 148 182 (49.48) |
| Women | 62 260 (54.93) | 108 798 (48.42) | 171 058 (50.61) | 151 301 (50.52) |
| Age at entry (%) | ||||
| 40–49 year | 45 636 (40.26) | 103 947 (46.27) | 149 583 (44.25) | 135 364 (45.20) |
| 50–59 year | 43 756 (38.61) | 75 252 (33.49) | 119 008 (35.21) | 112 468 (37.55) |
| 60–69 year | 23 948 (21.13) | 45 478 (20.24) | 69 426 (20.54) | 51 651 (17.25) |
| Area (%) | ||||
| Cixian | 22 135 (19.53) | 49 788 (22.16) | 71 923 (21.28) | 19 384 (6.47) |
| Feicheng | 23 470 (20.71) | 18 718 (8.33) | 42 188 (12.48) | 58 106 (19.40) |
| Linzhou | 19 805 (17.47) | 38 333 (17.06) | 58 138 (17.2) | 35 757 (11.94) |
| Yancheng | 12 460 (10.99) | 41 081 (18.28) | 53 541 (15.84) | 66 525 (22.21) |
| Yanting | 22 702 (20.03) | 43 500 (19.36) | 66 202 (19.59) | 33 325 (11.13) |
| Yangzhong | 12 768 (11.27) | 33 257 (14.80) | 46 025 (13.62) | 86 386 (28.85) |
| Person-years of follow-up* | 657 349 | 1 394 626 | 2 051 975 | 2 915 581 |
*Person-years are follow-up from cohort entry date to the date of last follow-up based on the analyses for upper gastrointestinal cancer incidence.
A total of 12 548 patients with 13 299 precancerous lesions, collectively, were diagnosed by endoscopy and pathological biopsy in the baseline screening. The number of lesions by anatomical site and centre are listed in online supplementary table S1. Among the 848 cancer cases diagnosed by screening, oesophageal squamous cell carcinoma accounted for 99.3% of oesophageal cancer (408/411) and adenocarcinoma accounted for 92.0% of gastric cancer (402/437). A total of 1191 patients received endoscopic mucosal resection for early cancer and precancerous lesions. Table 2 shows incidence and mortality for each cancer type by study cancer. During screening and follow-up, upper gastrointestinal cancer was diagnosed in 7797 individuals in the invited group and 12 333 in the control group, and 3626 individuals in the invited group versus 6386 individuals in the control group died from upper gastrointestinal cancer. The standardised incidence and mortality rates were 328 and 109 per 100 000 person-years in the screened group, respectively, and 373 and 168 per 100 000 person-years in the invited to screening group, respectively, compared with 430 and 238 per 100 000 person-years in the control group, respectively. The overall and truncated (40 to 69 years) age-standardised incidence and mortality by upper gastrointestinal sites and by centre were listed in online supplementary table S2.
Table 2.
Relative risks of incidence and mortality for each cancer type according to groups and the number needed to screen to prevent one event in the present follow-up period
| Characteristic | Invited to screening group (n=338 017) | Control group (n=299 483) | Screened versus control group | Invited to screening group versus control group | Number needed to screen (95% CI)‡ | ||||||
| Screened (n=113 340) | Not screened (n=224 677) | ||||||||||
| Cases | Rate (95% CI)* | Cases | Rate (95% CI)* | Cases | Rate (95% CI)* | Relative risk (95% CI)† | P value | Relative risk (95% CI)† | P value | ||
| Incidence | |||||||||||
| Upper gastrointestinal cancer¶ | 2201 | 327.54 (312.14 to 342.94) | 5596 | 394.48 (383.31 to 405.66) | 12 333 | 429.92 (421.97 to 437.86) | 0.77 (0.74 to 0.81) | <0.001 | 0.86 (0.84 to 0.89) | <0.001 | 98 (85 to 115) |
| Oesophageal cancer | 1127 | 160.60 (150.09 to 171.11) | 2896 | 202.60 (194.55 to 210.66) | 6305 | 219.98 (214.28 to 225.69) | 0.74 (0.69 to 0.79) | <0.001 | 0.85 (0.82 to 0.89) | <0.001 | 168 (143 to 206) |
| Non-cardia gastric cancer | 443 | 68.51 (61.23 to 75.79) | 1426 | 100.61 (94.99 to 106.23) | 3107 | 103.89 (100.09 to 107.69) | 0.66 (0.59 to 0.73) | <0.001 | 0.88 (0.83 to 0.94) | <0.001 | 283 (235 to 359) |
| Cardia gastric cancer | 634 | 96.29 (87.86 to 104.72) | 1277 | 88.48 (83.33 to 93.63) | 2928 | 100.39 (96.59 to 104.19) | 0.99 (0.90 to 1.09) | 0.213 | 0.90 (0.84 to 0.96) | 0.008 | NA§ |
| All-site cancer | 3899 | 572.95 (552.76 to 593.15) | 10 302 | 757.34 (741.36 to 773.31) | 22 051 | 777.46 (766.7 to 788.23) | 0.75 (0.73 to 0.78) | <0.001 | 0.89 (0.86 to 0.91) | <0.001 | 49 (45 to 54) |
| Mortality | |||||||||||
| Upper gastrointestinal cancer | 733 | 108.98 (99.99 to 117.96) | 2893 | 199.10 (191.21 to 206.99) | 6386 | 238.22 (232.06 to 244.38) | 0.43 (0.40 to 0.47) | <0.001 | 0.69 (0.66 to 0.72) | <0.001 | 77 (72 to 84) |
| Oesophageal cancer | 372 | 52.69 (46.64 to 58.74) | 1485 | 101.76 (96.10 to 107.42) | 3317 | 125.07 (120.57 to 129.58) | 0.40 (0.36 to 0.45) | <0.001 | 0.67 (0.63 to 0.71) | <0.001 | 138 (127 to 153) |
| Non-cardia gastric cancer | 175 | 27.50 (22.79 to 32.21) | 833 | 58.34 (54.03 to 62.65) | 1853 | 66.66 (63.49 to 69.83) | 0.38 (0.33 to 0.45) | <0.001 | 0.72 (0.66 to 0.78) | <0.001 | 255 (227 to 295) |
| Cardia gastric cancer | 186 | 28.79 (24.11 to 33.46) | 575 | 39.00 (35.60 to 42.40) | 1216 | 46.48 (43.73 to 49.23) | 0.58 (0.49 to 0.68) | <0.001 | 0.72 (0.65 to 0.80) | <0.001 | 565 (447 to 795) |
| All-site cancer | 1475 | 217.46 (204.85 to 230.06) | 5340 | 381.57 (370.37 to 392.78) | 11 786 | 433.91 (425.64 to 442.19) | 0.48 (0.45 to 0.50) | <0.001 | 0.72 (0.70 to 0.75) | <0.001 | 46 (44 to 49) |
| All cause | 3705 | 565.21 (547.30 to 583.70) | 13 713 | 1001.79 (985.16 to 1018.70) | 27 646 | 970.23 (958.86 to 981.74) | 0.49 (0.47 to 0.51) | <0.001 | 0.75 (0.74 to 0.77) | <0.001 | 25 (23 to 26) |
*Rates are per 100 000 person-years and standardised by age, sex and centre.
†Relative risks are standardised by age, sex and centre.
‡Number needed to screen to prevent one event in the control group.
§Not available.
¶Thirteen patients had both a oesophageal cancer and gastric cancer.
To evaluate the effectiveness of endoscopic screening, incidence and mortality of upper gastrointestinal cancer decreased by 23% (RR 0.77, 95% CI 0.74 to 0.81) and 57% (RR 0.43, 95% CI 0.40 to 0.47), respectively, in those having endoscopic examination. Significant reductions were also observed in incidence and mortality for oesophageal cancer, non-cardia gastric cancer, all-site cancer and in all-cause mortality. No significant effect on cardia gastric cancer incidence was noted, however reduction in mortality remained significant.
To evaluate the effectiveness of the screening programme, upper gastrointestinal cancer incidence and mortality were reduced by 14% (RR 0.86, 95% CI 0.84 to 0.89) and 31% (RR 0.69, 95% CI 0.66 to 0.72), respectively, in those invited to screening. Significant reductions were observed in incidence and mortality for all types of upper gastrointestinal cancer and all-site cancer, as well as in all-cause mortality.
The estimated NNS to prevent a single upper gastrointestinal cancer diagnosis and death over the study period was 98 (95% CI 85 to 115) and 77 (95% CI 72 to 84) (table 2), respectively.
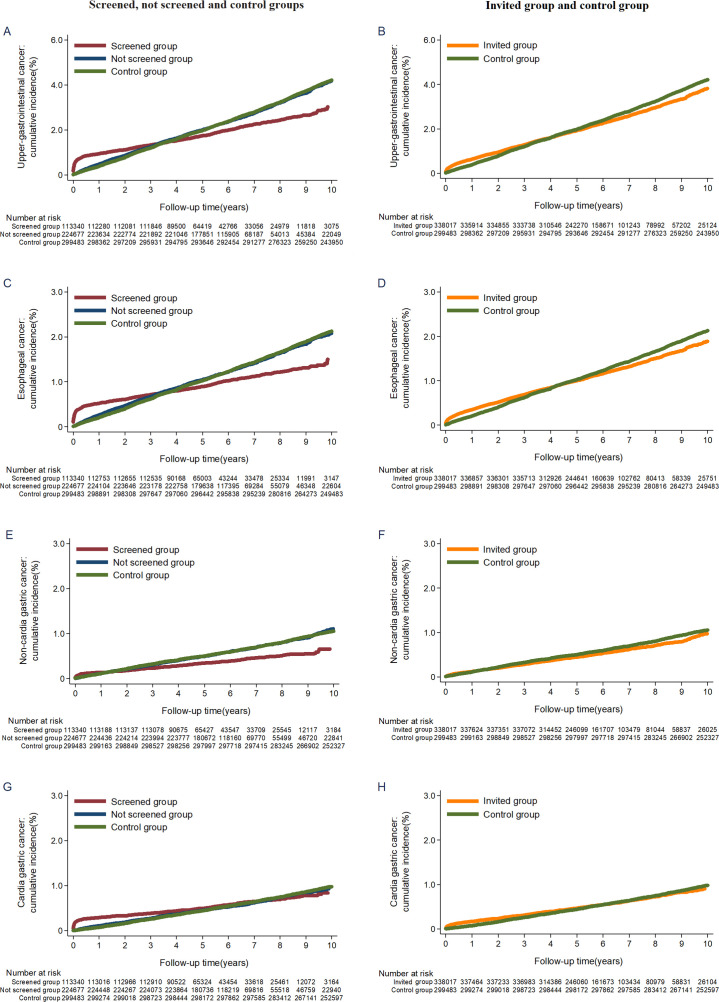
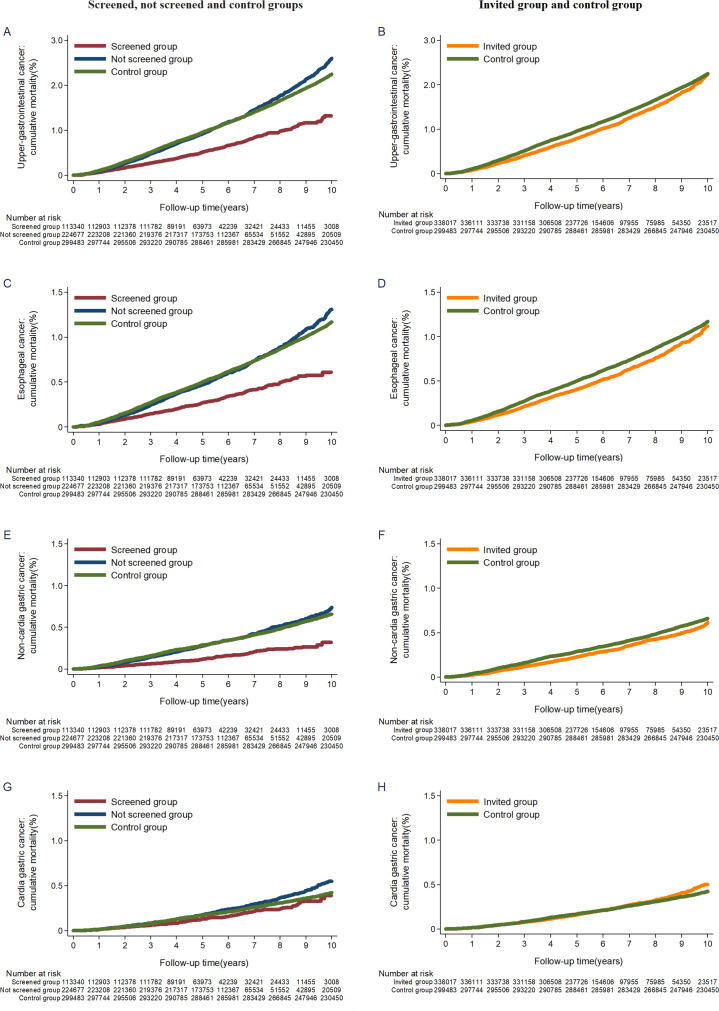
The cumulative incidence rate for upper gastrointestinal cancer was higher in the screened group during the first 3 to 4 years, after which the curves began to diverge (figure 2A). A reduction in mortality in the screened group was apparent after the first year of follow-up (figure 3A). The trends remained similar between the invited to screening group and the control group (figures 2B and 3B). There were similar patterns for incidence and mortality of oesophageal cancer, non-cardia gastric cancer and all-site cancer, except for cardia gastric cancer (figures 2C–2H and 3C–3H and online supplementary figure S2).
Figure 2.
Cumulative incidence of upper gastrointestinal cancer, oesophageal cancer, non-cardia gastric cancer and cardia gastric cancer.
Figure 3.
Cumulative mortality from upper gastrointestinal cancer, oesophageal cancer, non-cardia gastric cancer and cardia gastric cancer.
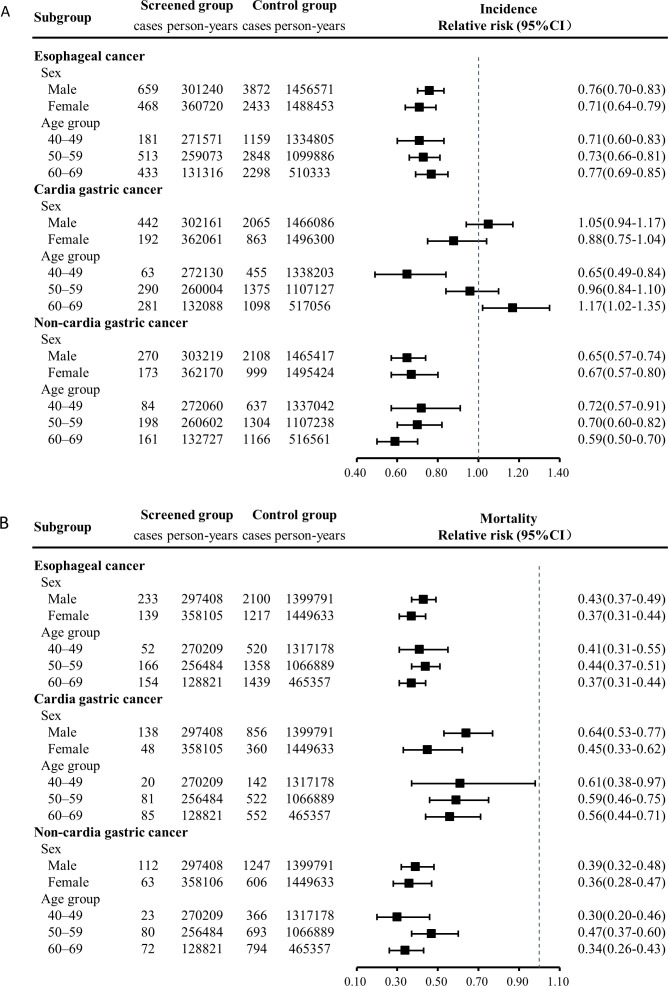
In subgroup analyses for age and sex, significant reductions were observed in incidence and mortality of oesophageal cancer and non-cardia gastric cancer, as well as the mortality of cardia gastric cancer (figure 4). Women had greater reductions in mortality than men for all types of upper gastrointestinal cancer; the relative risk of upper gastrointestinal cancer was 0.40 (95% CI 0.33 to 0.49) for women versus 0.50 (95% CI 0.43 to 0.57) for men. Mortality relative risks differed by age group across cancer sites. The relative risk of upper gastrointestinal cancer was 0.39 (95% CI 0.29 to 0.54), 0.53 (95% CI 0.44 to 0.62) and 0.43 (95% CI 0.36 to 0.51) for age groups 40 to 49, 50 to 59 and 60 to 69 years, respectively.
Figure 4.
Relative risks for different cancer type in screened versus control groups by age group and sex.
In the sensitivity analysis, significant reductions were observed for upper gastrointestinal cancer in the screened group after excluding one centre at a time and ranged from 18% to 28% and from 52% to 60% decreased risk for incidence and mortality, respectively (see online supplementary table S3). The results of propensity score matched analysis were consistent with those of our primary analysis (see online supplementary tables S4 and S5, figures S3 and fig S4).
Discussion
Findings from this multiple-centre population-based study have shown that one-time endoscopic screening programme was associated with a significant decrease in upper gastrointestinal cancer incidence and mortality. After up to 10 years of follow-up, upper gastrointestinal cancer incidence and mortality were reduced by 23% and 57%, respectively, in those screened and were reduced by 14% and 31%, respectively, in those invited to screen.
Results in relation to other studies
There is limited evidence from randomised controlled trial on the effectiveness of endoscopic screening. Only one study by Liu et al reported reductions in cumulative incidence and mortality from upper gastrointestinal cancer, in which the standardised incidence ratio was 0.57 (95% CI 0.38 to 0.86) and the standardised mortality ratio was 0.47 (95% CI 0.25 to 0.88).9 However, the results were largely limited by few events (23 incident cases and 10 deaths) in the screened group, while the control group was estimated through calculations.5
Evidence from observational studies shows inconsistent results in gastric cancer screening. Zhang et al’s meta-analysis included four cohort and six nested-control studies comprising 342 013 people from Japan, Korea and China.19 The combined results indicated a 40% reduction in gastric cancer mortality (RR 0.60, 95% CI 0.49 to 0.73), while no significant reduction was observed (RR 1.14, 95% CI 0.93 to 1.40) in incidence. Although factors such as gender and age were adjusted for, there was still considerable heterogeneity among studies (heterogeneity test I2=0.667, p<0.05). For example, Jun et al evaluated the effectiveness of the Korean National Cancer Screening Program in reducing gastric cancer mortality and found that the overall OR of mortality from gastric cancer among ever-screened subjects was 0.79 (95% CI, 0.77 to 0.81) compared with subjects who had never been screened.20 However, Hamashima et al investigated mortality from gastric cancer based on a large cohort in Japan and found a standardised mortality risk rate of 0.43 (95% CI 0.30 to 0.57) for the endoscopic screening group.21 In this study, we divided the gastric cancer into two types according to the lesion site considering the particularity of cardia cancer. Compared with the control group, mortality of both cardia and non-cardia gastric cancers in the screening group was significantly reduced, with RRs of 0.58 (95% CI 0.49 to 0.68) and 0.38 (95% CI 0.33 to 0.45), respectively. The results seem better than those of the screening cohort in South Korea and similar to those of the Japanese cohort. Moreover, differences in the effectiveness of screening were observed between the non-cardia and cardia gastric cancers, suggesting that the type of gastric cancer may be a potential explanation to the heterogeneity found in previous findings.
The most supportive evidence with respect to oesophageal cancer came from our study in Cixian.8 In that community assignment controlled endoscopic screening trial, significant reductions were found in both incidence and mortality from oesophageal cancer through 10 years of follow-up, with a HR of 0.61 (95% CI 0.42 to 0.89) and 0.45 (95% CI 0.54 to 0.95), respectively. The present study confirmed and improved our previous findings with a multicentre design and large sample of 0.64 million.
Possible explanations of findings
The effectiveness of endoscopic screening in reducing mortality related to upper gastrointestinal cancer may be due to the detection of precancerous lesions. It is well known that early-stage cancers including precancerous lesions accounted for the majority of screening-detected cancers, and if treated timely, they are less likely to progress than late-stage cancers. Therefore, long-term reduction in upper gastrointestinal cancer mortality that did not diminish substantially over time is consistent with the progression of upper gastrointestinal cancer and provides support for the sustained effect of early treatment. In addition, the reduction in cumulative incidence is most likely attributable to dominance of screen-detected prevalent cancers in the first 3 years of follow-up. After this point, the curves of the screened group and the control group began to deviate and a reduction in incidence becomes apparent. This finding is similar to that in studies of flexible sigmoidoscopy for colorectal cancer.22 23
This study is based on the existing screening cohort in high risk areas and it is aimed at evaluating the effectiveness of screening in a real-world situation. Unlike previous cohort studies using never screened or estimated cases as controls,9 21 24–26 we introduced a group of never invited subjects as the control group who were from remaining villages in each centre. This population-wide control significantly reduced the selection bias caused by non-random selection of controls. The invited to screening group and the control group have similar demographics and socioeconomic status, allowing for comparison of incidence and mortality. Therefore, we can evaluate the effectiveness of screening programme by comparing the outcomes between the invited group and the control group irrespective of compliance.
The effectiveness of endoscopic screening is diluted by non-compliance, resulting in a less effective of screening programme. The non-compliance rate in this study was 66.6%, but compared with endoscopic screening, the effectiveness of the screening programme was only reduced by 39% in incidence and 45% in mortality. In fact, the related cancer incidence and mortality in the non-screened group were slightly lower than that of the control group. The results of comparison of baseline risk factors showed that participants in the screened group had more frequently reported risk factors for developing upper gastrointestinal cancer, including poor drinking water, lower household income and a family history of cancer (see online supplementary table S6). Thus, it can be speculated that, screening directly reduced the incidence and mortality of screeners. Conversely, the screening programme was related to health education and other factors that indirectly reduced population risk of the non-screened group by encouraging high-risk individuals to participate in screening, which contributed to further reduction in incidence and mortality.
No significant effect on cardia gastric cancer incidence was observed in the analysis. Cardia cancer is difficult to detect because of its special anatomical location. Although the lesser curvature of the cardia was carefully examined with retroflexion during endoscopy, it can be easily be misdiagnosed or passed over, which reduces the possibility of early detection. Moreover, lesions of the cardia bleed easily during treatment, which increases difficulty of treatment. As a result, screening may not detect enough prevalent cardia cancers, and treatment of precancerous lesions in cardia may be less effective compared with those in the oesophagus and stomach. However, screening programmes are still effective for the incidence of cardia cancer due to indirect effects.
Strengths and limitations of this study
This is the first population-based cohort study to evaluate the effectiveness of one-time endoscopic screening on the incidence and mortality from upper gastrointestinal cancer through a long-term follow-up. Various data sources were used for identification of target population and ascertainment of cancer incidence and mortality. Sensitivity analysis showed that there was no significant difference in overall incidence and mortality in all participating study centres, and the results were generally similar in the propensity score matched cohort analysis. By introducing the control group, we evaluated the effectiveness of endoscopic screening and screening programme for upper gastrointestinal cancer. In addition, the effectiveness of screening on oesophageal cancer, non-cardia gastric cancer and cardia gastric cancer were evaluated separately, and it varied among different cancer types, indicating that the differences in cancer types need to be considered in the implementation of screening programmes.
One limitation of the study is that the cohort was based on the existing screening cohort and the unbalanced baseline caused by selection bias was inevitable. Risk factor investigation were prospectively collected from all the screened subjects, but for those who were not screened, only the first year of enrolment was investigated due to limited resources. The control group was retrospectively collected, and there was no information on risk factors, resulting to the inability to compare baseline characteristics other than age and sex. Furthermore, the recruitment was set up to 2012, thus the subjects enrolled in late 2012 were followed for only 3 years through the last follow-up. More time is needed to observe sufficient outcomes for these participants.
Conclusions and future research
Our findings show that one-time endoscopic examination is effective in prevention of upper gastrointestinal cancer in individuals aged 40 to 69 years. The screening programme in high risk areas of rural China has provided practical evidence for other developing countries with a heavy burden of upper gastrointestinal cancer. We will continue to follow-up the cohort and further analyses are needed to determine the optimal screening interval and age, identify high risk individuals and provide evidence for precise and individualised screening.
Acknowledgments
We are grateful to Hebei, Henan, Shandong, Jiangsu, Shanxi, Sichuan Provincial Disease Prevention and Control Organizations for their support of this research. We sincerely thank Dr. Guoqing Wang, Youlin Qiao, Ning Lv, Xiufeng Cao, Quanhong Wang, Guanrui Yang, Ruihua Shi and all the members of the expert committee of the National Early Diagnosis and Early Treatment Project of Esophageal Cancer. Thanks to all cooperating centers and their staff whose hard work made this study possible. We also thank Autumn Hullings from National Cancer Institute for language editorial support.
Footnotes
RC, YL, GS, BL, DZ, ZH, XW and JL contributed equally.
Contributors: RC, YL, WW and GW designed the study. GS, BYL, DZ, ZH, XW, JL, YL, XF and LL contributed to the data collection, data transmission and data correction after quality control and checking of the results. CH, LZ, SL, JW, JZ, YZ, BL and ZD contributed to management of centres. RC did the data analysis, under supervision of WW and GW. RC, GS, BYL, DZ, ZH, XW, JL, WW, and GW drafted the paper and interpreted the results. All authors contributed to data interpretation and rewriting the paper, and reviewed and approved the final version.
Funding: This study is supported by the National Key R&D Program of China (2016YFC0901400, 2016YFC1302800).
Disclaimer: The funders had no role in the design and conduct of the study; analysis, preparation, review and approval of the manuscript.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting or dissemination plans of this research.
Patient consent for publication: Not required.
Ethics approval: The study was approved by the independent ethics committee of National Cancer Center/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College (Approval No. 16-171/1250 and 17-124/1380) and the protocol of this retrospective cohort study was registered in the Chinese Clinical Trial Registry (Registration No. ChiCTR1800017163).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request. Data from the study are available on request from corresponding author, researchers can also apply for data through the official website (http://www.ncec-china.cn/).
References
- 1. Bray F, Ferlay J, Soerjomataram I, et al. . Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018;68:394–424. 10.3322/caac.21492 [DOI] [PubMed] [Google Scholar]
- 2. Zeng H, Chen W, Zheng R, et al. . Changing cancer survival in China during 2003–15: a pooled analysis of 17 population-based cancer registries. Lancet Global Health 2018;6:e555–67. 10.1016/S2214-109X(18)30127-X [DOI] [PubMed] [Google Scholar]
- 3. Zhou M, Wang H, Zeng X, et al. . Mortality, morbidity, and risk factors in China and its provinces, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet 2019;394:1145–58. 10.1016/S0140-6736(19)30427-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Zheng RS, Sun KX, Zhang SW, et al. . Report of cancer epidemiology in China, 2015]. Zhonghua Zhong Liu Za Zhi 2019;41:19–28. [DOI] [PubMed] [Google Scholar]
- 5. Wang G-Q, Jiao G-G, Chang F-B, et al. . Long-term results of operation for 420 patients with early squamous cell esophageal carcinoma discovered by screening. Ann Thorac Surg 2004;77:1740–4. 10.1016/j.athoracsur.2003.10.098 [DOI] [PubMed] [Google Scholar]
- 6. Kim Y, Jun JK, Choi KS, et al. . Overview of the National cancer screening programme and the cancer screening status in Korea. Asian Pac J Cancer Prev 2011;12:725–30. [PubMed] [Google Scholar]
- 7. Hamashima C. Systematic review G, Guideline development group for gastric cancer screening G. Update version of the Japanese guidelines for gastric cancer screening. Jpn J Clin Oncol 2018;48:673–83. [DOI] [PubMed] [Google Scholar]
- 8. Wei W-Q, Chen Z-F, He Y-T, et al. . Long-Term follow-up of a community assignment, one-time endoscopic screening study of esophageal cancer in China. J Clin Oncol 2015;33:1951–7. 10.1200/JCO.2014.58.0423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Liu M, He Z, Guo C, et al. . Effectiveness of intensive endoscopic screening for esophageal cancer in China: a community-based study. Am J Epidemiol 2019;188:776–84. 10.1093/aje/kwy291 [DOI] [PubMed] [Google Scholar]
- 10. Mansour-Ghanaei F, Sokhanvar H, Joukar F, et al. . Endoscopic findings in a mass screening program for gastric cancer in a high risk region - Guilan province of Iran. Asian Pac J Cancer Prev 2012;13:1407–12. 10.7314/APJCP.2012.13.4.1407 [DOI] [PubMed] [Google Scholar]
- 11. Roshandel G, Khoshnia M, Sotoudeh M, et al. . Endoscopic screening for precancerous lesions of the esophagus in a high risk area in northern Iran. Arch Iran Med 2014;17:246–52. doi:014174/AIM.006 [PMC free article] [PubMed] [Google Scholar]
- 12. Cho E, Kang MH, Choi KS, et al. . Cost-effectiveness outcomes of the National gastric cancer screening program in South Korea. Asian Pac J Cancer Prev 2013;14:2533–40. 10.7314/APJCP.2013.14.5.2533 [DOI] [PubMed] [Google Scholar]
- 13. He Z, Liu Z, Liu M, et al. . Efficacy of endoscopic screening for esophageal cancer in China (ESECC): design and preliminary results of a population-based randomised controlled trial. Gut 2019;68:198–206. 10.1136/gutjnl-2017-315520 [DOI] [PubMed] [Google Scholar]
- 14. Chen W, Zeng H, Chen R, et al. . Evaluating efficacy of screening for upper gastrointestinal cancer in China: a study protocol for a randomized controlled trial. Chin J Cancer Res 2017;29:294–302. 10.21147/j.issn.1000-9604.2017.04.02 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Dong Z. Guidelines for cancer screening and early diagnosis and treatment in China. Beijing: Peking University Medical Press, 2005. [Google Scholar]
- 16. Vandenbroucke JP, Pearce N. Incidence rates in dynamic populations. Int J Epidemiol 2012;41:1472–9. 10.1093/ije/dys142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Rembold CM. Number needed to screen: development of a statistic for disease screening. BMJ 1998;317:307–12. 10.1136/bmj.317.7154.307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Lunceford JK, Davidian M. Stratification and weighting via the propensity score in estimation of causal treatment effects: a comparative study. Stat Med 2004;23:2937–60. 10.1002/sim.1903 [DOI] [PubMed] [Google Scholar]
- 19. Zhang X, Li M, Chen S, et al. . Endoscopic screening in Asian countries is associated with reduced gastric cancer mortality: a meta-analysis and systematic review. Gastroenterology 2018;155:347–54. 10.1053/j.gastro.2018.04.026 [DOI] [PubMed] [Google Scholar]
- 20. Jun JK, Choi KS, Lee H-Y, et al. . Effectiveness of the Korean National cancer screening program in reducing gastric cancer mortality. Gastroenterology 2017;152:1319–28. 10.1053/j.gastro.2017.01.029 [DOI] [PubMed] [Google Scholar]
- 21. Hamashima C, et al. Impact of endoscopic screening on mortality reduction from gastric cancer. World J Gastroenterol 2015;21:2460–6. 10.3748/wjg.v21.i8.2460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Schoen RE, Pinsky PF, Weissfeld JL, et al. . Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N Engl J Med 2012;366:2345–57. 10.1056/NEJMoa1114635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Atkin W, Wooldrage K, Parkin DM, et al. . Long term effects of once-only flexible sigmoidoscopy screening after 17 years of follow-up: the UK flexible sigmoidoscopy screening randomised controlled trial. Lancet 2017;389:1299–311. 10.1016/S0140-6736(17)30396-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Kim H, Hwang Y, Sung H, et al. . Effectiveness of gastric cancer screening on gastric cancer incidence and mortality in a community-based prospective cohort. Cancer Res Treat 2018;50:582–9. 10.4143/crt.2017.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Hosokawa O, Miyanaga T, Kaizaki Y, et al. . Decreased death from gastric cancer by endoscopic screening: association with a population-based cancer registry. Scand J Gastroenterol 2008;43:1112–5. 10.1080/00365520802085395 [DOI] [PubMed] [Google Scholar]
- 26. Riecken B, Pfeiffer R, Ma JL, et al. . No impact of repeated endoscopic screens on gastric cancer mortality in a prospectively followed Chinese population at high risk. Prev Med 2002;34:22–8. 10.1006/pmed.2001.0925 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
gutjnl-2019-320200supp001.pdf (923.5KB, pdf)