Abstract
COVID-19, coronavirus disease 2019, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has become a pandemic. At the time of writing this (October 14, 2020), more than 38.4 million people have become affected, and 1.0 million people have died across the world. The death rate is undoubtedly correlated with the cytokine storm and other pathological pulmonary characteristics, as a result of which the lungs cannot provide sufficient oxygen to the body's vital organs. While diversified drugs have been tested as a first line therapy, the complexity of fatal cases has not been reduced so far, and the world is looking for a treatment to combat the virus. However, to date, and despite such promise, we have received very limited information about the potential of nanomedicine to fight against COVID-19 or as an adjunct therapy in the treatment regimen. Over the past two decades, various therapeutic strategies, including direct-acting antiviral drugs, immunomodulators, a few non-specific drugs (simple to complex), have been explored to treat Acute Respiratory Distress Syndrome (ARDS), Severe Acute Respiratory Syndrome (SARS) and Middle East Respiratory Syndrome (MERS), influenza, and sometimes the common flu, thus, correlating and developing specific drugs centric to COVID-19 is possible. This review article focuses on the pulmonary pathology caused by SARS-CoV-2 and other viral pathogens, highlighting possible nanomedicine therapeutic strategies that should be further tested immediately.
Keywords: Coronavirus; COVID-19; Influenza, Pulmonary drug delivery; SARS, SARS-CoV-2; MERS, Nanomedicine; Nanotherapeutics; Pathophysiology; Nanotechnology
Graphical Abstract
Nanomedicine Therapeutic Strategies for COVID-19 Like Infections

Introduction
Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) is the etiological source that causes Coronavirus Disease 2019, or COVID-19. Around 80% of cases of COVID-19 will be mild to moderate, close to 14% have severe disease and around 6% are critical. The possible symptoms include fever, headache, muscle pain, fatigue, and breath shortness followed by multi-organ failure. Nearly 20% of COVID-19 patients are experiencing Acute Respiratory Distress Syndrome (ARDS) [1], [2]. The high mortality rates depend on several clinical factors, such as ARDS, pneumonia, sepsis-induced by the ‘cytokine storm’ (e.g., an uncontrolled release of cytokines which causes systemic inflammation) followed by lung failure, coagulopathy and thrombotic events, secondary bacterial infections, and vital organ (heart/liver/kidney) injuries followed by failure, and others [1], [3], [4], [5]. Thus, considering the surge in new cases and death rate, the World Health Organization announced COVID-19 as a pandemic [6]. The origin of COVID-19 first arouse in Wuhan, China on December 16, 2019. Later, its genome was likely correlated with the bat's genome, nearly 96% similar; assuming that it has been transferred from animals, e.g., bats and pangolins (zoonotic disease) [7], [8].
SARS-CoV-2 infects both humans and animals, and it has been globally infecting humans with more than 38.4 million cases and more than 1,091,605 deaths (as per October 14, 2020), which is far greater than the SARS and Middle East Respiratory Syndrome (MERS). The infection is spreading through human-human and animal-human transmission via the oral-oral and oral-fecal route [9]. Recently, the Advisory Committee on Dangerous Pathogens (ACDP) in the UK categorized SARS-CoV-2 as a hazard group 3 organism.
Like other SARS-CoV, SARS-CoV-2 also uses the angiotensin 1-converting enzyme 2 (ACE2; a close homolog of ACE) for entry into the cells [10], [11], in addition to ACE2, it also uses other cellular proteases including transmembrane protease serine 2 (TMPRSS2) [12], [13], which is useful in priming of the process of SARS-CoV-2. Recently, one study concluded that SARS-CoV preferentially infects well-differentiated ciliated epithelial cells expressing ACE2. Since ACE2 is also the receptor for the coronavirus NL63, their findings are relevant to the biology of infection with this more common human pathogen [14]. Among other tissues, a high expression of ACE2 is found in the lungs and the small intestine [15]. For a SARS-CoV infection, expression of ACE2 in the airway epithelia appears to be both necessary and essential. The airway epithelial expression of ACE2 is complex and correlated with cell differentiation, a finding that may underlie susceptibility to infection [14]; thus, a probable source for viral loads. Besides, the recent single-cell transcriptome data on ACE2 and TMPRSS2 revealed that nasal epithelial cells (goblet/secretory cells and ciliated cells) exhibit high ACE2 gene expression when compared to other epithelial cells [16]. Equally important, alveolar type II cells also exhibit a high expression of ACE2. Besides, there is a concern for other associated receptors that act synergistically with infections, i.e., viral scavenging (DMBT1) receptors [17]. On the other hand, a high expression of furin, which exposes the binding and fusion domains of SARS-CoV-2, has also been observed in oral epithelial cells [18]. Interestingly, considering the human-human transmission through sneezing and coughing, it is clear that SARS-CoV-2 infects epithelial cells in the upper airway epithelium [16], later the alveoli (type II cells) [19], and oral mucosa [20].
Despite the recent developments in clinical modalities for multiple pulmonary infections, there is an unmet medical need for developing more effective targeted nano-delivery systems. This site-specific local delivery of nano-engineered therapeutic agents with controlled release and stabilization of the therapeutic agent play a crucial role in the management of COVID-19 like pulmonary infections. The foremost challenges with traditional pulmonary drug delivery include a lack of penetrability, non-targeted systemic delivery, and interaction with multiple targets at a single therapeutic site. Any advancements in pulmonary nano-drug delivery systems aim to provide maximum therapeutic efficacy with minimal off-targeted side effects, ease of functionalization (chemically, biologically), and personalized therapy which together have concomitant benefits. In addition to the potential therapeutic abilities, nanotechnology plays an essential role in bringing cost-effective, quick nano-diagnostics tools to detect pulmonary infection associated biomarkers.
With current approaches to avoid infection and the realization that transmission is the only possible intervention that can be implemented along with complementary medicine and support care, no feasible COVID-19 regimen is available. However, to counter the ongoing outbreaks of COVID-19, a vast number of medications (previously licensed and new drug candidates) targeting various stages of the virus cycle and host immune cells have been considered [21]. In this current review, we discuss the inflammatory response (cytokine storm) in a SARS-CoV2 infection, particularly in the lungs. In addition, we also highlight the currently repurposing of drugs and immunotherapeutic targets against COVID-19. Finally, we have placed a perspective towards nanomedicine by considering the previous investigations ensued in the field of COVID-like infectious diseases.
Inflammatory responses of COVID-19 and lung pathology
The majority of the COVID-19 symptoms and pathological features, at least to some degree, have also been observed in SARS and MERS infections [2], [22], [23], [24], which also come under the category of zoonotic diseases [25].
A macroscopic examination of COVID-19 post-mortem suggests pleurisy, pericarditis, lung contraction, and pulmonary edema [22]. There are also reports suggesting increased lung weight. Computed tomography (CT) scans are considered as the best diagnostic method for COVID-19 infections [26]. The CT scan report of COVID-19 patients showed bilateral lung opacities/infiltrates and hypoxemia with peripheral involvement of lower lobes [22]. Gross pathological observations clearly showed diffuse alveolar damage specifically air sac/alveolar tissue becoming more thickened and scarred permanently due to extensive fibrinogen deposition and associated fibrotic changes (elevation of D-dimer) in the extracellular matrix leading to shortening of breath, pulmonary hypertension and heart failure. Also, numerous pulmonary micro/macrothrombi have been observed, which are repressed by the use of heparin [27], [28] and tissue plasminogen activator [29]. These complex responses cause the lungs to lose their mechanical-stretchable properties, followed by interstitial fibrosis, bronchoconstriction, and other symptoms [2].
Dysfunction of the lungs can be characterized by the shortness of breath, which might be due to several reasons, including lungs occupied with a high amount of interstitial fluid, high content accumulation of collagen, not being able to produce enough surfactant, etc. These typical properties affect the lung tissue elasticity and gas transport (pulmonary circulation and diffusion of gas) functions. Lung histology showed the existence of damaged epithelial and endothelial cells, accumulation of exudates, red-blood cells, granulocytes (neutrophils), innate immune cells (monocytes, macrophages, dendritic cells), and lymphocytes (T helper and cytotoxic T cells) in the alveolar spaces that lead to a reduction in gas exchange function. Due to the inflammatory conditions, the rate of diffusion of gas across the alveoli changes, which leads to a hypoventilation condition in the lungs. During a poor gas exchange state, cells experience a high level of stress, which leads to apoptosis and/or necrosis. The role of neutrophils, such as the release of neutrophil elastases, and others, on lung epithelial cell apoptosis, has been documented in the literature [30]. Type II epithelial cells express several chemokines in response to the infection that recruits neutrophils and other innate cells to the lungs [31].
In general, during the infections, the lung epithelium protects the host by providing coordinated innate and adaptive immune responses [32] ( Box 1). The lung epithelium maintains the dry airspace by regulating the movement of proteins and fluids in and out of the lung. However, in acute infection conditions, like COVID-19, the activation of innate immune cells cross their threshold limit, which results in massive pro-inflammatory cytokine (IL-1β, IL-6, and tumor necrosis factor [TNF]-α) production followed by propagation as severe systemic inflammation. Alveolar macrophages and monocyte-derived macrophages are the primary source for pro-inflammatory cytokines, especially in SARS, MERS, and ARDS. In addition to alveolar macrophages, type II epithelial cells also produce large amounts of pro-inflammatory cytokines [33], [34]. The recurrent inflammatory cascades in the lungs further damage epithelial and endothelial membranes followed by air sac tissue matrix damage. In addition, the accumulation of cytokines, chemokines, and reactive oxygen species-rich fluid, damage the complete alveoli structure and function, resulting in a low level of oxygen supply to the blood. In this condition, intensive care unit (ICU) patients are supported by artificial air ventilators. Although the use of ventilators is the only option for severely affected COVID-19 patients, it has long been known that the continuous use of ventilation induces lung injury via alveoli cell apoptosis, i.e., ventilator-induced/associated lung injury [35]. Therefore, it is essential to use possible adjunct therapies to overcome treatment-induced injuries ( Fig. 1 and Box 2).
Box 1. Lung physiology.
Bronchi start from the trachea and extend to all parts of the lungs via bronchioles. The lung epithelium is comprised of different columns, which consist of mucus-secreting goblet cells, mucus clearance microvilli ciliated cells and cuboidal cells. The alveolar epithelium forms a tight barrier and regulates the movement of fluid and proteins from the circulatory system to the airspaces, and it regulates the entry of pathogenic substance that comes during the inhalation process. The alveolar epithelium contains two types of cells, alveolar type I (principal component) and II cells. Both cell types form a lining to the endothelial basement membrane and primarily the type 1 cell perform diverse functions, including barrier function, fluid reabsorption, gas exchange function, and others.
Nevertheless, type I cells are more susceptible to infections, injury, and cell death. Type II cells (pneumocytes) produce surfactants, which exist in the alveolar lining, which reduce the liquid surface area/surface tension [195]. Type II cells also act as the progenitor for type I cells during injury. The integrity of these two epithelial cells is maintained by the transmembrane proteins, such as claudins, occludins, and others.
Gas (O2/CO2) transport and exchange is the primary function of lungs in the human body. In physiological conditions, the lungs exhibit excellent elastic characteristics of the lung parenchyma and surface tension of alveoli. The elastic properties are acquired by the components, such as elastic fibers (elastin, microfibrils), fibril forming collagens (tensile strength, type I, II, III, V, XI), and geometric arrangement.
In general, during the infections, the lung epithelium senses the pathogens and/or injury and produces pro-inflammatory cytokines, chemokines, and other danger signals, which will recruit leukocytes [195]. Recruited antigen-presenting cells (APCs) also recognize the invading pathogens, and pathogen-associated molecular patterns (PAMP) by pattern recognition receptors (PRRs) followed by the activation of the innate immunity system and recruitment of innate immune cells, which can clear the pathogen via phagocytosis processes. Further, the activated innate immune system helps in the development of adaptive immune responses (both effector and memory) against a pathogen [32].
Fig. 1.
Lung pathology in COVID-19. The lungs of severely ill COVID-19 patients appear to be opaque in the CT scan. SARS-CoV-2 enters via the nose, mouth, or eyes and reaches the alveoli, where a high expression of ACE2 receptors are present. Alveoli exist in the form of balloon-shaped structures. In any lung infection, different cells and substances are involved in protective immunity as well as inflammation. Invading SARS-CoV-2 interacts with, especially, tracheobronchial and alveolar epithelium and subsequently induces damage (apoptosis/necrosis) to the cells. The damage affects the tight barrier integrity of both the endothelium and epithelium layers. The epithelium is composed of a monolayer of alveolar type I and alveolar type II cells, which perform gas exchange and the production of surfactant functions, respectively. These functions keep the air space dry in the lungs. The damaged cells produce danger signals, such as reactive oxygen/nitrogen species, which recruit the innate immune cells, such as monocytes, immature macrophages, neutrophils, and dendritic cells. Upon uncontrolled activation, immune cells, epithelial cells, and fibroblast cells secrete copious amounts of pro-inflammatory cytokines and chemokines, which in turn act as a causative factor for epithelial cell death. In addition, they block the functional Na+/K+-ATPase pump, which keeps the osmotic equilibrium in the alveolus. The impaired tight junctions lose their fluid resistance nature and allow the fluids into the alveolus leading to edematous inflammation, which obstructs the vital gas exchanges process. Note: This hypothetical figure illustration is based on the output obtained from different non-peer reviewed publications and in comparison of other lung diseases, such as ARDS, SARS, MERS, influenza. Abbreviations: ATI, alveolar type I cell; ATII, alveolar type II cell; BASC, bronchioalveolar stem cell; BM, basement membrane; EBM, endothelial basement membrane; ECM, extracellular matrix, NETs, neutrophil extracellular traps; RBC, red blood cell; ROS, reactive oxygen species.
Box 2. Key observations that need to be considered for designing COVID-19 therapeutics.
-
•
The RNA of the virus is protected by a lipid layer/envelope that allows the virus to penetrate the cells of the ocular, nasal, and buccal mucosa.
-
•
Spike proteins are responsible for the entry of the virus.
-
•
Unlike other CoVs, SARS-CoV-2 has a slight difference in their temperature withstanding capacity, humidity and type of material where it lies.
-
•
Observational studies of ABO blood groups on a large population confirmed that Rh (called Rhesus factor) positive and Rh/ABO blood groups are positively and significantly correlating to intubation and death [196]. Likely, the O blood group has a lower risk of infection compared to other blood groups and the B blood group has a higher risk of infection [196], [197]. However, earlier studies on SARS-CoV-1 has shown contrary results [198].
-
•
Although reasons are not known, children are likely to be resistant to SARS-CoV-2 infection and developing severe disease [199], [200]. One of the plausible reasons is the existence of rapid producing, broad reactive and variable affinity natural antibodies [201].
-
•
Pre-existing cross reactive T and B cells (including antibodies) against SARS-CoV-2 have been observed in unexposed healthy donors [202], [203], [204], [205], [206], [207], [208]. Probably, this contributes a clue to develop tailor-made, cross-protective vaccines against SARS-CoVs.
-
•
Age, gender and underlying chronic diseases have played a significant role in mortality by COVID-19.
-
•
Although the S protein is the best target for vaccine development, nucleic acid-based vaccines should not be undermined.
-
•
M protein in all CoV exhibits a N-terminal ectodomain glycosylation site. However, once glycosylated, the M protein becomes inactive. Therefore, enhancing the glycosylation is a possible intervention strategy [209].
-
•
It is still premature to confirm the effect of COVID-19 on diseases such as diabetes, hypertension, cardiovascular disease, cancer, inflammatory bowel disease, and others.
-
•
Substantial data are yet be obtained on the effects of COVID-19 in patients who are under immunosuppressive and/or anti-inflammatory/disease-modifying antirheumatic drugs.
Cytokine storm in COVID-19
SARS-CoV-2 interacts and induces alveoli tight epithelial cell barrier damage followed by lung injury, which is considered as the primary injury. In addition, infection triggers further damage of the tight junctions, production of reactive oxygen species and activates innate immune cells (e.g., macrophages) which worsen the condition. This type of immune cell activation induced damage is termed as the secondary injury, i.e., the inflammatory injury. Regardless of the direct primary injury, secondary injury is widely implicated in many other pathogenic infections, including but not limited to influenza, SARS, MERS, and others [24].
Upon infection (including SARS and MERS), the internalized virus uses host intracellular machinery to propagate while destroying host cells. In result, host cells deliver danger signals that recruit the innate immune cells to attack the virus. However, due to the presence of unseen viral signals, the innate immune system delivers copious amounts of pro-inflammatory cytokines (IL-1β, IL-6, TNF-α, interferon [IFN]-γ, and others), i.e., called the cytokine storm ( Fig. 2), which induces non-selective immunogenic cell death followed by organ failure. The above process has been recurrently observed in the lungs of SARS-CoV-2-infected patients, especially patients in the ICU [36]. Hyper activated lymphocytes are the additional culprits in causing the cytokine storm. Patients with severely ill COVID-19 infection show the existence of hyperactivated CD4+ and CD8+ CD38+HLA-DR+ T-cells. In vitro stimulation of COVID-19 patients’ peripheral blood mononuclear cells with a pool of peptides (resembling SARS-CoV-2 proteome) revealed the existence of SARS-CoV-2-specific CD4+ and CD8+ responses. Upon stimulation with peptides, CD4+ and CD8+ showed activation (CD69 and CD137) and memory (CD45RA- and CCR7+) responsive marker expression with predominantly Th1 biased cytokine secretion [37].
Fig. 2.
Therapeutic strategies for COVID-19. SARS-CoV-2 uses epithelial cells, particularly lung epithelial cells, for their propagation. During the replication process, virus particles induce cell death signals to release pro-inflammatory cytokines and DAMPs, which in turn are sensed by the macrophages, monocytes, and neutrophils followed by the activation of other bystander cells and the development of the systemic cytokine storm. Based on the life-cycle of SARS-CoVs, the above-proposed drugs have been used in clinical trials. Note: As detailed, the immunological responses of SARS-CoV-2 have not been established yet; thus, studies illustrated are in comparison with SARS and MERS. More details can be found in the text and Table 1. Abbreviations: ACE2, angiotensin 1-converting enzyme 2; CTL, cytotoxic T lymphocytes; DAMPs, danger-associated molecular patterns; ER, endoplasmic reticulum; NETs, neutrophil extracellular traps; RNA, ribonucleic acid; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Due to the inflammatory storm that occurs in COVID-19 patients, the development of ARDS results. The initiation of ARDS starts with the recruitment of immature alveolar macrophages at the airspaces in the alveoli; under the influence of growth factors (e.g., granulocyte-macrophage colony-stimulating factor (GM-CSF), cytokines, etc.) they differentiate into mature alveoli macrophages [38]. Upon maturation, they secrete chemotactic factors, such as IL-8 and other chemokines to attract various inflammatory immune cells [39]. However, each immune cell, e.g., neutrophils, macrophages, megakaryocytes [40], and others, have their own distinct role in infectious conditions. In addition, infection signals the recruitment of neutrophils to the edematous space, across the endothelium, interstitium, and epithelium, whereby they activate and perform regulatory functions, such as phagocytosis of pathogens. Comparably, an increased neutrophil count (i.e., neutrophilia) has been observed in the severely ill COVID-19 patients and is accompanied by the severity of the disease [36].
Nevertheless, due to the inflammatory microenvironment, the hyperactivation of neutrophils leads to secondary damage to the alveoli. Mediators of neutrophil-induced lung injury include proteases (neutrophil elastase), oxidants (reactive oxygen and nitrogen species), antimicrobial peptides (defensins), and Neutrophil Extracellular Traps (NET). Indeed, the same progression was observed in other lung injury diseases (ARDS, MERS, SARS, and influenza) [30]. Therefore, strategies, which target recruitment and hyperactivation of neutrophils or their inflammatory mediators, such as antagonism of CXCR2 (neutrophil chemokine receptor), and inhibition of neutrophil proteases, peptidylarginine deiminase IV, DNase-1, and others, would be beneficial for COVID-19 (reviewed elsewhere [30]).
In addition to neutrophils, macrophages have also been implicated in lung injury during pathogenic infections, e.g., influenza. Blocking monocyte-derived macrophage recruitment to the inflammatory site has attenuated secondary injury induced by macrophages [39], [41]. More importantly, pro-inflammatory cytokines, such as IL-1β, IL-6, TNF-α, and others, have a significant role in both primary and secondary injuries. Interestingly, monocyte-derived macrophage release of TRAIL (TNF-related apoptosis-inducing ligand) is one of the causative factors for epithelial cell apoptosis; infected epithelial cells express surface death receptors. It has been reported that SARS-CoV-2-induced lung injury consequently degenerates the spleen and lymph nodes. Among the clinical abnormalities, lymphopenia, or lymphocytopenia, is a primary significant condition observed in COVID-19 patients. Recent tissue histopathological studies of the spleen and lymph nodes from COVID-19 patients revealed that macrophages, especially ACE2 expressing CD169+ tissue-resident macrophages, are also directly infected by SARS-CoV-2. Probably, viruses envisage the capability of macrophages that deal with death [42]. However, unlike MERS-CoV [43], these viruses do not directly infect T and B cells [44]. Upon infection, these macrophages produce pro-inflammatory cytokines (IL-6, TNF-α) and activate lymphocytes. Subsequently, IL-6 has been used, along with other markers, as a potential inflammatory biomarker in COVID-19 to assess the severity and to provide a prognosis [45]. Historically, fatal cases of ARDS, influenza, MERS, and SARS are associated with IL-6 and IL-1β followed by macrophage-activation, and a similar trend has also been observed in COVID-19 [46]. Consequently, the activated lymphocytes exhaust [47] and express a high content of the Fas receptor, which mediates Fas/FasL signaling and induces activation-induced cell death, causing lymphocytopenia [44].
In addition to neutrophil and macrophage induced secondary injuries, recruited dendritic cells, and antigen presentation to the cytotoxic CD8+, T cells also accelerate the inflammatory process. Dendritic cells recruited to the inflammatory site access the virus, process the antigen, and present antigen-derived peptides to CD8+ T cells. Viral antigen-experienced CD8+ T cells clear the viral load by killing the infected epithelial cells through the release of perforin/granzyme-mediated or the Fas ligand process.
In addition to the mentioned irregularities and inflammatory injuries, another factor that causes lung tissue injury is the hypoxia condition. Due to the edematous condition of the alveoli, epithelial cells are more vulnerable to apoptosis, which allows for impaired tissue integrity and their functions (e.g., gas exchange). Lung epithelial cell apoptosis is mediated by several pathways, including a Fas ligand pathway, toll-like receptor (TLR) pathways, intracellular stress-induced, and others [48].
Current repurposed drugs and immunotherapeutic targets available against COVID-19
Unfortunately, at present, there are no newly-developed approved drugs for the treatment of COVID-19. However, knowing about the uncertainty of the efficacy of new drugs/vaccines and the lengthy discovery and developmental timeline, special efforts have been made to repurpose existing drugs including antiviral drugs, immunomodulators, and other adjunct therapies (Fig. 2) [49], [50]. The proposed drugs and their existing targets may or may not work in a similar fashion with SARS-CoV-2. Nevertheless, detailed studies need to be conducted to confirm their possible mechanism of action.
Virus attachment inhibitors
In addition to structural homology, SARS-CoV and SARS-CoV-2 spike (S) proteins exhibit high similarity sequences (nearly 76.5%) [51], [52], which preferentially bind to human ACE2 receptors. Earlier studies on SARS-CoVs have shown that overexpression of ACE2 and the injection of spike proteins exacerbated disease severity and lung injury, respectively. As mentioned-above, SARS-CoV-2 also uses ACE2 for its entry into cells [53]. Henceforward, strategies that hinder the virus-ACE2 interaction have been explored ( Table 1), such as serine protease (TMPRSS2) inhibitors (camostat mesylate) [12], the use of monoclonal antibodies against ACE2, the use of recombinant ACE2 protein (APN01, GSK2586881), and drugs that act on the renin-angiotensin system [52]. Along a similar line, the S protein has gained much attention as a target to develop monoclonal antibodies [54].
Table 1.
Therapeutic strategies for COVID-19a. Below is a list of drugs currently being explored for repurposing against COVID-19.
| Type of therapeutic strategy | Drug candidate | Comment |
|---|---|---|
| Antiviral therapy | ||
| Protease inhibitors | Camostat mesilate, lopinavir/ritonavir, darunavir/cobicistat, ASC09, Danoprevir, Boceprevir, GC376 |
|
| Nucleoside reverse transcriptase inhibitors | Azvudine, Emtricitabine/Tenofovir |
|
| Neuraminidase inhibitors | Oseltamivir |
|
| RNA polymerase inhibitors | Remdesivir, Ribavirin, Favipiravir |
|
| Viral fusion inhibitor | Umifenovir (Arbidol) | |
| Viral endonuclease inhibitor | Baloxavir marboxil (Xofluza) |
|
| Virus attachment inhibitors | Camostat mesylate, Nafamostat mesylate (Fusan) |
|
| Immunotherapies and inhibitors of inflammation | ||
| IL-1 receptor antagonist | Anakinra | |
| Janus kinase inhibitor | Baricitinib |
|
| TNF inhibitor | Adalimumab (Humira) |
|
| Janus kinase inhibitor | Ruxolitinib |
|
| Anti-VEGF | Bevacizumab |
|
| Complement inhibitor | Eculizumab (Soliris, Elizaria) | |
| DAMPs regulators | CD24Fc |
|
| Sphingosine-1-phosphate receptor modulator | Fingolimod (FTY720) |
|
| Viral replication inhibitors | Interferon β1α, Recombinant human interferon α1β, α2β |
|
| Viral entry inhibitors | Meplazumab |
|
| PD-1 antagonist | Camrelizumab |
|
| Antiviral peptide | CSA0001 |
|
| TLR agonists | PUL-042, Polyinosinic-polycytidylic acid (poly I:C) |
|
| IL-6 antagonist | Siltuximab |
|
| IL-6 receptor antagonist | Sarilumab, Tocilizumab | |
| GM-CSF antagonist | Gimsilumab, TJ003234 |
|
| Inhibition of SARS-CoV-2 induced inflammatory innate and adaptive immune responses | Intravenous immunoglobulin (IVIG) | Suppresses the inflammatory responses mediated by hyperactivated innate and adaptive immune cells [176], [177]. |
| Virus neutralization | Immunoglobulin from recovered patients, Anti-SARS-CoV-2 inactivated convalescent plasma | |
| Viral load clearance | NK cell treatment, NKG2D-ACE2 CAR-NK cells |
|
| Non-specific immunosuppressors/immunomodulators | ||
| Cellular therapy | Mesenchymal stromal cells |
|
| Steroid drugs | Corticosteroids |
|
| Anti-rheumatics | Leflunomide |
|
| Calcium channel blocker | Tetrandrine |
|
| Anti-parasitic drugs | Chloroquine/Hydroxychloroquine, Ivermectin |
|
| Antibiotics | Azithromycin, Carrimycin |
|
Abbreviations: CQ/HCQ, (hydroxy)chloroquine; DAMP, Danger-associated molecular patterns; GM-CSF, granulocyte-macrophage colony-stimulating factor; GvHD, graft versus host disease; MS, multiple sclerosis; STAT, signal transducers and activators of transcription; VEGF, vascular endothelial growth factor.
The above-mentioned drugs are currently evaluated in clinical trials against COVID-19 (for more information https://clinicaltrials.gov/).
Virus entry/fusion inhibitors
Although the spike protein in SARS-CoV and SARS-CoV-2 appears to be similar in homology, SARS-CoV-2 binds to different amino acids and shows high-affinity binding towards the human ACE2 receptor; glutamine residue (at 394 position) in the SARS-CoV-2 receptor-binding domain (RBD) interacts with the lysine (at 31 position) on the human ACE2 receptor [11], [52], [55]. On the other hand, SARS-CoV entry is facilitated by host cell proteases, such as elastase, cathepsin B, L and/or TMPRSS2. In addition to the proteases as mentioned above, SARS-CoV-2 exhibits a peculiar furin-like cleavage site, which is processed by the furin at the cell surface and trans-Golgi network (TGN) for entry and egress, respectively [18], [56], [57]. By using molecular modeling, diversified furin inhibitors have been proposed [57].
Antiviral therapeutics for COVID-19
A majority, but not all antiviral drugs come under the class of nucleoside analogs, which mimic the features of nucleic acid and disrupt virus replication. A list of antiviral drugs tabulated here is explored for many infections, even sometimes in human studies, such as HIV, influenza, hepatitis B (HBV), hepatitis C (HCV), and others. Several bioinformatic and/or in vitro studies on cells have been confirmed and proposed to use known antiviral drugs as anti-SARS-CoV-2 inhibitors (Table 1) [58], [59], [60].
Immunomodulation for COVID-19
Despite several reasons for mortality, mortality induced by the immune-compromised condition is confessed, especially in the older age population (~60 years). Various immunoregulatory functions are altered in COVID-19 like pathologies and in addition, the cytokine storm incurs severe damage to the host tissue. Thus, modulation of COVID-19-associated immune responses is mandatory.
Immunostimulatory drugs
A murine model of infection confirmed that CoVs modulate host immune responses by delaying type I interferon (IFN-I) to avoid the detection process [61], e.g., after infection, non-structural proteins (nsp; particularly nsp15) play a significant role in the inhibition of IFN-I responses, which is essential for the innate immune system to clear the virus-infected cells [62], [63]. Interestingly, the highest similarity (95%) of this protein was observed in SARS-CoV-2 as well with that of SARS-CoV [64], which raises the possibility to use novel approaches including TLR agonists, IFN agonists, and others (Table 1).
Immunotherapies that target inflammatory cytokines or hyperactivated immune cells
Antagonists to inflammatory mediators are widely used in autoimmune diseases in which hyperactivated immune cells damage different organs in the body. From the past few decades, autoimmune disease associated therapeutic interventions are accompanied by the inhibition of cytokines, including TNF, IL-17A, IL-6, IL-23, and others or blockade their receptors like IL-6R [65], [66]. Through this strategic targeting, the infiltration of monocytes, macrophages, and lymphocytes is regulated. Similarly, as explained earlier, COVID-19 infection also has shown hyperimmune activation; thus, the aforementioned strategies are useful as an adjunct therapy [67], [68]. Several clinical studies identified that pro-inflammatory cytokines are the causative factors for severe lung damage and coagulopathy observed in COVID-19 patients, particularly, predominant levels of IL-6 and TNF (Table 1) [4]. IL-6 and TNF-α antagonists have been used in the treatment of cytokine release syndrome. Thus, many clinical studies (Table 1) have been initiated with the same hypothesis.
Many other possible immunotherapeutic strategies (Table 1), such as passive immunotherapy to neutralize SARS-CoV-2, regulatory T cell-targeted therapies, cellular immunotherapies, immunomodulatory therapies and others, for COVID-19 have been recently reviewed [67], [68].
Other adjunct therapies for COVID-19
Renin-angiotensin system inhibitors
As explained above, ACE2 is expressed in many tissues. ACE2 plays a vital role in the renin-angiotensin system (RAS), especially in controlling electrolyte balance and blood pressure. Further, ACE2 also extends its role in regulating ARDS and virus-induced lung infections [69]. It is believed that a SARS-CoV-2 infection reduces the expression of ACE2, thereby disrupts the RAS. A study conducted on the use of ACE inhibitors and angiotensin-II type 1 receptor antagonists either alone or in combination with other antihypertensive drugs against COVID-19 have shown beneficial effects, such as a reduction in IL-6, and an increase in the CD3 and CD8 population, and also a reduction in the overall viral load. On top of that, the said results have not been observed in other anti-hypertensive drugs treatments [70]. However, a detailed mechanism of the efficacy has yet to be investigated. Considering the same mechanism, a few clinical trials are ongoing (NCT04353596, NCT04330300, NCT04367883, and NCT04345406).
Apart from the therapeutics/drugs mentioned in the table, other diversified drugs (including antimalarial drugs, antibiotics, antioxidants (α-Lipoic acid) [71], angiotensin-II type 1 receptor antagonists, vasodilators, and anti-coagulants) are being explored in clinical trials (reviewed elsewhere [72]).
Nanomedicine
Despite the wide availability of therapeutics, the use of various nanomedicine strategies has been successful in treating many ailments; precisely, however, its use in pulmonary drug/therapeutic targeting as an adjunct therapy awaits. Nanoengineering with potential drugs opens up the possibilities for improved treatment strategies in lung infections. Pulmonary nano-drug delivery systems offer unique physicochemical properties including mucosal penetrability, ease of ligand functionalization, enhanced permeation due to small size, increased local concentrations of drugs and high adjuvant properties for vaccine applications, which makes them ideal drug delivery systems for the treatment of COVID-19-like pulmonary infections ( Fig. 3).
Fig. 3.
Nanomedicine based strategies to prevent the pathologies associated with respiratory infections. A variety of viruses from different sources are responsible for respiratory infections. A few viruses, such as rhinovirus, parainfluenza virus, coronaviruses, adenoviruses, coxsackievirus, respiratory syncytial virus, herpesvirus, bocavirus, and others, particularly infect the upper respiratory tract. On the other hand, avian influenza virus, parainfluenza virus, respiratory syncytial virus, bocavirus, adenoviruses, metapneumovirus, and others infect the lower respiratory tract. In both the cases, they cause the common cold, bronchitis, bronchiolitis, and sometimes-severe pneumonia. Furthermore, the infection results in the dysfunction and damage to the other vital organs. Nanomedicine-based strategies that are explained above target the pathologies and reduce the severity of the disease. Abbreviations: NP, nanoparticle.
Do nanomedicine interventions play a vital role in the management of COVID-19-like pulmonary infections?
Regardless of the latest advances in therapeutic modalities for various pulmonary infections, there is still an unmet medical need to bring advanced nano-targeted therapeutics to the clinic. ARDS is one of the significant causes of morbidity and mortality across the world, characterized by acute, diffuse and inflammatory lung injury, which ultimately leads to refractory hypoxemia [73], [74]. Patients with ARDS combat multiple organ failure as they do not tolerate off-target side effects of various drugs. Besides, ARDS-associated infections are allied with heterogeneous pathophysiological lung environments [75], [76]. The main drawbacks of conventional pulmonary drug delivery are the lack of alveoli mucosa penetrability, lack of effectiveness of non-targeted delivery by a systemic route and association of multiple targets at a single therapeutic site. The past decade's rise in nano-research is now transforming into considerable commercialization attempts around the globe, and an increasing number of nanomedical therapies are now approved by the food and drug administration (FDA) and other agencies [77], from the first approved nano-pharmaceutical (Doxil® liposomal form of doxorubicin) in 1995 [78]. Nanoengineering with potential drugs opens up possibilities for improved treatment strategies in lung infections. Besides, the unique and/or abnormal signaling features of the pulmonary endothelium, including vasodilatory molecules (NO, prostacyclins) and enhanced expression of leukocyte-binding molecules (like ICAM and VCAM) [79] may play a potential role in nanocarrier binding for reducing ARDS-associated pulmonary embolisms (PE) and pulmonary arterial hypertension (PAH) diseases.
Nanoparticles have shown significant potential advantages for the delivery of antiviral molecules [80], [81]. In addition, nanoparticles display direct antiviral activity. Up to date, various forms of nanoparticle systems have been reported to exhibit antiviral activity including silver nanoparticles, functional gold nanoparticles, and quantum dots [82]. For instance, the widely used strategy by most of the 'antiviral' nanoparticles include blocking the viral attachment or viral entry into the host cells. Unfortunately, at the latter stage, the 'antiviral' nanoparticles have no suppressive effect on the progeny of the virus in their late phase of replication. In addition, despite challenges [83], [84], numerous studies have reported on nanoparticle based drug therapies against respiratory tract infections, e.g. selenium-, sliver-, PEG-PLGA-nanoparticles against the influenza (H1N1) virus. These nanoparticle based systems have shown an increased therapeutic efficacy and reduced associated toxicity compared to free drugs (reviewed in [85]). Interestingly, among these features, their mucoadhesive property presents a significant priority in designing nanoparticle-based therapies against respiratory infections; this could delineate the capacity of targeted delivery to the lungs. To achieve this high mucoadhesive property, several modifications of nanomaterials via various functionalization mechanisms have been developed (Fig. 3) [81].
Development of these disease-responsive nano delivery systems originated by considering their unique interactions, such as hydrogen bonding, hydrophobicity, ionic bonding, and others. Also, selective functionalization, including thiols, catechols, acrylates, and others, have proven their increased covalent interaction with mucin glycoproteins. Furthermore, polymers, such as cellulose, chitosan, pectin, alginate, and others have also bound in a noncovalent manner [84]. Among these polymers, chitosan nanoparticles have shown encouraging results for targeted drug delivery. Bioavanta-Bosti, a pioneer company in chitosan nanoparticle science, has begun a proof-of-concept study on a chitosan-based aerosol system namely “Novochizol aerosol". This system is used for the delivery of drugs like losartan, valsartan, telmisartan, and digoxin, which may play a potential role in the battle against the coronaviruses [86] Chitosan can be seen as a major player since it has unique mucoadhesive properties due to the interaction between positively charged chitosan and negatively charged mucus [87]. This is an effective way to improve the retention time of the drugs in the lungs, and hence might have enhanced clinical efficacy through prolonged lung retention.
Rationale applications of nanomedicine against coronaviruses
One of the possible strategies to fight against SARS-CoV-2 could implicate the prevention of viral entry into the host cell. It is well known that blockage of viral surface proteins can lead to virus inactivation, so that targeted nanoparticles specific to virus-expressed proteins, could minimize viral internalization. In our latter portion, we have unfolded the role of metallic and metallic-based nanoparticles in blocking viral proteins and, thus, preventing possible viral entry. Another way to deliver antivirals (such as zidovudine, acyclovir, dapivirine, and efavirenz) is to enhance drug bioavailability by using organic nanoparticles (such as albumin and gelatin) and virus-specific targeting proteins (reviewed elsewhere [85]). This approach would provide a dual benefit as it can lead to the successful delivery of drugs along with targeted antiviral activity.
Nanomedicine in immunotherapy, vaccines and vaccine adjuvants
Many flu (influenza) vaccines, such as Inflexal® V, Influvac® Plus, and others, were approved for clinical use against influenza infections [85], [88]. However, the use of vaccines is an excellent strategy to combat COVID-19 like diseases, their discovery time-line, stability, and delayed mechanism of action (production of immunological memory against target antigens) making them a second choice of therapies. Nevertheless, therapeutic and prophylactic strategies are under development to address current and potential future coronavirus infections, including effective vaccines. As previously discussed, the essential genomic match between SARS-CoV-2 and other coronaviruses help the pharma industry and academic institutions design appropriate vaccine candidates.
The development of COVID-19 vaccine candidates relyies on several high-tech platforms including attenuated and inactivated viruses, replicating and non-replicating viral vectors, DNA and mRNA, virus-like particles, and recombinant protein-based approaches. Some platforms offer vital advantages, such as viral vectors, which offer strong immune responses, superior protein expression, and prolonged stability. On the other hand, DNA or mRNA offer antigen manipulation flexibility [89]. In contrast, the recombinant protein-based development approach is more comfortable for scaling up vaccine doses using existing production capabilities. To date, 213 vaccines are in development, and 35 are in clinical testing [90]. The main strategy for most of the vaccine candidates is to induce antibodies against the viral S protein, averting the ACE2-mediated host uptake. In the case of SARS-CoV vaccine development, higher antigen-specific antibody titers and better protection were reported with the S protein subunit vaccines when compared to any other target strategy. SARS/MERS vaccine development research has suggested that S protein subunits and RBD of the S1 subunit as the most preferred target sites [91]. As complete knowledge on SARS-CoV-2 specific antigens is limited, traditional vaccinology methods in combination with nanotechnology would be beneficial.
Recent data from a team of researchers at UC San Diego highlighted the role of nanotechnology in COVID-19 vaccine development [90]. From a vaccine engineers’ point of view, nanomedicine offered them an ideal platform for the delivery of antigens, serving as adjuvant platforms, and mimicking viral structures. As cellular immune responses are essential to combat viral infections, vaccines and vaccine adjuvants, which elicit Th1-biased immune responses, they are preferred in COVID-19 like diseases. It is important to note here that in most of the cases, humoral immune responses have been observed for most adjuvants and delivery platforms while only viral vectors, immune stimulating complexes (ISCOMs) and Montanide™ have shown cytotoxic T cell responses in the clinic [92]. Recent reports indicate that nanoparticles allow multivalent antigen presentation and stabilization of antigens upon administration. They can also serve as adjuvants for boosting immune responses and as carriers for targeted antigen delivery [93]. Indeed, an mRNA vaccine delivered by a liposomal nanoparticle is amongst the best candidate currently awaiting regulatory approval against SARS-CoV-2 [94].
Among the different types of vaccines, subunit and DNA/RNA vaccines have gained much attention, due to their fewer side-effects, low cost, and ease of preparation [95]. Often subunit vaccines require vaccine adjuvants and substances/compositions that increase the immunogenicity of the targeted antigens. More information can be found in our recent review [92]. Further, the stability and tissue-specific targeting issues are limiting their success [93]. Therefore, the use of nanomedicine has become an attractive and quick strategy to address these limitations, mainly needed for COVID-19 like pandemics. For example, we have used an oleic acid nanoemulsion system to increase the antigen uptake capacity of the mucosa (in the nasal epithelial). These featured nanomaterials would be advantageous to use as targeted delivery vehicles for the respiratory tract [96].
Few exciting studies have been documented on nanotechnology-based vaccine developments against SARS-CoV-2. Erasmus et al. developed an Alphavirus-derived replicon RNA vaccine candidate (repRNA-CoV2S), which is composed of the SARS-CoV-2 spike protein replicons in the squalene-based emulsion of lipid inorganic nanoparticles (LION) [97]. Superparamagnetic iron oxide (Fe3O4) and cationic lipid 1,2-dioleoyl-3-trimethylammonium propane were used as an inorganic and lipid source in the nanoparticles, respectively. Immunization of this vaccine in mice and macaques produced antigen-specific antibody responses, which were comparable to the convalescent response from COVID-19 [97]. The superiority of the vaccine composition is that it elicited robust long-term antibody responses against SARS-CoV-2 upon single-site intramuscular injection. With squalene, a gold standard oil-based vaccine adjuvant, in the nanoparticle composition, it is anticipated to induce Th1-mediated cellular and robust antibody responses [92]. Besides, taking into consideration that older people are more prone to SARS-CoV-2 infections, the authors have studied the vaccine candidate responses in young and old mice. Vaccine candidates produced SARS-CoV-2 neutralizing antibodies effectively with and without multiple injection schedules. Furthermore, their two-vial approach, which contains a LION in one vial and repRNA in another vial, are easier to scale up than the complexed formulations [97].
In addition, carbon-nanomaterials have also been explored recently as novel anti-viral agents owing to their unique physicochemical properties. Garg et al. proposed conceptual insights into a series of bioisosteres derived from triazole functionalized heteroatom co-doped carbon quantum dots (TFH-CQDs) and designed peptide inhibitors to combat human coronavirus either by blocking viral entry or inhibiting the viral enzymes for replication, such as helicase and 3CLpro [98].
NVX-CoV2373 is another COVID-19 nanoparticle vaccine candidate [99], which is composed of trimeric full-length recombinant spike glycoproteins of SARS-CoV-2 and Matrix-M1 adjuvant (the combination of different purified fractions of saponins from the tree Quillaja saponaria Molina with cholesterol and phospholipid) [100]. Preclinical studies have shown its potency by eliciting anti-spike IgG antibodies with hACE2 receptor blocking, and virus neutralization capacity. Besides, it induced both T and B cell responses [99]. By considering the above merits, the vaccine candidate has been taken to clinical evaluation (Phase 1–2; NCT04368988). The safety and immunogenicity studies of this vaccine was conducted on 131 healthy adults [101]. Results revealed that the vaccine is safe and induced optimal immune responses. Preferably, the said adjuvant should enhance a Th1 polarized response. The vaccine benefited from the adjuvant in terms of dose sparing and an enhanced immune response [101].
Huan et al. studied the CoVaccine HT™ adjuvant effect against the SARS-CoV-2 spike S1 protein in mice [102]. The CoVaccine HT™ is an oil-in-water emulsion type, which is composed of negatively-charged sucrose fatty acid sulfate ester and squalene (source: plant-derived) [103]. Importantly, this vaccine adjuvant has proven its potency and efficacy in different vaccine formulations, including malaria, ebola, Zika, and others [104], [105], [106]. For comparison of adjuvant activity against SARS-CoV-2, alum (a gold standard in the family of adjuvants) and a Th2 adjuvant, were used in the study. The CoVaccine HT™ induced high antigen-specific antibody titers, class switching response, cell-mediated immune responses and virus-neutralizing antibody titers, which are more significant than the alum. One of the recent studies by Rao L et al., utilized the nano biotechnological approach to formulate nanodecoys with cellular membrane nanovesicles derived from genetically edited 293 T/ACE2 and THP-1 cells. The nanodecoys with biological properties have abundant ACE2 and cytokine receptors, which compete with host cells and significantly inhibited viral replication and infection [107]. Of note, a detailed discussion of nanotechnology-based vaccine development against COVID-19 has been covered in recent reviews [108], [109], [110], [111], [112].
Nanomedicine as therapeutic agents
As explained earlier, being the prime receptor of SARS-CoV-2, ACE2 has become a vibrant target for most therapeutic and prophylactic strategies. Recently, Wang et al. developed a biocompatible, cell membrane derived nano-antagonist particulate system prepared from ACE2-rich cells by a simple, cost-effective membrane extrusion method. These synthesized particles termed as HEK-293T-hACE2 NPs contained 265.1 ng/mg of ACE2 on the surface of the particles and acted as a plug to trap SARS-CoV-2 S1 in a competitive dose-dependent manner. Being highly interactive to ACE2, it inhibited SARS-CoV-2 pseudovirus entry into renal tubular cells [113]. Thus, it is clear that nanotechnology could support the conception of therapeutic tools for inhibiting viral and host interactions.
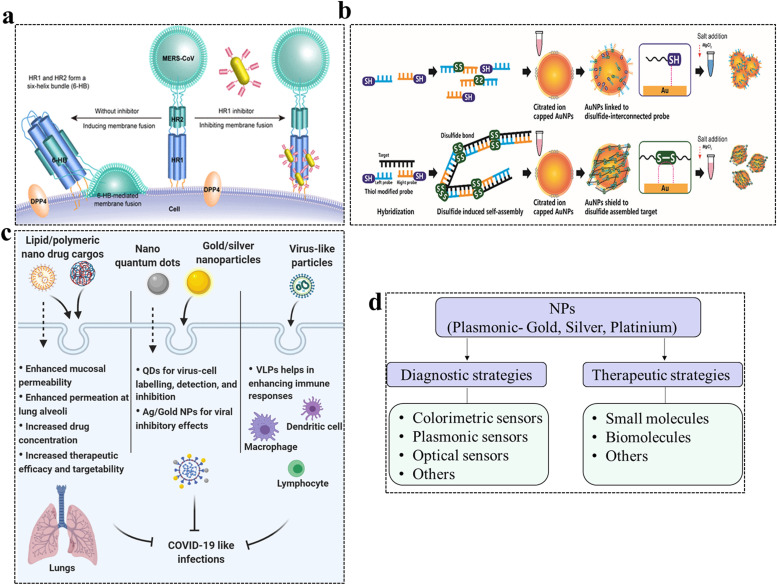
In an effort to improve the next generation of nano-engineered delivery mechanisms, research has been focused on incorporating therapeutic benefits in order to provide safe and efficient nano-platforms to combat COVID-19-like and other types of pulmonary infections. The success of nanomedicine as an antiviral therapy has been already proven in COVID-19-like pulmonary infections. One such example is Middle East Respiratory Syndrome Coronavirus (MERS-CoV), which has been identified as a virus with high pathogenicity and mortality rate [114]. MERS-CoV can cause severe respiratory illnesses and is recognized as a severe threat to public health. Currently, there has been no vaccine or effective treatment for MERS, and its treatment mainly relies on supportive measured and combination therapy of traditional antiviral drugs, such as interferon and ritonavir [115]. However, neither of them showed good antiviral effects in patients, leading to limited therapeutic applications. One of the possible reasons for an increasing number of MERS-CoV infections was due to spike protein (S protein)-mediated membrane fusion between MERS-CoV and host cells [116], [117], [118]. Recently, Ning and his team developed a novel gold nanorod-based peptide inhibitor to fight against MERS. The peptide showed good inhibitory activity against the S protein-mediated membrane fusion, and more importantly the designed gold nanorods enhanced inhibitory viral activity. The peptide-functionalized gold nanorods improved biostability and biocompatibility and had better physical and pharmaceutical profiles than those of the peptide alone, endowing potential clinical applications for the treatment of MERS (see Table 2 for more information, Fig. 4) [119]. Though nanotechnology offers a wide range of diagnostics techniques, SARS-CoV-2 specific diagnostic methods (e.g., biosensors) have yet to be fully developed [120], [121].
Table 2.
Expertise gained in nanomedicine approaches for pulmonary infections.
| Product/Formulation type | Indication | Therapeutic observations | Comments |
|---|---|---|---|
| Lipoquin® | Local antibiotic treatment for lung infections | Ciprofloxacin unilamellar nano liposome nebulized delivery system provides the reduction of dose frequency to a once daily treatment while maintaining a high local concentration in clinical evaluations compared to larger size liposomes [122]. | Commercial nano liposomal products for lung delivery are intended to increase the permeation, intra cellular drug enhancement for pulmonary infections, and these approaches provide insights for future therapeutics targeting systems with enhanced therapeutic response to fight pulmonary infections. |
| Arikayce® (liposomal Amikacin) | Non-tuberculous Mycobacterial Lung Disease | Liposomal Amikacin system is shown to increase intracellular drug concentration and overall therapeutic improvement [123], [191]. | |
| Ambisome® (liposomal amphotericin B) | Allergic bronchopulmonary aspergillosis | Currently in phase 2 clinical trials for Allergic Bronchopulmonary Aspergillosis treatment (ClinicalTrials.gov Identifier: NCT02273661). | |
| Liposomal amikacin for inhalation (LAI) | Bronchiectasis (Phase II clinical trial) | LAI produces improvement in sputum conversion with limited systemic toxicity in patients with refractory MAC (Mycobacterium avium complex) disease [124]. | Strategies with liposomal based nano or emulsified systems had high lung mucosa penetration capabilities. These strategies help in drug deposition with high retention in airways; these investigations provide clues for future COVID-19 like pulmonary infection associated drug delivery applications. |
| Tobramycin nano lipid carriers (NLCs) | P. aeruginosa infections associated with cystic fibrosis | Drug NLCs combat P. aeruginosa infection and also increase in vivo mucus penetration [192]. | |
| pGM169 drug with GL67A lipid solution | Cystic fibrosis (Phase IIb clinical trial) | Increased drug deposition and retention in the proximal airways [193]. | |
| MERS‐CoV S‐containing nanoparticles | Recombinant MERS-CoV S nanoparticle vaccine and Matrix-M1 adjuvant combination as a vaccine | MERS-CoV S nanoparticle vaccine produced high titter anti-S neutralizing antibody and protected mice from MERS-CoV infection in vivo [125]. | Introduction of vaccinated with MERS-CoV S nanoparticles developed a MERS-CoV neutralizing antibody response targeted to MERS-CoV S. |
| Immunoprophylactic strategy with protein cage nanoparticle (PCN) | Pulmonary viral infections including influenza viruses, a mouse-adapted SARS-coronavirus | Pulmonary instillation of PCN dramatically enhanced the subsequent host immune responses to primary viral infections of the lungs [126]. | Like liposomal strategies, polymeric nano systems also enhance localization of drug in the lung regions for a better therapeutic response. |
| PS-341 loaded PLGA-PEG NPs | Cystic fibrosis (Preclinical study in mice) | Enhanced drug localization with NPs to the lungs, helps in reducing immunosuppressive side effects resulting from PS-341 systemic administration over 11 days [127]. | |
| PRINT® (Particle Replication in Non-wetting Templates) technology | Influenza vaccine | To deliver influenza vaccine antigens, poly (lactide-co-glycolide) PRINT particles were designed to bind to a commercial trivalent injectable influenza vaccine electrostatically [194]. | Increased understanding of the delivery of the vaccine antigen. This approach has increased vaccine effectiveness and reduces the amount of antigen necessary to induce an immune response. |
| Gold nanorod-based heptad repeat (HR1) peptide inhibitor | Middle East respiratory syndrome coronavirus (MERS-CoV) | Pregnancy-induced hypertension (PIH), is a potent HR1 inhibitor and can selectively inhibit MERS-CoV S with an IC50 value of 1.171 μM. The developed pregnancy-induced hypertension (PIH) gold nanorods (PIH-AuNRs) exhibited a 10-fold higher inhibitory activity than PIH and can completely inhibit cell fusion at 1.171 μM with good biostability, excellent targeting ability and minimum off target effects. Therefore, PIH-AuNRs are a promising antiviral agent and may have a huge impact on developing pharmaceuticals in the clinic [119]. | HR1 peptide inhibitors have been developed to inhibit HR1/HR2-mediated membrane fusion between MERS-CoV and host cells, which is the major pathway of MERS-CoV-induced host infections. |
| Virus like nanoparticles (VLPs) | Enhancing or triggering the strong immune response in pulmonary infections | Virus mimicking empty particulate structures [128], [129]. | Hollow virus like particulate structures which lack in genetic material will provoke or enhance a strong immune response to combat lung associated infections. |
Fig. 4.
Diversified application of nanomedicine in combating respiratory infections. a) Schematic diagram of the inhibition of MERS-CoV S2 subunit-mediated membrane fusion with HR1 inhibitors. HR1 inhibitors can inhibit HR1/HR2 complex (6-HB)-mediated membrane fusion and prevent MERS-CoV infections. b) Strategy to detect the corona infection by colorimetric detection of double stranded DNA based on disulfide-induced self-assembly and shielding of AuNPs from salt-induced aggregation. In the absence of targets (virus), salt induces aggregation of AuNPs. c) Mechanisms of action of different nanoparticles. The design and use of nanomedicine approaches help in enhancing the delivery system targetability and therapeutic efficacy in lung-associated infections. The drug loaded nano vehicles can be passively or actively targeted to the pulmonary epithelium to enhance the permeation and localized drug release thereby reducing associated side effects. Inorganic nano-systems are useful in the diagnosis of virus infections and also have inhibitory effects on the virus. The virus like particle systems (VLPs) enhance the immune response to combat lung-associated infections. d) Strategies to combat the COVID-19-like respiratory infectious diseases. Nanomedicine can play a potential role in the diagnostic and therapeutic of COVID-19 like diseases. NPs are useful for the development of different sensors to detect SARS-CoV-2-like infections and, thus, can be used for an early real-time detection of a virus with precession. A therapeutics approach at the moment is based upon post functionalization strategies by using different biomolecules and small molecule inhibitors to prevent the entry of the viruses inside the host cells and to block viral replication. Abbreviations: AuNPs, gold nanoparticles; COVID-19, coronavirus disease 2019; DDP4, dipeptidyl peptidase four receptors; 6-HB, 6-helix bundle; HR, heptad repeat; QDs, quantum dots; MERS-CoV, Middle East respiratory syndrome-related coronavirus; NPs, nanoparticles; VLPs, virus-like particles.
(a) Adapted from ref [119]. Printed with permission from “Copyright 2019 American Chemical Society”. (b) Adapted from ref [141]. Printed with permission from “Copyright 2019 American Chemical Society”.
To deliver antibiotics locally with increased therapeutic efficacy, various nano-engineered approaches, such as liposomal formulations, have been reported for lung delivery [122], [123], [124], which are intended to increase permeation and intracellular drug delivery. Such strategies provide insights into future clinical targeting systems with improved therapeutic responses to fight pulmonary infections.
Interestingly, the MERS-CoV spike (S) protein is responsible for receptor binding and entry into cells. It is an immunodominant antigen and induces neutralizing antibodies in the host. Both of these characters make the S protein a perfect candidate for anti-MERS-CoV vaccines. Coleman et al. reported that MERS-CoV S nanoparticle-based vaccination in mice induces a higher neutralizing antibody response to the S protein and protects the mice against a viral challenge [125]. Such functionalized nanoparticle formulation systems could help to pave the way to develop new platform strategies to combat COVID-19-like respiratory diseases.
In addition to the liposomal strategies, polymeric nanosystems [126], [127] also enhance the localization of the drug in the lung regions and boost the therapeutic response. In an attempt to improve immunization approaches to strengthen immune responses, hollow viruses, such as particulate matter constructs (virus-like particles-VLPs) deficient in genetic material, could induce a robust immune response or improve the response to lung-related infections and are worth exploring in the immediate future for specific pulmonary infections [128], [129].
Semiconductor nanocrystals, such as quantum dots (QD), have also offered a wide range of peculiar properties attractive for treating viruses, including size-dependent optical and electronic properties [130]. Due to their distinguished luminescent properties, such as broad excitation spectroscopy, narrow and bright emission spectroscopy, long fluorescence lifetime, and size-dependent emission wavelengths, QDs have indeed showed hopes for virus-cell labeling, detection and image tracking. By using a pig coronavirus porcine epidemic diarrhoea virus (PEDV) as a model, recently Du et al. have demonstrated that Ag2S nanocrystals (NC) have an excellent viral inhibitory ability with a different mechanisms of action from other functional nanoparticles [82]. In addition to the inhibition of viral negative-strands generation and viral budding, Ag2S NC has also induced IFN-stimulated genes and the expression of several pro-inflammatory cytokines [82]. Moreover, the Ag2S NC were also shown to have comparable virus inhibitory effects on other RNA viruses, such as porcine reproductive and respiratory syndrome virus (PRRSV). Therefore, Ag2S NC has been identified to have broad-spectrum antiviral properties against RNA viruses [82]. Recent studies have shown that silver nanoparticles display antiviral activity against influenza A virus, hepatitis B virus, human parainfluenza virus, herpes simplex virus, and human immunodeficiency virus [131], which provide other types of therapeutic opportunities for combating pulmonary-associated infections. Recent reports describe the antiviral activity of silver or gold nanoparticles against DNA or RNA viruses. The possible mechanism of silver and other metal plasmonic nanoparticles is shown in Fig. 4. Apart from functionalization and targeting groups present on the nanoparticle surface, the mode of action is also dependent on the shape and size, which could interact with virus particles with a well-defined spatial arrangement. In recent years, scholars have paid more attention to the use of traditional medicine for fighting viruses [21]. The antiviral effects of traditional medicine might be associated with inhibiting the replication of viruses. Besides, they might improve respiratory virus-mediated lung damage [132].
Over the years, textile industries have been using different inorganic/metal nano-composites (alginate, copper, gold, zinc, magnesium, silver, titanium, and others), which would protect textiles from diversified microbes [133]. Although inorganic reagents have been used as broad-spectrum antimicrobial compounds, the drawback of their instability limits their use, e.g., silver vigorously reacts with Cl-, HS-, and SO4 2-, and others lose antimicrobial capacity [133]. To overcome this, a nanotechnology-based strategy, which increases the stability of the silver has gained much attention. Recently, silver nanoparticles prepared with the acrylic-based polymers in polycotton fabrics have shown a significant antimicrobial effect, including SARS-CoV-2 (SARS-CoV-2/human/BRA/SP02cc/2020-MT350282) [134]. The possible mechanism behind the antimicrobial, and in particular antiviral effect is explained by two pathways. First, sliver nanoparticles prevent the viral attachment to the host cells by interacting with virus surface glycoproteins via a sulfur linkage. Second, these nanoparticles interact with transcription factors, which are required for virus replication. However, the exact mechanism is yet to be established [134].
Du et al. have developed a unique one-step method in which they have used curcumin to prepare uniform and stable cationic carbon dots (CCM-CDs) with antiviral properties [135], [136]. Curcumin has already proven its effectiveness against various infections and exhibits antiviral activity by reducing viral RNA expression, protein synthesis, and virus titers. In addition, it was found to have a protective effect on cells against virus-induced apoptosis and cytopathic activity [137]. The authors studied the inhibitory performance of CCM-CDs on viral replication. In their study, they have selected PEDV as a coronavirus model. Most importantly, their findings suggest that the developed CCM-CDs inhibit the proliferation of PEDV with a much higher efficiency than non-curcumin modified carbon dots ( Fig. 5) [136].
Fig. 5.
The effect of EDA-CDs on PEDV. (a) The effect of different concentrations of EDA-CDs on PEDV-infected Vero cells by indirect immunofluorescence assay. Scale bar: 100 µm. (b) The titers of PEDV when exposed or unexposed to 125 µg/mL EDA-CDs or CCM-CDs. All error bars were determined according to the three replicate experiments. ** p < 0.01 and indicates superior antiviral activity of CCM-CDs to EDA-CDs treated and untreated, against PEDV. (c) Virus titers were calculated in the presence and absence of EDA-CDs or CCM-CDs. Pictures were taken at 12 hpi. Abbreviations: CCM-CDs, curcumin carbon dots; EDA-CDs, ethylenediamine carbon dots; PEDV, porcine epidemic diarrhoea virus.
Figure is reproduced from ref [136]. Copyright 2018 American Chemical Society.
Besides, nanotechnology applications have advanced to develop personalized patient management, including preparation of naso-delivery vehicles (for the easy administration of drugs by an intranasal route), insufflations (to deliver the drug directly to lungs), and others. The use of inorganic-polycotton fabrics (explained earlier) has been one of the best strategies for contagious diseases [134]. In order to combat the spread of coronavirus, the use of adequate personal protective equipment (PPE), including masks, gloves, medical aprons and kits, are also necessary [138]. Antimicrobial activity is imparted by the use of inorganic based nanoparticle coatings, which could be used in various types of fibers or materials, and also fabricating such cloths with anti-viral agents could be an interesting approach to minimize the spread of the virus. Moreover, there is a wide scope to consider the novel nano-strategies to combat emerging microbes, e.g., development of face/mouth masks, which are composed of stable and safe antimicrobial inorganic composites (with antibiotics/antiviral coatings, e.g., silicon nitride) [139], [140]. Although these nano-inorganic composites exhibit less allergic responses, detailed toxicity parameters should be sensibly evaluated. As composites are in the nano-range and there is a chance of ingestion and toxicity due to the accumulated composites.
Future nanocarrier research with specific ligand functionalization to target the aforementioned molecules are likely to enhance their therapeutic efficacy by avoiding off-targeted drug side effects. The advantages of their large surface-to-volume ratios, the high surface area activity and the size-dependent optical and electronic properties of the nanocarriers, which are widely explored in the field of biosensors and biomedicine, and their viricidal activity, have also been systematically investigated. All of these properties make nanoparticles a strong potential weapon to fight against COVID-19-like pulmonary infectious diseases in the near future.
Conclusion and future outlook
The complete genome sequencing of SARS-CoV-2 revealed a close homology with other CoVs, including SARS-CoV and MERS-CoV. In particular, viral enzymes such as 3CL protease, helicase, papain-like protease, and RNA-dependent RNA polymerase and their active sites are highly conserved across other CoVs. However, a better understanding of the SARS-CoV-2 tropism would benefit therapeutic purposes. In addition, the controversial effects of immune cells on secondary injuries warrant further investigation. Finally, many other endogenous protective factors that help in preventing tissue injury and innate repair functions are all unanswered questions that need to be explored further. The use of immunomodulatory therapies may not clear the virus load but have the potential to reduce the severity of the disease caused by the cytokine storm, details that we emphasized on the 'cytokine storm' should also support future vaccine research on COVID-19.
Until today, a few questions remain unclear including the best suitable therapeutic strategy to fight against COVID-19 and associated lung infections. The past decade, however, has witnessed the use of several combination strategies during numerous pandemics and other viral infections like HIV and influenza. Therefore, the use of a broad-spectrum antiviral agents in combination is advisable against COVID-19 and nanomedicine is primed to be central for any solutions due to its attractive properties as mentioned above. Currently, the scientific community is globally juggling many drugs to find suitable and/or manageable therapeutic options against COVID-19. Despite initial encouraging preliminary results in short cohorts, a tremendous effort is needed for targeted approaches, however, the time spent now will help with all future pandemics as nanomedicines can be a platform for numerous viruses, requiring simply to be functionalized with different regions that target different viruses. Worthwhile bioinformatic studies have come up with several druggable proteins and repurposing drugs, and rationalization of these drugs for the future use would be prudent [58], [59].
CRediT authorship contribution statement
Conception, S.R.B., T.J.W. Literature Survey and Writing, S.R.B., N.G.K., and R.A.B. Editing and Reviewing, T.J.W., Y.R., and J.B.
Final Approval, All the authors.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
JB acknowledges the support of Agence Nationale de la Recherche, France under the call "Flash COVID-19" (ANR-20-COVI-0093-COVIMUNE) and the European Union Horizon H2020 programme under Grant Agreement No. H2020-SC1-2019-874653-INDIGO and the Department of Biotechnology (DBT), Government of India. RB is supported through the Irish Research Council under the Government of Ireland Postdoctoral fellowship grant - GOIPD/2017/1283. NK, RB and YR also acknowledge the research grant from Science Foundation Ireland (SFI) co-funded under the European Regional Development Fund under grant Number 13/RC/2073. The figures were created with the support of BioRender.com under a paid subscription.
Biographies

Srinivasa Reddy Bonam is an immunopharmacologist. He obtained a B. Pharmacy and M. Pharmacy from Chalapathi Institute of Pharmaceutical Sciences, Guntur, India. He received a PhD (2018) from the AcSIR at the Indian Institute of Chemical Technology, India, under the guidance of Dr. H.M. Sampath Kumar, where he worked on the development of novel immunomodulators and vaccine adjuvants against infectious diseases. From 2018 to early 2020, he worked as a postdoctoral researcher at IBMC, University of Strasbourg, France under the supervision of Prof. Sylviane Muller. Currently, he is continuing his postdoctoral research on the ‘next-generation flu vaccines for the world’ with Prof. Jagadeesh Bayry at INSERM, CRC, Paris, France.

Niranjan G. Kotla is currently a doctoral researcher at the CURAM, National University of Ireland, Galway. He received his B. Pharmacy from Osmania University, India in 2011 and a M. Pharmacy from LPU, India in 2013. He worked as a Research Fellow at the Institute for Stem Cell Science and Regenerative Medicine (inStem), Bangalore, India on novel gut microbial metabolites for ulcerative colitis therapy. His current research focuses on disease responsive colon drug delivery systems, nanomedicine, biomaterial chemistry and translational medicine. He has published more than 20 journal articles, books and conference abstracts, also is involved in a number of industrial research collaborations on drug delivery.

Raghvendra A. Bohara is working as an Irish Research Council Fellow at the CÚRAM SFI Research Centre for Medical Devices, NUI Galway, Ireland. He is also affiliated as an Assistant Professor in the Department of Stem Cells and Regenerative Medicine, D. Y. Patil Education Society (Deemed to be a University), Kolhapur India. His research interests include targeted drug delivery and cancer nanomedicine. To date, he has published more than ~39 peer-reviewed articles, 6 book chapters, two books and two patents. He has supervised successfully three postgraduate students and one doctoral student. He has an H index of 18 with> 1000 citations. He is an Associate Editor for five journals and has served actively as a Guest Editor and Reviewer for a number of journals.

Yury A. Rochev obtained a Specialist Degree in Physics at Lomonosov Moscow State University, Biophysical Department, Russia. In 1990, he was awarded a Doctorate in Biophysics from the Institute of Biological Physics, the Soviet Academy of Science. He was appointed as a Lecturer in Biomedical Engineering Science at the National Centre for Biomedical Engineering Science, National University of Ireland, Galway in 2007. His research integrates material science and biological paradigms in the development of new smart biomaterials, polymer-based drug delivery systems, tissue engineering products, and medical devices.

Thomas J. Webster’s degrees are in chemical engineering from the University of Pittsburgh (B.S., 1995) and in biomedical engineering from Rensselaer Polytechnic Institute (M.S., 1997; Ph.D., 2000). His research explores the use of nanotechnology in numerous applications. Specifically, his research addresses the design, synthesis, and evaluation of nanophase materials (that is, materials with fundamental length scales less than 100 nm) as more effective biomedical materials. His research has led to the formation of several companies with numerous FDA approval implants. He has received a number of awards, including being a SCOPUS Highly Cited Researcher (representing the top 1% of citations in materials science) and is a fellow of 8 societies.

Jagadeesh Bayry (DVM, PhD) is a Director of Research at Institut National de la Santé et de la Recherche Médicale (INSERM), Paris, France and a Professor at Indian Institute of Technology Palakkad, Palakkad, India. His research is aimed at understanding the cellular and molecular mechanisms of immune homeostasis, immunotherapy and deciphering the host-pathogen interaction. His h-index is 59 and he has authored more than 250 articles with over 12,000 citations. He serves as an Associate Editor and Editorial Board member of various journals and has edited four books. He was also a member of a commission at the European Research Council. He has received several awards and distinctions including World's Top 2% Scientist by Citations (PLoS Biology 2020, https://doi.org/10.1371/journal.pbio.3000918).
References
- 1.Ji H.L., Zhao R., Matalon S., Matthay M.A. Elevated plasmin(ogen) as a common risk factor for COVID-19 susceptibility. Physiol. Rev. 2020;100:1065–1075. doi: 10.1152/physrev.00013.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Xu Z., Shi L., Wang Y., Zhang J., Huang L., Zhang C., Liu S., Zhao P., Liu H., Zhu L., Tai Y., Bai C., Gao T., Song J., Xia P., Dong J., Zhao J., Wang F.S. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir. Med. 2020;8:420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Zhang B., Zhou X., Qiu Y., Song Y., Feng F., Feng J., Song Q., Jia Q., Wang J. Clinical characteristics of 82 cases of death from COVID-19. PLoS One. 2020;15 doi: 10.1371/journal.pone.0235458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Ruan Q., Yang K., Wang W., Jiang L., Song J. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Intensive Care Med. 2020;46:846–848. doi: 10.1007/s00134-020-05991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.WHO, http://www.euro.who.int/en/health-topics/health-emergencies/coronavirus-covid-19/news/news/2020/3/who-announces-covid-19-outbreak-a-pandemic, 2020.
- 7.Liu P., Chen W., Chen J.P. Viral metagenomics revealed sendai virus and coronavirus infection of Malayan Pangolins (Manis javanica) Viruses. 2019;11:979. doi: 10.3390/v11110979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cyranoski D. Why children avoid the worst coronavirus complications might lie in their arteries. Nature. 2020;582:324–325. doi: 10.1038/d41586-020-01692-z. 〈https://www.nature.com/articles/d41586-020-01692-z〉 [DOI] [PubMed] [Google Scholar]
- 9.Hindson J. COVID-19: faecal–oral transmission. Nat. Rev. Gastroenterol. Hepatol. 2020;17:259. doi: 10.1038/s41575-020-0295-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yuan M., Wu N.C., Zhu X., Lee C.C.D., So R.T.Y., Lv H., Mok C., Wilson I.A. A highly conserved cryptic epitope in the receptor binding domains of SARS-CoV-2 and SARS-CoV. Science. 2020;368:630–633. doi: 10.1126/science.abb7269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wan Y., Shang J., Graham R., Baric R.S., Li F. Receptor recognition by the novel coronavirus from Wuhan: an analysis based on decade-long structural studies of SARS coronavirus. J. Virol. 2020;94 doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. e278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.T. Meng, H. Cao, H. Zhang, Z. Kang, D. Xu, H. Gong, et al., bioRxiv, 2020, 2020.2002.2008.926006, https://doi.org/10.1101/2020.02.08.926006.
- 14.Jia H.P., Look D.C., Shi L., Hickey M., Pewe L., Netland J. ACE2 receptor expression and severe acute respiratory syndrome coronavirus infection depend on differentiation of human airway epithelia. J. Virol. 2005;79:14614–14621. doi: 10.1128/JVI.79.23.14614-14621.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hamming I., Timens W., Bulthuis M.L., Lely A.T., Navis G., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sungnak W., Huang N., Bécavin C., Berg M., Queen R., Litvinukova M., Talavera-López C., Maatz H., Reichart D., Sampaziotis F., Worlock K.B., Yoshida M., Barnes J.L. SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together withinnate immune genes. Nat. Med. 2020;26:681–687. doi: 10.1038/s41591-020-0868-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.A. Sinjab, W. Treekitkarnmongkol, P. Brennan, K. Hara, K. Chang, et al., bioRxiv, 2020, 2020.2004.2016.045617, https://doi.org/10.1101/2020.04.16.045617.
- 18.M. Zhong, B.-P. Lin, H.-B. Gao, A.J. Young, X.-H. Wang, C. Liu, et al., bioRxiv, 2020, 2020.2004.2018.047951, 10.1101/2020.04.18.047951. [DOI]
- 19.Zhao Y., Zhao Z., Wang Y., Zhou Y., Ma Y., Zuo W. Single-Cell RNA Expression Profiling of ACE2, the Receptor of SARS-CoV-2. Am. J. Respir. Crit. Care Med. 2020;202:756–759. doi: 10.1164/rccm.202001-0179LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Xu H., Zhong L., Deng J., Peng J., Dan H., Zeng X., Li T., Chen Q. High expression of ACE2 receptor of 2019-nCoV on the epithelial cells of oral mucosa. Int. J. Oral Sci. 2020;12:8. doi: 10.1038/s41368-020-0074-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Callaway E. The race for coronavirus vaccines: a graphical guide. Nature. 2020;580:576–577. doi: 10.1038/d41586-020-01221-y. [DOI] [PubMed] [Google Scholar]
- 22.Hanley B., Lucas S.B., Youd E., Swift B., Osborn M. Autopsy in suspected COVID-19 cases. J. Clin. Pathol. 2020;73:239–242. doi: 10.1136/jclinpath-2020-206522. [DOI] [PubMed] [Google Scholar]
- 23.Yi Y., Lagniton P.N.P., Ye S., Li E., Xu R.H. COVID-19: what has been learned and to be learned about the novel coronavirus disease. Int. J. Biol. Sci. 2020;16:1753–1766. doi: 10.7150/ijbs.45134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lin L., Lu L., Cao W., Li T. Hypothesis for potential pathogenesis of SARS-CoV-2 infection–a review of immune changes in patients with viral pneumonia. Emerg. Microbes & Infect. 2020;9:727–732. doi: 10.1080/22221751.2020.1746199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lee P.I., Hsueh P.R. Emerging threats from zoonotic coronaviruses-from SARS and MERS to 2019-nCoV. J. Microbiol. Immunol. Infect. 2020;53:365–367. doi: 10.1016/j.jmii.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hani C., Trieu N.H., Saab I., Dangeard S., Bennani S., Chassagnon G., Revel M.P. COVID-19 pneumonia: a review of typical CT findings and differential diagnosis. Diagn. Interv. Imaging. 2020;101:263–268. doi: 10.1016/j.diii.2020.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tang N., Bai H., Chen X., Gong J., Li D., Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020;18:1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.E.M. Negri, B. Piloto, L.K. Morinaga, C.V.P. Jardim, S.A.E.-D. Lamy, M.A. Ferreira, et al., medRxiv, 2020, 2020.2004.2015.20067017, 10.1101/2020.04.15.20067017. [DOI]
- 29.H.D. Poor, C.E. Ventetuolo, T. Tolbert, G. Chun, G. Serrao, A. Zeidman, et al., medRxiv, 2020, 2020.2004.2017.20057125, 10.1101/2020.04.17.20057125. [DOI]
- 30.Narasaraju T., Tang B.M., Herrmann M., Muller S., Chow V.T.K., Radic M. Front. Pharmacol. 2020;11 doi: 10.3389/Fphar.2020.00870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Puneet P., Moochhala S., Bhatia M. Chemokines in acute respiratory distress syndrome. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2005;288:L3–L15. doi: 10.1152/ajplung.00405.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hippenstiel S., Opitz B., Schmeck B., Suttorp N. Lung epithelium as a sentinel and effector system in pneumonia – molecular mechanisms of pathogen recognition and signal transduction. Respir. Res. 2006;7:97. doi: 10.1186/1465-9921-7-97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Thorley A.J., Ford P.A., Giembycz M.A., Goldstraw P., Young A., Tetley T.D. Differential regulation of cytokine release and leukocyte migration by lipopolysaccharide-stimulated primary human lung alveolar type ii epithelial cells and macrophages. J. Immunol. 2007;178:463–473. doi: 10.4049/jimmunol.178.1.463. [DOI] [PubMed] [Google Scholar]
- 34.S. Tian, W. Hu, L. Niu, H. Liu, H. Xu, S.-Y. Xiao, Preprints, 2020, 10.20944/preprints202002.0220.v1 [DOI]
- 35.Dhanireddy S., Altemeier W.A., Matute-Bello G., O’Mahony D.S., Glenny R.W., Martin T.R., Liles W.C. Mechanical ventilation induces inflammation, lung injury, and extra-pulmonary organ dysfunction in experimental pneumonia. Lab. Investig. 2006;86:790–799. doi: 10.1038/labinvest.3700440. [DOI] [PubMed] [Google Scholar]
- 36.Liu J., Li S., Liu J., Liang B., Wang X., Wang H., Li W., Tong Q., Yi J., Zhao L., Xiong L., Guo C., Tian J., Luo J., Yao J., Pang R., Shen H., Peng C., Liu T., Zhang Q., Wu J., Xu L., Lu S., Wang B., Weng Z., Han C., Zhu H., Zhou R., Zhou H., Chen X., Ye P., Zhu B., Wang L., Zhou W., He S., He Y., Jie S., Wei P., Zhang J., Lu Y., Wang W., Zhang L., Li L., Zhou F., Wang J., Dittmer U., Lu M., Hu Y., Yang D., Zheng X. Longitudinal characteristics of lymphocyte responses and cytokine profiles in the peripheral blood of SARS-CoV-2 infected patients. EBioMedicine. 2020;55 doi: 10.1016/j.ebiom.2020.102763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Weiskopf D., Schmitz K.S., Raadsen M.P., Grifoni A., Okba N.M.A., Endeman H., van den Akker J., Molenkamp R., Koopmans M., van Gorp E., Haagmans B.L., de Swart R.L., Sette A., de Vries R.D. Phenotype and kinetics of SARS-CoV-2-specific T cells in COVID-19 patients with acute respiratory distress syndrome. Sci. Immunol. 2020;5:eabd2071. doi: 10.1126/sciimmunol.abd2071. 2020.2004.2011.20062349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zemans R.L., Hagood J.S. Diversity at the border. Nat. Immunol. 2020;21:112–114. doi: 10.1038/s41590-019-0585-9. [DOI] [PubMed] [Google Scholar]
- 39.Matthay M.A., Zemans R.L., Zimmerman G.A., Arabi Y.M., Beitler J.R., Mercat A., Herridge M., Randolph A.G., Calfee C.S. Acute respiratory distress syndrome. Nat. Rev. Dis. Primers. 2019;5:18. doi: 10.1038/s41572-019-0069-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.G. Li, S.E. Fox, B. Summa, B. Hu, C. Wenk, A. Akmatbekov, et al., bioRxiv, 2020, 2020.2004.2011.037473, https://doi.org/10.1101/2020.04.11.037473.
- 41.Lin K.L., Sweeney S., Kang B.D., Ramsburg E., Gunn M.D. CCR2-antagonist prophylaxis reduces pulmonary immune pathology and markedly improves survival during influenza infection. J. Immunol. 2011;186:508–515. doi: 10.4049/jimmunol.1001002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lemke G. How macrophages deal with death. Nat. Rev. Immunol. 2019;19:539–549. doi: 10.1038/s41577-019-0167-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chu H., Zhou J., Wong B.H.Y., Li C., Chan J.F.W., Cheng Z.S., Yang D., Wang D., Lee A.C., Li C., Yeung M.L., Cai J.P., Chan I.H., Ho W.K., To K.K., Zheng B.J., Yao Y., Qin C., Yuen K.Y. Middle east respiratory syndrome coronavirus efficiently infects human primary T lymphocytes and activates the extrinsic and intrinsic apoptosis pathways. J. Infect. Dis. 2016;213:904–914. doi: 10.1093/infdis/jiv380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Y. chen, Z. Feng, B. Diao, R. Wang, G. Wang, C. Wang, et al., medRxiv, 2020, 2020.2003.2027.20045427, 10.1101/2020.03.27.20045427. [DOI]
- 45.Zeng F., Huang Y., Guo Y., Yin M., Chen X., Xiao L., Deng G. Association of inflammatory markers with the severity of COVID-19: a meta-analysis. Int. J. Infect. Dis. 2020;96:467–474. doi: 10.1016/j.ijid.2020.05.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.McGonagle D., Sharif K., O’Regan A., Bridgewood C. The role of cytokines including interleukin-6 in COVID-19 induced pneumonia and macrophage activation syndrome-like disease. Autoimmun. Rev. 2020;19 doi: 10.1016/j.autrev.2020.102537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., Chen L., Li M., Liu Y., Wang G., Yuan Z., Feng Z., Zhang Y., Wu Y., Chen Y. Reduction and functional exhaustion of t cells in patients with coronavirus disease 2019 (COVID-19) Front. Immunol. 2020;11:827. doi: 10.3389/fimmu.2020.00827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Koizumi Y., Nagase H., Nakajima T., Kawamura M., Ohta K. Toll-like receptor 3 ligand specifically induced bronchial epithelial cell death in caspase dependent manner and functionally upregulated Fas expression. Allergol. Int. 2016;65 Suppl:S30–S37. doi: 10.1016/j.alit.2016.05.006. [DOI] [PubMed] [Google Scholar]
- 49.Pillaiyar T., Meenakshisundaram S., Manickam M. Recent discovery and development of inhibitors targeting coronaviruses. Drug Discov. Today. 2020;25:668–688. doi: 10.1016/j.drudis.2020.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ekins S., Mottin M., Ramos P.R.P.S., Sousa B.K.P., Neves B.J., Foil D.H., Zorn K.M., Braga R.C., Coffee M., Southan C., Puhl A.C., Andrade C.H. Déjà vu: stimulating open drug discovery for SARS-CoV-2. Drug Discov. Today. 2020;25:928–941. doi: 10.1016/j.drudis.2020.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Xu X., Chen P., Wang J., Feng J., Zhou H., Li X., Zhong W., Hao P. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci. China Life Sci. 2020;63:457–460. doi: 10.1007/s11427-020-1637-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhou P., Yang X.L., Wang X.G., Hu B., Zhang L., Zhang W., Si H.R., Zhu Y., Li B., Huang C.L., Chen H.D., Chen J., Luo Y., Guo H., Jiang R.D., Liu M.Q., Chen Y., Shen X.R., Wang X., Zheng X.S., Zhao K., Chen Q.J., Deng F., Liu L.L., Yan B., Zhan F.X., Wang Y.Y., Xiao G.F., Shi Z.L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mercurio I., Tragni V., Busto F., De Grassi A., Pierri C.L. Protein structure analysis of the interactions between SARS-CoV-2 spike protein and the human ACE2 receptor: from conformational changes to novel neutralizing antibodies. Cell. Mol. Life Sci. 2020 doi: 10.1007/s00018-020-03580-1. 2020.2004.2017.046185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wu K., Peng G., Wilken M., Geraghty R.J., Li F. Mechanisms of host receptor adaptation by severe acute respiratory syndrome coronavirus. J. Biol. Chem. 2012;287:8904–8911. doi: 10.1074/jbc.M111.325803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Coutard B., Valle C., de Lamballerie X., Canard B., Seidah N.G., Decroly E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antivir. Res. 2020;176 doi: 10.1016/j.antiviral.2020.104742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.W. Canrong, Y. Yueying, L. Yang, Z. Peng, W. Yali, W. Qiqi, et al., chinaXiv, 10.12074/202002.00062. [DOI]
- 58.Elfiky A.A. Ribavirin, remdesivir, sofosbuvir, galidesivir, and tenofovir against SARS-CoV-2 RNA dependent RNA polymerase (RdRp): a molecular docking study. Life Sci. 2020;253 doi: 10.1016/j.lfs.2020.117592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.D.E. Gordon, G.M. Jang, M. Bouhaddou, J. Xu, K. Obernier, M.J. O'Meara, et al., bioRxiv, 2020, 2020.2003.2022.002386, 10.1101/2020.03.22.002386. [DOI]
- 60.C. Ma, M.D. Sacco, B. Hurst, J.A. Townsend, Y. Hu, T. Szeto, et al., bioRxiv (2020) 2020.2004.2020.051581, 10.1101/2020.04.20.051581. [DOI]
- 61.Channappanavar R., Fehr A.R., Vijay R., Mack M., Zhao J., Meyerholz D.K., Perlman S. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-infected mice. Cell Host Microbe. 2016;19:181–193. doi: 10.1016/j.chom.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Deng X., Hackbart M., Mettelman R.C., O’Brien A., Mielech A.M., Yi G., Kao C.C., Baker S.C. Coronavirus nonstructural protein 15 mediates evasion of dsRNA sensors and limits apoptosis in macrophages. Proc. Natl. Acad. Sci. USA. 2017;114:E4251–E4260. doi: 10.1073/pnas.1618310114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Liu X., Fang P., Fang L., Hong Y., Zhu X., Wang D., Peng G., Xiao S. Porcine deltacoronavirus nsp15 antagonizes interferon-β production independently of its endoribonuclease activity. Mol. Immunol. 2019;114:100–107. doi: 10.1016/j.molimm.2019.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Kim D., Lee J.Y., Yang J.S., Kim J.W., Kim V.N., Chang H. The architecture of SARS-CoV-2 transcriptome. Cell. 2020;181:914–921. doi: 10.1016/j.cell.2020.04.011. e910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Schett G., Sticherling M., Neurath M.F. COVID-19: risk for cytokine targeting in chronic inflammatory diseases. Nat. Rev. Immunol. 2020;20:271–272. doi: 10.1038/s41577-020-0312-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Galeotti C., Bayry J. Autoimmune and inflammatory diseases following COVID-19. Nat. Rev. Rheumatol. 2020;16:413–414. doi: 10.1038/s41584-020-0448-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bonam S.R., Kaveri S.V., Sakuntabhai A., Gilardin L., Bayry J. Adjunct immunotherapies for the management of severely Ill COVID-19 patients. Cell Rep. Med. 2020;1 doi: 10.1016/j.xcrm.2020.100016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Stephen-Victor E., Das M., Karnam A., Pitard B., Gautier J.F., Bayry J. Potential of regulatory T-cell-based therapies in the management of severe COVID-19. Eur. Respir. J. 2020;56 doi: 10.1183/13993003.02182-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kuba K., Imai Y., Penninger J.M. Angiotensin-converting enzyme 2 in lung diseases. Curr. Opin. Pharmacol. 2006;6:271–276. doi: 10.1016/j.coph.2006.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Meng J., Xiao G., Zhang J., He X., Ou M., Bi J., Yang R., Di W., Wang Z., Li Z., Gao H., Liu L., Zhang G. Renin-angiotensin system inhibitors improve the clinical outcomes of COVID-19 patients with hypertension. Emerg. Microbes & Infect. 2020;9:757–760. doi: 10.1080/22221751.2020.1746200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.M. Zhong, A. Sun, T. Xiao, G. Yao, L. Sang, X. Zheng, et al., medRxiv, 2020, 2020.2004.2015.20066266, 10.1101/2020.04.15.20066266. [DOI]
- 72.Lythgoe M.P., Middleton P. Ongoing clinical trials for the management of the COVID-19 pandemic. Trends Pharmacol. Sci. 2020;41:363–382. doi: 10.1016/j.tips.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Johnson E.R., Matthay M.A. Acute lung injury: epidemiology, pathogenesis, and treatment. J. Aerosol. Med. Pulm. Drug Deliv. 2010;23:243–252. doi: 10.1089/jamp.2009.0775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Rubenfeld G.D., Caldwell E., Peabody E., Weaver J., Martin D.P., Neff M., Stern E.J., Hudson L.D. Incidence and outcomes of acute lung injury. N. Engl. J. Med. 2005;353:1685–1693. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- 75.Matthay M.A. Treatment of acute lung injury: clinical and experimental studies. Proc. Am. Thorac. Soc. 2008;5:297–299. doi: 10.1513/pats.200708-141DR. [DOI] [PubMed] [Google Scholar]
- 76.Fan E., Brodie D., Slutsky A.S. Acute respiratory distress syndrome: advances in diagnosis and treatment. J. Am. Med. Assoc. 2018;319:698–710. doi: 10.1001/jama.2017.21907. [DOI] [PubMed] [Google Scholar]
- 77.Ventola C.L. P & T Peer-rev. J. Formul. Manag. 2017;42:742–755. [PMC free article] [PubMed] [Google Scholar]
- 78.Barenholz Y. Doxil® — The first FDA-approved nano-drug: lessons learned. J. Control. Release. 2012;160:117–134. doi: 10.1016/j.jconrel.2012.03.020. [DOI] [PubMed] [Google Scholar]
- 79.Crosswhite P., Sun Z. Molecular mechanisms of pulmonary arterial remodeling. Mol. Med. 2014;20:191–201. doi: 10.2119/molmed.2013.00165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Lembo D., Donalisio M., Civra A., Argenziano M., Cavalli R. Nanomedicine formulations for the delivery of antiviral drugs: a promising solution for the treatment of viral infections. Expert Opin. Drug Deliv. 2018;15:93–114. doi: 10.1080/17425247.2017.1360863. [DOI] [PubMed] [Google Scholar]
- 81.Singh L., Kruger H.G., Maguire G.E.M., Govender T., Parboosing R. The role of nanotechnology in the treatment of viral infections. Ther. Adv. Infect. Dis. 2017;4:105–131. doi: 10.1177/2049936117713593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Du T., Liang J., Dong N., Lu J., Fu Y., Fang L., Xiao S., Han H. Glutathione-capped Ag2S nanoclusters inhibit coronavirus proliferation through blockage of viral RNA synthesis and budding. ACS Appl. Mater. Interfaces. 2018;10:4369–4378. doi: 10.1021/acsami.7b13811. [DOI] [PubMed] [Google Scholar]
- 83.Nguyen-Van-Tam J.S., Sellwood C. Intervention strategies for emerging respiratory virus infections: policy and public health considerations. Curr. Opin. Virol. 2013;3:192–198. doi: 10.1016/j.coviro.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Shaikh R., Raj Singh T.R., Garland M.J., Woolfson A.D., Donnelly R.F. Mucoadhesive drug delivery systems. J. Pharm. Bioall. Sci. 2011;3:89–100. doi: 10.4103/0975-7406.76478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Cojocaru F.D., Botezat D., Gardikiotis I., Uritu C.M., Dodi G., Trandafir L., Rezus C., Rezus E., Tamba B.I., Mihai C.T. Nanomaterials designed for antiviral drug delivery transport across biological barriers. Pharmaceutics. 2020;12:171. doi: 10.3390/pharmaceutics12020171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.〈https://www.swissbiotech.org/listing/bioavanta-bosti-announces-immediate-availability-of-its-chitosan-nanoparticle-technology-to-formulate-aerosol-anit-covid-19-drugs/〉, 2020.
- 87.Cheung R.C.F., Ng T.B., Wong J.H., Chan W.Y. Chitosan: an update on potential biomedical and pharmaceutical applications. Mar. Drugs. 2015;13:5156–5186. doi: 10.3390/md13085156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Herzog C., Hartmann K., Künzi V., Kürsteiner O., Mischler R., Lazar H., Glück R. Eleven years of Inflexal® V—a virosomal adjuvanted influenza vaccine. Vaccine. 2009;27:4381–4387. doi: 10.1016/j.vaccine.2009.05.029. [DOI] [PubMed] [Google Scholar]
- 89.Kaur S.P., Gupta V. COVID-19 vaccine: a comprehensive status report. Virus Res. 2020;288 doi: 10.1016/j.virusres.2020.198114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Florindo H.F., Kleiner R., Vaskovich-Koubi D., Acúrcio R.C., Carreira B., Yeini E., Tiram G., Liubomirski Y., Satchi-Fainaro R. Immune-mediated approaches against COVID-19. Nat. Nanotechnol. 2020;15:630–645. doi: 10.1038/s41565-020-0732-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jeyanathan M., Afkhami S., Smaill F., Miller M.S., Lichty B.D., Xing Z. Immunological considerations for COVID-19 vaccine strategies. Nat. Rev. Immunol. 2020;20:615–632. doi: 10.1038/s41577-020-00434-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bonam S.R., Partidos C.D., Halmuthur S.K.M., Muller S. An overview of novel adjuvants designed for improving vaccine efficacy. Trends Pharmacol. Sci. 2017;38:771–793. doi: 10.1016/j.tips.2017.06.002. [DOI] [PubMed] [Google Scholar]
- 93.Pati R., Shevtsov M., Sonawane A. Nanoparticle vaccines against infectious diseases. Front. Immunol. 2018;9 doi: 10.3389/fimmu.2018.02224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Shin M.D., Shukla S., Chung Y.H., Beiss V., Chan S.K., Ortega-Rivera O.A., Wirth D.M., Chen A., Sack M., Pokorski J.K., Steinmetz N.F. COVID-19 vaccine development and a potential nanomaterial path forward. Nat. Nanotechnol. 2020;15:646–655. doi: 10.1038/s41565-020-0737-y. [DOI] [PubMed] [Google Scholar]
- 95.Hobernik D., Bros M. DNA vaccines—how far from clinical use. Int. J. Mol. ci.S. 2018;19:3605. doi: 10.3390/ijms19113605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Sravanthi V., Pallavi M.C.P., Bonam S.R., Sathyabama S., Kumar H.M.S. Oleic acid nanoemulsion for nasal vaccination: impact on adjuvanticity based immune response. J. Drug Deliv. Sci. Technol. 2015;28:56–63. doi: 10.1016/j.jddst.2015.05.007. [DOI] [Google Scholar]
- 97.Erasmus J.H., Khandhar A.P., O’Connor M.A., Walls A.C., Hemann E.A., Murapa P., Archer J., Leventhal S., Fuller J.T., Lewis T.B., Draves K.E., Randall S., Guerriero K.A., Duthie M.S., Carter D., Reed S.G., Hawman D.W., Feldmann H., Gale M., Veesler D., Berglund P., Fuller D.H. AnAlphavirus-derived replicon RNA vaccine induces SARS-CoV-2 neutralizing antibody and T cell responses in mice and nonhuman primates. Sci. Transl. Med. 2020;12:eabc9396. doi: 10.1126/scitranslmed.abc9396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Garg P., Sangam S., Kochhar D., Pahari S., Kar C., Mukherjee M. Exploring the role of triazole functionalized heteroatom co-doped carbon quantum dots against human coronaviruses. Nano Today. 2020;35 doi: 10.1016/j.nantod.2020.101001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.J.-H. Tian, N. Patel, R. Haupt, H. Zhou, S. Weston, H. Hammond, et al., bioRxiv, 2020, 2020.2006.2029.178509, 10.1101/2020.06.29.178509. [DOI]
- 100.Magnusson S.E., Altenburg A.F., Bengtsson K.L., Bosman F., de Vries R.D., Rimmelzwaan G.F. Immunol. Res. 2018;66:224–233. doi: 10.1007/s12026-018-8991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Keech C., Albert G., Cho I., Robertson A., Reed P., Neal S. Phase 1–2 Trial of a SARS-CoV-2 Recombinant Spike Protein Nanoparticle Vaccine. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2026920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.B.K. Haun, C.-Y. Lai, C.A. Williams, T.A. Wong, M.M. Lieberman, L. Pessaint, et al., bioRxiv, 2020, 2020.2007.2024.220715, 10.1101/2020.07.24.220715. [DOI]
- 103.Lakhan N., Stevens N.E., Diener K.R., Hayball J.D. CoVaccine HTTM adjuvant is superior to Freund’s adjuvants in eliciting antibodies against the endogenous alarmin HMGB1. J. Immunol. Methods. 2016;439:37–43. doi: 10.1016/j.jim.2016.09.008. [DOI] [PubMed] [Google Scholar]
- 104.Medina L.O., To A., Lieberman M.M., Wong T.A.S., Namekar M., Nakano E., Andersen H., Yalley-Ogunro J., Greenhouse J., Higgs S., Huang Y.S., Vanlandingham D.L., Horton J.S., Clements D.E., Lehrer A.T. A recombinant subunit based zika virus vaccine is efficacious in non-human primates. Front. Immunol. 2018;9:2464. doi: 10.3389/fimmu.2018.02464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Lehrer A.T., Wong T.-A.S., Lieberman M.M., Humphreys T., Clements D.E., Bakken R.R., Hart M.K., Pratt W.D., Dye J.M. Recombinant proteins of Zaire ebolavirus induce potent humoral and cellular immune responses and protect against live virus infection in mice. Vaccine. 2018;36:3090–3100. doi: 10.1016/j.vaccine.2017.01.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kusi K.A., Remarque E.J., Riasat V., Walraven V., Thomas A.W., Faber B.W., Kocken C. Safety and immunogenicity of multi-antigen AMA1-based vaccines formulated with CoVaccine HTTM and Montanide ISA 51 in rhesus macaques. Malar. J. 2011;10:182. doi: 10.1186/1475-2875-10-182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rao L., Xia S., Xu W., Tian R., Yu G., Gu C. Decoy nanoparticles protect against COVID-19 by concurrently adsorbing viruses and inflammatory cytokines. Proc. Natl. Acad. Sci. USA. 2020 doi: 10.1073/pnas.2014352117. 202014352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Weiss C., Carriere M., Fusco L., Capua I., Regla-Nava J.A., Pasquali M., Scott J.A., Vitale F., Unal M.A., Mattevi C., Bedognetti D., Merkoçi A., Tasciotti E., Yilmazer A., Gogotsi Y., Stellacci F., Delogu L.G. Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano. 2020;14:6383–6406. doi: 10.1021/acsnano.0c03697. [DOI] [PubMed] [Google Scholar]
- 109.Chauhan G., Madou M.J., Kalra S., Chopra V., Ghosh D., Martinez-Chapa S.O. Nanotechnology for COVID-19: therapeutics and vaccine research. ACS Nano. 2020;14:7760–7782. doi: 10.1021/acsnano.0c04006. [DOI] [PubMed] [Google Scholar]
- 110.Nikaeen G., Abbaszadeh S., Yousefinejad S. Application of nanomaterials in treatment, anti-infection and detection of coronaviruses. Nanomedicine. 2020;15:1501–1512. doi: 10.2217/nnm-2020-0117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Nasrollahzadeh M., Sajjadi M., Soufi G.J., Iravani S., Varma R.S. Nanomaterials and nanotechnology-associated innovations against viral infections with a focus on coronaviruses. Nanomaterials. 2020;10:1072. doi: 10.3390/nano10061072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Heinrich M.A., Martina B., Prakash J. Nanomedicine strategies to target coronavirus. Nano Today. 2020;35 doi: 10.1016/j.nantod.2020.100961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.C. Wang, S. Wang, Y. Chen, J. Zhao, S. Han, G. Zhao, et al., bioRxiv, 2020, 2020.2008.2012.247338, 10.1101/2020.08.12.247338. [DOI]
- 114.de Groot R.J., Baker S.C., Baric R.S., Brown C.S., Drosten C., Enjuanes L., Fouchier R., Galiano M., Gorbalenya A.E., Memish Z.A., Perlman S., Poon L., Snijder E.J., Stephens G.M., Woo P., Zaki A.M., Zambon M., Ziebuhr J. Middle east respiratory syndrome coronavirus (MERS-CoV): announcement of the coronavirus study group. J. Virol. 2013;87:7790–7792. doi: 10.1128/JVI.01244-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Assiri A., McGeer A., Perl T.M., Price C.S., Al Rabeeah A.A., Cummings D.A.T., Alabdullatif Z.N., Assad M., Almulhim A., Makhdoom H., Madani H., Alhakeem R., Al-Tawfiq J.A., Cotten M., Watson S.J., Kellam P., Zumla A.I., Memish Z.A. Hospital outbreak of middle east respiratory syndrome coronavirus. N. Engl. J. Med. 2013;369:407–416. doi: 10.1056/NEJMoa1306742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Memish Z.A., Zumla A.I., Assiri A. Middle east respiratory syndrome coronavirus infections in health care workers. N. Engl. J. Med. 2013;369:884–886. doi: 10.1056/NEJMc1308698. [DOI] [PubMed] [Google Scholar]
- 117.Zaki A.M., van Boheemen S., Bestebroer T.M., Osterhaus A.D., Fouchier R.A. Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N. Engl. J. Med. 2012;367:1814–1820. doi: 10.1056/NEJMoa1211721. [DOI] [PubMed] [Google Scholar]
- 118.Assiri A., Al-Tawfiq J.A., Al-Rabeeah A.A., Al-Rabiah F.A., Al-Hajjar S., Al-Barrak A., Flemban H., Al-Nassir W.N., Balkhy H.H., Al-Hakeem R.F., Makhdoom H.Q., Zumla A.I., Memish Z.A. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect. Dis. 2013;13:752–761. doi: 10.1016/S1473-3099(13)70204-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Huang X., Li M., Xu Y., Zhang J., Meng X., An X., Sun L., Guo L., Shan X., Ge J., Chen J., Luo Y., Wu H., Zhang Y., Jiang Q., Ning X. Novel gold nanorod-based HR1 peptide inhibitor for middle east respiratory syndrome coronavirus. ACS Appl. Mater. Interfaces. 2019;11:19799–19807. doi: 10.1021/acsami.9b04240. [DOI] [PubMed] [Google Scholar]
- 120.Palestino G., García-Silva I., González-Ortega O., Rosales-Mendoza S. Can nanotechnology help in the fight against COVID-19? Expert Rev. Anti Infect. Ther. 2020:1–16. doi: 10.1080/14787210.2020.1776115. [DOI] [PubMed] [Google Scholar]
- 121.Talebian S., Wallace G.G., Schroeder A., Stellacci F., Conde J. Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nat. Nanotechnol. 2020;15:618–621. doi: 10.1038/s41565-020-0751-0. [DOI] [PubMed] [Google Scholar]
- 122.Cipolla D., Blanchard J., Gonda I. Development of liposomal ciprofloxacin to treat lung infections. Pharmaceutics. 2016;8:6. doi: 10.3390/pharmaceutics8010006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Shirley M. Amikacin liposome inhalation suspension: a review in Mycobacterium avium complex lung disease. Drugs. 2019;79:555–562. doi: 10.1007/s40265-019-01095-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Olivier K.N., Griffith D.E., Eagle G., McGinnis J.P., 2nd, Micioni L., Liu K., Daley C.L., Winthrop K.L., Ruoss S., Addrizzo-Harris D.J., Flume P.A., Dorgan D., Salathe M., Brown-Elliott B.A., Gupta R., Wallace R.J. Randomized trial of liposomal amikacin for inhalation in nontuberculous mycobacterial lung disease. Am. J. Respir. Crit. Care Med. 2017;195:814–823. doi: 10.1164/rccm.201604-0700OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Coleman C.M., Venkataraman T., Liu Y.V., Glenn G.M., Smith G.E., Flyer D.C., Frieman M.B. MERS-CoV spike nanoparticles protect mice from MERS-CoV infection. Vaccine. 2017;35:1586–1589. doi: 10.1016/j.vaccine.2017.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Wiley J.A., Richert L.E., Swain S.D., Harmsen A., Barnard D.L., Randall T.D., Jutila M., Douglas T., Broomell C., Young M., Harmsen A. Inducible bronchus-associated lymphoid tissue elicited by a protein cage nanoparticle enhances protection in mice against diverse respiratory viruses. PLoS One. 2009;4 doi: 10.1371/journal.pone.0007142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Vij N., Min T., Marasigan R., Belcher C.N., Mazur S., Ding H., Yong K.T., Roy I. Development of PEGylated PLGA nanoparticle for controlled and sustained drug delivery in cystic fibrosis. J. Nanobiotechnol. 2010;8:22. doi: 10.1186/1477-3155-8-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Shang W., Yang Y., Rao Y., Rao X. The outbreak of SARS-CoV-2 pneumonia calls for viral vaccines. NPJ Vaccines. 2020;5:18. doi: 10.1038/s41541-020-0170-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Le T.T., Cramer J.P., Chen R., Mayhew S. Evolution of the COVID-19 vaccine development landscape. Nat. Rev. Drug Discov. 2020;19:667–668. doi: 10.1038/d41573-020-00073-5. [DOI] [PubMed] [Google Scholar]
- 130.Du T., Liang J.G., Dong N., Liu L., Fang L.R., Xiao S.B. Carbon dots as inhibitors of virus by activation of type I interferon response. Carbon. 2016;110:278–285. doi: 10.1016/j.carbon.2016.09.032. [DOI] [Google Scholar]
- 131.Huy T.Q., Hien Thanh N.T., Thuy N.T., Chung P.V., Hung P.N., Le A.T., Hong Hanh N.T. Cytotoxicity and antiviral activity of electrochemical – synthesized silver nanoparticles against poliovirus. J. Virol. Methods. 2017;241:52–57. doi: 10.1016/j.jviromet.2016.12.015. [DOI] [PubMed] [Google Scholar]
- 132.Li Y., Liu X., Guo L., Li J., Zhong D., Zhang Y. Traditional Chinese herbal medicine for treating novel coronavirus (COVID-19) pneumonia: protocol for a systematic review and meta-analysis. Syst. Rev. 2020;9:75. doi: 10.1186/s13643-020-01343-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Rai M., Yadav A., Gade A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009;27:76–83. doi: 10.1016/j.biotechadv.2008.09.002. [DOI] [PubMed] [Google Scholar]
- 134.G.C. Tremiliosi, L.G.P. Simoes, D.T. Minozzi, R.I. Santos, D.C.B. Vilela, E.L. Durigon, et al., bioRxiv, 2020, 2020.2006.2026.152520, 10.1101/2020.06.26.152520. [DOI]
- 135.Liu H.B., Bai Y.L., Zhou Y.R., Feng C.C., Liu L.Z., Fang L.R. Blue and cyan fluorescent carbon dots: one-pot synthesis, selective cell imaging and their antiviral activity. RSC Adv. 2017;7:28016–28023. doi: 10.1039/c7ra03167j. [DOI] [Google Scholar]
- 136.Ting D., Dong N., Fang L., Lu J., Bi J., Xiao S., Han H. Multisite inhibitors for enteric coronavirus: antiviral cationic carbon dots based on curcumin. ACS Appl. Nano Mater. 2018;1:5451–5459. doi: 10.1021/acsanm.8b00779. [DOI] [PubMed] [Google Scholar]
- 137.Moghadamtousi S.Z., Kadir H.A., Hassandarvish P., Tajik H., Abubakar S., Zandi K. A review on antibacterial, antiviral, and antifungal activity of curcumin. Biomed. Res. Int. 2014;2014 doi: 10.1155/2014/186864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Campos E.V.R., Pereira A.E.S., de Oliveira J.L., Carvalho L.B., Guilger-Casagrande M., de Lima R. How can nanotechnology help to combat COVID-19? Opportunities and urgent need. J. Nanobiotechnol. 2020;18:125. doi: 10.1186/s12951-020-00685-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.G. Pezzotti, E. Ohgitani, M. Shin-Ya, T. Adachi, E. Marin, F. Boschetto, et al., bioRxiv, 2020, 2020.2006.2019.159970, 10.1101/2020.06.19.159970. [DOI]
- 140.Riediker M. Nano-safety research lessons for dealing with aerosol transmissions of COVID-19. Nanotoxicology. 2020;14:866–868. doi: 10.1080/17435390.2020.1786185. [DOI] [PubMed] [Google Scholar]
- 141.Kim H., Park M., Hwang J., Kim J.H., Chung D.R., Lee K., Kang M. Development of label-free colorimetric assay for MERS-CoV using gold nanoparticles. ACS Sens. 2019;4:1306–1312. doi: 10.1021/acssensors.9b00175. [DOI] [PubMed] [Google Scholar]
- 142.Cheng C.Y., Lee Y.L., Chen C.P., Lin Y.C., Liu C.E., Liao C.H., Cheng S.H. Lopinavir/ritonavir did not shorten the duration of SARS CoV-2 shedding in patients with mild pneumonia in Taiwan. J. Microbiol. Immunol. Infect. 2020;53:488–492. doi: 10.1016/j.jmii.2020.03.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Wang R.R., Yang Q.H., Luo R.H., Peng Y.M., Dai S.X., Zhang X.J., Chen H., Cui X.Q., Liu Y.J., Huang J.F., Chang J.B., Zheng Y.T. Azvudine, a novel nucleoside reverse transcriptase inhibitor showed good drug combination features and better inhibition on drug-resistant strains than lamivudine in vitro. PLoS One. 2014;9 doi: 10.1371/journal.pone.0105617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., Wang B., Xiang H., Cheng Z., Xiong Y., Zhao Y., Li Y., Wang X., Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. J. Am. Med. Assoc. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Lo M.K., Jordan R., Arvey A., Sudhamsu J., Shrivastava-Ranjan P., Hotard A.L., Flint M., McMullan L.K., Siegel D., Clarke M.O., Mackman R.L., Hui H.C., Perron M., Ray A.S., Cihlar T., Nichol S.T., Spiropoulou C.F. GS-5734 and its parent nucleoside analog inhibit Filo-, Pneumo-, and Paramyxoviruses. Sci. Rep. 2017;7:43395. doi: 10.1038/srep43395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Warren T.K., Jordan R., Lo M.K., Ray A.S., Mackman R.L., Soloveva V., Siegel D., Perron M., Bannister R., Hui H.C., Larson N., Strickley R., Wells J., Stuthman K.S., Van Tongeren S.A., Garza N.L., Donnelly G., Shurtleff A.C., Retterer C.J., Gharaibeh D., Zamani R., Kenny T., Eaton B.P., Grimes E., Welch L.S., Gomba L., Wilhelmsen C.L., Nichols D.K., Nuss J.E., Nagle E.R., Kugelman J.R., Palacios G., Doerffler E., Neville S., Carra E., Clarke M.O., Zhang L., Lew W., Ross B., Wang Q., Chun K., Wolfe L., Babusis D., Park Y., Stray K.M., Trancheva I., Feng J.Y., Barauskas O., Xu Y., Wong P., Braun M.R., Flint M., McMullan L.K., Chen S.S., Fearns R., Swaminathan S., Mayers D.L., Spiropoulou C.F., Lee W.A., Nichol S.T., Cihlar T., Bavari S. Therapeutic efficacy of the small molecule GS-5734 against Ebola virus in rhesus monkeys. Nature. 2016;531:381–385. doi: 10.1038/nature17180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Sheahan T.P., Sims A.C., Graham R.L., Menachery V.D., Gralinski L.E., Case J.B., Leist S.R., Pyrc K., Feng J.Y., Trantcheva I., Bannister R., Park Y., Babusis D., Clarke M.O., Mackman R.L., Spahn J.E., Palmiotti C.A., Siegel D., Ray A.S., Cihlar T., Jordan R., Denison M.R., Baric R.S. Broad-spectrum antiviral GS-5734 inhibits both epidemic and zoonotic coronaviruses. Sci. Transl. Med. 2017;9:eaal3653. doi: 10.1126/scitranslmed.aal3653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Morse J.S., Lalonde T., Xu S., Liu W.R. Learning from the past: possible urgent prevention and treatment options for severe acute respiratory infections caused by 2019‐nCoV. ChemBioChem. 2020;21:730–738. doi: 10.1002/cbic.202000047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Stockman L.J., Bellamy R., Garner P. SARS: systematic review of treatment effects. PLoS Med. 2006;3 doi: 10.1371/journal.pmed.0030343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Morra M.E., Van Thanh L., Kamel M.G., Ghazy A.A., Altibi A.M.A., Dat L.M., Thy T., Vuong N.L., Mostafa M.R., Ahmed S.I., Elabd S.S., Fathima S., Le Huy Vu T., Omrani A.S., Memish Z.A., Hirayama K., Huy N.T. Clinical outcomes of current medical approaches for Middle East respiratory syndrome: a systematic review and meta-analysis. Rev. Med. Virol. 2018;28 doi: 10.1002/rmv.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.〈https://www.fujifilm.com/jp/en/news/hq/3211?_ga=2.98370583.926946726.1587470916–273032141.1587470916〉, 2020.
- 152.Blaising J., Polyak S.J., Pecheur E.I. Arbidol as a broad-spectrum antiviral: an update. Antivir. Res. 2014;107:84–94. doi: 10.1016/j.antiviral.2014.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Q. Liu, X. Fang, L. Tian, X. Chen, U. Chung, K. Wang, et al., medRxiv, 2020, 2020.2004.2011.20056523, https://doi.org/10.1101/2020.04.11.20056523.
- 154.O’Hanlon R., Shaw M.L. Baloxavir marboxil: the new influenza drug on the market. Curr. Opin. Virol. 2019;35:14–18. doi: 10.1016/j.coviro.2019.01.006. [DOI] [PubMed] [Google Scholar]
- 155.Cvetkovic R.S., Keating G. Anakinra. Biodrugs. 2002;16:303–311. doi: 10.2165/00063030-200216040-00005. discussion 313-304. [DOI] [PubMed] [Google Scholar]
- 156.Cauchois R., Koubi M., Delarbre D., Manet C., Carvelli J., Blasco V.B., Jean R., Fouche L., Bornet C., Pauly V., Mazodier K., Pestre V., Jarrot P.A., Dinarello C.A., Kaplanski G. Early IL-1 receptor blockade in severe inflammatory respiratory failure complicating COVID-19. Proc. Natl. Acad. Sci. USA. 2020;117:18951–18953. doi: 10.1073/pnas.2009017117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Cavalli G., De Luca G., Campochiaro C., Della-Torre E., Ripa M., Canetti D., Oltolini C., Castiglioni B., Tassan Din C., Boffini N., Tomelleri A., Farina N., Ruggeri A., Rovere-Querini P., Di Lucca G., Martinenghi S., Scotti R., Tresoldi M., Ciceri F., Landoni G., Zangrillo A., Scarpellini P., Dagna L. Interleukin-1 blockade with high-dose anakinra in patients with COVID-19, acute respiratory distress syndrome, and hyperinflammation: a retrospective cohort study. Lancet Rheumatol. 2020;2:e325–e331. doi: 10.1016/S2665-9913(20)30127-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Bronte V., Ugel S., Tinazzi E., Vella A., De Sanctis F., Cane S. Baricitinib restrains the immune dysregulation in patients with severe COVID-19. J. Clin. Investig. 2020 doi: 10.1172/JCI141772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Titanji B.K., Farley M.M., Mehta A., Connor-Schuler R., Moanna A., Cribbs S.K., O’Shea J., DeSilva K., Chan B., Edwards A., Gavegnano C., Schinazi R.F., Marconi V.C. Use of baricitinib in patients with moderate to severe coronavirus disease 2019. Clin. Infect. Dis. 2020:ciaa879. doi: 10.1093/cid/ciaa879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Lapadula G., Marchesoni A., Armuzzi A., Blandizzi C., Caporali R., Chimenti S., Cimaz R., Cimino L., Gionchetti P., Girolomoni G., Lionetti P., Marcellusi A., Mennini F.S., Salvarani C. Adalimumab in the treatment of immune-mediated diseases. Int. J. Immunopathol. Pharmacol. 2014;27:33–48. doi: 10.1177/03946320140270S103. [DOI] [PubMed] [Google Scholar]
- 161.Harrison C., Vannucchi A.M. Ruxolitinib: a potent and selective Janus kinase 1 and 2 inhibitor in patients with myelofibrosis. An update for clinicians. Ther. Adv. Hematol. 2012;3:341–354. doi: 10.1177/2040620712459746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kazazi-Hyseni F., Beijnen J.H., Schellens J.H. Bevacizumab. Oncologist. 2010;15:819–825. doi: 10.1634/theoncologist.2009-0317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Parker C.J., Kar S., Kirkpatrick P. Eculizumab. Nat. Rev. Drug Discov. 2007;6:515–516. doi: 10.1038/nrd2369. [DOI] [PubMed] [Google Scholar]
- 164.Giudice V., Pagliano P., Vatrella A., Masullo A., Poto S., Polverino B.M. Combination of Ruxolitinib and Eculizumab for treatment of severe SARS-CoV-2-related acute respiratory distress syndrome: a controlled study. Front. Pharmacol. 2020;11:857. doi: 10.3389/fphar.2020.00857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Diurno F., Numis F.G., Porta G., Cirillo F., Maddaluno S., Ragozzino A. Eculizumab treatment in patients with COVID-19: preliminary results from real life ASL Napoli 2 Nord experience. Eur. Rev. Med. Pharmacol. Sci. 2020;24:4040–4047. doi: 10.26355/eurrev_202004_20875. [DOI] [PubMed] [Google Scholar]
- 166.Zheng P., Liu Y., Chen H.G., Devenport M., Reddy P., Farag S.S. Targeting Danger Associated Molecular Pattern (DAMP) with CD24Fc to reduce acute Gvhd: study design on a randomized double blind placebo controlled Phase III clinical trial (CATHY Study) Biol. Blood Marrow Transpl. 2020;26:S180–S181. doi: 10.1016/j.bbmt.2019.12.741. [DOI] [Google Scholar]
- 167.Giovannoni G., Hawkes C., Lechner-Scott J., Levy M., Waubant E., Gold J. The COVID-19 pandemic and the use of MS disease-modifying therapies. Mult. Scler. Related Disord. 2020;39 doi: 10.1016/j.msard.2020.102073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Z. Meng, T. Wang, C. Li, X. Chen, L. Li, X. Qin, et al., medRxiv, 2020, 2020.2004.2011.20061473, https://doi.org/10.1101/2020.04.11.20061473.
- 169.H. Bian, Z.-H. Zheng, D. Wei, Z. Zhang, W.-Z. Kang, C.-Q. Hao, et al., medRxiv (2020) 2020.2003.2021.20040691, 10.1101/2020.03.21.20040691. [DOI]
- 170.Markham A., Keam S.J. Camrelizumab: first global approval. Drugs. 2019;79:1355–1361. doi: 10.1007/s40265-019-01167-0. [DOI] [PubMed] [Google Scholar]
- 171.Casanova V., Sousa F.H., Shakamuri P., Svoboda P., Buch C., D’Acremont M., Christophorou M.A., Pohl J., Stevens C., Barlow P.G. Citrullination alters the antiviral and immunomodulatory activities of the human cathelicidin LL-37 during rhinovirus infection. Front. Immunol. 2020;11:85. doi: 10.3389/fimmu.2020.00085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Liu B., Li M., Zhou Z., Guan X., Xiang Y. Can we use interleukin-6 (IL-6) blockade for coronavirus disease 2019 (COVID-19)-induced cytokine release syndrome (CRS) J. Autoimmun. 2020;111 doi: 10.1016/j.jaut.2020.102452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.M. Roumier, R. Paule, M. Groh, A. Vallee, F. Ackermann, medRxiv, 2020, 2020.2004.2020.20061861, 10.1101/2020.04.20.20061861. [DOI]
- 174.Canziani L.M., Trovati S., Brunetta E., Testa A., De Santis M., Bombardieri E. Interleukin-6 receptor blocking with intravenous tocilizumab in COVID-19 severe acute respiratory distress syndrome: A retrospective case-control survival analysis of 128 patients. J. Autoimmun. 2020 doi: 10.1016/j.jaut.2020.102511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Klopfenstein T., Zayet S., Lohse A., Selles P., Zahra H., Kadiane-Oussou Nd.J. Impact of tocilizumab on mortality and/or invasive mechanical ventilation requirement in a cohort of 206 COVID-19 patients. Int. J. Infect. Dis. 2020 doi: 10.1016/j.ijid.2020.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Mohtadi N., Ghaysouri A., Shirazi S., sara Ansari A., Shafiee E., Bastani E., Kokhazadeh T., Tavan H. Recovery of severely ill COVID-19 patients by intravenous immunoglobulin (IVIG) treatment: a case series. Virology. 2020;548:1–5. doi: 10.1016/j.virol.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Cao W., Liu X., Bai T., Fan H., Hong K., Song H., Han Y., Lin L., Ruan L., Li T. High-dose intravenous immunoglobulin as a therapeutic option for deteriorating patients with coronavirus disease 2019. Open Forum Infect. Dis. 2020;7:ofaa102. doi: 10.1093/ofid/ofaa102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Sheridan C. Convalescent serum lines up as first-choice treatment for coronavirus. Nat. Biotechnol. 2020;38:655–658. doi: 10.1038/d41587-020-00011-1. [DOI] [PubMed] [Google Scholar]
- 179.Shen C., Wang Z., Zhao F., Yang Y., Li J., Yuan J., Wang F., Li D., Yang M., Xing L., Wei J., Xiao H., Yang Y., Qu J., Qing L., Chen L., Xu Z., Peng L., Li Y., Zheng H., Chen F., Huang K., Jiang Y., Liu D., Zhang Z., Liu Y., Liu L. Treatment of 5 critically Ill patients with COVID-19 with convalescent plasma. J. Am. Med. Assoc. 2020;323:1582–1589. doi: 10.1001/jama.2020.4783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Duan K., Liu B., Li C., Zhang H., Yu T., Qu J. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc. Natl. Acad. Sci. USA. 2020 doi: 10.1073/pnas.2004168117. 202004168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.G. Rada, J. Corbalan, P. Rojas, medRxiv, 2020, 2020.2004.2013.20064162, 10.1101/2020.04.13.20064162. [DOI]
- 182.Russell C.D., Millar J.E., Baillie J.K. Clinical evidence does not support corticosteroid treatment for 2019-nCoV lung injury. Lancet. 2020;395:473–475. doi: 10.1016/S0140-6736(20)30317-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Xiong Y., Liu Y., Cao L., Wang D., Guo M., Jiang A., Guo D., Hu W., Yang J., Tang Z., Wu H., Lin Y., Zhang M., Zhang Q., Shi M., Liu Y., Zhou Y., Lan K., Chen Y. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerg. Microbes & Infect. 2020;9:761–770. doi: 10.1080/22221751.2020.1747363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Breedveld F.C., Dayer J.M. Leflunomide: mode of action in the treatment of rheumatoid arthritis. Ann. Rheum. Dis. 2000;59:841–849. doi: 10.1136/ard.59.11.841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Fantini J., Di Scala C., Chahinian H., Yahi N. Structural and molecular modelling studies reveal a new mechanism of action of chloroquine and hydroxychloroquine against SARS-CoV-2 infection. Int. J. Antimicrob. Agents. 2020;55 doi: 10.1016/j.ijantimicag.2020.105960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Mahévas M., Tran V.T., Roumier M., Chabrol A., Paule R., Guillaud C., Fois E., Lepeule R., Szwebel T.A., Lescure F.X., Schlemmer F., Matignon M., Khellaf M., Crickx E., Terrier B., Morbieu C., Legendre P., Dang J., Schoindre Y., Pawlotsky J.M., Michel M., Perrodeau E., Carlier N., Roche N., de Lastours V., Ourghanlian C., Kerneis S., Ménager P., Mouthon L., Audureau E., Ravaud P., Godeau B., Gallien S., Costedoat-Chalumeau N. Clinical efficacy of hydroxychloroquine in patients with covid-19 pneumonia who require oxygen: observational comparative study using routine care data. Br. Med. J. 2020;369:m1844. doi: 10.1136/bmj.m1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.J. Magagnoli, S. Narendran, F. Pereira, T. Cummings, J.W. Hardin, S.S. Sutton, et al., medRxiv, 2020, 2020.2004.2016.20065920, 10.1101/2020.04.16.20065920. [DOI]
- 188.A. Shamshirian, A. Hessami, K. Heydari, R. Alizadeh-Navaei, M.A. Ebrahimzadeh, G.W. Yip, et al., medRxiv, 2020, 2020.2004.2014.20065276, 10.1101/2020.04.14.20065276. [DOI]
- 189.Caly L., Druce J.D., Catton M.G., Jans D.A., Wagstaff K.M. The FDA-approved drug ivermectin inhibits the replication of SARS-CoV-2 in vitro. Antivir. Res. 2020;178 doi: 10.1016/j.antiviral.2020.104787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Momekov G., Momekova D. Ivermectin as a potential COVID-19 treatment from the pharmacokinetic point of view: antiviral levels are not likely attainable with known dosing regimens. Biotechnol. Biotechnol. Equip. 2020;34:469–474. doi: 10.1080/13102818.2020.1775118. [DOI] [Google Scholar]
- 191.Olivier K.N., Shaw P.A., Glaser T.S., Bhattacharyya D., Fleshner M., Brewer C.C., Zalewski C.K., Folio L.R., Siegelman J.R., Shallom S., Park I.K., Sampaio E.P., Zelazny A.M., Holland S.M., Prevots D.R. Inhaled amikacin for treatment of refractory pulmonary nontuberculous mycobacterial disease. Ann. Am. Thorac. Soc. 2014;11:30–35. doi: 10.1513/AnnalsATS.201307-231OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Moreno-Sastre M., Pastor M., Esquisabel A., Sans E., Viñas M., Fleischer A., Palomino E., Bachiller D., Pedraz J.L. Pulmonary delivery of tobramycin-loaded nanostructured lipid carriers for Pseudomonas aeruginosa infections associated with cystic fibrosis. Int. J. Pharm. 2016;498:263–273. doi: 10.1016/j.ijpharm.2015.12.028. [DOI] [PubMed] [Google Scholar]
- 193.Alton E.W.F.W., Armstrong D.K., Ashby D., Bayfield K.J., Bilton D., Bloomfield E.V., Boyd A.C., Brand J., Buchan R., Calcedo R., Carvelli P., Chan M., Cheng S.H., Collie D., Cunningham S., Davidson H.E., Davies G., Davies J.C., Davies L.A., Dewar M.H., Doherty A., Donovan J., Dwyer N.S., Elgmati H.I., Featherstone R.F., Gavino J., Gea-Sorli S., Geddes D.M., Gibson J., Gill D.R., Greening A.P., Griesenbach U., Hansell D.M., Harman K., Higgins T.E., Hodges S.L., Hyde S.C., Hyndman L., Innes J.A., Jacob J., Jones N., Keogh B.F., Limberis M.P., Lloyd-Evans P., Maclean A.W., Manvell M.C., McCormick D., McGovern M., McLachlan G., Meng C., Montero M.A., Milligan H., Moyce L.J., Murray G.D., Nicholson A.G., Osadolor T., Parra-Leiton J., Porteous D.J., Pringle I.A., Punch E.K., Pytel K.M., Quittner A.L., Rivellini G., Saunders C.J., Scheule R.K., Sheard S., Simmonds N.J., Smith K., Smith S.N., Soussi N., Soussi S., Spearing E.J., Stevenson B.J., Sumner-Jones S.G., Turkkila M., Ureta R.P., Waller M.D., Wasowicz M.Y., Wilson J.M., Wolstenholme-Hogg P. Repeated nebulisation of non-viral CFTR gene therapy in patients with cystic fibrosis: a randomised, double-blind, placebo-controlled, phase 2b trial. Lancet Respir. Med. 2015;3:684–691. doi: 10.1016/S2213-2600(15)00245-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Galloway A.L., Murphy A., DeSimone J.M., Di J., Herrmann J.P., Hunter M.E., Kindig J.P., Malinoski F.J., Rumley M.A., Stoltz D.M., Templeman T.S., Hubby B. Development of a nanoparticle-based influenza vaccine using the PRINT® technology. Nanomed. Nanotechnol. Biol. Med. 2013;9:523–531. doi: 10.1016/j.nano.2012.11.001. [DOI] [PubMed] [Google Scholar]
- 195.Manicone A.M. Role of the pulmonary epithelium and inflammatory signals in acute lung injury. Expert Rev. Clin. Immunol. 2009;5:63–75. doi: 10.1586/177666X.5.1.63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.M. Zietz, N.P. Tatonetti, medRxiv, 2020, 2020.2004.2008.20058073, 10.1101/2020.04.08.20058073. [DOI]
- 197.J. Zhao, Y. Yang, H. Huang, D. Li, D. Gu, X. Lu, et al., medRxiv, 2020, 2020.2003.2011.20031096, 10.1101/2020.03.11.20031096. [DOI]
- 198.Guillon P., Clément M., Sébille V., Rivain J.-G., Chou C.-F., Ruvoën-Clouet N., Le Pendu J. Inhibition of the interaction between the SARS-CoV Spike protein and its cellular receptor by anti-histo-blood group antibodies. Glycobiology. 2008;18:1085–1093. doi: 10.1093/glycob/cwn093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Fischer A. Resistance of children to Covid-19. How. Mucosal Immunol. 2020;13:563–565. doi: 10.1038/s41385-020-0303-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Salje H., Tran Kiem C., Lefrancq N., Courtejoie N., Bosetti P., Paireau J., Andronico A., Hozé N., Richet J., Dubost C.L., Le Strat Y., Lessler J., Levy-Bruhl D., Fontanet A., Opatowski L., Boelle P.Y., Cauchemez S. Estimating the burden of SARS-CoV-2 in France. Science. 2020;369:208–211. doi: 10.1126/science.abc3517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Carsetti R., Quintarelli C., Quinti I., Piano Mortari E., Zumla A., Ippolito G., Locatelli F. The immune system of children: the key to understanding SARS-CoV-2 susceptibility. Lancet Child Adolesc. Health. 2020;4:414–416. doi: 10.1016/S2352-4642(20)30135-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.K.W. Ng, N. Faulkner, G.H. Cornish, A. Rosa, R. Harvey, S. Hussain, et al., bioRxiv, 2020, 2020.2005.2014.095414, 10.1101/2020.05.14.095414. [DOI]
- 203.Lv H., Wu N.C., Tsang O.T.-Y., Yuan M., Perera R.A.P.M., Leung W.S., So R., Chan J., Yip G.K., Chik T., Wang Y., Choi C., Lin Y., Ng W.W., Zhao J., Poon L., Peiris J., Wilson I.A., Mok C. Cross-reactive antibody response between SARS-CoV-2 and SARS-CoV infections. Cell Rep. 2020;31 doi: 10.1016/j.celrep.2020.107725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Mateus J., Grifoni A., Tarke A., Sidney J., Ramirez S.I., Dan J.M. Selective and cross-reactive SARS-CoV-2 T cell epitopes in unexposed humans. Science. 2020:eabd3871. doi: 10.1126/science.abd3871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Sekine T., Perez-Potti A., Rivera-Ballesteros O., Strålin K., Gorin J.-B., Olsson A. Robust T cell immunity in convalescent individuals with asymptomatic or mild COVID-19. Cell. 2020 doi: 10.1016/j.cell.2020.08.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Grifoni A., Weiskopf D., Ramirez S.I., Mateus J., Dan J.M., Moderbacher C.R., Rawlings S.A., Sutherland A., Premkumar L., Jadi R.S., Marrama D., de Silva A.M., Frazier A., Carlin A.F., Greenbaum J.A., Peters B., Krammer F., Smith D.M., Crotty S., Sette A. Targets of T cell responses to SARS-CoV-2 coronavirus in humans with COVID-19 disease and unexposed individuals. Cell. 2020;181:1489–1501. doi: 10.1016/j.cell.2020.05.015. e1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.J. Braun, L. Loyal, M. Frentsch, D. Wendisch, P. Georg, F. Kurth, et al., medRxiv, 2020, 2020.2004.2017.20061440, 10.1101/2020.04.17.20061440. [DOI]
- 208.N.L. Bert, A.T. Tan, K. Kunasegaran, C.Y.L. Tham, M. Hafezi, A. Chia, et al., bioRxiv, 2020, 2020.2005.2026.115832, 10.1101/2020.05.26.115832. [DOI]
- 209.Ujike M., Taguchi F. Incorporation of spike and membrane glycoproteins into coronavirus virions. Viruses. 2015;7:1700–1725. doi: 10.3390/v7041700. [DOI] [PMC free article] [PubMed] [Google Scholar]