Abstract
Purpose
To study the association between paternal age and schizophrenia in offspring.
Methods
This report describes a nationwide population-based cohort study from 1997 to 2013. Data from Taiwan’s National Health Insurance Research Database were utilized to answer the research question. A total of 17,649 offspring with schizophrenia were selected from 11 million offspring in the general population. Additionally, we established the offspring without schizophrenia as the comparison group by matching the study cohort by age, gender in a 1:4 ratio (n = 70,596).
Results
The median age at first presentation with schizophrenia was 20 years (interquartile range (IQR), 17 to 24). Comparison of the schizophrenia and non-schizophrenia groups indicated that father’s age at birth (30.0 (IQR), 27 to 33 vs. 29.0 (IQR), 26 to 32 years), mother’s age at birth (26.0 (IQR), 24 to 29 vs. 26.0 (IQR), 23 to 29 years), paternal schizophrenia (2.6% vs. 0.6%), and maternal schizophrenia (4.4% vs. 0.7%) were all significantly greater in the schizophrenia group. In addition, each 5-year increase in father’s age increased the odds of being diagnosed with schizophrenia (model 1: aOR = 1.22; 95% CI 1.20, 1.24; model 2: aOR = 1.20; 95% CI 1.18, 1.23). Subgroup analysis showed that each 5-year increase in father’s age increased the odds of being diagnosed with schizophrenia in male and female offspring, as well as in offspring of mothers and fathers with or without schizophrenia (aOR = 1.20 to 2.20, all p values < 0.01).
Conclusion
This study indicated that advanced paternal age increased the risk of schizophrenia in offspring. Offspring born to fathers older by 5-year increments were at heightened risk of schizophrenia.
Keywords: Paternal age, Offspring, Schizophrenia, Cumulative risk
Introduction
Recent studies have reported that increased paternal age is associated with adverse obstetric outcomes, birth defects, autism spectrum disorder, and schizophrenia in children [1–3], each of which has substantial impact on public health worldwide [4]. Schizophrenia is a severe neuropsychiatric syndrome that has a prevalence of 0.30–0.66% and an incidence of 10.2–22.0 per 100,000 people per year [5].
A meta-analysis of 12 studies reported that advanced paternal age was a potential risk factor for schizophrenia [6]. Moreover, other research has reported that the age of the father and grandfather was linked to an elevated risk of schizophrenia in offspring, thereby demonstrating an impact of fathers over 2 generations [7]. Another meta-analysis, which included 19 original studies and 3 systemic reviews, showed there was moderate certainty of evidence regarding the association of high paternal age with schizophrenia in offspring [1]. However, these studies reported different risk estimates because they used different paternal age categories [4]. In addition, most of these epidemiological studies did not adjust for confounding by factors associated with schizophrenia risk in offspring [8].
A 2019 study in Taiwan found that the association between paternal age and early-onset schizophrenia in offspring was not confounded by parental polygenic risk score for schizophrenia [8], which is a factor that partially captures parental genetic vulnerability to schizophrenia. These findings indicate that paternal age may independently increase the risk of early onset of schizophrenia in offspring [9]. However, the study had a number of limitations. First, compared with case-control genome-wide association study (GWAS) of other diseases, the sample size of this GWAS was relatively small. Second, despite efforts to recruit patients with schizophrenia throughout the country to ensure that the ascertained samples were generalizable to the patient population with schizophrenia in Taiwan, some ascertainment bias may have been present [9]. For instance, patients with early-onset schizophrenia have longer duration of illness and greater chances for referral than those with late-onset schizophrenia.
In this study, we used the Taiwan National Health Insurance Research Database (NHIRD) to determine the association between paternal age and schizophrenia in a single large population–based sample. Our study is the largest schizophrenia dataset from a nationalized health care system, which adds value to the literature. We focused on the influence of parental schizophrenia and compared the effect of maternal and paternal age with the risk of schizophrenia in offspring.
Materials and methods
Data source
Taiwan has had a single-payer compulsory National Health Insurance (NHI) program since 1995. By the end of 2009, approximately 99% of all 23,500,000 residents were enrolled in the NHI program. All claims data are collected in the NHIRD, which is maintained by the Department of Health and the National Health Research Institutes (NHRI). This database consists of deidentified personal data that has been available for research purposes since 2000 and is among the largest administrative health care databases worldwide. The NHIRD provides comprehensive patient data and important information, such as beneficiaries, catastrophic illness and medical service registries, demographic data, dates and number of clinical visits, dates of admission and discharge, prescribed drugs and dosages, prescription orders and durations, operations, examinations, diagnostic codes (according to the International Classification of Disease, Revision 9, Clinical Modification (ICD-9-CM)), procedure codes, and physician specialties [10]. The NHRI reported that there are no significant differences in age or sex between a randomly sampled group and all beneficiaries of the NHI program. The encryption procedures used to ensure patient privacy were consistent with those used for other datasets such that all claims data could be linked to obtain the medical data required for this study.
Study subjects and design
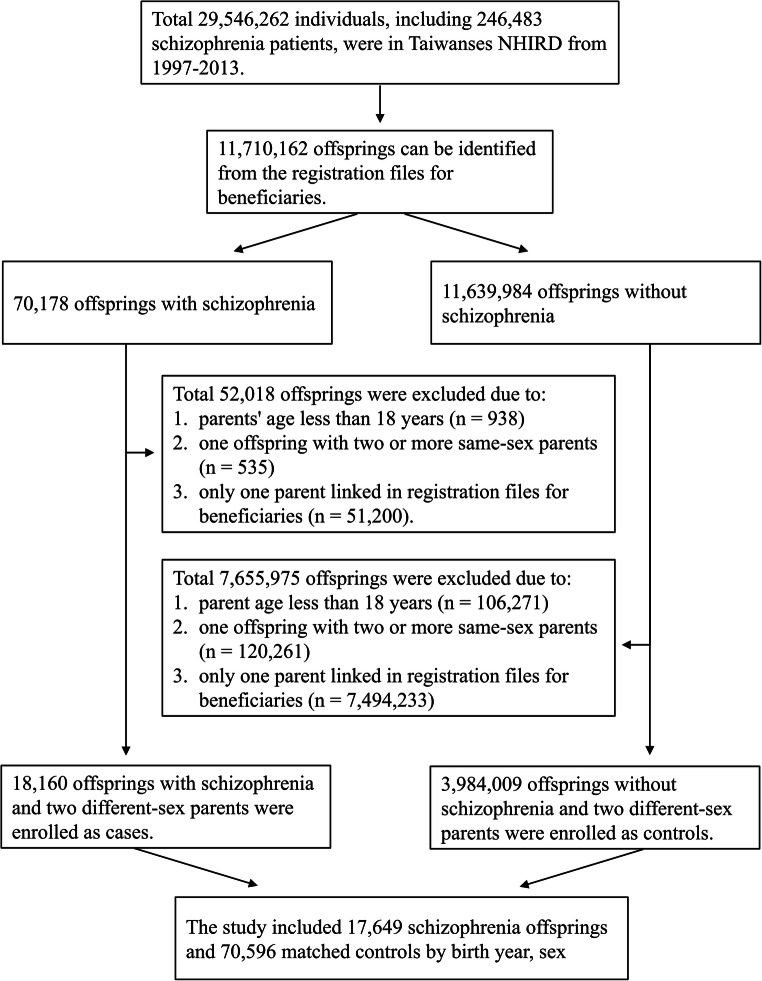
Study subjects comprised 11,710,162 insured offspring between 1997 and 2013, where 70,178 had received a diagnosis of schizophrenia (ICD-9-CM code), and 11,639,984 did not have a diagnosis of schizophrenia. We used the underwriting data file [title code] field to determine whether it is an “offspring linked to parents” relationship. After exclusion of those born to parents younger than 18 years, one offspring with two or more same-sex parents or those with only one parent linked in registration files for beneficiaries, there were 18,160 individuals with schizophrenia and 3,984,009 without schizophrenia. The schizophrenia and non-schizophrenia groups were randomly 1:4 frequency-matched according to offspring age (birth year) and sex (Fig. 1).
Fig. 1.
Flowchart of the patient enrollment for the schizophrenia group and the matched non-schizophrenia group
Validity of ICD codes
The diagnosis of schizophrenia was made by Taiwan psychiatrists based on the DSM-IV criteria [11]. A previous study suggested that the accuracy rates of disease-specific claims data may increase up to 96% if the claims data are modified by valid methods [12]. Another study assessed the accuracy of NHI claims data and its associated factors [13]. Further attention to the association of inaccurate claims in cases with infrequent outpatient visits, young age, and those attending non-accredited hospitals are needed to address the efficiency of diagnosis in Taiwan. That study indicated that compared with patients with < 1 outpatient visit, the odds ratio of accurate diagnosis of diabetes based on the ICD codes for patients with ≥ 2 outpatient visits was 2.12 (95% CI = 1.24–3.65). Compared with those who had not been hospitalized, the odds ratio of an accurate diagnosis for patients with ≥ 1 hospitalization was 4.14 (95% CI = 2.38–7.19). Thus, we ensured that “cases” of schizophrenia, who were defined as having the diagnostic code for “schizophrenia” for two ambulatory visits in 1 year or had the diagnostic code for “schizophrenia” for one inpatient claim code, were identified and collected for analysis.
Ethics statement
The study was approved by the Institutional Review Board (IRB) of Chang Gung Memorial Hospital (CGMH 201801162B0). Exclusion conditions were based on IRB and research purposes on the genetic inheritance of the parent and offspring.
Level of urbanization
For study of urbanization, all 365 townships in Taiwan were stratified into 4 levels according to the standards established by the Taiwanese NHRI based on a cluster analysis of Taiwan census data from 2000. In this classification, “1” referred to the most urbanized area and “4” referred to the least urbanized area. This classification was based on population density (persons/km2), number of physicians per 100,000 people, percentage of people with a college education, percentage of people over 65 years old, and percentage of agricultural workers.
Statistical analysis
The distributions of demographic factors were compared between the two groups. Pearson’s chi-square test was used to compare categorical variables and the Mann-Whitney-Wilcoxon test was used to compare continuous variables. The parental age of offspring at birth was mainly classified into seven age categories, which ranged from 18 to 50 years or older in 5-year intervals with 21 to 25 years as the reference group. Conditional logistic regression was used to investigate the risk factors for schizophrenia, including parents’ age, parents’ schizophrenia status, family urbanization, and income. Sensitivity analyses for conditional logistic regression were performed to assess the heterogeneous effects of schizophrenia and non-schizophrenia. SAS 9.0 for Windows (SAS Institute, Cary, North Carolina, USA) was used for data management and analysis. A p value below 0.05 was considered to be significant.
Results
Baseline characteristics of individuals with and without schizophrenia (Table 1)
Table 1.
Baseline characteristics of individuals with and without schizophrenia
| Characteristic | Total | Schizophrenia | Non-schizophrenia | p value* | |||
|---|---|---|---|---|---|---|---|
| 88,245 | 17,649 | 70,596 | |||||
| Age (first schizophrenia record in NHIRD) | NS | ||||||
| Median (IQR) | 20.0 | 17–24 | 20.0 | 17–24 | 20.0 | 17–24 | |
| Gender | |||||||
| Female, N (%) | 38,035 | 43.1% | 7607 | 43.1% | 30,428 | 43.1% | NS |
| Age (1st schizophrenia record in NHIRD) | |||||||
| Median (IQR) | 20.0 | 17–24 | 20.0 | 17–24 | 20.0 | 17–24 | |
| Male, N (%) | 50,210 | 56.9% | 10,042 | 56.9% | 40,168 | 56.9% | |
| Age (1st schizophrenia record in NHIRD) | |||||||
| Median (IQR) | 21.0 | 18–24 | 21.0 | 18–24 | 21.0 | 18–24 | |
| Mother’s age at birth | < 0.001 | ||||||
| Median (IQR) | 26.0 | 23–29 | 26.0 | 24–29 | 26.0 | 23–29 | |
| Age group, years | |||||||
| ≤ 20 | 5510 | 6.2% | 1083 | 6.1% | 4427 | 6.3% | |
| 21–25 | 34,375 | 39.0% | 6289 | 35.6% | 28,086 | 39.8% | |
| 26–30 | 35,259 | 40.0% | 7136 | 40.4% | 28,123 | 39.8% | |
| 31–35 | 10,804 | 12.2% | 2486 | 14.1% | 8318 | 11.8% | |
| 36–40 | 1952 | 2.2% | 551 | 3.1% | 1401 | 2.0% | |
| 41–45 | 258 | 0.3% | 79 | 0.4% | 179 | 0.3% | |
| 46–50 | 30 | 0.0% | 10 | 0.1% | 20 | 0.0% | |
| ≥ 50 | 57 | 0.1% | 15 | 0.1% | 42 | 0.1% | |
| Father’s age at birth | < 0.001 | ||||||
| Median (IQR) | 29.0 | 27–32 | 30.0 | 27–33 | 29.0 | 26–32 | |
| Age group, years | |||||||
| ≤ 20 | 984 | 1.1% | 171 | 1.0% | 813 | 1.2% | |
| 21–25 | 13,063 | 14.8% | 2152 | 12.2% | 10,911 | 15.5% | |
| 26–30 | 40,714 | 46.1% | 7461 | 42.3% | 33,253 | 47.1% | |
| 31–35 | 23,214 | 26.3% | 5005 | 28.4% | 18,209 | 25.8% | |
| 36–40 | 6599 | 7.5% | 1627 | 9.2% | 4972 | 7.0% | |
| 41–45 | 1971 | 2.2% | 597 | 3.4% | 1374 | 1.9% | |
| 46–50 | 873 | 1.0% | 307 | 1.7% | 566 | 0.8% | |
| ≥ 50 | 827 | 0.9% | 329 | 1.9% | 498 | 0.7% | |
| Mother with schizophrenia, N (%) | < 0.001 | ||||||
| Yes | 1275 | 1.4% | 782 | 4.4% | 493 | 0.7% | |
| No | 86,970 | 98.6% | 16,867 | 95.6% | 70,103 | 99.3% | |
| Father with schizophrenia, N (%) | < 0.001 | ||||||
| Yes | 884 | 1.0% | 457 | 2.6% | 427 | 0.6% | |
| No | 87,361 | 99.0% | 17,192 | 97.4% | 70,169 | 99.4% | |
| Both parents with schizophrenia, N (%) | 59 | 0.1% | 45 | 0.3% | 14 | 0.0% | |
| Family urbanization, N (%) | |||||||
| Very high | 7071 | 8.0% | 2837 | 16.1% | 4234 | 6.0% | |
| High | 10,962 | 12.4% | 4197 | 23.8% | 6765 | 9.6% | |
| Moderate | 3119 | 3.5% | 1221 | 6.9% | 1898 | 2.7% | |
| Low/unknown | 67,093 | 76.0% | 9394 | 53.2% | 57,699 | 81.7% | |
| Family income (TWD per month), N (%) | |||||||
| 0/unknown | 71,071 | 80.5% | 12,146 | 68.8% | 58,925 | 83.5% | |
| 1–15,840 | 3344 | 3.8% | 1203 | 6.8% | 2141 | 3.0% | |
| 15,841–25,000 | 8509 | 9.6% | 2690 | 15.2% | 5819 | 8.2% | |
| ≧ 25,001 | 5321 | 6.0% | 1610 | 9.1% | 3711 | 5.3% | |
*Pearson’s chi-square test for categorical variables (except for family urbanization and income) and Mann-Whitney-Wilcoxon test for continuous variables
IQR, interquartile range; TWD, Taiwan dollar; N, number; NS, not significant
Ultimately, we analyzed 17,649 individuals with schizophrenia and 70,596 matched individuals without schizophrenia from 1997 to 2013. Because of matching, each group was 43.1% female and had a median age of 20 years (interquartile range (IQR), 17 to 24). However, median maternal age (26.0 (IQR), 24 to 29 vs. 26.0 (IQR), 23 to 29) and median paternal age (30.0 (IQR), 27 to 33 vs. 29.0 (IQR), 26 to 32) at birth were significantly higher in the schizophrenia group than in the non-schizophrenia group (both p values < 0.001). More individuals with schizophrenia had a mother with schizophrenia (4.4% vs. 0.7%, p values < 0.001), a father with schizophrenia (2.6% vs. 0.6%, p values < 0.001) or both parents with schizophrenia (0.3% vs. 0.0%). The number and percentage of schizophrenia and controls in each parental age category are also presented in Table 1.
Conditional logistic regression analysis of parental factors associated with schizophrenia
Conditional logistic regression analysis (Table 2) with adjustment for confounding by mother’s age at 5-year increments, father’s age at 5-year increments, maternal schizophrenia, and paternal schizophrenia indicated that the odds of being diagnosed with schizophrenia increased with the father’s age in 5-year increments (model 1: aOR = 1.22; 95% CI 1.20, 1.24; model 2: aOR = 1.20; 95% CI 1.18, 1.23), but the mother’s age in 5-year increments had no such effect (model 1: aOR = 0.99; 95% CI 0.97, 1.02; model 2: aOR = 0.98; 95% CI 0.96, 1.01). Maternal schizophrenia (model 1: aOR = 5.93; 95% CI 5.27, 6.67; model 2: aOR = 6.55; 95% CI 5.79, 7.42) and paternal schizophrenia (model 1: aOR = 4.13; 95% CI 3.60, 4.74; model 2: aOR = 4.60; 95% CI 3.97, 5.33) also significantly increased the odds of being diagnosed with schizophrenia in the offspring (both p values < 0.001). When the odds of schizophrenia were estimated in the extended parental age groups (paternal age, 41–45 years, 46–50 years, and > 50 years; maternal age, 41–45 years and 46–50 years), we found paternal age greater than 40 years (model 1: aOR = 2.09 to 2.96; model 2: aOR = 2.01 to 2.57; all p values < 0.001) was associated with increased odds of being diagnosed with schizophrenia. Compared with the offspring of mothers ages 21 to 25 years, the offspring of mothers with age less than 20 years had increased odds of being diagnosed with schizophrenia (model 1: aOR = 1.13; 95% CI 1.05, 1.22; model 2: aOR = 1.13; 95% CI 1.05, 1.23).
Table 2.
Conditional logistic regression analysis of the association of offspring schizophrenia with potential risk factors
| Variables | Univariable analysis | Multivariable analysis | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| OR | 95% CI | p value | aOR* | 95% CI | p value | aOR** | 95% CI | p value | ||||
| Mother’s age at birth (5 year increments) | 1.16 | 1.13 | 1.18 | < 0.001 | 0.99 | 0.97 | 1.02 | 0.529 | 0.98 | 0.96 | 1.01 | 0.179 |
| Father’s age at birth (5 year increments) | 1.23 | 1.21 | 1.25 | < 0.001 | 1.22 | 1.20 | 1.24 | < 0.001 | 1.20 | 1.18 | 1.23 | < 0.001 |
| Maternal schizophrenia | 6.65 | 5.92 | 7.46 | < 0.001 | 5.93 | 5.27 | 6.67 | < 0.001 | 6.55 | 5.79 | 7.42 | < 0.001 |
| Paternal schizophrenia | 4.35 | 3.81 | 4.97 | < 0.001 | 4.13 | 3.60 | 4.74 | < 0.001 | 4.60 | 3.97 | 5.33 | < 0.001 |
| Mother’s age at birth group, years | ||||||||||||
| ≤ 20 | 1.09 | 1.02 | 1.17 | 0.018 | 1.13 | 1.05 | 1.22 | 0.002 | 1.13 | 1.05 | 1.23 | 0.002 |
| 21–25 (reference) | 1.00 | - | - | - | 1.00 | - | - | - | 1.00 | - | - | - |
| 26–30 | 1.14 | 1.10 | 1.18 | < 0.0001 | 1.03 | 0.99 | 1.07 | 0.167 | 1.03 | 0.98 | 1.07 | 0.272 |
| 31–35 | 1.35 | 1.28 | 1.42 | < 0.0001 | 1.03 | 0.97 | 1.10 | 0.344 | 1.03 | 0.96 | 1.10 | 0.462 |
| 36–40 | 1.78 | 1.61 | 1.97 | < 0.0001 | 1.08 | 0.96 | 1.21 | 0.207 | 1.08 | 0.96 | 1.22 | 0.206 |
| 41–45 | 2.00 | 1.53 | 2.61 | < 0.0001 | 1.04 | 0.79 | 1.39 | 0.767 | 1.01 | 0.75 | 1.36 | 0.942 |
| 46–50 | 2.22 | 1.04 | 4.74 | 0.040 | 1.86 | 0.85 | 4.10 | 0.123 | 1.71 | 0.75 | 3.89 | 0.203 |
| ≥ 50 | 1.58 | 0.88 | 2.86 | 0.127 | 1.27 | 0.69 | 2.34 | 0.434 | 1.06 | 0.55 | 2.03 | 0.869 |
| Father’s age at birth group, years | ||||||||||||
| ≤ 20 | 1.07 | 0.90 | 1.27 | 0.449 | 1.01 | 0.85 | 1.21 | 0.907 | 0.95 | 0.79 | 1.14 | 0.558 |
| 21–25 (reference) | 1.00 | - | - | - | 1.00 | - | - | - | 1.00 | - | - | - |
| 26–30 | 1.14 | 1.08 | 1.20 | < 0.0001 | 1.15 | 1.09 | 1.22 | < 0.0001 | 1.17 | 1.10 | 1.24 | < 0.0001 |
| 31–35 | 1.40 | 1.33 | 1.48 | < 0.0001 | 1.41 | 1.32 | 1.50 | < 0.0001 | 1.42 | 1.33 | 1.52 | < 0.0001 |
| 36–40 | 1.68 | 1.56 | 1.81 | < 0.0001 | 1.67 | 1.53 | 1.82 | < 0.0001 | 1.66 | 1.52 | 1.82 | < 0.0001 |
| 41–45 | 2.27 | 2.04 | 2.53 | < 0.0001 | 2.09 | 1.86 | 2.36 | < 0.0001 | 2.01 | 1.78 | 2.28 | < 0.0001 |
| 46–50 | 2.81 | 2.42 | 3.26 | < 0.0001 | 2.53 | 2.16 | 2.96 | < 0.0001 | 2.39 | 2.03 | 2.82 | < 0.0001 |
| ≥ 50 | 3.36 | 2.90 | 3.90 | < 0.0001 | 2.96 | 2.53 | 3.47 | < 0.0001 | 2.57 | 2.18 | 3.03 | < 0.0001 |
*Logistic model 1: adjustment for mother’s age, father’s age, maternal schizophrenia, and paternal schizophrenia
**Logistic model 2: adjustment for all model 1 factors and family urbanization and family income
We stratified individuals by sex, maternal schizophrenia, paternal schizophrenia, and both parents with or without schizophrenia to verify the impact of paternal age on schizophrenia in 5-year increments (Table 3). The results show that father’s age in 5-year increments significantly increased the odds of being diagnosed with schizophrenia in all groups (model 1: aOR = 1.22 to 2.18; model 2: aOR = 1.20 to 2.20; all p values < 0.05). However, there is no significant increase in odds of being diagnosed with schizophrenia associated with mother’s age in 5-year increments in all groups.
Table 3.
Subgroup analysis and adjusted odds ratios of the effect of parental age on offspring schizophrenia (conditional logistic regression)
| Stratified variable | aOR* | 95% CI | p value | aOR** | 95% CI | p value | |||
|---|---|---|---|---|---|---|---|---|---|
| Female | Mother’s age (5-year increments) | 1.00 | 0.97 | 1.04 | 0.976 | 0.99 | 0.95 | 1.03 | 0.575 |
| Father’s age (5-year increments) | 1.22 | 1.18 | 1.25 | < 0.001 | 1.20 | 1.17 | 1.24 | < 0.001 | |
| Male | Mother’s age (5-year increments) | 0.96 | 0.93 | 0.99 | 0.010 | 0.94 | 0.91 | 0.98 | 0.001 |
| Father’s age (5-year increments) | 1.26 | 1.23 | 1.29 | < 0.001 | 1.25 | 1.22 | 1.28 | < 0.001 | |
| Mother with schizophrenia | Mother’s age (5-year increments) | 1.01 | 0.41 | 2.49 | 0.980 | 0.95 | 0.83 | 1.09 | 0.455 |
| Father’s age (5-year increments) | 2.48 | 0.90 | 6.88 | 0.081 | 1.24 | 1.14 | 1.35 | < 0.001 | |
| Mother without schizophrenia | Mother’s age (5-year increments) | 1.00 | 0.97 | 1.02 | 0.691 | 0.98 | 0.96 | 1.01 | 0.221 |
| Father’s age (5-year increments) | 1.22 | 1.19 | 1.24 | < 0.001 | 1.20 | 1.18 | 1.22 | < 0.001 | |
| Father with schizophrenia | Mother’s age (5-year increments) | 2.88 | 0.06 | 136.73 | 0.591 | 0.90 | 0.72 | 1.11 | 0.313 |
| Father’s age (5-year increments) | 1.13 | 0.01 | 210.13 | 0.963 | 1.32 | 1.11 | 1.56 | 0.002 | |
| Father without schizophrenia | Mother’s age (5-year increments) | 0.98 | 0.95 | 1.00 | 0.049 | 0.97 | 0.94 | 0.99 | 0.006 |
| Father’s age (5-year increments) | 1.24 | 1.22 | 1.26 | < 0.001 | 1.23 | 1.20 | 1.25 | < 0.001 | |
| Both parents without schizophrenia | Mother’s age (5-year increments) | 0.99 | 0.97 | 1.02 | 0.654 | 1.00 | 0.97 | 1.02 | 0.693 |
| Father’s age (5-year increments) | 1.22 | 1.19 | 1.24 | < 0.001 | 1.20 | 1.17 | 1.22 | < 0.001 | |
| Both parents with schizophrenia | Mother’s age (5-year increments) | 1.46 | 0.81 | 2.61 | 0.207 | 1.41 | 0.78 | 2.55 | 0.258 |
| Father’s age (5-year increments) | 2.18 | 1.26 | 3.76 | 0.005 | 2.20 | 1.22 | 3.98 | 0.009 | |
*Logistic model 1: adjustment for mother’s age, father’s age
**Logistic model 2: adjustment for mother’s age, father’s age, family urbanization, family income
Discussion
Principal findings of the study
The key result of the present study, which examined 17,649 schizophrenia patients and 70,596 matched controls from the population of Taiwan, is that children of older fathers have increased odds of being diagnosed with schizophrenia. Our finding of a significant association of schizophrenia in offspring with advanced paternal age but not advanced maternal age is consistent with many previous studies and meta-analyses [2, 6, 14–20]. However, some previous studies have reported that advanced maternal age also increased the risk of schizophrenia in offspring [21, 22].
There is some uncertainty regarding whether a gradual increase in father’s age increases the risk of schizophrenia or whether the increased risk only applies to the oldest group of fathers. Knowledge regarding this risk is important in counseling of potential parents and in determining public health policy [23]. Our analyses of different paternal age categories showed that the odds of being diagnosed with schizophrenia in offspring increased as paternal age gradually increased. The risk is even more significant if the father is older than 40. Our stratified analysis, which considered parental schizophrenia and father’s age in 5-year increments, also indicated an increased risk of schizophrenia in the offspring of older fathers but no such effect for mother’s age. McGrath et al. [2] found a U-shaped relationship between paternal age and risk of schizophrenia, whereas the offspring of older fathers were at increased risk of schizophrenia, and to a lesser extent, young fathers were at increased risk for schizophrenia. The authors also reported the offspring of more adolescent mothers are at risk of different mental health disorders. We also found the offspring of mothers with age less than 20 years had increased odds of being diagnosed with schizophrenia (Table 2).
Results of the study in the context of other observations
The biological basis underlying the relationship between paternal age and risk of schizophrenia in offspring has not been elucidated. Nonetheless, previous researchers have proposed several hypotheses for this association. For example, older fathers would be more affected by de novo mutations that disrupt DNA repair, lower testosterone levels, altered mitotic divisions leading to new mutations in sperm, epigenetic alterations [23–25], and other environmental factors. There could also be selection into late fatherhood, in which fathers with a genetic tendency for schizophrenia are more likely to be older when having children.
Our main analyses adjusted for key variables (age of the other parent and parental schizophrenia), and we also included socioeconomic factors and urbanization. The etiology of schizophrenia is poorly understood, but accumulating evidence has revealed a wide range of brain abnormalities. These factors may reflect both genetic and environmental effects as well as the clinical heterogeneity of schizophrenia [26]. Among the environmental risk factors for schizophrenia, evidence supports urbanicity [27, 28]. Income inequality is associated with numerous adverse health outcomes. There is also evidence that ecological level socioenvironmental factors may increase the risk of schizophrenia [29].
Because there are many divisions in spermatogonial stem cells during a man’s lifetime, late fatherhood could yield more mutations [30], which can lead to aberrant biological sequelae [31]. Moreover, recent whole-genome sequencing studies have reported a linear relationship between paternal age and the rate of de novo mutations in offspring, with approximately 2 additional de novo mutations per year [32]. In addition, the father transfers 3 to 4 times more de novo mutations to the offspring than the mother [32, 33]. Many researchers favor the de novo mutation hypothesis as the explanation for the effect of father’s age on schizophrenia in offspring; however, there is currently more evidence in favor of the selection into late fatherhood hypothesis. All previous epidemiological studies that used a matched control group performed matching of cases and controls based on features of the children. An analysis using matching based on the environmental characteristics of the father himself may provide additional information [23].
Offspring of parents with schizophrenia
Our results also showed having a mother or a father or both parents diagnosed with schizophrenia resulted in more offspring being diagnosed with schizophrenia. As seen in Table 1, there are 59 where both parents have a diagnosis of schizophrenia, and 45 (76.3%) of these offspring have a schizophrenia diagnosis. This outcome is consistent with previous reports. The offspring of parents with severe mental illness, including schizophrenia, have a one-in-three risk of developing severe mental illness themselves [34]. The heritability of schizophrenia is up to 80%. If one parent suffers from the condition, the probability that it will be passed down to the offspring is 13%. If this trail is present in both parents, the risk is greater than 20%. Genetic factors are assumed to play a role in the development of schizophrenia [35].
A significant body of longitudinal research has followed the offspring of parents with schizophrenia. This combination of results suggests that the offspring of parents with schizophrenia are at high risk not only for schizophrenia but also for poor developmental and general mental health outcomes [36]. Although family history remains the single most reliable predictor of illness, molecular genetic tools are becoming increasingly informative [34].
Strengths and limitations
Our study has a number of strengths. First, this study is the largest study to date to examine the relationship of paternal age with schizophrenia in offspring. Second, the results suggest that unmeasured genetic and environmental selection factors shared by siblings, as well as the influence of several measured covariates, do not account for the association of schizophrenia with paternal age, which is consistent with a causal relationship. Third, and perhaps most importantly, the current study suggests that with father’s age in 5-year increments there are increased odds of being diagnosed with schizophrenia in offspring rather than an increased risk, which is due solely to the effect of very old fathers. Although for every 5 years in age after the father is age 40, the increased odds of a diagnosis of schizophrenia in offspring are more significant. Moreover, the magnitudes of most of the associations in our study were stronger than in previous studies and few previous studies were from Asia. In contrast to the previous study by Wang et al. [9], our study included all individuals with schizophrenia in the complete population of Taiwan. Therefore, there is little risk of selection bias, which may occur in clinically selected samples.
However, our study has limitations in detailed personal history, and Taiwan NHIRD does not provide perinatal information, such as birth weight and weeks of pregnancy and lifestyle, e.g., smoking/alcohol abuse, body mass index, and functional capacity. In contrast, there are many unknown data when considering family urbanization and income classification. In addition, the loss of 52,018 offspring diagnosed with schizophrenia, which was due to the study exclusion criteria, could have resulted in some bias.
Conclusions
The results of the present study suggest that advanced paternal age increases the risk for schizophrenia in offspring. In addition, the offspring born to fathers older by 5-year increments were at heightened risk of schizophrenia, indicating this increased risk was not due to the effect of a small number of very old fathers.
Acknowledgments
We sincerely appreciate the Biostatistics Center, Kaohsiung Chang Gung Memorial Hospital, for performing the statistical analysis.
Abbreviations
- CGMH
Chang Gung Memorial Hospital
- GWAS
Genome-wide association study
- NHI
National Health Insurance
- NHIRD
National Health Insurance Research Database
Authors’ contributions
Conceived and supervised this work, KCL: methodology, software, and validation, HJC and YHY; writing—original draft preparation, KCL, TYH, and YCO; writing—review and editing, KCL, TLH, and YJC; funding acquisition, KCL. All authors read and approved the final manuscript.
Funding
This study was supported by CMRPG8G0071-73, CMRPG8H0981, and CMRPG 8B0771 from the Chang Gung Memorial Hospital.
Compliance with ethical standards
The study was approved by the Institutional Review Board (IRB) of Chang Gung Memorial Hospital (CGMH 201801162B0). Exclusion conditions were based on IRB and research purposes on the genetic inheritance of the parent and offspring.
Competing interests
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Kuo-Chung Lan, Email: lankuochung@gmail.com.
Hsin-Ju Chiang, Email: n22370@cgmh.org.tw.
Tiao-Lai Huang, Email: a540520@adm.cgmh.org.tw.
Yu-Jie Chiou, Email: yujie6666@gmail.com.
Te-Yao Hsu, Email: tyhsu@cgmh.org.tw.
Yu-Che Ou, Email: tedycou@gmail.com.
Yao-Hsu Yang, Email: r95841012@adm.cgmh.org.tw.
References
- 1.Oldereid NB, Wennerholm UB, Pinborg A, Loft A, Laivuori H, Petzold M, Romundstad LB, Söderström-Anttila V, Bergh C. The effect of paternal factors on perinatal and paediatric outcomes: a systematic review and meta-analysis. Hum Reprod Update. 2018;24(3):320–389. doi: 10.1093/humupd/dmy005. [DOI] [PubMed] [Google Scholar]
- 2.McGrath JJ, Petersen L, Agerbo E, Mors O, Mortensen PB, Pedersen CB. A comprehensive assessment of parental age and psychiatric disorders. JAMA Psychiatry. 2014;71(3):301–309. doi: 10.1001/jamapsychiatry.2013.4081. [DOI] [PubMed] [Google Scholar]
- 3.Nybo Andersen AM, Urhoj SK. Is advanced paternal age a health risk for the offspring? Fertil Steril. 2017;107(2):312–318. doi: 10.1016/j.fertnstert.2016.12.019. [DOI] [PubMed] [Google Scholar]
- 4.Bergh C, Pinborg A, Wennerholm UB. Parental age and child outcomes. Fertil Steril. 2019;111(6):1036–1046. doi: 10.1016/j.fertnstert.2019.04.026. [DOI] [PubMed] [Google Scholar]
- 5.Malaspina D, Gilman C, Kranz TM. Paternal age and mental health of offspring. Fertil Steril. 2015;103(6):1392–1396. doi: 10.1016/j.fertnstert.2015.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Miller B, Messias E, Miettunen J, Alaraisanen A, Jarvelin MR, Koponen H, et al. Meta-analysis of paternal age and schizophrenia risk in male versus female offspring. Schizophr Bull. 2011;37(5):1039–1047. doi: 10.1093/schbul/sbq011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Frans EM, McGrath JJ, Sandin S, Lichtenstein P, Reichenberg A, Langstrom N, et al. Advanced paternal and grandpaternal age and schizophrenia: a three-generation perspective. Schizophr Res. 2011;133(1–3):120–124. doi: 10.1016/j.schres.2011.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McGrath J, Saha S, Chant D, Welham J. Schizophrenia: a concise overview of incidence, prevalence, and mortality. Epidemiol Rev. 2008;30:67–76. doi: 10.1093/epirev/mxn001. [DOI] [PubMed] [Google Scholar]
- 9.Wang SH, Hsiao PC, Yeh LL, Liu CM, Liu CC, Hwang TJ, Hsieh MH, Chien YL, Lin YT, Huang YT, Chen CY, Chandler SD, Faraone SV, Neale B, Glatt SJ, Tsuang MT, Hwu HG, Chen WJ. Advanced paternal age and early onset of schizophrenia in sporadic cases: not confounded by parental polygenic risk for schizophrenia. Biol Psychiatry. 2019;86:56–64. doi: 10.1016/j.biopsych.2019.01.023. [DOI] [PubMed] [Google Scholar]
- 10.Yeh H-Y, Wu J-C, Haschler I, Chen T-J, Wetter T. Taiwan’s National Health Insurance Research Database: administrative health care database as study object in bibliometrics. Scientometrics. 2011;86(2):365–380. doi: 10.1007/s11192-010-0289-2. [DOI] [Google Scholar]
- 11.McLean D, Thara R, John S, Barrett R, Loa P, McGrath J, Mowry B. DSM-IV “criterion A” schizophrenia symptoms across ethnically different populations: evidence for differing psychotic symptom content or structural organization? Cult Med Psychiatry. 2014;38(3):408–426. doi: 10.1007/s11013-014-9385-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Quam L, Ellis LB, Venus P, Clouse J, Taylor CG, Leatherman S. Using claims data for epidemiologic research. The concordance of claims-based criteria with the medical record and patient survey for identifying a hypertensive population. Med Care. 1993;31(6):498–507. doi: 10.1097/00005650-199306000-00003. [DOI] [PubMed] [Google Scholar]
- 13.Lin CC, Lai MS, Syu CY, Chang SC, Tseng FY. Accuracy of diabetes diagnosis in health insurance claims data in Taiwan. J Formos Med Assoc. 2005;104(3):157–163. [PubMed] [Google Scholar]
- 14.Miller B, Suvisaari J, Miettunen J, Jarvelin MR, Haukka J, Tanskanen A, et al. Advanced paternal age and parental history of schizophrenia. Schizophr Res. 2011;133(1–3):125–132. doi: 10.1016/j.schres.2011.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sipos A, Rasmussen F, Harrison G, Tynelius P, Lewis G, Leon DA, Gunnell D. Paternal age and schizophrenia: a population based cohort study. BMJ. 2004;329(7474):1070. doi: 10.1136/bmj.38243.672396.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Byrne M, Agerbo E, Ewald H, Eaton WW, Mortensen PB. Parental age and risk of schizophrenia: a case-control study. Arch Gen Psychiatry. 2003;60(7):673–678. doi: 10.1001/archpsyc.60.7.673. [DOI] [PubMed] [Google Scholar]
- 17.Zammit S, Allebeck P, Dalman C, Lundberg I, Hemmingson T, Owen MJ, Lewis G. Paternal age and risk for schizophrenia. Br J Psychiatry. 2003;183:405–408. doi: 10.1192/bjp.183.5.405. [DOI] [PubMed] [Google Scholar]
- 18.Brown AS, Schaefer CA, Wyatt RJ, Begg MD, Goetz R, Bresnahan MA, Harkavy-Friedman J, Gorman JM, Malaspina D, Susser ES. Paternal age and risk of schizophrenia in adult offspring. Am J Psychiatry. 2002;159(9):1528–1533. doi: 10.1176/appi.ajp.159.9.1528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Malaspina D, Harlap S, Fennig S, Heiman D, Nahon D, Feldman D, Susser ES. Advancing paternal age and the risk of schizophrenia. Arch Gen Psychiatry. 2001;58(4):361–367. doi: 10.1001/archpsyc.58.4.361. [DOI] [PubMed] [Google Scholar]
- 20.Dalman C, Allebeck P. Paternal age and schizophrenia: further support for an association. Am J Psychiatry. 2002;159(9):1591–1592. doi: 10.1176/appi.ajp.159.9.1591. [DOI] [PubMed] [Google Scholar]
- 21.Ekeus C, Olausson PO, Hjern A. Psychiatric morbidity is related to parental age: a national cohort study. Psychol Med. 2006;36(2):269–276. doi: 10.1017/S0033291705006549. [DOI] [PubMed] [Google Scholar]
- 22.Krishnaswamy S, Subramaniam K, Indran H, Ramachandran P, Indran T, Indran R, Aziz JA. Paternal age and common mental disorders. World J Biol Psychiatry. 2009;10(4 Pt 2):518–523. doi: 10.1080/15622970802653691. [DOI] [PubMed] [Google Scholar]
- 23.de Kluiver H, Buizer-Voskamp JE, Dolan CV, Boomsma DI. Paternal age and psychiatric disorders: a review. Am J Med Genet B Neuropsychiatr Genet. 2017;174(3):202–213. doi: 10.1002/ajmg.b.32508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Perrin MC, Brown AS, Malaspina D. Aberrant epigenetic regulation could explain the relationship of paternal age to schizophrenia. Schizophr Bull. 2007;33(6):1270–1273. doi: 10.1093/schbul/sbm093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Atsem S, Reichenbach J, Potabattula R, Dittrich M, Nava C, Depienne C, Böhm L, Rost S, Hahn T, Schorsch M, Haaf T, el Hajj N. Paternal age effects on sperm FOXK1 and KCNA7 methylation and transmission into the next generation. Hum Mol Genet. 2016;25(22):4996–5005. doi: 10.1093/hmg/ddw328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Cunningham C, Peters K. Aetiology of schizophrenia and implications for nursing practice: a literature review. Issues Ment Health Nurs. 2014;35(10):732–738. doi: 10.3109/01612840.2014.908441. [DOI] [PubMed] [Google Scholar]
- 27.Vassos E, Pedersen CB, Murray RM, Collier DA, Lewis CM. Meta-analysis of the association of urbanicity with schizophrenia. Schizophr Bull. 2012;38(6):1118–1123. doi: 10.1093/schbul/sbs096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Heinz A, Deserno L, Reininghaus U. Urbanicity, social adversity and psychosis. World Psychiatry. 2013;12(3):187–197. doi: 10.1002/wps.20056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Burns JK, Tomita A, Kapadia AS. Income inequality and schizophrenia: increased schizophrenia incidence in countries with high levels of income inequality. Int J Soc Psychiatry. 2014;60(2):185–196. doi: 10.1177/0020764013481426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Buwe A, Guttenbach M, Schmid M. Effect of paternal age on the frequency of cytogenetic abnormalities in human spermatozoa. Cytogenet Genome Res. 2005;111(3–4):213–228. doi: 10.1159/000086892. [DOI] [PubMed] [Google Scholar]
- 31.Goriely A, McGrath JJ, Hultman CM, Wilkie AO, Malaspina D. “Selfish spermatogonial selection”: a novel mechanism for the association between advanced paternal age and neurodevelopmental disorders. Am J Psychiatry. 2013;170(6):599–608. doi: 10.1176/appi.ajp.2013.12101352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kong A, Frigge ML, Masson G, Besenbacher S, Sulem P, Magnusson G, Gudjonsson SA, Sigurdsson A, Jonasdottir A, Jonasdottir A, Wong WSW, Sigurdsson G, Walters GB, Steinberg S, Helgason H, Thorleifsson G, Gudbjartsson DF, Helgason A, Magnusson OT, Thorsteinsdottir U, Stefansson K. Rate of de novo mutations and the importance of father’s age to disease risk. Nature. 2012;488(7412):471–475. doi: 10.1038/nature11396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goldmann JM, Wong WS, Pinelli M, Farrah T, Bodian D, Stittrich AB, et al. Parent-of-origin-specific signatures of de novo mutations. Nat Genet. 2016;48(8):935–939. doi: 10.1038/ng.3597. [DOI] [PubMed] [Google Scholar]
- 34.Sandstrom A, Sahiti Q, Pavlova B, Uher R. Offspring of parents with schizophrenia, bipolar disorder, and depression: a review of familial high-risk and molecular genetics studies. Psychiatr Genet. 2019;29(5):160–169. doi: 10.1097/YPG.0000000000000240. [DOI] [PubMed] [Google Scholar]
- 35.Janoutova J, Janackova P, Sery O, Zeman T, Ambroz P, Kovalova M, et al. Epidemiology and risk factors of schizophrenia. Neuro Endocrinol Lett. 2016;37(1):1–8. [PubMed] [Google Scholar]
- 36.Hameed MA, Lewis AJ. Offspring of parents with schizophrenia: a systematic review of developmental features across childhood. Harv Rev Psychiatry. 2016;24(2):104–117. doi: 10.1097/HRP.0000000000000076. [DOI] [PubMed] [Google Scholar]