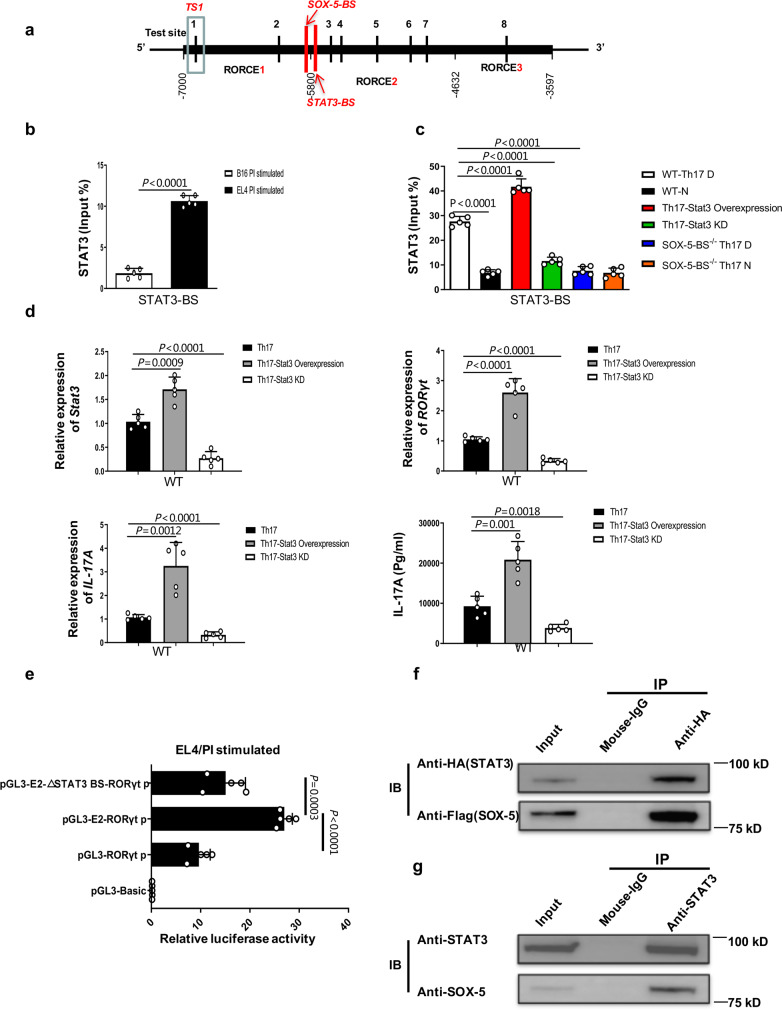
Fig. 10. STAT3 is necessary for the activation of RORCE2.
a Red vertical line shows STAT3 binding site (STAT3-BS) in RORCE2, which is located approximately 50 bp downstream of the SOX-5-BS. b, c A ChIP-qPCR assay was performed to determine the enrichment of STAT3 in its binding site in RORCE2 in EL4 cells (b), WT Th17-polarized cells before and after STAT3 overexpression or knockdown (KD) and Th17-polarized cells generated from SOX-5-BS-deficient mice (c). Th17 D represents Th17-polarized cells, and N represents naïve cells. d Relative expression of Stat3, RORγt, and IL-17A genes in WT Th17-polarized cells and IL-17A production in culture supernatants before and after Stat3 overexpression or knockdown (Stat3 KD) were determined by RT-qPCR and ELISA, respectively. e Luciferase assays were performed with different reporter constructs in EL4 cells. RORCE2 with a STAT3-BS deletion (E2-∆Stat3BS) was cloned upstream of RORγt promoter in the pGL3 vector, and transcriptional activity was assessed with a dual-luciferase system. f Hela cells were transfected with pcDNA3.1-SOX-5 Flag vector together with pcDNA3.1-STAT3 HA vector and then cultured for 48 h. After stimulated with PI and Biochanin A, whole-cell lysates from Hela cells were subjected to IP with anti-HA antibody or control mouse IgG, and IB with anti-HA or anti-Flag HRP antibody. Input proteins (input) were also IB with anti-HA or anti-Flag HRP antibody. g Whole-cell lysates from Th17 cells were subjected to IP with anti-STAT3 antibody or control mouse IgG and IB with anti-SOX-5 or anti-STAT3 antibody. Input proteins (input) were also IB with anti-SOX-5 or anti-STAT3 antibody. Mean ± SEM are shown, n = 5 independent experiments, unpaired two-tailed Student’s t-test (b–e). Experimental mice were between 8 and 12 weeks of age, with no preference to gender and were maintained on a C57BL/6 background. Source data are provided as a Source Data file.