Abstract
Sarcopenia and frailty are associated with aging. In older burn patients, frailty has been associated with mortality and discharge disposition, but sarcopenia has not been examined. This study aims to investigate the relationship between frailty and computed tomography (CT)-derived sarcopenia with length of stay and mortality in older burn patients. Burn patients ≥60 years old admitted between 2008 and 2017 who had chest or abdomen CT scans within 1 week of admission were evaluated. Frailty was assessed using the Canadian Study of Health and Aging Clinical Frailty Scale (CFS). Sarcopenia was assessed on CT exams by measuring skeletal muscle index (SMI) of paraspinal muscles at T12 and all skeletal muscles at L3. The relationship between frailty scores and SMI with length of stay (LOS) and mortality was determined using logistic regression. Eighty-three patients (59 men; mean age 70.2 ± 8.5 years) had chest (n = 50) or abdomen (n = 60) CT scans. Mean TBSA = 14.3 ± 14.0%, LOS = 25.8 ± 21.3 days, CFS = 4.36 ± 0.99. Sixteen patients (19.3%) died while in the hospital. CT-derived measurement of SMI at T12 was significantly associated with LOS (P < .05), but not with mortality (P = .561). CT-derived metrics at L3 were not significantly associated with outcomes. CFS was not associated with LOS (P = .836) or mortality (P = .554). In older burn patients, low SMI of the paraspinal muscles at T12 was associated with longer LOS.
The growth rate of the elderly population in the United States is increasing more rapidly than the growth rate of the general population. By 2030, the elderly population is expected to exceed 20% of the total population, compared to 13% in 2010.1 A study of nearly 200,000 trauma admissions reported an increased mortality rate in patients over the age of 40 years.2 In older adults who sustain a burn injury, frailty has been associated with increased mortality, increased length of stay (LOS), and poorer discharge disposition.3,4 CT-derived sarcopenia has not been examined in older burn patients.
Multiple outcome models have been used in burn patients to guide treatment decisions and inform discussions with families and patients.5,6,7 However, the accuracy of these models in older adults has not been examined. Frailty is an important predictor of clinical outcomes in elderly patients.8 Frailty is characterized by physical and cognitive decline, which results in increased risks of adverse outcomes, including mortality, falls, and increased LOS.9,10,11 The most commonly studied frailty scale in burn patients is the Canadian Study on Health and Aging Clinical Frailty Scale (CFS).12 In elderly burn and trauma patients, every 1-unit increase CFS increases the likelihood of death by 23%.3,4,13
While clinical frailty scores are useful, they are subjective and require patient participation. For burn patients, clinical scoring of frailty is often not possible, particularly during intensive care and various life-sustaining treatments (eg, mechanical ventilation and emergent surgery). Thus, there is a need for an objective assessment of frailty that does not rely on patient participation. Ideally, this assessment would incorporate clinical frailty scales as well as a direct assessment of physiologic status.
Sarcopenia is generally defined by loss of skeletal muscle mass and muscle function.14 Although sarcopenia has been shown to predict mortality and LOS in postoperative patients,15 it has not been studied in elderly burn patients. CT scans have become widely used to measure muscle mass because measurements can be obtained easily on routine clinical scans obtained in the course of patient care.16,17,18 A study of 450 trauma patients reported that CT-derived sarcopenia was associated with increased mortality.19 In another study of 252 elderly patients who had sustained blunt trauma, CT-derived sarcopenia was associated with frailty, as manifested by a decreased likelihood of independent living after hospital discharge.20
The purpose of our study was to determine whether CT-derived sarcopenia predicts outcomes in elderly patients with a burn injury. We hypothesized that CFS scores, and CT-derived sarcopenia, would predict mortality and increased LOS in elderly burn patients.
MATERIALS AND METHODS
Patients
Following approval from the Institutional Review Board, a retrospective study was conducted in all patients aged 60 years and older admitted with a burn injury between 2008 and 2017. Patients who had a CT scan of the chest or abdomen (or both) within 1 week of admission were included in the study. Electronic health record was reviewed to determine age, sex, burn size (% TBSA), inhalation injury, LOS, and mortality.
Calculation of CFS
Two authors independently assessed frailty scores to determine preburn functional status using the CHSA CFS. Both authors determined comorbidities, mobility, and ability to perform activities of daily living (ADLs) prior to the burn injury to establish a frailty score for each patient. The score was extrapolated based on clinical deficits indicated in admission history, physicals, physical therapy and occupational therapy consults, social work notes, and discharge planning notes. Frailty scores were calculated on a scale from 1 (very fit) to 7 (severely frail) (Supplementary Table 1).12
CT Protocol
Patients were scanned on one of the five multidetector CT scanners: four 64-row CT scanners (one Somatom Definition, Siemens Healthcare; one Somatom Sensation, Siemens Healthcare; two LightSpeed VCT, GE Healthcare) and one 128-row CT scanner (Somatom Definition AS+, Siemens Healthcare). Calibration of the CT scanners was completed daily using a quality assurance phantom to ensure consistency in measurements of attenuation (also referred to as “radiodensity” or “density”), following specifications of the CT manufacturer and the American College of Radiology. Scans were performed routinely at 120 kV. Scanning was conducted with automated dose modulation (typical effective tube current of 220–260 mA). All region-of-interest measurements were made on axial images reconstructed using a filtered back-projection technique with the standard body filter (on GE scanners) or a B40f kernel (on Siemens scanners). Slice thickness varied depending on the type of study (chest, 2.5–5 mm; abdomen, 1.25–5 mm).
CT Measurements
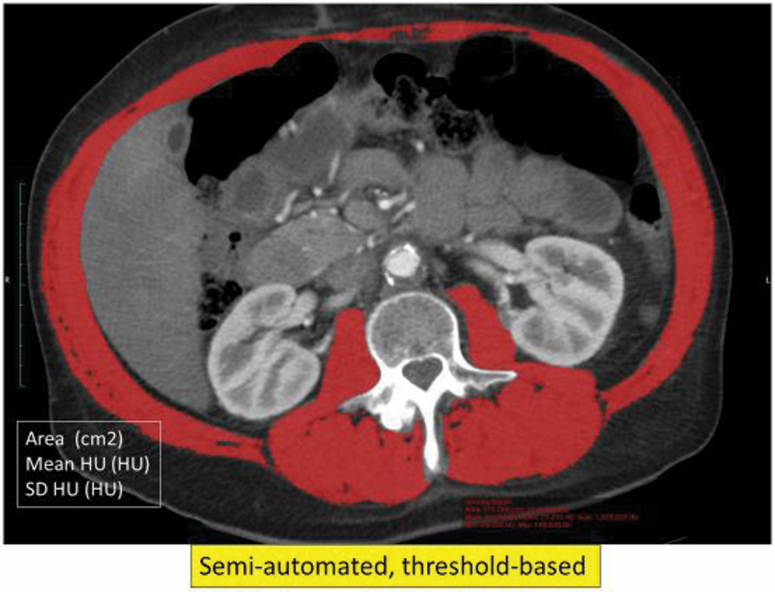
Measurements were made independently on every CT scan by a musculoskeletal radiologist with 3 years of experience and reviewed by a second musculoskeletal radiologist with 22 years of experience. CTs were excluded if they were limited by artifact, such as beam-hardening artifact from spinal instrumentation. Discrepancies were resolved by consensus. Radiologists were blinded to clinical data. Muscle measurements were performed with commonly used software (OsiriX MD version 9.5.1; Pixmeo, Bernex, Switzerland), which allows muscle regions of interest to be identified and thresholded using a standard range (ie, −29 to +150 HU). Within the segmented muscle region-of-interest, the software outputs measurements of skeletal muscle cross-sectional area (CSA, measured in cm2) and skeletal muscle density (SMD, measured in Hounsfield units [HU]). Based on prior work,16 CT scans were analyzed: 1) at the level of T12 pedicle on CT scans of the chest and abdomen, where all posterior paraspinal skeletal muscle was segmented (Figure 1) and 2) at the level of L3 pedicle on CT scans of the abdomen, where all skeletal muscle was segmented (Figure 2).
Figure 1.
Axial CT scan at the T12 level shows the posterior paraspinal muscle in red. Muscle is segmented to include tissue with a CT density between −29 HU and +150 HU.
Figure 2.
Axial CT scan at the L3 level shows skeletal muscle in red. Muscle is segmented to include tissue with a CT density between −29 HU and +150 HU.
CSA of posterior paraspinal muscles at T12 (CSA-T12) (the sum of the right and left posterior paraspinal muscle areas at T12 [cm2]) was measured and then adjusted for patient height to yield a skeletal muscle index at T12 (SMI-T12, calculated as CSA-T12 [cm2] divided by patient height [m2]). Skeletal muscle density at T12 (SMD-T12) also was recorded (the average density of the right and left posterior paraspinal muscles measured in Hounsfield units, HU). Similarly, CSA of skeletal muscles at L3 (CSA-L3, measured in cm2), skeletal muscle index at L3 (SMI-L3, calculated as CSA-L3 [cm2] divided by patient height [m2]), and skeletal muscle density at L3 (SMD-L3, measured in HU) were recorded. The intra-rater reliability of CT measurements was assessed from 25% of the exams after a minimum latency period of 4 weeks.
Statistical Analysis
Quantitative variables were summarized as means ± standard deviation (SD) and categorical variables as proportions. Logistic regression was used to evaluate the effect of predictors on mortality. One patient was discharged to hospice and, for the sake of analysis, was considered to have died by the time of analysis. Linear regression was used to evaluate LOS. Although LOS can be right-skewed, Q–Q plots of residuals from the linear regressions indicated sufficient conformity to normality. All subjects were used in the LOS evaluation because LOS did not differ significantly (Wilcoxon rank-sum test, P = .11) between survivors (median = 2210,39) and nonsurvivors (median = 173,32). First, we evaluated the effect of age, TBSA, and inhalation injury as these are commonly associated with mortality and LOS in burn patients. Second, to assess whether frailty scores or sarcopenia metrics could predict mortality and/or LOS as well as TBSA, age, and inhalation injury, we evaluated the univariate effects of frailty scores and sarcopenia metrics. Due to the small sample size of this study, we examined not only the hypothesis tests for the data but also the point estimates and 95% confidence intervals (CI).
CT muscle metrics are known to differ markedly between men and women.21 To account for the sex differences, each muscle metric was scaled to mean of 0 and a standard deviation of 1. This scaling removed sex differences and the results of the analyses express the effect of a 1 standard deviation difference from the sex-specific mean of the outcome. In addition, CT muscle metrics were dichotomized into normal and abnormal based on published cutpoints for abnormally low muscle mass (myopenia)22,23 and density (myosteatosis) (Tables 1 and 2).24,25 Two cutpoints were used for skeletal muscle index (SMI) values at the L3 level.22,23 For skeletal muscle density (SMD), the cutpoint from Aubrey et al 25 was used because the Derstine et al24 L3 density cutpoint only identified two abnormal patients. For patients receiving intravenous contrast material, the CT values for muscle density were adjusted.21
Table 1.
Cutpoints for CT-derived skeletal muscle index (SMI) metrics proposed in the literature
| CT Metric (cm2/m2) | Women | Men | Reference |
|---|---|---|---|
| L3 SMI | 34.4 | 45.4 | [22] |
| L3 SMI | 38.5 | 52.4 | [23] |
| T12 SMI | 7.8 | 10.9 | [22] |
Values were defined as abnormal if they were below the specified thresholds.
Table 2.
Cutpoints for CT-derived skeletal muscle density (SMD) metrics proposed in the literature
| CT Metric (HU) | Women | Men | Reference |
|---|---|---|---|
| T12 SMD | 30 | 30 | [25] |
| L3 SMD | 30 | 30 | [25] |
| L3 SMD | 34.3 | 38.5 | [24] |
Values were defined as abnormal if they were below the specified thresholds. Values corrected for administration of contrast agents.
In addition to the primary objective of evaluating the relationships between frailty and sarcopenia metrics and patient outcomes, data were collected allowing estimation of inter- and intra-rater reliability of these metrics. Frailty was assessed by two raters allowing estimation of interrater reliability for this measure. For CT metrics, inter- and intra-rater reliability was estimated by the intraclass correlation (ICC) with random subjects and fixed raters.
All analyses were conducted using R Version 3.5.1 Statistical Computing Software. Statistical tests were two-sided with a significance level of 0.05.
RESULTS
There were 653 patients over the age of 60 years admitted to the burn unit from 2008 to 2017. A total of 83 patients (12.7% of this total population of burn unit admissions) had a thoracic CT scan (n = 50) and/or abdomen CT scan (n = 60) and were included in the study. Patient demographics are shown in Table 3. Means and standard deviations for each metric are shown in Table 4.
Table 3.
Patient characteristics (Mean ± SD)
| n = 83 | |
|---|---|
| Age (years) | 70.2 ± 8.5 |
| Male (N [%]) | 59 (71.1%) |
| %TBSA burn | 14.3 ± 14.0 |
| Inhalation injury (%) | 22.90% |
| CFS | 4.36 ± 0.99 |
| OR trips | 1.13 ± 1.32 |
| ICU days | 22.7 ± 21.0 |
| Ventilator days | 14.7 ± 20.2 |
| Mortality (%) | 20.50% |
| LOS (days) | 25.76 ± 21.3 |
Table 4.
Mean and standard deviations for CT-derived metrics in burn patients
| Mean | SD | ||
|---|---|---|---|
| T12 | CSA (cm2) | 27.24 | 8.62 |
| SMI (cm2/m2) | 9.11 | 2.44 | |
| SMD (HU) | 18.07 | 6.59 | |
| L3 | CSA (cm2) | 128.13 | 32.84 |
| SMI (cm2/m2) | 43.43 | 9.96 | |
| SMD (HU) | 29.07 | 10.76 |
The study included 24 women and 59 men with a mean age of 70.2 ± 8.5 years. The mean TBSA was 14.3 ± 14.0% and mean LOS was 25.8 ± 21.3 days. Inhalation injury was reported in 19 patients (22.9%) and 16 patients (19.3%) died while in the hospital. The mean CFS was 4.36 ± 0.99. Frailty scores were not significantly associated with mortality (OR = 0.848 [0.492, 1.467], P = .554), or LOS (estimate =−0.496 [−5.254, 4.262], P = .836).
T12 CT Results
Of the 50 patients who had CT scans that included the T12 level, 14 (25%) died, including 7 women and 7 men. Five metrics for the T12 CT images were evaluated, but none of these were significant predictors of mortality (Table 5). For mortality, the odds ratio (OR) for SMI was 0.694 [0.194, 2.487, P = .575], suggesting that as the muscle size increases, the risk of mortality decreases. However, this did not reach the level of statistical significance. The OR for scaled SMD was 1.52 [0.79, 2.922, P = .210], suggesting that as density increases, the risk of mortality increases, but again this was not a statistically significant result. The findings for CT metrics with relation to LOS are shown in Table 6. The estimate for the relationship between LOS and CSA was −7.38 [−14.08, −0.675, P = .032]. After accounting for patient height, the relationship between LOS and SMI was −14.93 [−28.21, −1.644, P = .028], and for scaled SMI was −9.212 [−15.723, −2.702, P = .007]. These results suggest that the average LOS was 7.28 days shorter for subjects in the normal range when compared with those in the abnormal range but could be as small as less than one day or as big as 14 days based on the 95% CI. Conversely, as SMI decreased, the LOS for patients increased. SMD at the T12 level was not statistically associated with LOS. For each 1-unit increase in SMD (HU), the average LOS increased by 0.710 days [−6.325, 7.745], P = .84].
Table 5.
Logistic regression for mortality using patients with T12 CT scans
| Predictor | Estimate | Std. Error | P |
|---|---|---|---|
| SMI (Normal) | −0.365 | 0.651 | .575 |
| Scaled SMI | −0.191 | 0.329 | .561 |
| Scaled CSA | −0.201 | 0.327 | .537 |
| Scaled SMD | 0.418 | 0.334 | .210 |
Table 6.
Linear regression for LOS using patients with T12 CT scans
| Predictor | Estimate | Std. Error | P |
|---|---|---|---|
| SMI (Normal) | −14.928 | 6.607 | .028* |
| Scaled SMI | −9.212 | 3.238 | .007* |
| Scaled CSA | −7.380 | 3.335 | .032* |
| Scaled SMD | 0.710 | 3.499 | .840 |
*P < .05.
L3 CT Results
Of the 60 patients who had CT scans that included the L3 level, 15 (25%) died (6 women and 9 men). Eight different CT metrics were independently evaluated for their relationship with mortality and LOS. As shown in Tables 1 and 2, there were two different thresholds for SMI and SMD at the L3 level. The first SMI threshold22 identified 24 patients as abnormal and 36 patients as normal, while the second threshold23 identified 40 patients as abnormal and 20 patients as normal. The first SMD threshold25 identified 49 patients as abnormal and 11 patients as abnormal, while the second SMD threshold24 identified 58 patients as abnormal and 2 patients as normal. Because only two normal patients were identified using the second SMD threshold, this threshold was eliminated from the analysis. These were analyzed separately and as scaled values. The results of the logistic regression analysis for the CT metrics and mortality are shown in Table 7. None of the L3 CT image metrics were significant predictors of mortality. The OR for the scaled SMI at L3 was 0.60 [0.313, 1.144, P = .120] and for the scaled CSA the OR was 0.63 [0.328, 1.199, P = .158]. While not statistically significant in this study, the odds ration suggests that as the SMI and CSA increased, the risk of mortality decreased. This suggests that those with higher muscle mass may have improved odds of survival, but that our current study does not have adequate power. The association between SMD and mortality also did not reach the level of significance. The OR for mortality with respect to SMD was 1.16 [0.628, 2.129, P = .64].
Table 7.
Logistic regression for mortality using patients with L3 CT scans
| Estimate | Std. Error | P | |
|---|---|---|---|
| SMI ref [22] cutoff | −0.365 | 0.602 | .544 |
| SMI ref [23] cutoff | −1.466 | 0.818 | .073 |
| SMD ref [25] cutoff | 0.145 | 0.754 | .847 |
| Scale CSA | −0.466 | 0.330 | .158 |
| Scaled SMI | −0.514 | 0.331 | .120 |
| Scaled SMD | 0.146 | 0.311 | .640 |
The results of the linear regression analysis for the CT metrics and LOS are shown in Table 8. None of the CT metrics for muscle size or density were significant for LOS. The estimate for scaled CSA at L3 was −2.143 [−7.957, 3.672, P = .464] and the scaled SMI at L3 was −2.452 [−8.258, 3.354, P = .404]. While neither of these results was significant, both were negative, indicating that larger muscle size may be associated with a shorter LOS. The same is true for SMD and according to the linear regression as the density increases, the LOS decreases [estimate =−2.219, −8.032, 3.593, P = .448].
Table 8.
Linear regression for LOS using patients with L3 CT scans
| Predictor | Estimate | Std. Error | P |
|---|---|---|---|
| SMI ref [22] cutoff | −11.167 | 5.670 | .054 |
| SMI ref [23] cutoff | −6.000 | 6.036 | .324 |
| SMD ref [25] cutoff | −13.642 | 7.196 | .063 |
| Scaled CSA | −2.143 | 2.905 | .464 |
| Scaled SMI | −2.452 | 2.901 | .401 |
| Scaled SMD | −2.219 | 2.904 | .448 |
Inter- and Intra-Rater Reliability of Frailty and CT Metrics
The process of data collection allowed investigations into inter- and intra-rater reliability of aspects of clinical frailty and CT metrics. Since frailty was assessed for each patient by two observers, the interrater reliability of the two frailty scores was 0.93 [0.9, 0.95]. For a subset of patients who had a second analysis of CT scans by the same person, intra-rater reliability was greater than 0.95.
DISCUSSION
As the population ages, the desire to better prognosticate which elderly patients will do well following injury or surgery has led to the development of numerous frailty scales and sarcopenia metrics. In the burn population, frailty has been shown to predict mortality and discharge to a SNF.3,4,13 This study demonstrates that routinely acquired chest or abdomen CT scans that include the paraspinal muscles at the T12 level may provide information that can predict LOS in an elderly burn population.
To date, there are no studies of sarcopenia in an elderly burn population; however, there have been many studies in other surgical subspecialties that have examined the relationship between sarcopenia and a variety of patient outcomes.20,26,27 These studies have mixed results. Some studies have found that sarcopenia is associated with increased mortality, loss of independence, discharge to a SNF, and increased LOS.20,26,27 Other studies, including Hamidi et al28 in middle-aged Emergency General Surgery patients, have been less promising. They reported that psoas SMI was not associated with in-hospital major complications, mortality or readmissions, but was associated with minor complications and LOS.28 The same group examined psoas size with a quartile-based approach in geriatric trauma patients and reported a weak correlation between frailty and sarcopenia (R2 = 0.04), with sarcopenia predictive of adverse discharge disposition, but not mortality or in-hospital complications.29
In our study, the correlation between frailty and SMI was similar at the T12 level (0.05) and decreased at the L3 level (0.007). The lack of an association between frailty and sarcopenia may seem surprising as these two entities are often discussed together. Although interrelationships and partial overlaps exist between physical frailty and sarcopenia, the association between these two entities is weaker than might be expected, likely because of different operational definitions 30. While there is some overlap between frailty and sarcopenia, these two entities are distinct. 31 Frailty is defined as a clinical state of increased vulnerability of an older person to stressors in physical, psychological, or social domains, and sarcopenia is often considered a component of physical frailty. In other words, “frailty” is generally considered a broader term used to indicate reduced homeostatic reserves, while “sarcopenia” generally refers to a loss of muscle mass, quality, and function. Both physical frailty and sarcopenia are more common with age, have multifactorial pathogenesis, have been associated with adverse outcomes (eg, falls, hospitalization, and decreased survival), and are active areas of research to determine whether prevention and treatment interventions can minimize adverse clinical outcomes. Generally, frailty is observed less often than sarcopenia (often with a ratio of approximately 2 to 1), and frail older people are more likely to be sarcopenic than those who were not frail.32 On the other hand, patients with some medical conditions (eg, cancer or burns) may not be overtly frail at the time of presentation, but may have latent, objective features of sarcopenia on CT that are associated with a diminished physiologic reserve, even in younger patients.
Similar to Mccusker et al,29 our goal was to compare the association of frailty and sarcopenia with mortality, LOS, and discharge disposition. In our elderly population of burn patients, we were unable to find an association between mortality and frailty or between mortality and CT-derived sarcopenia at the T12 and L3 levels. This is likely because the mortality in this study was driven by the severity of the burn and the presence of concomitant trauma rather than the baseline condition of the patient as patients who underwent CT scans had a higher mortality rate (20%) than those who did not have a CT scan (~10%). In this study, frailty was not predictive of discharge disposition and we were unable to evaluate the association of discharge disposition and sarcopenia due to the small sample size in each group. Prior work by our group did show an association between increased frailty scores and discharge to a SNF, but the population who received CT scans was only a small portion of the total burn population.4,13 In a study of patients with pancreatic cancer who underwent pancreatectomy, a decrease in psoas SMI of 1.5 cm/m2 was associated with a LOS greater than 10 days with an odds ratio of 1.75 (95% CI: 1.10–2.78).27 We also found an association between SMI and LOS at the T12 level (odds ratio = 2.22).
Our study is unique compared to previously discussed studies of sarcopenia,28,29 which either did not assess CT metrics that have been more widely validated16 and/or did not define sarcopenia based on specific CT cutpoints. CT measurements of muscle size (SMI) and density (SMD) are continuous variables, but there is considerable interest in operationalizing specific diagnostic cutpoints to define normal versus abnormal in clinical practice. For this study, to examine the effects of SMI and SMD as fully as possible, we chose to evaluate the effects of SMI and SMD on mortality and length of stay for both the continuous variable as well as for various previously established cutpoints of abnormal vs. normal. For SMI at the L3 level, we analyzed the performance of two cutpoints: 1) two standard deviations below the mean SMI in a healthy population (604 healthy kidney donors in the United States, ages 18–40) 22 and 2) the most commonly used CT sarcopenia cutpoint in the medical literature (using optimal stratification analysis for predicting mortality in 250 patients with respiratory and gastrointestinal cancers) 23. For SMD at the L3 level, we also evaluated the performance of two cutpoints: 1) the widely accepted cut point of 30 HU defined as two standard deviations below the mean in young, healthy persons 25 and 2) a recently proposed cutpoint defined as two standard deviations below the mean in 735 kidney donors 24. Sarcopenia at the T12 level is not as commonly studied. As such, only one cutpoint for SMI and SMD were evaluated at T12. For SMI at the T12 level, we analyzed the performance of a cutpoint of two standard deviations below the mean SMI in a healthy population22 and for SMD at the T12 level the widely accepted cut point of 30 HU defined as two standard deviations below the mean in young, healthy persons was used.25 In our analysis, the use of cutpoints revealed similar results at both the T12 and L3 levels and for mortality and LOS to the results seen using the continuous data. Although we tested the use of specific cutpoints in our burn cohort, we recognize that any binary classification may not fully capture the magnitude of patient risk, both near and far from cutoff values.
One of the limitations of this study was its retrospective nature. We were also unable to track patient outcomes after they were discharged from the hospital. Another limitation is that few burn patients received a CT scan (only 12.7% of patients over age 60 years admitted during the period studied), and those that did have scans are inherently different than those who did not have scans. Routine CT scanning is not standard of care for patients with a burn injury, which limits the generalizability of these findings. We chose to limit the time interval between admission and CT scanning to only 1 week, because of the increasingly recognized phenomenon of “acute sarcopenia.” 33 Both the prevalence of CT scans in the burn population and limiting the timing of the scans made the sample size for this study small. This significantly limits the power of the study to detect and estimate relationships. Therefore, there may be differences in outcomes that are related to frailty or sarcopenia that we are unable to detect in this study. It is also plausible that the rate and magnitude of change over time in individual patients may be predictive of outcomes that were not evaluated with our study design.
This study also has several strengths. First, it is the first and largest study to examine frailty and sarcopenia in older burn patients. Second, we used CT, a widely used technique that has been validated for quantifying sarcopenia (eg, SMI) and myosteatosis (eg, SMD) and applied it to older patients with burn injuries using reference values from the literature. Finally, we maintained a short time interval between admission and CT scans to minimize temporal changes in muscle metrics expected to occur during hospitalization.
This study serves as one of the first attempts to examine frailty and sarcopenia together in an elderly burn population. CT-derived sarcopenia appears to predict LOS in this population. A prospective analysis of the relationship between frailty and sarcopenia is warranted. Future studies could include the assessment of temporal muscle changes in individual patients.
CONCLUSION
CT-derived measurements of sarcopenia at T12 were predictive of hospital length of stay in older burn patients.
Supplementary Material
Funding: This project was supported by the National Center for Advancing Translational Sciences, National Institutes of Health, through grant number UL1 TR001860. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH. This project was also supported by the University of California, Davis Collaborative for Diagnostic Innovation seed grant.
REFERENCES
- 1. Ortman JM Velkoff VA, Hogan H. an aging nation: the older population in the United States. U.S. Department of Commerce 2014. P25–1140. [Google Scholar]
- 2. Morris JA Jr, MacKenzie EJ, Damiano AM, Bass SM. Mortality in trauma patients: the interaction between host factors and severity. J Trauma 1990;30:1476–82. [PubMed] [Google Scholar]
- 3. Masud D, Norton S, Smailes S, Shelley O, Philp B, Dziewulski P. The use of a frailty scoring system for burns in the elderly. Burns 2013;39:30–6. [DOI] [PubMed] [Google Scholar]
- 4. Romanowski KS, Barsun A, Pamlieri TL, Greenhalgh DG, Sen S. Frailty score on admission predicts outcomes in elderly burn injury. J Burn Care Res 2015;36:1–6. [DOI] [PubMed] [Google Scholar]
- 5. Gomez M, Wong DT, Stewart TE, Redelmeier DA, Fish JS. The FLAMES score accurately predicts mortality risk in burn patients. J Trauma 2008;65:636–45. [DOI] [PubMed] [Google Scholar]
- 6. Ryan CM, Schoenfeld DA, Thorpe WP, Sheridan RL, Cassem EH, Tompkins RG. Objective estimates of the probability of death from burn injuries. N Engl J Med 1998;338:362–6. [DOI] [PubMed] [Google Scholar]
- 7. Williams DJ, Walker JD. A nomogram for calculation of the Revised Baux Score. Burns 2015;41:85–90. [DOI] [PubMed] [Google Scholar]
- 8. Joseph B, Pandit V, Sadoun M, et al. Frailty in surgery. J Trauma Acute Care Surg 2014;76:1151–6. [DOI] [PubMed] [Google Scholar]
- 9. Torpy JM, Lynm C, Glass RM. JAMA patient page. Frailty in older adults. JAMA 2006;296:2280. [DOI] [PubMed] [Google Scholar]
- 10. Joseph B, Pandit V, Zangbar B, et al. Validating trauma-specific frailty index for geriatric trauma patients: a prospective analysis. J Am Coll Surg 2014;219:10–17.e1. [DOI] [PubMed] [Google Scholar]
- 11. Clegg A, Young J, Iliffe S, Rikkert MO, Rockwood K. Frailty in elderly people. Lancet 2013;381:752–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Rockwood K, Song X, MacKnight C, et al. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005;173:489–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Curtis E, Romanowski K, Sen S, Hill A, Cocanour C. Frailty score on admission predicts mortality and discharge disposition in elderly trauma patients over the age of 65 y. J Surg Res 2018;230:13–19. [DOI] [PubMed] [Google Scholar]
- 14. Cruz-Jentoft AJ, Bahat G, Bauer J, et al. ; Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2 Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 2019;48:16–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. DeAndrade J, Pedersen M, Garcia L, Nau P. Sarcopenia is a risk factor for complications and an independent predictor of hospital length of stay in trauma patients. J Surg Res 2018;221:161–6. [DOI] [PubMed] [Google Scholar]
- 16. Amini B, Boyle SP, Boutin RD, et al. Approaches to assessment of muscle mass and myosteatosis on computed tomography: a systematic review. J Geront: Series A. 74, Issue October 10, 2019, p. 1671–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lenchik L, Lenoir KM, Tan J, et al. Opportunistic measurement of skeletal muscle size and muscle attenuation on computed tomography predicts 1-year mortality in medicare patients. J Gerontol A Biol Sci Med Sci 2019;74:1063–069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Boutin RD, Bamrungchart S, Bateni CP, et al. CT of patients with hip fracture: muscle size and attenuation help predict mortality. AJR Am J Roentgenol 2017;208:W208–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Kaplan SJ, Pham TN, Arbabi S, et al. Association of radiologic indicators of frailty with 1-year mortality in older trauma patients: opportunistic screening for sarcopenia and osteopenia. JAMA Surg 2017;152:e164604. [DOI] [PubMed] [Google Scholar]
- 20. Fairchild B, Webb TP, Xiang Q, Tarima S, Brasel KJ. Sarcopenia and frailty in elderly trauma patients. World J Surg 2015;39:373–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Boutin RD, Kaptuch JM, Bateni CP, Chalfant JS, Yao L. Influence of IV contrast administration on ct measures of muscle and bone attenuation: implications for sarcopenia and osteoporosis evaluation. AJR Am J Roentgenol 2016;207:1046–054. [DOI] [PubMed] [Google Scholar]
- 22. Derstine BA, Holcombe SA, Goulson RL, et al. Quantifying sarcopenia reference values using lumbar and thoracic muscle areas in a healthy population. J Nutr Health Aging 2017;21:180–5. [DOI] [PubMed] [Google Scholar]
- 23. Prado CM, Lieffers JR, McCargar LJ, et al. Prevalence and clinical implications of sarcopenic obesity in patients with solid tumours of the respiratory and gastrointestinal tracts: a population-based study. Lancet Oncol 2008;9:629–35. [DOI] [PubMed] [Google Scholar]
- 24. Derstine BA, Holcombe SA, Ross BE, Wang NC, Su GL, Wang SC. Skeletal muscle cutoff values for sarcopenia diagnosis using T10 to L5 measurements in a healthy US population. Sci Rep 2018;8:11369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Aubrey J, Esfandiari N, Baracos VE, et al. Measurement of skeletal muscle radiation attenuation and basis of its biological variation. Acta Physiol (Oxf) 2014;210:489–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Friedman J, Lussiez A, Sullivan J, Wang S, Englesbe M. Implications of sarcopenia in major surgery. Nutr Clin Pract 2015;30:175–9. [DOI] [PubMed] [Google Scholar]
- 27. Joglekar S, Asghar A, Mott SL, et al. Sarcopenia is an independent predictor of complications following pancreatectomy for adenocarcinoma. J Surg Oncol 2015;111:771–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Hamidi M, Ho C, Zeeshan M, et al. Can sarcopenia quantified by computed tomography scan predict adverse outcomes in emergency general surgery? J Surg Res 2019;235:141–7. [DOI] [PubMed] [Google Scholar]
- 29. Mccusker A, Khan M, Kulvatunyou N, et al. Sarcopenia defined by a computed tomography estimate of the psoas muscle area does not predict frailty in geriatric trauma patients. Am J Surg 2019;218:261–5. [DOI] [PubMed] [Google Scholar]
- 30. Reijnierse EM, Trappenburg MC, Blauw GJ, et al. Common ground? the concordance of sarcopenia and frailty definitions. J Am Med Dir Assoc 2016;17:371.e7–371.12. [DOI] [PubMed] [Google Scholar]
- 31. Williams GR, Deal AM, Muss HB, et al. Frailty and skeletal muscle in older adults with cancer. J Geriatr Oncol 2018;9:68–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Mijnarends DM, Schols JM, Meijers JM, et al. Instruments to assess sarcopenia and physical frailty in older people living in a community (care) setting: similarities and discrepancies. J Am Med Dir Assoc 2015;16:301–8. [DOI] [PubMed] [Google Scholar]
- 33. Welch C, K Hassan-Smith Z, A Greig C, M Lord J, A Jackson T. Acute sarcopenia secondary to hospitalisation–an emerging condition affecting older adults. Aging Dis 2018;9:151–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.