Abstract
Bats harbor diverse viruses and manifest distinct antiviral immune responses. Recently in Cell Host & Microbe, Boys et al. demonstrated that bat receptor transporter protein 4 (RTP4) is an innate antiviral effector that inhibits flavivirus replication, revealing an evolutionary arms race between flaviviruses and their hosts.
Bats harbor diverse viruses and manifest distinct antiviral immune responses. Recently in Cell Host & Microbe, Boys et al. demonstrated that bat receptor transporter protein 4 (RTP4) is an innate antiviral effector that inhibits flavivirus replication, revealing an evolutionary arms race between flaviviruses and their hosts.
Main Text
The Red Queen hypothesis posits that organisms must constantly change and adapt to selective pressures, including pathogens (Van Valen, 1973). This process leads to an evolutionary arms race at a molecular level that prevents either pathogens or hosts from achieving complete victory over each other (Daugherty and Malik, 2012). In vertebrate animals, interferons and interferon-stimulated genes (ISGs) are key players in the innate antiviral immune response. In a recent issue of Cell Host & Microbe, Boys et al. demonstrate that a bat ortholog of the ISG RTP4 (receptor transporter protein 4) inhibits flavivirus replication, which suggests a molecular arms race waged between flaviviruses and mammalian innate immunity (Boys et al., 2020).
Bats serve as reservoir hosts for a wide diversity of viruses, including some that are highly pathogenic in humans, such as Marburg virus, Nipah virus, and SARS-CoV-2 (Letko et al., 2020). Despite expanding research on bat virology and bat immunology, interactions between bats and flaviviruses have not been studied extensively (Fagre and Kading, 2019). Flaviviruses include significant human pathogens such as dengue virus (DENV), Zika virus (ZIKV), West Nile virus (WNV), and yellow fever virus (YFV) (Pierson and Diamond, 2020). The flaviviruses known to cause human disease are all transmitted by arthropod vectors (mosquitoes or ticks), but some flaviviruses circulating in nature have no known arthropod vector. Many of these no-known-vector flaviviruses were isolated from bats (Blitvich and Firth, 2017). Since there are unusual features of the bat antiviral response that may contribute to their ability to serve as reservoir hosts (Hayman, 2016), Boys et al. set out to identify genes in the black flying fox bat (Pteropus alecto) that restrict flavivirus infection. They expressed a P. alecto cDNA library (enriched for ISGs) in a human hepatocyte cell line, then infected with DENV or ZIKV. Antiviral genes were identified by sequencing the P. alecto transcripts from cells that survived viral infection. This approach identified some ISGs that had previously been shown to inhibit flaviviruses in studies with their human orthologs. However, one of the most potent inhibitors of both DENV and ZIKV was RTP4, whose human ortholog had shown only modest antiviral activity in prior studies. To confirm this observation, the group demonstrated that P. alecto RTP4 (paRTP4) inhibited YFV infection more potently than Homo sapiens RTP4 (hsRTP4) in both P. alecto and human cell lines.
To define the scope of paRTP4 antiviral activity, the group tested diverse viruses in cells expressing paRTP4 or hsRTP4. They found that both paRTP4 and hsRTP4 broadly inhibit flaviviruses, and paRTP4 additionally inhibited hepatitis C virus (HCV, a Flaviviridae family member not in the Flavivirus genus), and two nidoviruses (equine arteritis virus, and human coronavirus OC43 [HCoV-OC43]). The inhibited viruses are all enveloped positive-sense RNA viruses that replicate on endoplasmic reticulum (ER) membranes. Boys et al. confirmed that RTP4 localized to the ER, suggesting an interaction with the viral replication complex. However, paRTP4 mutants lacking their transmembrane domains retained full antiviral activity.
Next, Boys et al. asked what stage of flavivirus replication was inhibited by paRTP4. They demonstrated a direct interaction between paRTP4 and viral RNA using cross-linking immunoprecipitation. This interaction was distributed throughout the viral genome, on both positive-sense and negative-sense viral RNA. paRTP4 did not inhibit translation of viral RNA, as shown by translation reporter assays and a polysome-association assay. Instead, they found that paRTP4 inhibits flavivirus RNA replication by binding the double-stranded RNA replication intermediate and disrupting the viral replicase complex.
Having shown how paRTP4 inhibits flavivirus replication, the authors next returned to the Red Queen concept, asking why paRTP4 was so much more potent than hsRTP4. Amino acid sequence analysis revealed that paRTP4 is over 70% larger than hsRTP4 and that neither ortholog was an extreme example of mammalian RTP4 ortholog sizes. Such a wide range of protein sizes is unusual and is consistent with the idea that RTP4 evolves rapidly. To investigate this further, the group tested additional mammalian RTP4 orthologs (cow, pig, dog, and mouse, as well as additional bat and primate species), for antiviral activity against DENV, ZIKV, WNV, YFV, HCV, and a bat-origin flavivirus with no known arthropod vector, Entebbe bat virus (ENTV). This array of infections revealed that evolutionary relatedness of RTP4 orthologs correlates with their antiviral activity against a given virus. Additionally, it showed that viruses were most resistant to RTP4 from their natural host, e.g., HCV and YFV to human RTP4 and ENTV to bat RTP4. These relationships further support the rapid evolution of RTP4 and flaviviruses in a virus-host arms race (Figure 1 ). The group further demonstrated this by reconstructing an inferred ancestral RTP4 ortholog, which exhibited slightly reduced potency against flaviviruses compared to paRTP4 but increased potency against HCoV-OC43 compared to other mammalian orthologs. Collectively, these data suggest that modern RTP4 orthologs evolved to more specifically target flaviviruses and highlight the trade-offs between potency and specificity for antiviral effectors.
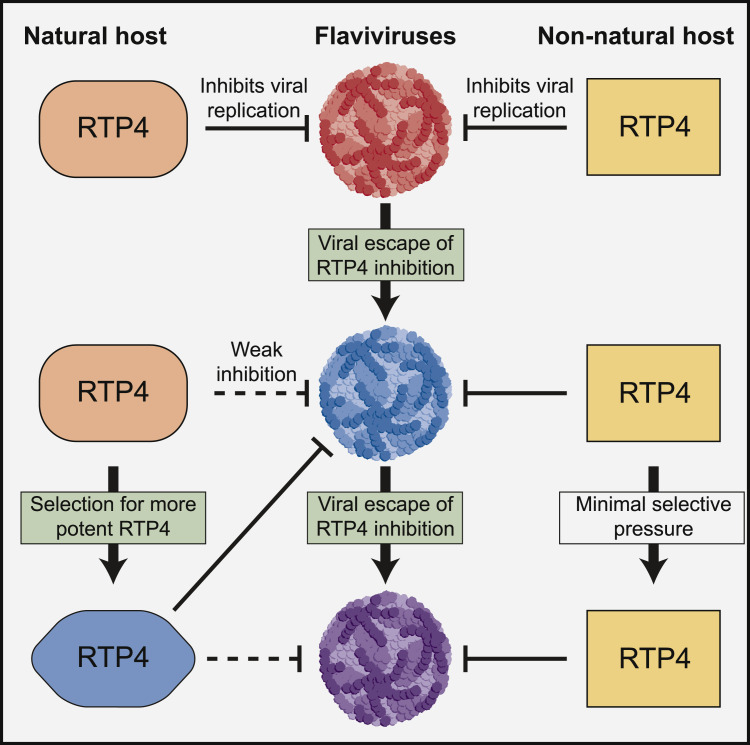
Figure 1.
An Evolutionary Arms Race between Flaviviruses and RTP4
Flaviviruses replicate in their natural host species, but non-natural host species also may be exposed to infection, for example through a mosquito or tick bite. Boys et al. found that the antiviral effector RTP4 inhibits flaviviruses in a species-specific manner, with RTP4 orthologs from natural hosts being less potent against particular flaviviruses than orthologs from non-natural hosts. Their work suggests a molecular arms race, wherein RTP4 inhibits flavivirus replication, creating selective pressure for flaviviruses to evolve to antagonize RTP4 in their natural hosts, and in turn pressuring RTP4 to evolve greater potency against the specific flaviviruses that target that host.
Interestingly, ENTV was the only virus tested that was efficiently inhibited by hsRTP4, but it was efficiently inhibited by only one of three bat orthologs tested. Conversely, YFV, the closest relative to ENTV tested, showed the opposite pattern, being strongly inhibited by all three bat RTP4 orthologs but only weakly by hsRTP4. This is consistent with a model wherein flaviviruses evolve to antagonize the RTP4 of the specific hosts they infect. To test the viral side of the evolutionary arms race, the authors serially passaged YFV in cells expressing a bat RTP4 ortholog. After several passages, YFV replication increased due to the emergence of an escape mutant located in a component of the viral replicase complex, consistent with the observation that RTP4 disrupts this complex. Although the escape mutant replicated better than the parental virus in the presence of the specific bat RTP4 ortholog it arose against, the mutant was attenuated in the presence of other RTP4 orthologs. This observation again reveals the potential trade-offs involved in evolving to more potently antagonize an antiviral effector in a particular host species.
The work by Boys et al. supports the idea that flavivirus host range is determined, in part, by the ability of viruses to antagonize the antiviral response in particular host species. For example, ZIKV, DENV, and YFV naturally infect humans and other primates. These viruses efficiently antagonize the ISG-inducing transcription factor STAT2 in human cells, but not mouse cells, and accordingly, they replicate poorly in immune-competent mice (Best, 2017). Consistent with this paradigm, DENV and YFV were relatively resistant to the antiviral effects of hsRTP4 compared to other orthologs (although ZIKV inhibition was less species specific). These flaviviruses circulate between humans and mosquitoes, but some mosquito-borne flaviviruses require other animals for circulation in nature. For example, WNV circulates between birds and mosquitoes, with humans as incidental dead-end hosts. Since WNV does not replicate to sufficiently high levels in humans to be transmitted back to mosquitoes, humans do not contribute to the WNV transmission cycle, so there is no selective pressure for WNV to specifically antagonize human RTP4. Although Boys et al. found that WNV was poorly inhibited by primate orthologs of RTP4, and more efficiently inhibited by other mammalian orthologs, any specificity is unlikely to derive from selective pressures on WNV evolution. Since many flaviviruses use birds as amplifying hosts, it will be interesting to determine whether avian RTP4 orthologs have similar diversity and antiviral activity as their mammalian counterparts. Since bird species vary in their susceptibility to flavivirus disease and their ability to sustain high levels of flavivirus replication, comparing avian RTP4 orthologs may yield evidence of additional evolutionary arms races. Likewise, many tick-borne flaviviruses use rodents as amplifying hosts and may exhibit species-specific antagonism of rodent RTP4.
The Red Queen hypothesis can describe an ongoing arms race between a virus and a particular host species. This relationship is more complicated for vector-borne viruses, such as mosquito- and tick-borne flaviviruses, which must maintain the ability to replicate in both an arthropod vector and a vertebrate host, thus constraining the ability of these viruses to adapt to a particular host species. These constraints may be relaxed for flaviviruses such as ENTV that are thought not to require an arthropod vector for their transmission. It will be interesting to investigate the antiviral activity of bat RTP4 orthologs against additional no-known-vector flaviviruses, which may have a greater ability to engage in an arms race with their vertebrate host compared to vector-borne flaviviruses.
In conclusion, Boys et al. demonstrate that the ortholog of RTP4 found in P. alecto is a potent antiviral effector, particularly against flaviviruses. They defined the antiviral domain of the protein and showed that it targets viral double-stranded RNA, ultimately disrupting viral replication. They then compared RTP4 activity across diverse sets of mammals and flaviviruses, identifying relationships consistent with an evolutionary virus-host arms race. These data raise exciting new questions for the fields of innate antiviral immunity, flavivirus host-pathogen interactions, and mammalian and viral evolution.
References
- Best S.M. The Many Faces of the Flavivirus NS5 Protein in Antagonism of Type I Interferon Signaling. J. Virol. 2017;91:e01970-16. doi: 10.1128/JVI.01970-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blitvich B.J., Firth A.E. A Review of Flaviviruses that Have No Known Arthropod Vector. Viruses. 2017;9:154. doi: 10.3390/v9060154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boys I.N., Xu E., Mar K.B., De La Cruz-Rivera P.C., Eitson J.L., Moon B., Schoggins J.W. RTP4 Is a Potent IFN-Inducible Anti-flavivirus Effector Engaged in a Host-Virus Arms Race in Bats and Other Mammals. Cell Host Microbe. 2020;28:712–723.e9. doi: 10.1016/j.chom.2020.09.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daugherty M.D., Malik H.S. Rules of engagement: molecular insights from host-virus arms races. Annu. Rev. Genet. 2012;46:677–700. doi: 10.1146/annurev-genet-110711-155522. [DOI] [PubMed] [Google Scholar]
- Fagre A.C., Kading R.C. Can Bats Serve as Reservoirs for Arboviruses? Viruses. 2019;11:215. doi: 10.3390/v11030215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayman D.T.S. Bats as Viral Reservoirs. Annu. Rev. Virol. 2016;3:77–99. doi: 10.1146/annurev-virology-110615-042203. [DOI] [PubMed] [Google Scholar]
- Letko M., Seifert S.N., Olival K.J., Plowright R.K., Munster V.J. Bat-borne virus diversity, spillover and emergence. Nat. Rev. Microbiol. 2020;18:461–471. doi: 10.1038/s41579-020-0394-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierson T.C., Diamond M.S. The continued threat of emerging flaviviruses. Nat. Microbiol. 2020;5:796–812. doi: 10.1038/s41564-020-0714-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Valen L. A New Evolutionary Law. Evol. Theory. 1973;1:1–30. [Google Scholar]